Enablex Tablets
Brand Information
| Brand name | Enablex Tablets |
| Active ingredient | Darifenacin |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Enablex Tablets.
Summary CMI
ENABLEX tablets
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using ENABLEX?
ENABLEX contains the active ingredient darifenacin (as darifenacin hydrobromide). ENABLEX is used to decrease the urgency and the frequency of urination.
For more information, see Section 1. Why am I using ENABLEX? in the full CMI.
2. What should I know before I use ENABLEX?
Do not use if you have ever had an allergic reaction to darifenacin or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use ENABLEX? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with ENABLEX and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use ENABLEX?
- The usual starting dose is 7.5mg once a day. This may be increased to 15 mg once a day if necessary.
- Swallow the tablet whole with a glass of water. Do not chew, crush, or divide the tablet.
More instructions can be found in Section 4. How do I use ENABLEX? in the full CMI.
5. What should I know while using ENABLEX?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using ENABLEX? in the full CMI.
6. Are there any side effects?
Common side effects of ENABLEX include constipation; diarrhoea; stomach ache or indigestion; nausea or vomiting; nasal dryness; flatulence; dry or itchy skin; dry eyes; blurred or changed vision; dryness of the mouth, nose, and throat; sore throat or cough; change in taste; mouth ulcers; swelling of the hands, ankles or feet; unusual tiredness, confusion, or weakness; difficulty sleeping; dizziness; pain in the joints; discharge or itching in the vagina; signs of a possible urinary tract infection, such as pain on urination; urinary retention; fast or irregular heart beats; weight gain; excessive sweating; and impotence.
Serious side effects of ENABLEX include sudden signs of allergy such as rash, itching, or hives on the skin; swelling of the face, lips, tongue, or other parts of the body; and shortness of breath, wheezing or trouble breathing.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
ENABLEX tablets
Active ingredient: darifenacin (as darifenacin hydrobromide)
Consumer Medicine Information (CMI)
This leaflet provides important information about using ENABLEX.
You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using ENABLEX.
Where to find information in this leaflet:
1. Why am I using ENABLEX?
2. What should I know before I use ENABLEX?
3. What if I am taking other medicines?
4. How do I use ENABLEX?
5. What should I know while using ENABLEX?
6. Are there any side effects?
7. Product details
1. Why am I using ENABLEX?
ENABLEX contains the active ingredient darifenacin (as darifenacin hydrobromide). ENABLEX is used to decrease the urgency and the frequency of urination.
ENABLEX works by relaxing the bladder smooth muscle and helps to decrease muscle spasm. This delays the initial desire and the urge to urinate.
Ask your doctor if you have any questions about why this medicine has been prescribed for you.
Your doctor may have prescribed it for another reason.
This medicine is not addictive.
2. What should I know before I use ENABLEX?
Warnings
Do not use ENABLEX if:
- you are allergic to darifenacin or any of the ingredients listed at the end of this leaflet.
Symptoms of an allergic reaction may include shortness of breath, wheezing or difficulty in breathing; swelling of the face, lips, tongue, or any other parts of the body; rash, itching or hives on the skin. - you have any of the following health conditions:
- a disorder of the eye called narrow angle glaucoma that is not being controlled with medicine.
- a blockage in the stomach or intestine.
- urinary tract blockage. - the expiry date printed on the pack has passed.
- the packaging is torn or shows signs of tampering.
Check with your doctor if you:
- have any other medical conditions, especially the following:
- heart problems
- liver problems
- narrow-angle glaucoma that is controlled with medicine.
- stomach, intestinal or large bowel problems, including chronic constipation. - have had any allergy to any other medicines, or any other substances, such as foods, preservatives, or dyes.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
There is no information on the use of this medicine during pregnancy. Your doctor will discuss the possible risks and benefits involved.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
It is not known if the active ingredient of ENABLEX passes into breast milk and whether it could affect your baby. Your doctor will discuss the possible risks and benefits of breast-feeding while taking ENABLEX.
Use in children
ENABLEX is not recommended for use in children because there have been no studies of its effects in this age group.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with ENABLEX and affect how it works. These include:
- medicines used to treat fungal infections, such as ketoconazole, itraconazole and miconazole
- medicines used to treat depression, such as paroxetine, fluoxetine, nefazodone and tricyclic antidepressants
- medicines used to treat heart problems, such as digoxin and flecainide
- cimetidine, a medicine used to treat stomach ulcers
- ritonavir, a medicine used to treat HIV/AIDS
- thioridazine, a medicine used to treat schizophrenia
- other medicines that work in a similar way to ENABLEX.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect ENABLEX.
4. How do I use ENABLEX?
How much to take
- The usual starting dose is one 7.5 mg tablet once a day.
- If necessary, your doctor may increase your dose to one 15 mg tablet once a day, as early as two weeks after starting therapy.
- If you have a problem with your liver or are taking certain medicines, your doctor may limit your dose to one 7.5 mg tablet once a day.
When to take it
Take ENABLEX at about the same time each day.
Taking your tablet at the same time each day will have the best effect. It will also help you remember when to take it.
How to take it
- Swallow the tablet whole with a glass of water. Do not chew, crush, or divide the tablet.
- It can be taken with or without food.
How long to take it for
Continue taking this medicine for as long as your doctor tells you to.
ENABLEX helps control your condition, but does not cure it.
If you forget to use it
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Otherwise, take it as soon as you remember, and then go back to taking your medicine as you would normally.
Do not take a double dose to make up for the dose you missed.
This may increase the chance of you getting an unwanted side effect.
If you're not sure what to do, ask your doctor or pharmacist.
If you take too much ENABLEX
If you think that you have taken too many ENABLEX tablets, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using ENABLEX?
Things you should do
- If you become pregnant while taking ENABLEX, tell your doctor.
Your doctor can discuss with you the risks of taking it while you are pregnant. - Remind any doctor, dentist or pharmacist you visit that you are using ENABLEX.
- If you are about to be started on any new medicine, tell your doctor, dentist, or pharmacist that you are taking ENABLEX.
Things you should not do
- Do not give this medicine to anyone else, even if their condition seems like yours.
- Do not take it to treat any other complaints unless your doctor tells you to.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how ENABLEX affects you.
This medicine may cause dizziness or blurred vision in some people.
Looking after your medicine
- Keep your tablets in the pack protected from light until it is time to take them.
- Keep ENABLEX tablets in a cool dry place, away from moisture or heat where the temperature stays below 25°C. For example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Heat and dampness can destroy some medicines.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Some side effects such as raised blood pressure may not give you any symptoms and can only be found when tests are done.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor or pharmacist if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Stop taking ENABLEX and tell your doctor immediately or go to Accident and Emergency at your nearest hospital. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What ENABLEX contains
| Active ingredient (main ingredient) |
|
| Other ingredients (inactive ingredients) |
|
Do not take this medicine if you are allergic to any of these ingredients.
What ENABLEX tablets look like
ENABLEX 7.5 mg tablets are round, white film coated, modified release tablets marked with "DF" on one side and "7.5" on the other side. Available in blister packs of 28 tablets.
ENABLEX 15 mg tablets are round, light peach film coated, modified release tablets marked with "DF" on one side and "15" on the other side. Available in blister packs of 28 tablets.
Australian Registration numbers:
ENABLEX 7.5 mg: AUST R 99418
ENABLEX 15 mg: AUST R 99439
Who distributes ENABLEX
Aspen Pharmacare Australia Pty Ltd
34-36 Chandos St
St Leonards NSW 2065
Australia
This leaflet was revised in October 2025.
Brand Information
| Brand name | Enablex Tablets |
| Active ingredient | Darifenacin |
| Schedule | S4 |
MIMS Revision Date: 01 December 2020
1 Name of Medicine
Darifenacin hydrobromide.
2 Qualitative and Quantitative Composition
The active ingredient of Enablex is darifenacin hydrobromide. Enablex modified-release tablets contain 7.5 mg or 15 mg darifenacin (as hydrobromide).
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Enablex 7.5 mg modified-release tablets are round, shallow, convex, white tablets with "DF" on one side and "7.5" on the reverse.
Enablex 15 mg modified-release tablets are round, shallow, convex, light peach-coloured tablets with "DF" on one side and "15" on the reverse.
4 Clinical Particulars
4.1 Therapeutic Indications
Enablex modified-release tablets are indicated for the treatment of overactive bladder, with the symptoms of urgency, urge urinary incontinence or frequency of micturition.
4.2 Dose and Method of Administration
The recommended starting dose of Enablex modified-release tablets is 7.5 mg once daily. For those patients requiring greater symptom relief, the dose may be increased to 15 mg daily, as early as two weeks after starting therapy, based on individual response.
Enablex modified-release tablets should be taken once daily with liquid. They may be taken with or without food and should be swallowed whole and not chewed, divided or crushed.
Use in patients with hepatic impairment. For patients with moderate hepatic impairment (Child-Pugh B) or when co-administered with potent CYP3A4 inhibitors (e.g. ketoconazole, itraconazole, miconazole, nefazodone and ritonavir), the daily dose of Enablex should not exceed 7.5 mg.
Enablex is not recommended for use in patients with severe hepatic impairment (Child-Pugh C) (see Section 4.4 Special Warnings and Precautions for Use).
Use in patients with renal impairment. There are no special dosage requirements for patients with renal impairment.
4.3 Contraindications
Hypersensitivity to the active substance or to any of the excipients. Enablex is contraindicated in patients with urinary retention, gastric retention or uncontrolled narrow angle glaucoma.
4.4 Special Warnings and Precautions for Use
Identified precautions. Concomitant conditions. Enablex should be administered with caution to patients with autonomic neuropathy, hiatus hernia, clinically significant bladder outflow obstruction, risk for urinary retention, severe constipation (defined as two or less bowel movements per week) or gastrointestinal obstructive disorders such as pyloric stenosis (see Section 4.3 Contraindications).
Enablex should be used with caution in patients being treated for narrow angle glaucoma (see Section 4.3 Contraindications).
As with other antimuscarinics, patients should be instructed to discontinue Enablex and seek immediate medical attention if they experience edema of the tongue or laropharynx, or difficulty breathing (see Section 4.8 Adverse Effects (Undesirable Effects)).
Enablex should be used with caution in patients with risk of decreased gastrointestinal motility (see Section 4.3 Contraindications), gastro-oesophagal reflux and/or who are concurrently taking medicinal products (such as oral bisphosphonates) that can cause or exacerbate oesophagitis.
Caution should be used when prescribing antimuscarinics to patients with pre-existing cardiac diseases.
Use in hepatic impairment. There is a risk of increased exposure in this population (see Section 5.2 Pharmacokinetic Properties). No dose adjustment is required in patients with mild hepatic impairment (Child-Pugh A). For patients with moderate hepatic impairment (Child-Pugh B) or when co-administered with potent CYP3A4 inhibitors (e.g. ketoconazole, itraconazole, miconazole, nefazodone and ritonavir), the daily dose of Enablex should not exceed 7.5 mg. Enablex is not recommended for use in patients with severe hepatic impairment (Child-Pugh C) (see Section 4.2 Dose and Method of Administration).
Pharmacokinetics in patients with impaired hepatic function. Darifenacin pharmacokinetics were investigated in subjects with mild (Child Pugh A) or moderate (Child Pugh B) impairment of hepatic function given darifenacin 15 mg once daily to steady state. Mild hepatic impairment had no effect on the pharmacokinetics of darifenacin. However, protein binding of darifenacin was affected by moderate hepatic impairment. The fraction of unbound darifenacin increased from 1.7% in subject with normal hepatic function to 4.5% in subject with moderate hepatic impairment. After adjusting for plasma protein binding, unbound darifenacin exposure was estimated to be 4.7-fold higher in subjects with moderate hepatic impairment than in subjects with normal hepatic function.
Use in renal impairment. Pharmacokinetics in patients with impaired renal function. A small study of subjects (n=24) with varying degrees of renal impairment (creatinine clearance between 10 and 136 mL/min) given darifenacin 15 mg once daily to steady state demonstrated no relationship between renal function and darifenacin clearance.
Use in the elderly. Pharmacokinetics in elderly patients. There are no special dosage requirements for the elderly. A Phase III population pharmacokinetic analysis of patient data (patients aged 60-89 years) indicated a trend for clearance to decrease with age by 19% per decade. The safety and efficacy profiles were not affected by age.
Paediatric use. No studies have been performed in children. Therefore, until more information is available, Enablex is not recommended for use in children.
Pharmacokinetics in paediatric patients. The pharmacokinetics of darifenacin have not been established in the paediatric population.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Effects of other medicinal products on darifenacin. Darifenacin metabolism is primarily mediated by the cytochrome P450 enzymes CYP2D6 and CYP3A4. Therefore, drugs which inhibit these enzymes may alter darifenacin pharmacokinetics when they are co-administered (see Section 5.2 Pharmacokinetic Properties).
CYP2D6 inhibitors. No special dosing requirements are necessary in the presence of CYP2D6 inhibitors, although the risk of adverse events may be increased following concomitant treatment of darifenacin with CYP2D6 inhibitors such as paroxetine, cimetidine and fluoxetine and caution should be exercised.
CYP3A4 inhibitors. No special dosing requirements are necessary in the presence of moderate CYP3A4 inhibitors (e.g. fluconazole, erythromycin). The daily dose of darifenacin should not exceed 7.5 mg when co-administered with potent CYP3A4 inhibitors (e.g. ketoconazole, itraconazole, miconazole, nefazodone, ritonavir).
Ketoconazole. In a drug interaction study, when a 7.5 mg once daily dose of Enablex was given to steady state and co-administered with the potent CYP3A4 inhibitor ketoconazole 400 mg, mean darifenacin Cmax increased to 11.2 nanogram/mL in EM (n = 10) and 55.4 nanogram/mL in one PM subject (n = 1). Mean AUC increased to 143 and 939 nanogram.hour/mL in EM and in one PM subject, respectively. When a 15 mg daily dose of Enablex was given with ketoconazole, mean darifenacin Cmax increased to 67.6 nanogram/mL and 58.9 nanogram/mL in EMs (n = 3) and in one PM subject (n = 1), respectively. Mean AUC increased to 1,110 and 931 nanogram.hour/mL in EMs and in one PM subject, respectively.
CYP2D6 substrates. Caution should be exercised when darifenacin is used concomitantly with medications that are predominantly metabolised by CYP2D6 and which have a narrow therapeutic window such as flecainide, thioridazine, or tricyclic antidepressants such as imipramine.
CYP3A4 substrates. Darifenacin had no clinically relevant effect on the exposure of the CYP3A4 substrate midazolam and had no effect on the pharmacokinetics of the oral contraceptives, levonorgestrel or ethinylestradiol.
P-glycoprotein inhibitors. Darifenacin is a substrate of the drug efflux transporter P-glycoproteins. The in vivo effect of P-glycoproteins inhibition on darifenacin exposure has not been studied.
Warfarin. Standard therapeutic prothrombin time monitoring for warfarin should be continued. The effect of warfarin on prothrombin time was not altered when co-administered with darifenacin.
Digoxin. Therapeutic drug monitoring for digoxin should be performed when initiating and ending darifenacin treatment as well as changing the darifenacin dose. Darifenacin 30 mg once daily (two times greater than the recommended daily dose), co-administered with digoxin at steady state resulted in a small increase in digoxin exposure.
Antimuscarinic agents. The concomitant use of darifenacin with other antimuscarinic agents may increase the frequency and/or severity of antimuscarinic pharmacological effects such as dry mouth, constipation and blurred vision. The potentiation of anticholinergic effects with anti-parkinson agents and tricyclic antidepressants may also occur if antimuscarinic agents are used concurrently with such medicinal products. However, no studies involving the interaction with anti-parkinson agents and tricyclic antidepressants have been performed.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available in humans. No significant effects on fertility were observed in male or female rats treated with darifenacin (2 weeks [female] or 9 weeks [male] prior to and throughout mating) at oral doses up to 50 mg/kg/day, corresponding to approximately 55 times the human AUC at the MRHD.
Use in pregnancy. (Category B3)
Darifenacin and/or its metabolites cross the placenta in rats and rabbits. There was no evidence of teratogenicity in rats or rabbits following oral administration of darifenacin during the period of organogenesis and foetogenesis at doses up to 50 and 30 mg/kg/day, respectively (59 and 28 times the MRHD, based on unbound AUC).
At these maternotoxic doses, darifenacin caused delayed ossification of sacral and caudal vertebrae in rats and increased postimplantation loss/ decreased foetal viability in rabbits.
When oral treatment of rats at this dose was extended throughout organogenesis and foetogenesis, gestation and lactation, additional findings included increased gestation length, dystocia, reductions in birth weight, postnatal survival and growth of offspring and altered indices of offspring behaviour.
At the NOEL for pup developmental effects (10 mg/kg/day), the relative systemic exposure to darifenacin was approximately 9 times that anticipated at the MRHD (based on AUC).
Animal studies do not indicate direct or indirect harmful effects with respect to fertility, pregnancy and embryofoetal development. The safety of darifenacin in human pregnancy has not been established and, because animal reproduction studies are not always predictive of human response, this drug should be used during pregnancy only if the benefit to the mother outweighs the potential risk to the foetus.
Use in lactation. Darifenacin is excreted into the milk of rats. It is not known whether darifenacin is excreted into human milk and, therefore, caution should be exercised before Enablex is administered to a nursing woman.
4.7 Effects on Ability to Drive and Use Machines
No studies of the effects of Enablex on the ability to drive and use machines have been performed. However, antimuscarinics such as Enablex, may produce dizziness or blurred vision. Patients should not drive vehicles, use machines or perform other tasks which require alertness if they experience these adverse events.
4.8 Adverse Effects (Undesirable Effects)
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
During the clinical development of Enablex, a total of 7,271 patients and volunteers have been exposed to phase I to III clinical trials for up to one year duration of therapy for overactive bladder and other indications.
The phase II and III overactive bladder clinical trial program for Enablex included 2,678 patients who were treated with Enablex modified-release tablets 3.75 mg to 45 mg once daily for up to 12 months.
Of this total, 1,059 patients participated in three twelve-week, phase III, fixed dose efficacy and safety studies. Of this total, 337 and 334 patients were treated with Enablex modified-release tablets, 7.5 mg daily and 15 mg daily, respectively.
Adverse events in clinical trials. Table 1 lists the adverse events reported (regardless of causality) in 1% or more patients treated with 7.5 or 15 mg Enablex modified-release tablets in fixed dose, placebo controlled phase III studies. Adverse events were reported by 54.0% and 65.6% of patients receiving 7.5 and 15 mg once daily Enablex modified-release tablets, respectively, and by 48.7% of patients receiving placebo.
The majority of adverse events in Enablex treated subjects were mild or moderate and mostly occurred during the first two weeks of treatment. The incidence of serious adverse events was similar for 7.5 mg, 15 mg and placebo. The profile of adverse events remained consistent across all populations and doses studied.
The most frequently reported adverse events were dry mouth and constipation. However, the patient discontinuation rates due to these events were low.
Consistent with M3-muscarinic receptor selectivity, the incidence of central nervous system adverse events at all doses was similar to placebo. The incidence of cardiovascular adverse events, such as tachycardia, was less than 1% at all doses and did not increase with dose.
In clinical trials of volunteers and patients (n = 964 treated, n = 261 placebo), clinically significant changes in QT interval were not observed with Enablex up to and including 60 mg once daily, the highest dose studied.
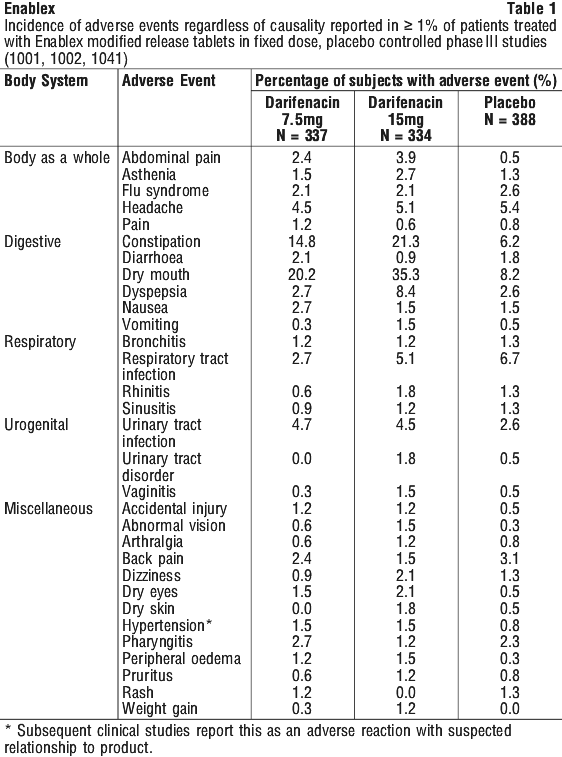
Long-term safety was evaluated in 410 patients for 6 months and 106 patients for 1 year. The long-term safety profile of darifenacin was similar to that observed in the short-term pivotal studies.
Adverse reactions with suspected relationship to product. The following adverse drug reactions, listed in Table 2, have been reported in clinical studies following treatment with Enablex and are listed according to system organ classes in MedDRA. Within each system organ class, the adverse drug reactions are ranked by frequency, with the most frequent reactions first. Within each frequency grouping, adverse drug reactions are presented in order of decreasing seriousness. In addition, the corresponding frequency category using the following convention (CIOMS III) is also provided for each adverse drug reaction: very common (≥ 1/10); common (≥ 1/100, < 1/10); uncommon (≥ 1/1,000, < 1/100); rare (≥ 1/10,000, < 1/1,000) very rare (< 1/10,000), including isolated reports.
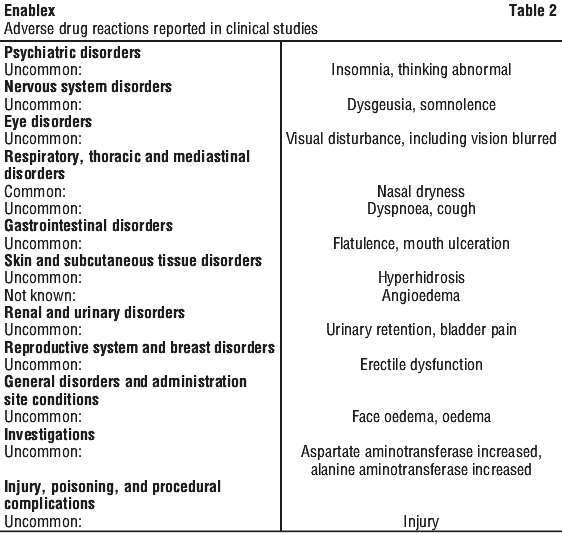
Generalised hypersensitivity reactions including angioedema, with or without airway obstruction (also see Section 4.4 Special Warnings and Precautions for Use) have been reported;
urinary retention;
palpitations.
Because these spontaneously reported events are from the worldwide post-marketing experience, the frequency of events and the role of darifenacin in their causation cannot be reliably determined.
4.9 Overdose
Overdosage with antimuscarinic agents, including Enablex, can result in severe antimuscarinic effects. Treatment should be symptomatic and supportive. In the event of overdosage, ECG monitoring is recommended. Enablex has been administered in clinical trials at doses up to 75 mg (five times the maximum therapeutic dose) and signs of overdose were limited to abnormal vision.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Darifenacin is a selective muscarinic M3-receptor antagonist that exhibits 9 to 59-fold selectivity for the human M3-receptor over human muscarinergic M1, M2, M4 and M5-receptors. The M3-receptor is the major subtype that controls urinary bladder muscle contraction.
Cystometric studies performed with darifenacin in patients with involuntary bladder contractions showed increased bladder capacity, increased volume threshold for unstable contractions and diminished frequency of unstable detrusor contractions after darifenacin treatment. These findings are consistent with the clinical observations of reduced frequency of incontinence, reduced frequency of micturition, reduced frequency of urgency and increased functional bladder capacity.
Consistent with its selectivity profile, the incidence of central nervous system adverse events at all doses was similar to placebo. The incidence of cardiovascular adverse events such as tachycardia was less than 1% for all doses and did not increase with dose. As expected from this class of drugs, prolonged colonic transit and reduced salivary flow were observed in a dose dependent manner.
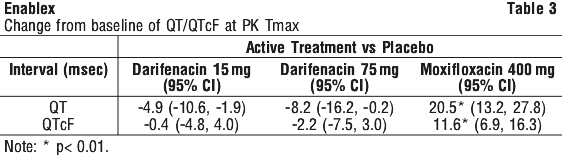
Electrophysiology. The effect of six day treatment with 15 mg and 75 mg Enablex on QT/QTc interval was evaluated in a multiple dose, double blind, randomised, placebo and active controlled (moxifloxacin 400 mg) parallel arm design study in 179 healthy adults (44% male, 56% female) aged 18 to 65. Subjects included 18% poor metabolisers and 82% extensive metabolisers. The QT interval was measured over a 24 hour period both pre-dosing and at steady state.
The 75 mg dose of Enablex was chosen because this achieves exposure similar to that observed in CYP2D6 poor metabolisers administered the highest recommended dose (15 mg) of darifenacin in the presence of a potent CYP3A4 inhibitor. At the doses studied, Enablex did not result in QT/QTc interval prolongation at any time during steady state, while moxifloxacin treatment resulted in a mean increase from baseline QTcF of about 7.0 millisecond when compared to placebo. See Table 3.
Clinical trials. Enablex modified-release tablets were evaluated for the treatment of patients with overactive bladder whose signs and symptoms included urgency, urge urinary incontinence, increased frequency, nocturia and reduced functional bladder capacity in 4 randomised, placebo controlled, multicentre, double blind, 12 week studies. The majority of patients were white (94%) and female (84%), with a mean age of 58 years, range 19 to 93 years. 33% of patients were ≥ 65 years of age. These characteristics were well balanced across treatment groups. 60% of patients had never received prior pharmacotherapy for overactive bladder and the intentional randomisation of subjects known to be responsive to, or tolerant of, anticholinergic therapy was avoided.
The three fixed dose studies included 1,059 patients. Of this total, 337 and 334 patients were treated with Enablex modified-release tablets 7.5 mg daily and 15 mg daily, respectively. In addition, a dose titration study included 395 patients, of whom 268 initially received Enablex modified-release tablets 7.5 mg daily with the option to increase to 15 mg daily after 2 weeks.
Tables 4 and 5 show the efficacy data from the placebo controlled studies of patients treated with 7.5 and 15 mg once daily Enablex for 12 weeks. A significant decrease in symptoms of urge urinary incontinence was observed.
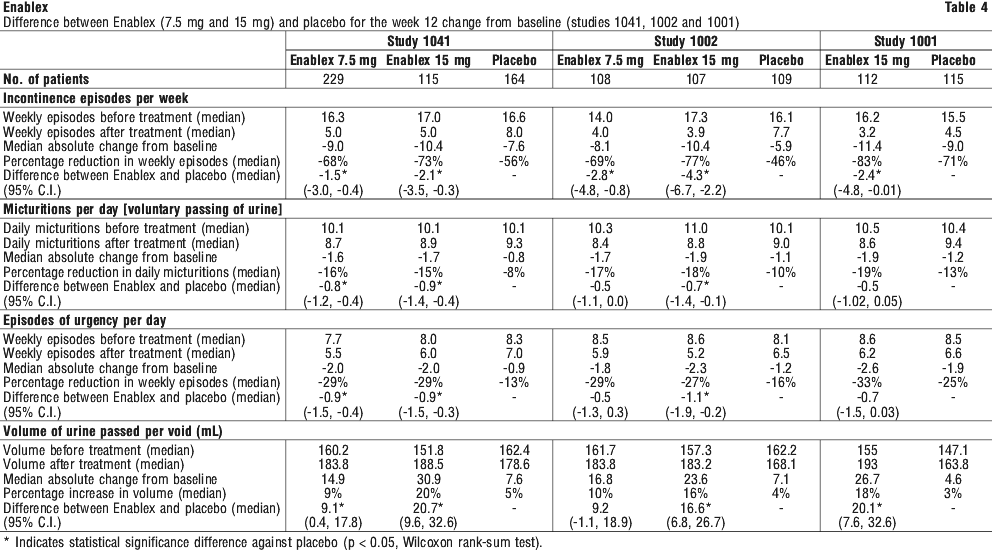
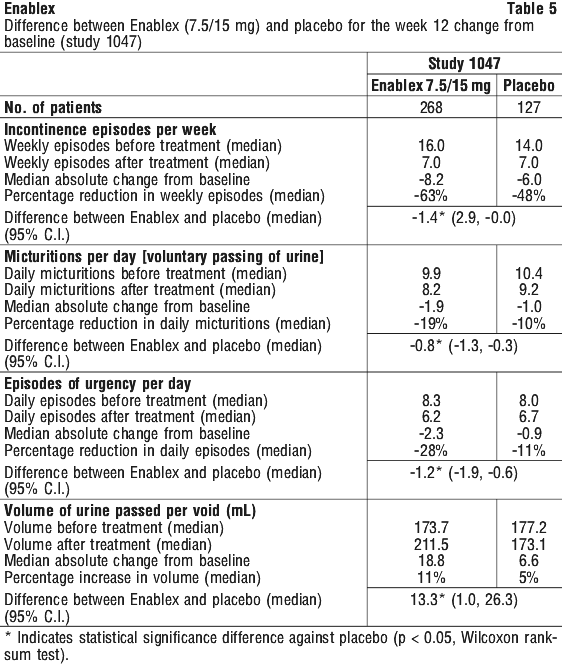

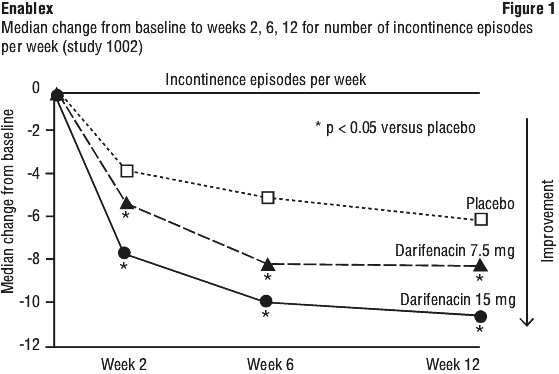
In a clinical study of 12 months duration at doses ranging from 7.5 to 30 mg, sustained improvements from baseline were observed in the number of incontinence episodes per week and in other key secondary efficacy endpoints including number of micturitions per day, episodes of urgency and average volume of urine passed per void.
On quality of life measures, darifenacin 7.5 mg and 15 mg were associated with statistically and clinically meaningful improvements over placebo in the incontinence impact, role limitations, social limitation and severity measures domains, as defined by the King's Health Questionnaire (KHQ).
Darifenacin 15 mg was also associated with improvements on the emotions domain of the KHQ.
5.2 Pharmacokinetic Properties
Absorption. Darifenacin is rapidly and completely (> 98%) absorbed after oral administration, although oral bioavailability is limited by first pass metabolism (see Metabolism). The estimated mean oral bioavailability of darifenacin in extensive metabolisers at steady state is 15% and 19% for 7.5 and 15 mg modified-release tablets, respectively. Maximum plasma levels are reached approximately seven hours after administration of the modified-release tablets and steady-state plasma levels are achieved by the sixth day of dosing. At steady state, peak to trough fluctuations in darifenacin concentrations are small, thereby maintaining therapeutic plasma levels over the 24 hour dosing interval (see Figure 2).
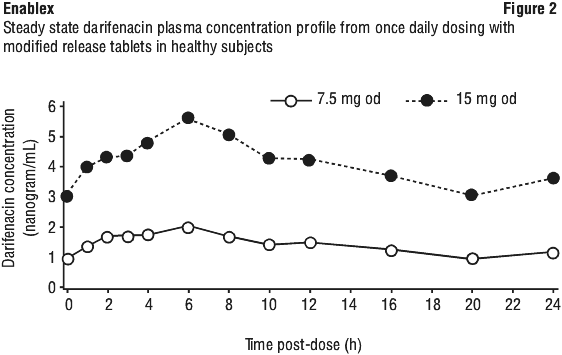
Based on free drug levels in animal cerebrospinal fluid and plasma, darifenacin shows negligible penetration of the blood brain barrier.
Metabolism. Darifenacin is extensively metabolised by the liver following oral administration.
Metabolism is mediated by cytochrome P450 enzymes CYP2D6 and CYP3A4. The three main metabolic routes are as follows:
monohydroxylation in the dihydrobenzofuran ring;
i) dihydrobenzofuran ring opening;
ii) N-dealkylation of the pyrrolidine nitrogen.
The initial products of the hydroxylation and N-dealkylation pathways are major circulating metabolites but none contributes significantly to the overall clinical effect of darifenacin.
Variability in metabolism. A subset of individuals (approximately 7% of the Caucasian population) are poor metabolisers of substrate for CYP2D6. Therefore, the metabolism of darifenacin in these poor metabolisers will be principally mediated via CYP3A4. Individuals with full CYP2D6 activity are referred to as extensive metabolisers.
Population pharmacokinetic analyses of phase III data indicated that, on average, steady-state exposure is 66% higher in poor metabolisers than in extensive metabolisers. However, there is considerable overlap between the ranges of exposures seen in these two populations and clinical experience confirms that there are no special dosing requirements for poor metabolisers.
Excretion. Following administration of an oral dose of 14C-darifenacin solution to healthy volunteers, approximately 60% of the radioactivity was recovered in the urine and 40% in the faeces. Only a small percentage of the excreted dose was unchanged darifenacin (3%). Estimated darifenacin clearance is 40 L/h for extensive metabolisers and 32 L/h for poor metabolisers. The estimated half-life for darifenacin following chronic dosing is 12.8 to 18.7 hrs.
The effect of food on pharmacokinetics. Food had no effect on darifenacin pharmacokinetics during multiple dose administration of modified-release tablets.
Gender influence on pharmacokinetics. No special dosage requirements are necessary based on gender. A population pharmacokinetic analysis of patient data indicated that darifenacin exposure was 23% lower in males than females. In clinical studies, the safety and efficacy profiles were not affected by gender.
5.3 Preclinical Safety Data
Genotoxicity. Darifenacin was neither mutagenic (bacterial and mammalian cell mutation assays) nor clastogenic (human lymphocyte assay) when tested in vitro in the presence and absence of metabolic activation. No chromosomal aberrations were observed in an in vivo mouse bone cytogenetics assay following systemic exposure to darifenacin at levels > 32 times human exposures at the MRHD.
Carcinogenicity. Two year carcinogenicity studies with dietary administration of darifenacin were conducted in mice and rats. No evidence of drug related carcinogenicity was revealed in mice (up to 100 mg/kg/day) or rats (up to 15 mg/kg/day). These doses correspond to unbound darifenacin exposure levels approximately 32 times (mice) and 10 times (rats) human exposure at the maximum recommended human dose (MRHD): 15 mg.
6 Pharmaceutical Particulars
6.1 List of Excipients
Enablex modified-release tablets 7.5 mg. Calcium hydrogen phosphate, hypromellose, magnesium stearate, Opadry complete film coating system 00F18296 White.
Enablex modified-release tablets 15 mg. Calcium hydrogen phosphate, hypromellose, magnesium stearate, Opadry complete film coating system 00F12951 Yellow, Opadry complete film coating system 00F15613 Red and Opadry complete film coating system 00F18296 White.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Protect from light.
6.5 Nature and Contents of Container
Both strengths of Enablex tablets are available in PA/Al/PVC/Al, PVC/PCTFE (Aclar)/Al or PVC/PVDC/Al blister packs containing 7, 14, 28, 56 or 98 tablets.
Not all pack sizes or container types may be marketed.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Darifenacin hydrobromide is a white to almost white monomorphic crystalline solid. The solubility of darifenacin hydrobromide in water is 6.03 mg/mL at 37°C, with a resulting pH of 5.5.
Chemical structure. Schematic structure.
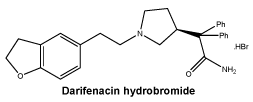
Molecular formula: C28H30N2O2.HBr.
Molecular weight: 507.5.
CAS number. 133099-07-7.
7 Medicine Schedule (Poisons Standard)
S4 (Prescription only medicine).
Date of First Approval
09 August 2010
Date of Revision
16 October 2020
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.