Epiduo Gel
Brand Information
| Brand name | Epiduo Gel |
| Active ingredient | Adapalene + Benzoyl peroxide |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Epiduo Gel.
Summary CMI
EPIDUO® Gel
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using EPIDUO?
EPIDUO contains the active ingredients adapalene and benzoyl peroxide. EPIDUO is used to treat acne. It is used on the face, chest or back
For more information, see Section 1. Why am I using EPIDUO? in the full CMI.
2. What should I know before I use EPIDUO?
Do not use if you have ever had an allergic reaction to EPIDUO or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use EPIDUO? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with EPIDUO and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use EPIDUO?
Apply a thin film of gel evenly over the entire acne affected area(s) once a day, on a clean and dry skin. If applying to the face, avoid the eyes and lips
More instructions can be found in Section 4. How do I use EPIDUO? in the full CMI.
5. What should I know while using EPIDUO?
| Things you should do |
|
| Things you should not do |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using EPIDUO? in the full CMI.
6. Are there any side effects?
Common side effects include dry skin, skin irritation, skin burning sensation, skin exfoliation (scaling) superficial reddening of skin.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
EPIDUO® Gel
Active ingredient(s): 0.1% adapalene / 2.5% benzoyl peroxide
Consumer Medicine Information (CMI)
This leaflet provides important information about using EPIDUO. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using EPIDUO.
Where to find information in this leaflet:
1. Why am I using EPIDUO?
2. What should I know before I use EPIDUO?
3. What if I am taking other medicines?
4. How do I use EPIDUO?
5. What should I know while using EPIDUO?
6. Are there any side effects?
7. Product details
1. Why am I using EPIDUO?
EPIDUO contains the active ingredients adapalene and benzoyl peroxide.
- Adapalene is a topical retinoid. Its main actions are to normalise skin cell development, prevent pores from being blocked and promote skin renewal.
- Benzoyl peroxide kills bacteria (antibacterial) and also works by softening and peeling the outer layer of the skin.
Adapalene and benzoyl peroxide work together to rapidly reduce inflammation.
EPIDUO is used to treat acne. It is used on the face, chest or back.
Your doctor, however, may prescribe EPIDUO for another purpose.
Ask your doctor if you have any questions about why EPIDUO has been prescribed for you.
This medicine is available only with a doctor's prescription.
2. What should I know before I use EPIDUO?
Warnings
Do not use EPIDUO if:
- you are allergic to adapalene, benzoyl peroxide or any of the ingredients listed at the end of this leaflet. Always check the ingredients to make sure you can use this medicine.
- you have cuts, scrapes or eczema on your skin
- your skin is sunburnt
- your child is below 12 years of age.
Check with your doctor if you:
- you are allergic to any other medicines or other substances such as foods, dyes or preservatives.
- have or have had any other health problems.
- plan to become pregnant or to breastfeed.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Do not use EPIDUO if you are pregnant or are planning a pregnancy. If you fall pregnant while using EPIDUO, the treatment must be discontinued and you should inform your doctor as soon as possible for further follow-up. Check with your doctor if you are pregnant or intend to become pregnant.
Do not apply EPIDUO onto your chest if you are breastfeeding. Talk to your doctor if you are breastfeeding or intend to breastfeed.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with EPIDUO and affect how it works.
Your doctor or pharmacist may have more information on medicines to be careful with or avoid while taking EPIDUO. Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect EPIDUO.
4. How do I use EPIDUO?
- Follow all directions given to you by your doctor and pharmacist carefully. They may differ from the information contained in this leaflet.
- If you do not understand the instructions on the tube/bottle / box, ask your doctor or pharmacist for help.
When to use EPIDUO
- EPIDUO should be applied once daily.
- Depending on your condition and how you react to EPIDUO, your doctor may ask you to use it differently.
- If you use more EPIDUO than you should on your skin, you will not get rid of your acne quicker, but your skin may become irritated and red.
How to apply EPIDUO
- Cleanse the skin before applying EPIDUO – a non-soap cleanser is useful. Allow the skin to dry completely before applying EPIDUO.
- Apply a thin film of gel evenly over the entire acne affected area(s) once a day, on a clean and dry skin. If applying to the face, avoid the eyes and lips.
- Wash your hands afterwards.
- If the product enters the eye, wash immediately with luke-warm water.
If you forget to use EPIDUO
Do not apply a double dose to make up for forgotten individual doses.
If you have trouble remembering to use your medicine, ask your pharmacist for some hints.
If you swallow EPIDUO
If you think that you have swallowed EPIDUO, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26 in Australia; 0800 764 766 in NZ), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using EPIDUO?
Things you should do
- If you feel that the effect of EPIDUO is too strong or too weak, talk to your doctor or pharmacist.
- If you experience irritation when applying EPIDUO, a non-comedogenic moisturiser applied daily may help reduce this sensation. If symptoms persist, speak to your doctor or pharmacist. You may be asked to use the gel less often or to stop for a short time.
Call your doctor straight away if you:
- become or plan to become pregnant while using EPIDUO.
- are about to start using any new medicine.
Remind any doctor, dentist or pharmacist you visit that you are using EPIDUO.
Things you should not do
- Do not give EPIDUO to anyone else, even if they have the same condition as you.
- Do not use EPIDUO to treat any other medical complaints unless your doctor tells you to.
- Do not use in children below 12 years of age.
Things to be careful of
- Please be aware that EPIDUO contains benzoyl peroxide which may cause bleaching and discolouration to the hair and dyed fabrics.
- When using EPIDUO on the face, don't get it into your eyes, mouth or nostrils, and avoid other very sensitive areas of the body.
- Other acne products (containing benzoyl peroxide and/or retinoids) should not be used with EPIDUO.
- Avoid waxing as a means of hair removal on the skin treated with EPIDUO.
- Avoid using other potentially irritating topical products (medicated or abrasive soaps and cleansers, soaps and cosmetics that have strong skin-drying effect and products with high concentrations of alcohol, astringents, spices, or limes) while being treated with EPIDUO.
- EPIDUO contains propylene glycol (E1520) that may cause skin irritation.
- EPIDUO contains polysorbate 80 (E433), which can cause allergic reactions
- Cosmetics with irritant or drying effect should not be used with EPIDUO.
- Avoid excessive exposure to sunlight and UV lamps.
- Protect your skin when you are in the sun, especially between 10 am and 3 pm. If outdoors, wear protective clothing and use a non-comedogenic, broad spectrum, SPF 50+ sunscreen.
Looking after your medicine
- Keep EPIDUO in a cool, dry place where the temperature stays below 25°C.
- Do not refrigerate.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
If skin irritation appears after the application of EPIDUO, the intensity is generally mild or moderate with some local symptoms (redness, dryness, scaling, stinging and burning) peaking during the first two weeks and then subsiding spontaneously.
EPIDUO may occasionally cause some unwanted effects at the site of application
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems or to Medsafe at pophealth.my.site.com/carmreportnz/s/. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What EPIDUO contains
| Active ingredient (main ingredient) | Adapalene 0.1% w/w Benzoyl peroxide 2.5% w/w |
| Other ingredients (inactive ingredients) | copolymer of acrylamide and sodium acryloyldimethyltaurate disodium edetate docusate sodium glycerol Isohexadecane poloxamer 124 polysorbate 80 (E433) propylene glycol (E1520) sorbitan oleate purified water |
| Potential allergens | polysorbate 80 (E433) propylene glycol (E1520) |
Do not take this medicine if you are allergic to any of these ingredients.
What EPIDUO looks like
EPIDUO Gel is a white to very pale yellow opaque gel. It is supplied in two type of containers
- Plastic tubes with a screw cap containing 30g of gel (AUST R 143869)
- Plastic bottles with a pump dispenser and plastic cap containing 30g of gel (AUST R 222977)
Who distributes EPIDUO
Galderma Australia Pty Ltd
Level 18, 1 Denison Street
North Sydney, NSW 2060
Telephone 1800 800 765
Distributed in New Zealand by:
Healthcare Logistics
58 Richard Pearse Drive
Airport Oaks
Auckland
Telephone (09) 9185100
Made in France
® Registered Trademark
This leaflet was prepared in January 2026.
Brand Information
| Brand name | Epiduo Gel |
| Active ingredient | Adapalene + Benzoyl peroxide |
| Schedule | S4 |
MIMS Revision Date: 01 December 2025
1 Name of Medicine
Adapalene, benzoyl peroxide.
2 Qualitative and Quantitative Composition
Each g of Epiduo contains adapalene 1 mg (0.1%) and benzoyl peroxide 25 mg (2.5%).
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Epiduo is a white to very pale-yellow opaque gel.
4 Clinical Particulars
4.1 Therapeutic Indications
Cutaneous treatment of acne vulgaris on the face, chest and back when comedones, papules and pustules are present, and the condition has not responded to first line treatment.
4.2 Dose and Method of Administration
Epiduo should be applied to the entire acne affected areas once a day on a clean and dry skin. A thin film of gel should be applied, with the fingertips, avoiding the eyes, lips and mucous membranes (see Section 4.4 Special Warnings and Precautions for Use).
Patients should be instructed to wash their hands after applying Epiduo. Cosmetics may be applied after Epiduo has dried.
If irritation occurs, the patient should be directed to apply noncomedogenic moisturisers, to use the medication less frequently (e.g. every other day), to suspend use temporarily, or to discontinue use altogether.
The duration of treatment should be determined by the doctor on the basis of the clinical condition. Early signs of clinical improvement usually appear after 1 to 4 weeks of treatment.
4.3 Contraindications
Hypersensitivity to the active substances or to any of the excipients.
Pregnancy.
Women planning a pregnancy.
4.4 Special Warnings and Precautions for Use
Identified precautions. Epiduo is for external use only. Epiduo should not be applied to damaged skin, either broken (cuts or abrasions), sunburnt or eczematous skin.
Epiduo should not come into contact with the eyes, mouth, nostrils or mucous membranes. If product enters the eye, wash immediately with warm water.
This product contains propylene glycol (E1520) that may cause skin irritation.
This medicine contains polysorbate 80 (E433), which can cause allergic reactions.
If a reaction suggesting sensitivity to any component of the formula occurs, the use of Epiduo should be discontinued.
Excessive exposure to sunlight or UV radiation (e.g. sunlamps) should be avoided.
Epiduo should not come into contact with any coloured material including hair and dyed fabrics as this may result in bleaching and discolouration.
Depending upon the severity of local cutaneous adverse reactions, patients should be instructed to use a moisturiser, reduce the frequency of the application of Epiduo, or discontinue use.
As with other topical retinoids, use of "waxing" as a depilatory method should be avoided on skin treated with Epiduo.
Avoid concomitant use of other potentially irritating topical products (medicated or abrasive soaps and cleansers, soaps and cosmetics that have strong skin-drying effect and products with high concentrations of alcohol, astringents, spices, or limes).
Use in the elderly. No data available.
Paediatric use. The safety and effectiveness of Epiduo have not been studied in children below 12 years of age.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
No interaction studies have been conducted with Epiduo.
From previous experience with adapalene and benzoyl peroxide, there are no known interactions with other medicinal products which might be used cutaneously and concurrently with Epiduo. However, other retinoids or benzoyl peroxide or drugs with a similar mode of action should not be used concurrently. Caution should be exercised if cosmetics with desquamative, irritant or drying effects are used, as they may produce additive irritant effects with Epiduo.
Absorption of adapalene through human skin is low (see Section 5.2 Pharmacokinetic Properties), and therefore interaction with systemic medicinal products is unlikely.
The percutaneous penetration of benzoyl peroxide in the skin is low and the drug substance is completely metabolised into benzoic acid which is rapidly eliminated. Therefore, the potential interaction of benzoic acid with systemic medicinal products is unlikely to occur.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Fertility testing of Epiduo gel has not been performed in any species.
Oral adapalene had no effect on the fertility of rats at doses up to 20 mg/kg/day (> 500 times the maximum recommended human dose, based on Cmax).
Benzoyl peroxide administered orally to male rats at 1 g/kg/day resulted in testicular atrophy; there was no effect on fertility in male rats at oral doses of 500 mg/kg/day or in female rats at 1 g/kg/day.
Use in pregnancy. (Category D)
Orally administered retinoids have been associated with congenital abnormalities. When used in accordance with the prescribing information, topically administered retinoids are generally assumed to result in low systemic exposure due to minimal dermal absorption. However, there could be individual factors (e.g. damaged skin barrier, excessive use) that contribute to an increased systemic exposure.
Animal embryofoetal developmental studies have not been conducted with Epiduo gel.
No adequate or well controlled studies in pregnant women have been conducted with topical benzoyl peroxide. Benzoyl peroxide at oral doses of 1 g/kg/day resulted in lower birthweights of rat pups and lower pup bodyweight gain after birth; no reproductive toxicity was observed in rats at oral doses of 500 mg/kg/day benzoyl peroxide. It is unknown whether benzoyl peroxide can cause foetal harm when used by pregnant women.
Adapalene administered orally at high doses (≥ 25 mg/kg/day) to pregnant rats and rabbits was found to induce foetal abnormalities. In addition the incidences of various skeletal variations were increased at lower oral doses in rats. Topical administration at doses up to 6 mg/kg, resulting in an exposure level ≈ 230 times (based on Cmax) that anticipated clinically, was not associated with teratogenicity. Nevertheless, increased incidences of various naturally occurring skeletal variations were still observed following topical administration to rats at 2 mg/kg (Cmax exposure ≈ 60 times that anticipated clinically) and to rabbits at 6 mg/kg (Cmax exposure ≈ 76 times that anticipated clinically); topical no effect levels were 0.6 and 2 mg/kg respectively (Cmax exposure ≈ 19 and 48 times that anticipated clinically).
There have been isolated reports of birth defects in babies born to women using topical drugs with a similar mechanism of action to adapalene during pregnancy. However, there are no adequate or well controlled studies in pregnant women. Because of the potential risk of adverse effects on foetal development, adapalene should not be used by women who are pregnant or who plan to become pregnant during treatment. In the case of unexpected pregnancy, treatment should be discontinued.
Use in lactation. It is not known whether adapalene is distributed into human milk. After IV or oral administration, adapalene was excreted in rat milk. Oral treatment of rat dams with up to 15 mg/kg/day adapalene (300 times that anticipated clinically, based on Cmax) after organogenesis and during lactation was not associated with adverse effects on functional development of rat pups. It is unknown whether benzoyl peroxide or its metabolite benzoic acid is distributed into breast milk. Benzoyl peroxide has not been reported to cause problems in breastfed babies. Epiduo should be used with caution in breastfeeding women, and to avoid contact exposure of the infant, application of Epiduo should only be used on areas away from the chest when used during breastfeeding.
4.7 Effects on Ability to Drive and Use Machines
Epiduo has no or negligible influence on the ability to drive and use machines.
4.8 Adverse Effects (Undesirable Effects)
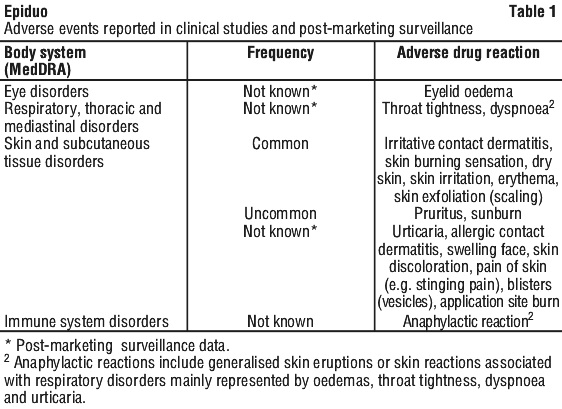
Epiduo may cause the following adverse reactions, ranked in Table 1 by frequency and within each frequency grouping by decreasing medical seriousness.
If skin irritation appears after application of Epiduo, the intensity is generally mild or moderate, with local tolerability signs and symptoms (erythema, dryness, scaling, burning1 and pain of skin (including stinging)) peaking during the first two weeks and then subsiding spontaneously.
1 Cases of application site burn included superficial burns, second degree burns and severe burn reactions.
Tabulated summary of adverse reactions. The adverse reactions are classified by System Organ Class and frequency, using the following convention: very common (≥ 1/10), common (≥ 1/100 to < 1/10), uncommon (≥ 1/1,000 to < 1/100), rare (≥ 1/10,000 to < 1/1,000), very rare (< 1/10,000), not known (cannot be estimated from the available data) and were reported with Epiduo in clinical studies.
4.9 Overdose
Epiduo is for once daily cutaneous use only.
In case of accidental ingestion, appropriate symptomatic measures should be taken.
If the medications are applied excessively, no more rapid or better results will be obtained and marked redness, peeling or discomfort may occur.
The acute oral toxicity of adapalene topical gel, 0.1% in mice and rats is greater than 10 mL/kg (10 mg/kg). Inadvertent oral ingestion of adapalene may lead to the same adverse effects as those associated with excessive oral intake of vitamin A, including teratogenesis in women of childbearing years. Therefore, pregnancy testing should be carried out in women of childbearing potential who have ingested the product.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: D10A anti-acne preparations for topical use.
ATC code: D10AD53.
Mechanism of action. Epiduo combines two active substances, which act through different, but complementary mechanisms of action.
Adapalene. Adapalene is a chemically stable, naphthoic acid derivative with retinoid-like activity. Biochemical and pharmacological profile studies have demonstrated that adapalene alters the pathology of acne vulgaris: it is a potent modulator of cellular differentiation and keratinisation and has anti-inflammatory properties. Mechanistically, adapalene binds to specific retinoic acid nuclear receptors. Current evidence indicates that topical adapalene normalises the differentiation of follicular epithelial cells resulting in decreased microcomedone formation. Adapalene inhibits the chemotactic responses of human polymorphonuclear leucocytes in vitro; it also inhibits the metabolism of arachidonic acid to inflammatory mediators. In vitro studies have shown inhibition of the AP-1 factors and the inhibition of the expression of toll-like receptors 2. This profile indicates that the cell mediated inflammatory component of acne is reduced by adapalene.
Benzoyl peroxide (BPO). Benzoyl peroxide has been shown to have antimicrobial activity; particularly against P. acnes, which is abnormally present in the acne affected pilosebaceous unit. Additionally benzoyl peroxide has demonstrated exfoliative and keratolytic activities acting against comedones at all stages of their development. Benzoyl peroxide is also sebostatic, counteracting the excessive sebum production associated with acne.
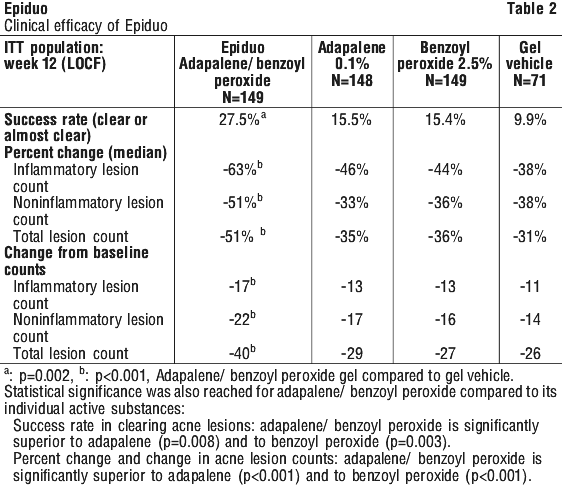
Clinical trials. Clinical efficacy was established in a multicentre, double blind, active and vehicle controlled 12 week trial with 517 acne patients, who were randomised to the 4 parallel arms of the study. The patients with acne vulgaris enrolled in this study were approximately 60% male and 40% female subjects, mean age of study subjects: 16.5 years; range 12-56, presenting 20 to 50 inflammatory lesions with no nodules and cysts and 30 to 100 non-inflammatory lesions at baseline. Success rate results among the treatment groups began to diverge early in favour of Epiduo and continued to separate throughout the course of the study. After 12 weeks of treatment the efficacy of Epiduo was statistically significantly better than that of its individual components and its vehicle in total, inflammatory and non-inflammatory lesion counts (all p < 0.001). Significant differences in speed of onset of action for Epiduo were demonstrated as early as week 1 (Epiduo 19.7%; adapalene 13% [p = 0.001]; BPO 11.3% [p = 0.01]; vehicle 7.8% [p = 0.002]). Early onset of action was also observed in inflammatory lesion count reductions at week 1 (Epiduo 25.7%; adapalene 14.7% [p < 0.001]; BPO 20% [p = 0.001]; vehicle 13.6% [p < 0.001]). The net beneficial effect (active minus vehicle) obtained from Epiduo was greater than the sum of the net benefits obtained from the individual components, thus indicating a potentiation of the therapeutic activities of these substances when used in a fixed dose combination (Table 2).
5.2 Pharmacokinetic Properties
The pharmacokinetic (PK) profile of adapalene in Epiduo is similar to the PK profile of adapalene 0.1% gel alone.
In a 30 day clinical PK study conducted in patients with acne who were tested with either the fixed combination gel or with an adapalene 0.1% matched formula under maximised conditions (with application of 2 g gel per day), adapalene was not quantifiable in the majority of plasma samples (limit of quantification 0.1 nanogram/mL). Low levels of adapalene (Cmax between 0.1 and 0.2 nanogram/mL) were measured in two blood samples taken from the subjects treated with Epiduo and in three samples from the subjects treated with adapalene 0.1% gel. The highest adapalene AUC0-24h determined in the fixed combination group was 1.99 nanogram.h/mL.
These results are comparable to those obtained in previous clinical PK studies on various adapalene 0.1% formulations, where systemic exposure to adapalene was consistently low.
The percutaneous penetration of benzoyl peroxide is low; when applied on the skin, it is completely converted into benzoic acid which is rapidly eliminated. Benzoic acid also has a wide margin of safety and is an approved food additive.
5.3 Preclinical Safety Data
Genotoxicity. Genotoxicity testing of Epiduo gel has not been conducted.
Adapalene did not demonstrate mutagenic or clastogenic activity in in vitro tests with bacterial and mammalian cells and showed no clastogenic activity in mammalian cells in vitro or in an in vivo test in mice.
Benzoyl peroxide was not mutagenic in bacteria or clastogenic in Chinese hamster lung cells in vitro, but did induce DNA damage in vitro in the unscheduled DNA synthesis test and the E. coli SOS chromotest, probably by a reactive oxygen species (ROS) mechanism. Protective mechanisms against ROS are known to exist in vivo. Benzoyl peroxide was not genotoxic in vivo in a dominant lethal mutation study in mice, a cytogenetic assay in rats or a host mediated assay in rats.
Carcinogenicity. Studies have not been conducted to investigate the carcinogenic potential of the combination product Epiduo.
Carcinogenicity studies with adapalene have been conducted in mice at topical doses up to 6 mg/kg/day, and in rats at oral doses up to 1.5 mg/kg/day. These doses are up to 760 times (mice) and 120 times (rats) the exposure at the maximum recommended human dose of 2 grams Epiduo gel, based on plasma concentration data. In the rat oral dosing study, there was an increased incidence of phaeochromocytomas in the adrenal medullas of male rats at 1.5 mg/kg/day but not at lower doses (≤ 0.5 mg/kg/day; about 85 times the clinical exposure based on Cmax). This finding was not observed in female rats or in mice. Epiduo at the recommended clinical dose is unlikely to induce phaeochromocytomas in acne vulgaris patients.
Animal studies on compounds with a similar mode of action to adapalene have indicated that these may enhance the development of skin cancers caused by UV light. Adapalene is essentially stable to oxygen and light and is chemically nonreactive. While short-term studies have shown no phototoxic or photoallergic potential of adapalene, small numbers of reactions consistent with phototoxicity were reported in clinical studies, and the safety of using adapalene during long or repeated exposures to sunlight or UV radiation has not been established in animals or humans. Exposure to excessive sunlight or UV irradiation (including sunlamps) should be avoided during treatment with adapalene.
Benzoyl peroxide has been shown to be a tumour promoter and progression agent in a number of animal studies. Studies in mice have shown that benzoyl peroxide does not increase the growth of tumours initiated by UV light. The clinical significance of this is unknown.
6 Pharmaceutical Particulars
6.1 List of Excipients
Acrylamide/sodium acryloyldimethyltaurate copolymer, disodium edetate, docusate sodium, glycerol isohexadecane, poloxamer, polysorbate 80 (E433), propylene glycol (E1520), sorbitan oleate, purified water.
See Section 2 Qualitative and Quantitative Composition.
6.2 Incompatibilities
Not applicable.
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
Shelf life is 2 years.
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Do not refrigerate.
6.5 Nature and Contents of Container
2 g, 5 g, 30 g, 45 g and 60 g HDPE tubes, closed with a PP screw-cap.
5 g, 30 g, 45 g and 60 g PP bottles with pump, closed with a PP cap.
Not all pack sizes may be marketed.
6.6 Special Precautions for Disposal
No special requirements.
Any unused product or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure. Adapalene.
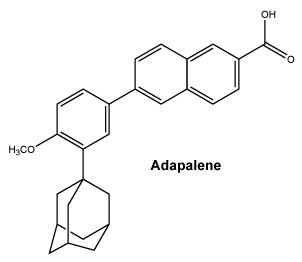
Molecular formula: C28H28O3.
Molecular weight: 412.52.
CAS number. 106685-40-9.
Chemical structure. Benzoyl peroxide.
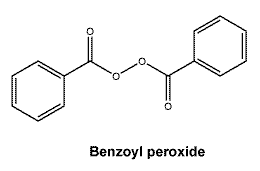
Molecular formula: C14H10O4.
Molecular weight: 242.2.
CAS number. 94-36-0.
7 Medicine Schedule (Poisons Standard)
Prescription medicine (S4).
Date of First Approval
22 January 2009
Date of Revision
08 October 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.