Estrogel
Brand Information
| Brand name | Estrogel |
| Active ingredient | Estradiol |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Estrogel.
What is in this leaflet
This leaflet answers some common questions about Estrogel.It does not contain all the available information. It does not take the place of talking to your pharmacist or doctor.
All medicines have risks and benefits. Your pharmacist or doctor has weighed the risks of you taking Estrogel against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your pharmacist or doctor.
Keep this leaflet with the medicine. You may need to read it again.
What Estrogel is used for
Estrogel is a Hormone Replacement Therapy (HRT). It contains the female hormone estrogen. Estrogel is used in postmenopausal women who have not had a natural period within the last 6 months.
Estrogel is used for:
Relief of symptoms occurring after menopause
During menopause, the amount of the estrogen produced by a woman’s body drops. This can cause symptoms such as hot face, neck and chest (“hot flushes”). Estrogel alleviates these symptoms after menopause. You will only be prescribed Estrogel if your symptoms seriously hinder your daily life.
Prevention of osteoporosis
After menopause some women may develop fragile bones (osteoporosis). You should discuss all available options with your doctor.
If you are at an increased risk of fractures due to osteoporosis and other medicines are not suitable for you, you can use Estrogel to prevent osteoporosis after menopause.
Estrogel works by replacing the estrogen in your body. This is so that you have a similar amount of estrogen as before your menopause.
Ask your doctor if you have any questions about why this medicine has been prescribed for you.
Your doctor may have prescribed it for another reason.
Before you use Estrogel
When you must not use it
Do not use Estrogel if you have an allergy to:
- any medicine containing estradiol
- any of the ingredients listed at the end of this leaflet.
Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin
Do not use this medicine if you have any of the following conditions:
- If you have or have ever had breast cancer, or if you are suspected of having it,
- If you have cancer which is sensitive to estrogen, such as cancer of the womb lining (endometrium), or if you are suspected of having it,
- If you have any unexplained vaginal bleeding,
- If you have excessive thickening of the womb lining (endometrial hyperplasia) that is not being treated,
- If you have or have ever had blood clots in a vein (thrombosis), such as in the legs (deep venous thrombosis) or lungs (pulmonary embolism),
- If you have a blood clotting disorder (such as protein C, protein S, or antithrombin deficiency),
- If you have or recently have had a disease caused by blood clots in the arteries, such as a heart attack, stroke or angina,
- If you have or have ever had a liver disease and your liver function tests have not returned to normal,
- If you have a rare blood problem called “porphyria” which is passed down in families (inherited).
Do not use this medicine after the expiry date printed on the pack or if the packaging is torn or shows signs of tampering. If it has expired or is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start using this medicine, talk to your doctor.
Before you start to use it
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
Tell your doctor if you have or have had any of the following medical conditions, as these may return or become worse during treatment with Estrogel:
- liver or kidney disease
- difficulty breathing, wheezing, chronic cough, asthma or other chronic breathing conditions
- fibrosis inside your womb,
- growth of womb lining outside your womb (endometriosis) or a history of excessive growth of the womb lining (endometrial hyperplasia),
- increased risk of developing blood clots (see “Blood clots in a vein (thrombosis)”),
- increased risk of getting an estrogen-sensitive cancer (such as having a mother, sister or grandmother who has had breast cancer),
- high blood pressure,
- a liver disorder, such as a benign liver tumour,
- diabetes,
- gallstones,
- migraine or severe headaches,
- a disease of the immune system that affects many organs of the body (Systemic Lupus Erythematosus, SLE),
- epilepsy,
- asthma,
- a disease affecting the eardrum and hearing (otosclerosis),
- a very high level of fat in your blood (triglycerides),
- fluid retention due to heart or kidney problems,
- if you receive thyroid hormone therapy.
- if you have hereditary or acquired angioedema.
Stop using Estrogel and see a doctor immediately
If you notice any of the following when taking HRT:
- yellowing of your skin or the whites of your eyes (jaundice). These may be signs of a liver disease
- a large rise in your blood pressure (symptoms may be headache, tiredness, dizziness)
- migraine-like headaches which happen for the first time
- if you become pregnant
- if you notice signs of a blood clot, such as: painful swelling and redness of the legs, or sudden chest pain
- difficulty in breathing
Note: Estrogel is not a contraceptive. If it is less than 12 months since your last menstrual period or you are under 50 years old, you may still need to use additional contraception to prevent pregnancy. Speak to your doctor for advice.
HRT and cancer
Excessive thickening of the lining of the womb (endometrial hyperplasia) and cancer of the lining of the womb (endometrial cancer)
Using estrogen-only HRT will increase the risk of excessive thickening of the lining of the womb (endometrial hyperplasia) and cancer of the womb lining (endometrial cancer).
Using a progestogen (eg. progesterone) in addition to the estrogen for at least 12 days of each 28 day cycle protects you from this extra risk. Your doctor will prescribe a progestogen separately if you still have your womb. If you have had your womb removed (a hysterectomy), discuss with your doctor whether you can safely take this product without a progestogen.
In women who still have a womb and who are not using HRT, on average 5 in 1000 will be diagnosed with endometrial cancer between the ages of 50 and 65.
For women aged 50 to 65 who still have a womb and who take estrogen-only HRT, between 10 and 60 women in 1000 will be diagnosed with endometrial cancer (i.e. between 5 and 55 extra cases), depending on the dose and for how long it is taken.
Irregular bleeding
You may have irregular bleeding or drops of blood (spotting) during the first 3-6 months of using Estrogel. However, if the irregular bleeding:
- carries on for more than the first 6 months
- starts after you have been using Estrogel for more than 6 months
- carries on after you have stopped using Estrogel see your doctor as soon as possible.
Breast cancer
Evidence shows that using combined estrogen-progestogen or estrogen-only HRT increases the risk of breast cancer. The extra risk depends on how long you use HRT. The additional risk becomes clear within 3 years of use. After stopping HRT, the extra risk will decrease with time, but the risk may persist for 10 years or more if you have used HRT for more than 5 years.
Compare
Women aged 50 to 54 who are not taking HRT, on average 13 to 17 in 1000 will be diagnosed with breast cancer over a 5-year period.
For women aged 50 who start taking estrogen only HRT for 5 years there will be 16-17 cases in 1000 users (i.e. an extra 0 – 3 cases).
For women aged 50 who start taking estrogen-progestogen HRT over 5 years, there will be 21 cases in 1000 users (i.e. an extra 4 to 8 cases).
For women aged 50 -59 who are not taking HRT, on average 27 in 1000 will be diagnosed with breast cancer over a 10 year period.
For women aged 50 who start taking estrogen only HRT for 10 years there will be 34 cases in 1000 users (i.e. an extra 7 cases).
For women aged 50 who start taking estrogen-progestogen HRT over 10 years, there will be 48 cases in 1000 users (i.e. an extra 21 cases).
Regularly check your breasts. See your doctor if you notice any changes such as:
- dimpling of the skin
- changes in the nipple
- any lumps you can see or feel.
Additionally, you are advised to join mammography screening programs when offered to you. For mammogram screening, it is important that you inform the nurse/healthcare professional who is actually taking the x-ray that you use HRT, as this medication may increase the density of your breasts which may affect the outcome of the mammogram. Where the density of the breast is increased, mammography may not detect all lumps.
Ovarian cancer
Ovarian cancer is rare, much rarer than breast cancer. The use of estrogen-only or combined estrogen-progestogen HRT has been associated with a slightly increased risk of ovarian cancer. The risk of ovarian cancer varies with age. For example, in women aged 50 to 54 who are not taking HRT, about 2 women in 2000 will be diagnosed with ovarian cancer over a 5-year period. For women who have been using HRT for 5 years, there will be about 3 cases per 2000 users (i.e. about 1 extra case).
Effect of HRT on heart and circulation
Blood clots in a vein (thrombosis)
The risk of blood clots in the veins is about 1.3 to 3- times higher in HRT users than in non-users, especially during the first year of using it.
Blood clots can be serious, and if one travels to the lungs, it can cause chest pain, breathlessness, fainting or even death.
You are more likely to get a blood clot in your veins as you get older. Inform your doctor if any of these situations applies to you:
- you are unable to walk for a long time because of major surgery, injury or illness (see also section 3, If you need to have surgery)
- you are seriously overweight (BMI >30 kg/m2)
- you have any blood clotting problem that needs long-term treatment with a medicine used to prevent blood clots
- if any of your close relatives has ever had a blood clot in the leg, lung or another organ
- you have systemic lupus erythematosus (SLE)
- you have cancer.
Compare
Looking at women in their 50s who are not using HRT, on average, over a 5-year period, 4 to 7 in 1000 would be expected to get a blood clot in a vein.
For women in their 50s who have been using estrogen-progestogen HRT for over 5 years, there will be 9 to 12 cases in 1000 users (i.e.an extra 5 cases).
For women in their 50s who have had their womb removed and have been using estrogen-only HRT for over 5 years, there will be 5 to 8 cases in 1000 users (i.e. 1 extra case).
Heart disease (heart attack)
There is no evidence that HRT will prevent a heart attack.
Women over the age of 60 years who use estrogen-progestogen HRT are slightly more likely to develop heart disease than those not using any HRT.
For women who have had their womb removed and are using estrogen-only therapy there is no increased risk of developing a heart disease.
Stroke
The risk of getting stroke is about 1.5 times higher in HRT users than in nonusers. The number of extra cases of stroke due to use of HRT will increase with age.
Compare
Looking at women in their 50s who are not using HRT, on average, 8 in 1000 would be expected to have a stroke over a 5-year period. For women in their 50s who are using HRT, there will be 11 cases in 1000 users, over 5 years (i.e. an extra 3 cases).
Other conditions
HRT will not prevent memory loss. There is some evidence of a higher risk of memory loss in women who start using HRT after the age of 65. Speak to your doctor for advice.
Do not use this medicine if you are pregnant. Estrogel is for use in postmenopausal women only. If you become pregnant, stop using Estrogel and contact your doctor.
Do not breastfeed if you are using this medicine.
If you have not told your doctor about any of the above, tell them before you start taking Estrogel.
Taking other medicines
Tell your pharmacist or doctor if you are taking any other medicines, including any that you get without a prescription from your pharmacy, supermarket or health food shop.
Estrogel and some other medicines may interfere with each other. These include:
- Skin cleansers and detergents e.g. products containing benzalkonium chloride or sodium lauryl sulphate.
- Other skin products containing alcohol e.g. astringents or sunscreens.
- Products to treat skin and scalp disorders e.g. products to cure warts, acne or dandruff.
- Other skin medications which change how skin is made, e.g. anti-cancer products.
- Medicines for epilepsy (such as lamotrigine, phenobarbital, phenytoin and carbamazepine).
- Medicines for tuberculosis (such as rifampicin, rifabutin).
- Medicines for HIV infection (such as nevirapine, efavirenz, ritonavir and nelfinavir).
- Herbal products containing St John’s wort (Hypericum perforatum).
HRT can affect the way some other medicines work:
- A medicine for epilepsy (lamotrigine), as this could increase frequency of seizures.
- Medicines for Hepatitis C virus (HCV) (such as combination regimen ombitasvir/paritaprevir/ ritonavir with or without dasabuvir as well as a regimen with glecaprevir/pibrentasvir) may cause increases in liver function blood test results (increase in ALT liver enzyme) in women using Combined Hormonal Contraceptives (CHCs) containing ethinylestradiol. Estrogel contains estradiol instead of ethinylestradiol. It is not known whether an increase in ALT liver enzyme can occur when using Estrogel with this HCV combination regimen.
These medicines may be affected by Estrogel or may affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines.
Your doctor and pharmacist will have more information on these and other medicines to be careful with or avoid while taking this medicine.
How to use Estrogel
Follow all directions given to you by your doctor or pharmacist carefully. They may differ from the information contained in this leaflet.
If you do not understand the instructions printed on the pharmacist label, ask your pharmacist or doctor for help.
How much to use
- Your doctor will prescribe the lowest effective dose for the shortest time to treat your symptoms.
- One pump is equal to one measure
- The usual daily dose is 2 measures (i.e. 2 pumps) of gel. The Pump pack will last four weeks.
- If 4 measures (i.e. 4 pumps) of gel have been prescribed, the Pump Pack will last two weeks.
Spread the gel on a large area of skin on each shoulder, outer arm and/or each mid-inner thigh.
How to use it
- If you have never used any other HRT medicines or you are switching to Estrogel from a period-free HRT product, you can start using Estrogel on any convenient day.
- If you are currently using another type of HRT where you have a period, finish your current medicine pack before you start using Estrogel.
- Do not ask anyone else to apply the gel. Only you should apply your medicine.
- Do not use strong skin cleaners or detergents when washing the area where you will apply the gel.
- Avoid close skin contact with your partner for one hour after application.
- Do not wash the skin or apply other skin care products until at least one hour after application.
- If the prescribed dose does not provide relief, tell your doctor. Do not use more than the prescribed dose.
Your doctor will aim to prescribe the lowest effective dose to treat your symptoms for as short as necessary. Speak to your doctor if you think this dose is too strong or not strong enough.
Preparing your new Pump Pack
Before using your new Pump Pack for the first time, you need to prepare it for use as follows:
- Remove the cap from the canister.
- Press the plunger down a few times until the gel comes out.
- Do not use this first dose of gel from your Pump Pack. This dose should be discarded.
- Your Pump Pack is now ready to use.
How much to use and when to use
- Apply the gel once a day, either in the morning or evening.
- Try to use the gel at about the same time each day.
- Your doctor will prescribe the lowest effective dose for the shortest time to treat your symptoms.
- One pump is equal to one measure.
Make sure to push the pump firmly down to ensure a full pump is delivered. - The usual daily dose is 2 measures (i.e. 2 pumps) of gel. The Pump pack will last four weeks.
- If 4 measures (i.e. 4 pumps) of gel have been prescribed, the Pump Pack will last two weeks.
- The correct dose of gel should be dispensed and applied to clean, dry, intact areas of skin e.g. on the arms and shoulders, and/or inner thighs. The area of application should be as large as possible see table below.

How to apply the gel
- Make sure that your hands and the skin where you are going to apply the gel are clean, dry and unbroken.
- Remove the canister cap to reveal the plunger.
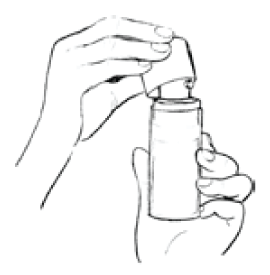
- Hold the Estrogel Pump Pack in one hand and place your other hand under the spout, ready to collect the gel.
- Push the plunger down firmly. This will dispense one measure of the gel.
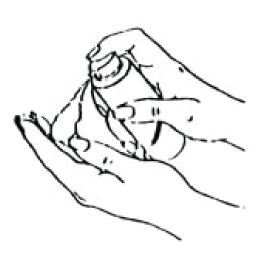
- Apply the gel as per table above, to either:
- the outer arm and shoulder of one or both arms,
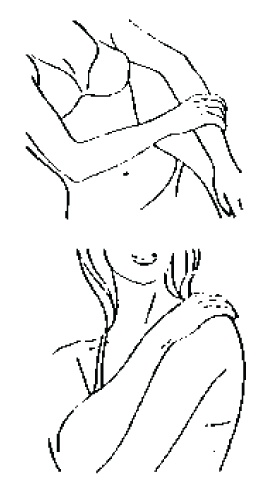
or
- the mid-inner thigh of one or both legs.
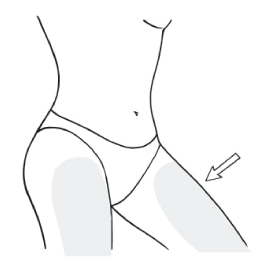
- Do not apply on or near the breasts, or near the genital area.
- Spread the gel over a large area of skin on each shoulder, outer arm or mid-inner thigh keeping the area of application as constant as possible from day to day.
- If your doctor has prescribed 2 measures of gel, spread 1 measure over each outer arm and shoulder, or each mid-inner thigh. If 3 measures of gel have been prescribed, spread the gel over each arm and shoulder or each mid-inner thigh. If 4 measures of gel have been prescribed, spread 2 measures over each outer arm and shoulder or each mid-inner thigh. (See steps 4-8).
- Replace the cap.
- Leave to dry for 5 minutes before covering with clothes.
- Wash hands with soap and water after applying the gel.
If you believe Estrogel has been transferred to another person (man or child):
Wash the area of skin onto which may have been affected immediately with soap and water.
How you know when your Pump Pack is empty
The Pump Pack is nearly empty when the plunger does not return back to its original position after you have pushed it down.
When this happens, do not use the Pump Pack any longer. Start using a new Pump Pack
When to take it
- Apply the gel once a day, either in the morning or evening.
- Try to use the gel at about the same time each day.
How long to take it
Your doctor will prescribe the lowest effective dose for the shortest time to treat your symptoms.
If you forget to take it
- If it is more than 12 hours until your next dose, apply the gel as soon as you remember and apply the next dose at the normal time.
- If it is less than 12 hours until your next dose, skip the missed dose and apply the next dose at the normal time.
- Do not use a double dose (two doses at the same time) to make up for a forgotten dose.
- If you forget a dose you may have breakthrough bleeding or spotting.
If you take too much (overdose)
Immediately telephone your doctor or the Poisons Information Centre (telephone 13 11 26) for advice, or go to Accident and Emergency at the nearest hospital, if you think that you or anyone else may have taken too much Estrogel.
The effects of overdosing are generally: breast tenderness, nausea and vaginal bleeding. These symptoms disappear when treatment is stopped or the dose is reduced.
While you are using Estrogel
Things you must do
Talk to your pharmacist or doctor if your symptoms do not improve. Your pharmacist or doctor will assess your condition and decide if you should continue to take the medicine.
The use of HRT carries risks which need to be considered when deciding whether to start using it, or whether to carry on using it.
The experience in treating women with a premature menopause (due to ovarian failure or surgery) is limited. If you have a premature menopause the risks of using HRT may be different. Please talk to your doctor.
Before you start (or restart) HRT your doctor will ask you about your own and your family’s medical history. Your doctor may decide to perform a physical examination. This may include an examination of your breasts and/or an internal examination, if necessary.
Once you have started on Estrogel you should see your doctor for regular check-ups (at least once a year). At these check-ups, discuss with your doctor the benefits and risks of continuing with Estrogel.
Go for regular breast screening, as recommended by your doctor.
If you need to have surgery
If you are going to have surgery, tell the surgeon that you are using Estrogel. You may need to stop using Estrogel about 4 to 6 weeks before the operation to reduce the risk of a blood clot. Ask your doctor when you can start using Estrogel again.
Things you must not do
Do not give your medicine to anyone else, even if they have the same condition as you.
Do not stop using your medicine or change the dosage without checking with your doctor.
Side effects
Tell your pharmacist or doctor as soon as possible if you do not feel well while you are taking Estrogel. All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Ask your pharmacist or doctor to answer any questions you may have.
Tell your pharmacist or doctor if you notice any of the following and they worry you:
- Headache.
- Nausea, abdominal pain.
- Breast swelling or pain, breast enlargement, menstrual cramps, heavy menstrual bleeding, vaginal discharge.
- Weight change (increase or decrease), water retention with peripheral oedema.
- Depression, mood swings.
- Vertigo.
- Flatulence, vomiting.
- Benign breast or uterine tumour,
- Increased volume of uterine leiomyoma,
- Vaginitis or vaginal candidiasis.
- Feeling weak (asthenia).
- Glucose intolerance.
- Change in libido.
- Hypertension.
- Skin decolouration, acne.
- Appearance of milk secretion (galactorrhea).
The above list includes the more common side effects of your medicine. They are usually mild.
If any of the following happen, tell your doctor immediately or go to Accident and Emergency at your nearest hospital:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin
- Sudden chest pain
- Pain in your chest that spreads to your arm or neck
- Yellowing of the eyes and face, darkening of urine, itchy skin (jaundice)
- Unexpected vaginal bleeding or spotting (breakthrough bleeding) after using Estrogel for some time, or after you stop treatment
- Breast change, including dimpling of the breast skin, change in the nipple, lumps that you can see or feel (breast cancer)
- Painful menstrual periods
- Unexplained migraine-like headaches
- Rash with target-shaped reddening or sores (erythema multiforme)
- Anaphylactic reaction (in women with past history of allergic reaction)
The above list includes very serious side effects. You may need urgent medical attention or hospitalisation.
Tell your pharmacist or doctor if you notice anything that is making you feel unwell.
Other side effects not listed above may also occur in some people.
After using Estrogel
Storage
Keep your medicine in the original pack until it is time to use it.
Keep your medicine in a cool dry place where the temperature stays below 30°C.
Do not store Estrogel or any other medicine in the bathroom or near a sink. Do not leave it on a window sill or in the car. Heat and dampness can destroy some medicines.
Keep it where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
Ask your pharmacist what to do with any medicine that is left over, or if the expiry date has passed.
Product description
What it looks like
Estrogel is a non-greasy, non-staining, clear, colourless, transdermal gel with an odour of alcohol.
Estrogel is a gel for transdermal use provided in a 80g bottle with a metered dose pump. Each Pump Pack delivers 64 metered doses.
Ingredients
The active ingredient is estradiol hemihydrate. This is a manmade form of the female hormone estrogen.
Each 1.25g dose of Estrogel gel contains 0.75 mg of estradiol (as hemihydrate).
It also contains:
- Carbomer 980, trolamine, ethanol and purified water.
This medicine does not contain lactose, sucrose, gluten, tartrazine or any other azo dyes.
Supplier
Estrogel is supplied in Australia by:
Besins Healthcare Australia Pty Ltd
Level 16, Tower 2, Darling Park,
201 Sussex Street,
Sydney, NSW 2000
® = Registered Trademark
Australian Registration Number
AUST R 301620
This leaflet was prepared in August 2023
Brand Information
| Brand name | Estrogel |
| Active ingredient | Estradiol |
| Schedule | S4 |
MIMS Revision Date: 01 March 2024
1 Name of Medicine
Estradiol hemihydrate.
2 Qualitative and Quantitative Composition
Estrogel contains the active ingredient estradiol (as hemihydrate). One gram of gel contains 0.6 mg (0.06% w/w) estradiol.
Each pump actuation delivers 1.25 g of gel which contains 0.75 mg of estradiol.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Estrogel transdermal gel is a clear, colourless gel with an odour of alcohol.
4 Clinical Particulars
4.1 Therapeutic Indications
Hormone replacement therapy (HRT) for estrogen deficiency symptoms in postmenopausal women.
Prevention of osteoporosis in postmenopausal women at high risk of future fractures who are intolerant of, or contraindicated for, other medicinal products approved for the prevention of osteoporosis (see Section 4.4 Special Warnings and Precautions for Use).
The experience treating women older than 65 years is limited. The lowest effective dose should be used for the shortest duration (see Section 4.2 Dose and Method of Administration; Section 4.4 Special Warnings and Precautions for Use).
4.2 Dose and Method of Administration
Dosage. Estrogel as an estrogen only product is indicated only for women without a uterus. Estrogel should be administered daily on a continuous basis.
In women with an intact uterus it is recommended to add a progestogen (e.g. progesterone) for at least 12 days of each month, in accordance with the manufacturers' recommendations.
Menopausal symptoms. Each metered dose (1 pump actuation) from the dispenser is 1.25 g of Estrogel. Although some women will respond to 1.25 g daily, the most usual starting dose is two pumps (2.5 g which contains 1.5 mg estradiol) of Estrogel once daily. In the majority of women this dose will provide effective relief of menopausal symptoms. If after one month's treatment effective relief is not obtained, the dosage may be increased to three or to a maximum of four pumps (5 g which contains 3.0 mg estradiol) of Estrogel daily.
The lowest effective dose should be used for maintenance therapy. The optimal daily maintenance dose needs to be reevaluated on a regular (e.g. annually) basis. Estrogel should only be continued for as long as the benefit outweighs the risk.
For initiation and continuation of treatment of postmenopausal symptoms, the lowest effective dose should be used for the shortest duration (see Section 4.4 Special Warnings and Precautions for Use).
Long-term safety of daily doses of Estrogel above 2.5 g (1.5 mg estradiol) has not been established.
Prevention of osteoporosis. The minimum effective dose is 2.5 g of Estrogel once daily for most patients. Use with progestogen:
In women with an intact uterus the recommended dose of a progestogen should be administered for at least 12 days of each month, in accordance with the manufacturers' recommendations. Unless there is a previous diagnosis of endometriosis, it is not recommended to add a progestogen in hysterectomised women.
Initiation of treatment. Women who are post-menopausal or have very infrequent menstrual cycles. Treatment with Estrogel can be started on any day.
Switching from a continuous estrogen-progestogen combined HRT. Treatment with Estrogel can be started on any day of the cycle.
Switching from a cyclic or continuous sequential HRT treatment. Finish the therapeutic sequence before beginning treatment with Estrogel.
Method of administration. The pump pack will require priming before using a new pump pack for the first time. The first dose dispensed should be discarded.
The correct dose of gel should be dispensed and applied to clean, dry, intact areas of skin e.g. on the arms and shoulders, and/or inner thighs. The area of application should be as large as possible.
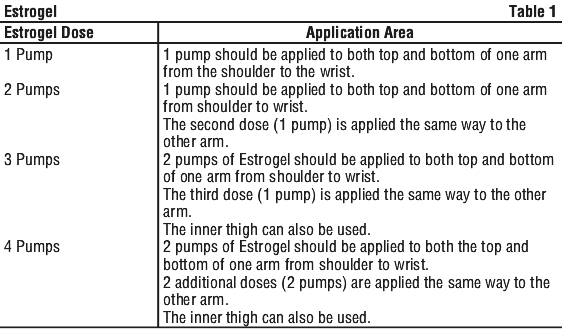
For low dose Estrogel (1 pump), Estrogel should be applied to both top and bottom of one arm from the shoulder to the wrist.
For standard dose Estrogel (2 pumps), 1 pump should be applied to both top and bottom of one arm from shoulder to wrist. The second dose is applied the same way to the other arm.
For a higher dose of Estrogel, 2 pumps of Estrogel should be applied to both top and bottom of one arm from shoulder to wrist. The third dose (1 pump) is applied the same way to the other arm. The inner thigh can also be used.
For the highest approved dose (4 pumps), 2 pumps of Estrogel should be applied to both the top and bottom of one arm from shoulder to wrist. The 2 additional doses (2 pumps) are applied the same way to the other arm. The inner thigh can also be used. See Table 1.
Estrogel should be allowed to dry for 5 minutes before covering the skin with clothing.
The gel should be applied by the patient herself, not by anyone else, and skin contact, particularly with a male partner, should be avoided for one hour after application. Wash hands with soap and water after applying the gel. Washing the skin or contact with other skin products should be avoided until at least one hour after application of Estrogel.
For people not being treated with Estrogel. In the event of contact with an application area, which has not been washed or is not covered with clothing, wash the area of skin onto which Estrogel may have been transferred as soon as possible, using soap and water.
If the patient forgets to apply a dose and it is more than 12 hours until the next dose, the missed dose should be applied and normal dosing resumed the next day. If the next dose is less than 12 hours away, it is best just to wait and apply the next dose normally. Patients should be advised not to apply two doses at the same time.
Forgetting a dose may increase the likelihood of break-through bleeding and spotting.
Method of administration. For transdermal use.
4.3 Contraindications
Known, past or suspected breast cancer.
Known or suspected estrogen-dependent malignant tumours (e.g. endometrial cancer).
Undiagnosed genital bleeding.
Untreated endometrial hyperplasia.
Previous or current venous thromboembolism (e.g. deep venous thrombosis, pulmonary embolism).
Known thrombophilic disorders (e.g. protein C, protein S, or antithrombin deficiency, see Section 4.4 Special Warnings and Precautions for Use).
Active or recent arterial thromboembolic disease (e.g. angina, myocardial infarction).
Acute liver disease, or a history of liver disease as long as liver function tests have failed to return to normal.
Known or suspected pregnancy.
Lactation.
Known hypersensitivity to the active substances or to any of the excipients.
Porphyria.
4.4 Special Warnings and Precautions for Use
For the treatment of postmenopausal symptoms, HRT should only be initiated for symptoms that adversely affect quality of life. In all cases, a careful appraisal of the risks and benefits should be undertaken at least annually, and HRT should only be continued as long as the benefit outweighs the risk.
Evidence regarding the risks associated with HRT in the treatment of premature menopause is limited. Due to the low level of absolute risk in younger women, however, the balance of benefits and risks for these women may be more favourable than in older women.
Medical examination and follow-up. Before initiating or reinstituting HRT, a complete personal and family medical history should be taken. Physical (including pelvic and breast) examination should be guided by this and by contraindications and warnings for use. During treatment, periodic check-ups are recommended of a frequency and nature adapted to the individual woman.
Women should be advised what changes in their breasts should be reported to their doctor or nurse (see "Breast cancer" below). Investigations, including appropriate imaging tools, e.g. mammography should be carried out in accordance with currently accepted screening practices, modified to the clinical needs of the individual.
Conditions which need supervision. If any of the following conditions are present, have occurred previously, and/or have been aggravated during pregnancy or previous hormone treatment, the patient should be closely supervised. It should be taken into account that these conditions may recur or be aggravated during treatment with Estrogel, in particular:
Leiomyoma (uterine fibroids) or endometriosis.
Risk factors for thromboembolic disorders (see below).
Risk factors for estrogen dependent tumours, e.g. 1st degree heredity for breast cancer.
Hypertension.
Liver disorders (e.g. liver adenoma).
Diabetes mellitus with or without vascular involvement.
Cholelithiasis.
Migraine or (severe) headache.
Systemic lupus erythematosus (SLE).
A history of endometrial hyperplasia (see below).
Epilepsy.
Asthma.
Otosclerosis.
Reasons for immediate withdrawal of therapy. Therapy should be discontinued in case a contraindication is discovered and in the following situations:
jaundice or deterioration in liver function;
significant increase in blood pressure;
new onset of migraine-type headache;
pregnancy.
Endometrial hyperplasia and carcinoma. In women with an intact uterus the risk of endometrial hyperplasia and carcinoma is increased when estrogens are administered alone for prolonged periods. The reported increase in endometrial cancer risk among estrogen-only users varies from 2- to 12-fold greater compared with non-users, depending on the duration of treatment and estrogen dose (see Section 4.8 Adverse Effects (Undesirable Effects)). After stopping treatment risk may remain elevated for at least 10 years.
The addition of a progestogen cyclically for at least 12 days per month/28 day cycle or continuous combined estrogen-progestogen therapy in non-hysterectomised women prevents the excess risk associated with estrogen-only HRT.
Break-through bleeding and spotting may occur during the first months of treatment. If break-through bleeding or spotting appears after some time on therapy, or continues after treatment has been discontinued, the reason should be investigated, which may include endometrial biopsy to exclude endometrial malignancy.
Unopposed estrogen stimulation may lead to premalignant or malignant transformation in the residual foci of endometriosis. Therefore, the addition of progestogens to estrogen replacement therapy should be considered in women who have undergone hysterectomy because of endometriosis if they are known to have residual endometriosis.
Breast cancer. The overall evidence shows an increased risk of breast cancer in women taking combined estrogen-progestogen or estrogen-only HRT, that is dependent on the duration of taking HRT.
Estrogen-only therapy. The WHI trial found no increase in the risk of breast cancer in hysterectomised women using estrogen-only HRT. Observational studies have mostly reported a small increase in risk of having breast cancer diagnosed that is lower than that found in users of estrogen-progestogen combinations (see Section 4.8 Adverse Effects (Undesirable Effects)).
Combined estrogen-progestogen therapy. The randomised placebo-controlled trial, the Women's Health Initiative study (WHI), and a meta-analysis of prospective epidemiological studies are consistent in finding an increased risk of breast cancer in women taking combined estrogen-progestogen for HRT that becomes apparent after about 3 years (1-4) (see Section 4.8 Adverse Effects (Undesirable Effects)).
Results from a large meta-analysis showed that after stopping treatment, the excess risk will decrease with time and the time needed to return to baseline depends on the duration of prior HRT use. When HRT was taken for more than 5 years, the risk may persist for 10 years or more. HRT, especially estrogen-progestogen combined treatment, increases the density of mammographic images which may adversely affect the radiological detection of breast cancer.
Ovarian cancer. Ovarian cancer is much rarer than breast cancer. Epidemiological evidence from a large meta-analysis suggests a slightly increased risk in women estrogen-only or combined estrogen-progestogen HRT which becomes apparent within 5 years of use and diminishes over time after stopping.
Some other studies, including the WHI trial suggest that use of combined HRTs may be associated with a similar, or slightly smaller risk (see Section 4.8 Adverse Effects (Undesirable Effects)).
Venous thromboembolism. HRT is associated with a 1.3 - 3 fold risk of developing venous thromboembolism (VTE), i.e. deep vein thrombosis or pulmonary embolism. The occurrence of such an event is more likely in the first year of HRT than later (see Section 4.8 Adverse Effects (Undesirable Effects)).
Patients with known thrombophilic states have an increased risk of VTE and HRT may add to this risk. HRT is therefore contraindicated in these patients (see Section 4.3 Contraindications).
Generally recognised risk factors for VTE include, use of estrogens, older age, major surgery, prolonged immobilisation, obesity (BMI > 30 kg/m2), pregnancy/postpartum period, systemic lupus erythematosus (SLE) and cancer. There is no consensus about the possible role of varicose veins in VTE. As in all postoperative patients, prophylactic measures need be considered to prevent VTE following surgery. If prolonged immobilisation is to follow elective surgery temporarily stopping HRT 4 to 6 weeks earlier is recommended. Treatment should not be restarted until the woman is completely mobilised.
In women with no personal history of VTE but with a first degree relative with a history of thrombosis at young age, screening may be offered after careful counseling regarding its limitations (only a proportion of thrombophilic defects are identified by screening).
If a thrombophilic defect is identified which segregates with thrombosis in family members or if the defect is 'severe' (e.g. antithrombin, protein S, or protein C deficiencies or a combination of defects) HRT is contraindicated.
Women already on chronic anticoagulant treatment require careful consideration of the benefit risk of use of HRT.
If VTE develops after initiating therapy, the drug should be discontinued.
Patients should be told to contact their doctors immediately when they are aware of a potential thromboembolic symptom (e.g. painful swelling of a leg, sudden pain in the chest, dyspnoea).
Coronary artery disease (CAD). There is no evidence from randomised controlled trials of protection against myocardial infarction in women with or without existing CAD who received combined estrogen-progestogen or estrogen-only HRT.
Estrogen-only. Randomised controlled data found no increased risk of CAD in hysterectomised women using estrogen-only therapy.
Combined estrogen-progestogen therapy. The relative risk of CAD during use of combined estrogen+progestogen HRT is slightly increased. As the baseline absolute risk of CAD is strongly dependent on age, the number of extra cases of CAD due to estrogen+progestogen use is very low in healthy women close to menopause, but will rise with more advanced age.
Ischaemic stroke. Combined estrogen-progestogen and estrogen-only therapy are associated with an up to 1.5-fold increase in risk of ischemic stroke. The relative risk does not change with age or time since menopause. However, as the baseline risk of stroke is strongly age-dependent, the overall risk of stroke in women who use HRT will increase with age (see Section 4.8 Adverse Effects (Undesirable Effects)).
Other conditions. Estrogens may cause fluid retention, and therefore patients with cardiac or renal dysfunction should be carefully observed.
Women with pre-existing hypertriglyceridaemia should be followed closely during estrogen replacement or hormone replacement therapy, since rare cases of large increases of plasma triglycerides leading to pancreatitis have been reported with estrogen therapy in this condition.
Exogenous estrogens may induce or exacerbate symptoms of hereditary and acquired angioedema.
Effects on thyroid function. Women with normal thyroid function can compensate for the increased TBG by making more thyroid hormone, thus maintaining free T4 and T3 serum concentrations in the normal range. Women dependent on thyroid hormone replacement therapy who are also receiving estrogens may require increased doses of their thyroid-replacement therapy. These women should have their thyroid function monitored in order to maintain an acceptable range.
Use in the elderly. The experience treating women older than 65 years is limited.
HRT use does not improve cognitive function. There is some evidence from the WHI trial of increased risk of probable dementia in women who start using continuous combined or estrogen-only HRT after the age of 65.
ALT elevations. During clinical trials with patients treated for hepatitis C virus (HCV) infections with the combination regimen ombitasvir/paritaprevir/ritonavir with and without dasabuvir, ALT elevations greater than 5 times the upper limit of normal (ULN) were significantly more frequent in women using ethinylestradiol-containing medicinal products such as CHCs. Additionally, also in patients treated with glecaprevir/pibrentasvir, ALT elevations were observed in women using ethinylestradiol-containing medications such as CHCs. Women using medicinal products containing estrogens other than ethinylestradiol, such as estradiol, had a rate of ALT elevation similar to those not receiving any estrogens; however, due to the limited number of women taking these other estrogens, caution is warranted for co-administration with the combination drug regimen ombitasvir/paritaprevir/ritonavir with or without dasabuvir and also the regimen glecaprevir/pibrentasvir. See Section 4.5.
Paediatric use. Use in children is not recommended.
There is limited data available supporting the use of transdermal estradiol in paediatric indications such as Turners Syndrome and Pubertal Induction. The treating physician should determine the benefits and risks of use of Estrogel in children prior to initiating treatment.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Treatment with surface active agents (e.g. sodium lauryl sulphate), or other drugs which alter barrier structure or function, could remove drug bound to the skin, altering transdermal flux. Therefore, patients should avoid the use of strong skin cleansers and detergents (e.g. benzalkonium or benzothonium chloride products), skin care products of high alcoholic content (astringents, sunscreens) and keratolytics (e.g. salicylic acid, lactic acid).
The use of any concomitant skin medication which alters skin production (e.g. cytotoxic drugs) should be avoided at the site of application of Estrogel.
Sun exposure and photo-irritation. Estrogel did not exhibit reactions that were indication of photo-irritation or photoallergy. Estrogel was well tolerated by the subjects with no clinically relevant findings.
The metabolism of estrogens may be increased by concomitant use of substances known to induce drug-metabolising enzymes, specifically cytochrome P450 enzymes, such as anticonvulsants (e.g. phenobarbital, phenytoin, carbamazepine) and anti-infectives (e.g. rifampicin, rifabutin, nevirapine, efavirenz).
Ritonavir and nelfinavir, although known as strong inhibitors, by contrast exhibit inducing properties when used concomitantly with steroid hormones. Herbal preparations containing St. John's wort (Hypericum perforatum) may induce the metabolism of estrogens.
At transdermal administration, the first-pass effect in the liver is avoided and thus, transdermally applied estrogens HRT might be less affected than oral hormones by enzyme inducers.
Clinically, an increased metabolism of estrogens and progestogens may lead to decreased effect and changes in the uterine bleeding profile.
Effect of HRT with estrogens on other medicinal products. Hormone contraceptives containing estrogens have been shown to significantly decrease plasma concentrations of lamotrigine when co-administered due to induction of lamotrigine glucuronidation. This may reduce seizure control. Although the potential interaction between hormone replacement therapy and lamotrigine has not been studied, it is expected that a similar interaction exists, which may lead to a reduction in seizure control among women taking both medicinal products together.
Pharmacodynamic interactions. During clinical trials with the HCV combination drug regimen ombitasvir/paritaprevir/ritonavir with and without dasabuvir, ALT elevations greater than 5 times the upper limit of normal (ULN) were significantly more frequent in women using ethinylestradiol-containing medicinal products such as CHCs. Women using medicinal products containing estrogens other than ethinylestradiol, such as estradiol, had a rate of ALT elevation similar to those not receiving any estrogens; however, due to the limited number of women taking these other estrogens, caution is warranted for co-administration with the combination drug regimen ombitasvir/paritaprevir/ritonavir with or without dasabuvir and also the regimen with glecaprevir/pibrentasvir (see Section 4.4).
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No specific studies on fertility have been conducted with Estrogel. Estradiol has been shown to impair fertility in animals.
Use in pregnancy. (Category B3)
Estrogel must not be used during pregnancy (see Section 4.3 Contraindications). If pregnancy occurs during medication with Estrogel, treatments should be withdrawn immediately.
The results of most epidemiological studies to date relevant to inadvertent fetal exposure to estrogens indicate no teratogenic or fetotoxic effects.
Use in lactation. Estrogel must not be used during breast-feeding (see Section 4.3 Contraindications). Estradiol is excreted in human breast milk.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Summary of the safety profile. The undesirable effects are generally mild and rarely require treatment withdrawal. Undesirable effects, if any, usually occur during the first months of treatment.
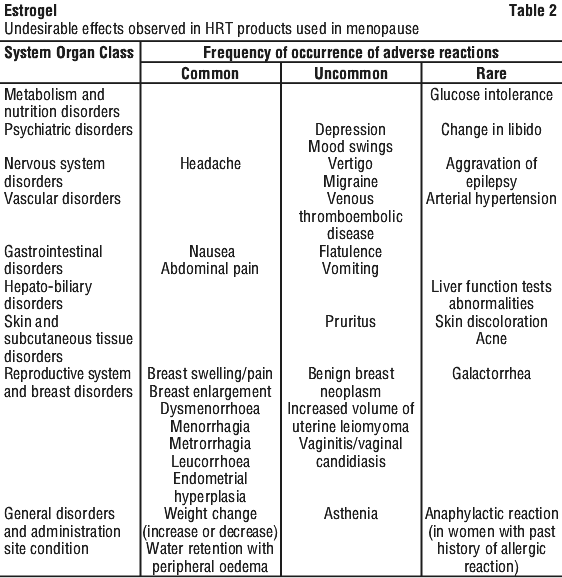
Undesirable effects observed with HRT products used in menopause are reported in Table 2.
Adverse reactions were categorised as very common (≥ 1/10), common (≥ 1/100 to ≤ 1/10), uncommon (≥ 1/1,000 to ≤ 1/100) or rare (≥ 1/10,000 to ≤ 1/1,000).
gall bladder disease;
skin and subcutaneous disorders: chloasma, erythema multiforme, erythema nodosum, vascular purpura;
probable dementia over the age of 65 (see Section 4.4 Special Warnings and Precautions for Use).
Exacerbation of hereditary and acquired angioedema.
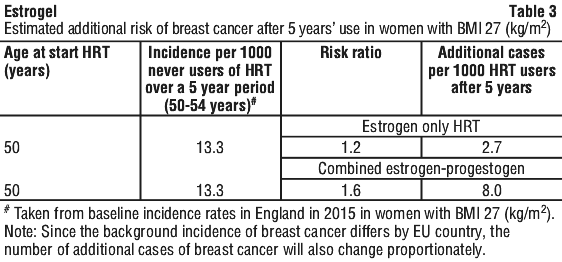
Breast cancer risk. An up to 2-fold increased risk of having breast cancer diagnosed is reported in women taking combined estrogen-progestogen therapy for more than 5 years.
The increased risk in users of estrogen-only therapy is substantially lower than that seen in users of estrogen-progestogen combinations.
The level of risk is dependent on the duration of use (see Section 4.4 Special Warnings and Precautions for Use).
Absolute risk estimations based on results of the largest randomised placebo-controlled trial (WHI-study) and the largest meta-analysis of prospective epidemiological studies are presented in Table 3, Table 4 and Table 5.
Largest meta-analysis of prospective epidemiological studies.


In women with a uterus, use of estrogen-only HRT is not recommended because it increases the risk of endometrial cancer (see Section 4.4 Special Warnings and Precautions for Use).
Depending on the duration of estrogen-only use and estrogen dose, the increase in risk of endometrial cancer in epidemiology studies varied from between 5 and 55 extra cases diagnosed in every 1000 women between the ages of 50 and 65.
Adding a progestogen to estrogen-only therapy for at least 12 days per cycle can prevent this increased risk. In the Million Women Study the use of five years of combined (sequential or continuous) HRT did not increase risk of endometrial cancer (RR of 1.0 (0.8-1.2)).
Ovarian cancer. Use of estrogen-only or combined estrogen-progestogen HRT has been associated with a slightly increased risk of having ovarian cancer diagnosed (see Section 4.4 Special Warnings and Precautions for Use).
A meta-analysis from 52 epidemiological studies reported an increased risk of ovarian cancer in women currently using HRT compared to women who have never used HRT (RR 1.43, 95% CI 1.31-1.56). For women aged 50 to 54 years taking 5 years of HRT, this results in about 1 extra case per 2000 users. In women aged 50 to 54 who are not taking HRT, about 2 women in 2000 will be diagnosed with ovarian cancer over a 5-year period.
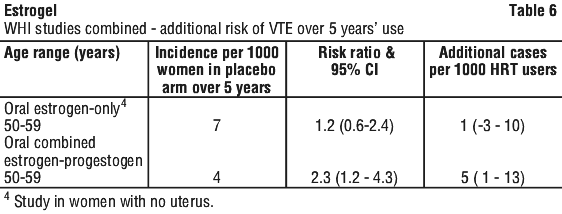
Risk of venous thromboembolism. HRT is associated with a 1.3-3-fold increased relative risk of developing venous thromboembolism (VTE), i.e. deep vein thrombosis or pulmonary embolism. The occurrence of such an event is more likely in the first year of using HT (see Section 4.4 Special Warnings and Precautions for Use). Results of the WHI studies are presented in Table 6.
Risk of ischaemic stroke. The use of estrogen-only and estrogen + progestogen therapy is associated with an up to 1.5 fold increased relative risk of ischaemic stroke. The risk of haemorrhagic stroke is not increased during use of HRT.
This relative risk is not dependent on age or on duration of use, but as the baseline risk is strongly age-dependent, the overall risk of stroke in women who use HRT will increase with age see Table 7, see Section 4.4 Special Warnings and Precautions for Use.

4.9 Overdose
Pain in the breasts or excessive production of cervical mucus may be indicative of too high a dosage, but acute overdosage has not been reported and is unlikely to be a problem. Overdosages of estrogen may cause nausea, and withdrawal bleeding may occur. There are no specific antidotes and treatment should be symptomatic.
For information on the management of overdose, contact the Poisons Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
ATC Code: G03CA03.
Sex Hormones and Modulators of the Genital System - Natural and Semisynthetic Estrogens, plain.
Mechanism of action. The active ingredient, estradiol, is chemically and biologically identical to endogenous human estradiol, the major and most potent estrogenic hormone secreted by the ovary from menarche to menopause. Estrogens are important for the development and maintenance of the female reproductive system and secondary sexual characteristics, and modulate various physiological processes, including bone resorption. Exogenous estradiol substitutes for the loss of endogenous estrogen production in menopausal women to alleviate menopausal symptoms, and prevents bone loss following menopause or ovariectomy.
Clinical trials. Estrogen deficiency symptoms. Estrogen supplementation is recognised as an effective treatment for reducing the frequency and severity of hot flushes. A Cochrane review estimated that oral HRT reduces the frequency of hot flushes by 75% and eases severity (MacLennan 2004), a subsequent meta-analysis showed that oral estradiol resulted in 16.8 fewer hot flushes per week (Nelson 2004). Estrogen with or without progesterone is recommended for management of vasomotor menopausal symptoms by HRT guidelines worldwide (Santen 2010, Sturdee 2011, Panay 2013, de Villiers 2016); the Global Consensus Statement on menopausal hormone therapy specifies HRT as the most effective treatment for vasomotor symptoms (de Villiers 2016).
Efficacy data for Estrogel in the treatment of estrogen deficiency symptoms in postmenopausal women was provided by 9 company sponsored studies (Archer 2003, CV141-002 1999, Archer 2012, Foidart 1994, Kornafel 1992, Christiansen 1988, Jensen 1987b, Dupont 1991, Sentrakul 1991) and 11 non-company sponsored studies (Mizunuma 2011, Akhila 2006, Foidart 1997, Pelissier 1999 and Pelissier 2001, Faguer de Moustier 1989, Holst 1987, Holst 1983a, Vihtamaki 1998, Elkik 1982, Polo-Kantola 1999, Basdevant 1991).
Compared with placebo, Estrogel produced a significantly greater decrease in the frequency and severity of hot flushes (Archer 2012 (0.75-1.5 mg estradiol), Archer 2003 (0.75-1.5 mg estradiol), Kornafel 1992 (1.5 mg estradiol), Christiansen 1988 (3 mg estradiol), Polo-Kantola 1999 (1.5 mg estradiol)); a larger decrease with Estrogel versus placebo was also reported by Jensen 1987b (3 mg estradiol) although a statistical analysis was not reported. Mizunuma 2011 showed that, after an induction phase in which Estrogel decreased the incidence of hot flushes (1.08 mg estradiol), subjects who switched to placebo saw an increase in the incidence of hot flushes back to baseline levels (0.54 mg estradiol maintenance phase). Estrogel also significantly improved vaginal atrophy markers when compared with placebo (Archer 2012 (0.75-1.5 mg estradiol), Archer 2003 (0.75-1.5 mg estradiol), Kornafel 1992 (1.5 mg estradiol)).
Estrogel efficacy in the relief of menopausal symptoms was not significantly different from estradiol transdermal patches (Akhila 2006 (1.5 mg estradiol), Foidart 1997 (1.5 mg estradiol)).
When compared with other transdermal regimens, the efficacy of Estrogel at a dose of 1.5 mg/day in the relief of menopausal symptoms was not significantly different from estradiol transdermal patches (CV141-002 1999 (0.375-1.5 mg estradiol), Akhila 2006 (1.5 mg estradiol), Foidart 1997 (1.5 mg estradiol)), although the lower 0.75 mg/day Estrogel dose was not as effective as a 50 microgram/day transdermal patch (CV141-002 1999). When compared with oral estrogen regimens, Akhila 2006 determined Estrogel (1.5 mg estradiol) efficacy to be significantly better than oral conjugated estrogen when assessed by the incidence of complete relief of vasomotor symptoms, psychological disturbances, genital symptoms, and urinary symptoms. However, other studies reported that improvements in symptoms were similar with Estrogel and oral regimens (Jensen 1987b, Dupont 1991, Sentrakul 1991, Faguer de Moustier 1989, Elkik 1982, Basdevant 1991).
Improvements in vaginal atrophy markers were also similar between Estrogel (1.5 mg estradiol) and oral regimens (0.625 mg conjugated estrogens) (Dupont 1991). One Estrogel study noted potential efficacy of Estrogel in managing insomnia (Dupont 1991).
Where reported, the addition of oral progesterone did not alter the Estrogel response.
In the 4-year KEEPS study, recently postmenopausal women had similar and substantial reductions in hot flashes and night sweats with lower-than-conventional doses of oral (0.45 mg conjugated estrogens) or transdermal patch estrogen (50 microgram/daily). These reductions were sustained during 4 years. Insomnia was intermittently reduced compared with placebo for both hormone regimens (Santoro 2017). A comparison, performed by the company, of data from 2 Estrogel company-sponsored studies (Archer 2012 (0.75-1.5 mg estradiol), Brennan 2001 (0.375-1.5 mg estradiol)) against data from the KEEPS study showed Estrogel to be equivalent to the Climara patch in the alleviation of hot flushes and similar in systemic estradiol exposure (Piette 2014).
Overall, there is strong support for the efficacy of Estrogel in the treatment of menopausal symptoms.
Prevention of osteoporosis. Estrogen supplementation prevents the usual accelerated increase in bone loss if HRT is started early in menopause, while starting estrogen treatment later stops and to a degree reverses bone loss (Santen 2010), preserves and increases bone mineral density (Santen 2010), and reduces fracture risk by up to 27% (Christenson 2012). In the KEEPS study, both oral and transdermal HRT groups showed favourable effects on bone mineral density compared to the placebo group (Cobin 2017). The Global Consensus Statement on menopausal hormone therapy (de Villiers 2016) and most international guidelines (NICE 2015, Stuenkel 2016, Baber 2016, NAMS 2017, Cobin 2017) state that HRT has been shown to significantly lower the risk of hip, vertebral, and other osteoporosis-related fractures in postmenopausal women.
Efficacy data for Estrogel for the prevention of osteoporosis in postmenopausal women was provided by 3 company-sponsored studies (Jensen 1988 and Johansen 1988, Riis 1987a, Riis 1987b) and 8 non-company sponsored studies (Ng 1993, Yang 2007, Sun 2002, Devogelaer 1998, Wimalawansa 1995, Palacios 1995, Ribot 1990, Tremollieres 1990). Bone mineral content and density remained steady or increased during Estrogel (3 mg estradiol) treatment but decreased during placebo treatment, resulting in significantly higher bone mineral content and density in Estrogel-treated subjects versus placebo-treated subjects (Jensen 1988, Riis 1987a, Riis 1987b). One study showed no change in bone mineral density with either Estrogel or placebo (Ng 1993); however, the study was of shorter duration than the company-sponsored studies (1 year versus 2 years) and used a lower dose of Estrogel (0.75-1.5 mg/day versus 3 mg/day of estradiol).
No studies were identified that compared Estrogel with other transdermal regimens in the prevention of osteoporosis.
When compared with oral regimens, improvements in bone mineral content and density were similar between Estrogel (3 mg estradiol) and oral estradiol valerate (2 mg estradiol valerate) (Jensen 1988, Riis 1987b), oral conjugated estradiol (0.625 mg/day) (Palacios 1994), and oral intermittent cyclically-administered etidronate (400 mg/day) (Wimalawansa 1995). The improvement in bone mineral density with Estrogel dose range (0.75-1.5 mg estradiol) was significantly greater than with oral estriol (2 mg/day) (Yang 2007, Devogelaer 1998).
The remaining study, which compared Estrogel doses, also supported the efficacy of Estrogel in preventing the postmenopausal decline in bone mineral density when administered at doses of 1.5 mg/day of estradiol or above (Sun 2002).
Where reported, the addition of oral progesterone did not alter the Estrogel response.
Overall, the efficacy studies strongly support the efficacy of Estrogel in the prevention of postmenopausal osteoporosis.
In summary, the efficacy of Estrogel in the treatment of estrogen deficiency symptoms has been demonstrated for treatment durations of up to 2 years (Christiansen 1988 and Jensen 1987b) and the effect of Estrogel on osteoporosis has been demonstrated for up to 4 years (Sun 2002). No evidence of tolerance to the efficacious effects of Estrogel was reported.
5.2 Pharmacokinetic Properties
Absorption. Pharmacokinetic studies indicate that, when applied topically to a large area of skin in a volatile solvent, approximately 10% of the estradiol is percutaneously absorbed into the vascular system, regardless of the age of the patient. Daily application of 2.5 g or 5 g Estrogel over a surface area of 400-750 cm2 results in a gradual increase in estrogen blood levels to steady state after approximately 3-5 days and provides circulating levels of both estradiol and estrone equivalent in absolute concentrations and in their respective ratio to those obtained during the early-mid follicular phase of the menstrual cycle.
Estrogel was administered to 17 postmenopausal women once daily on the posterior surface of one arm from wrist to shoulder for 14 consecutive days. Maximum serum concentrations (Cmax) of estradiol and estrone on Day 12 were 117 picogram/mL and 128 picogram/mL, respectively.
The time-averaged serum estradiol and estrone concentrations (Caverage) over the 24 hour dose interval after administration of 2.5 g of Estrogel on Day 12 were 76.8 picogram/mL and 95.7 picogram/mL, respectively.
When treatment is stopped, estradiol and urinary conjugated estradiol concentrations return to baseline in about 76 hours.
Distribution. Estradiol is extensively bound to plasma proteins, mainly to sex hormone binding globulin (SHBG) and to a lesser degree albumin. Only around 2% is free and biologically active. Tissue distribution of unbound estradiol is rapid and wide.
Metabolism. Once systemically absorbed, transdermally applied estradiol is metabolised in the same way as the endogenous hormone. Estradiol is metabolised primarily in the liver to estrone, then later to estriol, epioestriol and catechol estrogens, which are then conjugated to sulfates and glucuronides. Metabolism involves multiple CYP isozymes, but is predominantly mediated by CYP3A4. Estriol is glucuronidated by UGT1A1. Estradiol metabolites are subject to enterohepatic circulation.
Excretion. Excretion is mainly via the urine (as conjugated metabolites). Only a small amount is excreted in faeces.
5.3 Preclinical Safety Data
Genotoxicity. Estradiol shows some genotoxic activity in vitro. While mutagenicity was not observed with estradiol in bacterial cells (Ames test), positive results have been obtained with the compound in assays for mutagenicity, induction of chromosomal aberrations, aneuploidy, sister chromatid exchange (indicative of DNA damage), and single-strand DNA breaks in mammalian cells. No clastogenicity was observed with estradiol in vivo in rodent bone marrow micronucleus assays. Reactive catechol metabolites of estradiol have been found to form DNA adducts.
Carcinogenicity. Long-term, continuous administration of natural and synthetic estrogens in laboratory animal species increases the frequency of carcinomas of the breast, uterus, cervix, vagina, testis, and liver. Carcinogenicity by estradiol may involve gene mutation induced by reactive metabolites or the activation of estrogen receptor-mediated signaling pathways that sustain the growth and survival of preneoplastic and malignant cells.
6 Pharmaceutical Particulars
6.1 List of Excipients
Ethanol, carbomer 980, trolamine, purified water.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C. Protect from light.
6.5 Nature and Contents of Container
Metering canister composed of a polypropylene bottle, a LDPE pouch, a polypropylene metering pump and closed with a polypropylene cap, containing 80 g of gel and are available in packs of 1, 2 and 3 pump(s).
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure.
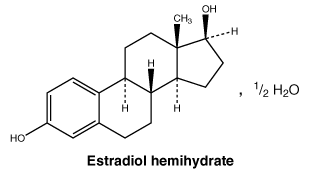
CAS number. 35380-71-3.
7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Date of First Approval
08 May 2019
Date of Revision
18 December 2023
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.