Eumovate
Brand Information
| Brand name | Eumovate |
| Active ingredient | Clobetasone butyrate |
| Schedule | S3 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Eumovate.
Summary CMI
EUMOVATE
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using EUMOVATE?
EUMOVATE contains the active ingredient clobetasone butyrate. EUMOVATE is used to treat eczema and dermatitis. It can help you to control patches of dry, red, itchy, flaky or inflamed skin caused by eczema and dermatitis.
For more information, see Section 1. Why am I using EUMOVATE? in the full CMI.
2. What should I know before I use EUMOVATE?
Do not use if you have ever had an allergic reaction to clobetasone butyrate, other similar medicines such as hydrocortisone or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use EUMOVATE? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with EUMOVATE and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use EUMOVATE?
- Follow the instructions provided on how to use EUMOVATE.
- The minimum amount of EUMOVATE cream should be used for the shortest amount of time. Do not use EUMOVATE for more than 7 days.
More instructions can be found in Section 4. How do I use EUMOVATE? in the full CMI.
5. What should I know while using EUMOVATE?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using EUMOVATE? in the full CMI.
6. Are there any side effects?
Side effects which have been reported include rash, itch, local skin burning, skin thinning, skin bleaching or increased skin pigmentation, hair disorders, worsening of eczema or dermatitis symptoms. Serious side effects include allergic reaction, infection, high blood pressure, glaucoma, cataract, a decrease in the level of the hormone cortisol in your blood, increased blood sugar levels, weakening of the bones through gradual loss of mineral and glucose in the urine, increased weight, rounding of the face, obesity, increased body hair, delayed weight gain (in children) and slow growth (in children). For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
EUMOVATE
Active ingredient: clobetasone butyrate 0.05% w/w cream
Consumer Medicine Information (CMI)
This leaflet provides important information about using EUMOVATE. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using EUMOVATE.
Where to find information in this leaflet:
1. Why am I using EUMOVATE?
2. What should I know before I use EUMOVATE?
3. What if I am taking other medicines?
4. How do I use EUMOVATE?
5. What should I know while using EUMOVATE?
6. Are there any side effects?
7. Product details
1. Why am I using EUMOVATE?
EUMOVATE contains the active ingredient clobetasone butyrate. EUMOVATE is a topical corticosteroid. It is used to control inflammation on the skin.
EUMOVATE is used to treat eczema and dermatitis. It can help you to control patches of dry, red, itchy, flaky or inflamed skin caused by eczema and dermatitis.
EUMOVATE works to stop the skin's over-reaction to the triggers that cause skin flare ups. It suppresses the inflammation that causes eczema or dermatitis.
EUMOVATE cream base also has moisturising properties.
2. What should I know before I use EUMOVATE?
Warnings
Do not use EUMOVATE if:
- you are allergic to clobetasone butyrate, other similar medicines such as hydrocortisone, or any of the ingredients listed at the end of this leaflet. Always check the ingredients to make sure you can use this medicine. Some of the symptoms of an allergic reaction may include:
- shortness of breath.
- wheezing or difficulty breathing.
- swelling of the face, lips, tongue or other parts of the body.
- rash, itching or hives on the skin.
Do not use EUMOVATE on:
- cuts or open wounds.
- infected skin.
- inflamed skin near ulcers.
- delicate skin areas such as the face, groin, genitals or between the toes.
Do not use EUMOVATE unless advised by a doctor to treat any conditions other than eczema and dermatitis.
Do not use the cream on the wrong skin conditions. It could make it worse.
Do not use EUMOVATE to treat:
- rosacea (skin condition of the face where the nose and cheeks are unusually red).
- acne, spots or pimples.
- pruritis (itching) without rash.
- perioral dermatitis.
- untreated bacterial infections such as cellulitis (infection of the skin), folliculitis (infection of the hair follicle), furunculosis (boils) or impetigo (blisters).
- fungal infections such as athlete's foot and jock itch.
- viral infections such as cold sores, chicken pox or shingles.
- parasitic infestations such as scabies.
- psoriasis unless recommended by your doctor.
If you have any of these conditions, ask your doctor or pharmacist for advice.
If you are not sure what is causing your skin problem, ask your doctor or pharmacist.
Check with your doctor or pharmacist if you:
- have any other medical conditions, particularly kidney or liver conditions.
- take any medicines for any other condition.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor or pharmacist if you are pregnant or intend to become pregnant.
Talk to your doctor or pharmacist if you are breastfeeding or intend to breastfeed.
You should not use EUMOVATE if you are pregnant or may be pregnant or if you are breastfeeding unless advised by your doctor.
Your doctor or pharmacist will discuss the benefits and risks of using EUMOVATE whilst pregnant or breastfeeding.
If you do use EUMOVATE when breastfeeding, do not use EUMOVATE on your breast area to ensure that the baby does not accidentally get EUMOVATE in their mouth.
Children
Do not use EUMOVATE on children under the age of 12 years except on the advice of a doctor.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with EUMOVATE and affect how it works. These include:
- other corticosteroid medicines which may include some eczema creams, asthma inhalers, tablets, injections, nasal sprays, and eye or nose drops.
- ritonavir (a medicine for the treatment of HIV).
- itraconazole (a medicine for the treatment of fungal infections).
Do not use other corticosteroids (like hydrocortisone) on the skin while you are using this cream. You would be doubling the dose.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect EUMOVATE.
4. How do I use EUMOVATE?
How much to use
- EUMOVATE is for external use only.
- The minimum amount of EUMOVATE should be used for the shortest amount of time. Do not use EUMOVATE for more than 7 days.
- Squeeze out the cream along the top of your index finger (see picture).
- From the crease in the finger, squeeze the cream halfway to your fingertip. This will cover a patch of skin the same size as the palm of your hand.
- Use the fingertip unit as a guide. For smaller areas, use a smaller amount. This cream is not meant to treat large areas.
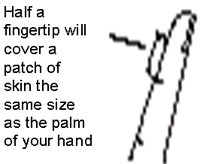
How to use EUMOVATE
Adults and children over 12 years of age:
- Wash your hands and dry them.
- Squeeze out the correct amount of cream to cover the affected area on your index finger. The picture above gives you an idea of how much to use.
- Gently rub cream onto the patch of skin you are treating.
- Wash your hands again (unless it is your hands you are treating).
- Don't cover the treated patch of skin with anything (bandages, dressings, gloves or plastic wrap). It can cause more of the medicine to pass through the skin.
- Be careful not to get the cream in your eyes.
Use in children under 12 years of age only on the advice of a doctor.
When to use EUMOVATE
- Use the cream twice a day for up to 7 days.
- Using it at the same time each day will have the best effect. It will also help you remember when to use it.
If you forget to use EUMOVATE
If you forget to use EUMOVATE or miss an application, use it when you remember.
Do not try to make up for missed applications by using the cream more often as this may increase the chance of you getting a side effect.
If you use too much EUMOVATE
If you use a bit too much of the cream by mistake, don't worry but try to keep to the fingertip unit. Using corticosteroids on the skin continuously over many weeks or months can cause skin thinning and permanent skin damage.
If you or anyone else swallows EUMOVATE, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using EUMOVATE?
Things you should do
Remind any doctor, dentist or pharmacist you visit that you are using EUMOVATE. See your doctor straight away if:
- you become pregnant while using EUMOVATE.
- you experience blurred vision or other visual disturbances.
- you experience any side effects.
- you notice your rash/ irritation gets better but then comes back. Don't treat the same patch more than twice without taking advice from your doctor.
- you notice your skin gets worse or it does not improve within a week.
Things you should not do
- Do not use EUMOVATE to treat any other complaints unless your doctor or pharmacist tells you to.
- Do not give your medicine to anyone else, even if they have the same condition as you.
- Do not use for more than 7 days except on advice of a doctor.
EUMOVATE is meant to control skin conditions that improve within a week of treating yourself. If you think you need further treatment after that, see a doctor for advice. Do not keep on using it.
Other skin conditions
It is possible that you are treating the wrong skin condition or that your skin may still be reacting to something that it is coming in contact with. Some of the common triggers are:
- earrings or studs – especially gold-plated earrings.
- other jewellery.
- coins.
- watch buckles, metal straps or the metal back of a watch.
- metal studs or fastenings on jeans, bras or underwear.
All of these may have a metal in them called nickel that is a very common trigger. If you react badly to nickel, all of the triggers in the list could be a problem.
Other common triggers include rubber and pine tree sap, which are used in things we touch every day. You might find triggers:
- in the home: such as plasters, furniture polish, varnishes, rubber gloves or elastic in clothes.
- in substances you use at work: like glues, oils, lubricants or cement.
- in the garden: certain plants and weeds, gardening gloves.
Even if it is not practical to avoid triggers, there are often practical steps you can take to minimise possible irritation.
If your skin condition clears up in less than a week, stop using the cream. Think about emollient (moisturising) products to help stop it coming back.
Skin specialists often advise people with eczema or dermatitis to use emollient (or moisturising) skin products, including creams and bath oils, to keep moisture in the skin. This can make your skin more resistant to flare ups. Avoid using soap and heavily scented products. Ask your doctor or pharmacist for further information.
Looking after your medicine
Keep your cream in the pack until it is time to use it.
Keep your medicine in a cool dry place where the temperature stays below 25°C.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
Do not use this medicine if the packaging is torn or shows signs of tampering.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
If your skin gets worse during treatment, you may have a skin infection or a trigger you have not recognised or even an allergy to the cream. Stop using EUMOVATE and see a doctor or pharmacist as soon as possible.
Using more than the correct amount of cream may cause changes to your skin e.g. thinning or allow the active ingredient to pass through the skin and affect other parts of the body. This is not a problem if you use the right amount of cream and for the correct period of time.
Some side effects may show up in blood tests or when your doctor gives you a medical examination.
Do not be alarmed by the following list of side effects. You may not experience any of them.
Less serious side effects
| Less serious side effects | What to do |
Skin and subcutaneous tissue disorders:
| Speak to your doctor or pharmacist if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Allergic reaction: Symptoms of an allergic reaction include some or all of the following:
Symptoms of an infection include some or all of the following:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
The following are very rare side effects and may occur if you use more than the correct amount of cream or for longer than recommended:
| Serious side effects | What to do |
Metabolism and nutrition disorders:
| Contact your doctor if you experience any of these side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is available over-the-counter without a doctor's prescription.
What EUMOVATE contains
| Active ingredient (main ingredient) | clobetasone butyrate (0.05% w/w) |
| Other ingredients (inactive ingredients) | glycerol glyceryl monostearate cetostearyl alcohol beeswax substitute 6621 arlacel 165 dimeticone 20 chlorocresol sodium citrate dihydrate citric acid monohydrate water-purified |
Do not take this medicine if you are allergic to any of these ingredients.
What EUMOVATE looks like
EUMOVATE is a smooth white cream for topical use that is available in a 15 g or 30 g tube (AUST R 178288).
A 5 g sample tube is also available from your doctor only.
Not all pack sizes may be distributed in Australia.
Who distributes EUMOVATE
GlaxoSmithKline Australia Pty Ltd
Level 4, 436 Johnston Street,
Abbotsford, Victoria, 3067
Phone: 1800 033 109
www.gsk.com.au
Trade marks are owned by or licensed to the GSK group of companies.
© 2025 GSK group of companies or its licensor.
This leaflet was prepared on 4 August 2025.
Version 12.0
Brand Information
| Brand name | Eumovate |
| Active ingredient | Clobetasone butyrate |
| Schedule | S3 |
MIMS Revision Date: 01 October 2023
1 Name of Medicine
Clobetasone butyrate.
2 Qualitative and Quantitative Composition
Eumovate cream contains 0.05% w/w clobetasone butyrate as the active ingredient.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Cream.
White cream for topical use.
4 Clinical Particulars
4.1 Therapeutic Indications
Short-term (up to 7 days) treatment of milder forms of eczema, dermatitis and other steroid responsive skin conditions.
4.2 Dose and Method of Administration
Adults and children 12 years and over. Apply a thin film and gently rub in, using only enough to cover the affected area twice daily for up to 7 days.
If the condition resolves within 7 days, treatment with Eumovate cream should be stopped.
If the condition does not improve within the first 7 days or becomes worse, the patient should see a doctor.
If after 7 days of treatment, improvement is seen but further treatment is required, the patient should see a doctor.
After application, the hands should be washed unless they are the site being treated.
Patients advised by their doctors to use this cream for prolonged periods should be advised to tell subsequent doctors about this use.
All patients should be warned against prolonged used on one area of skin, or use of excessive quantities.
All patients should also be informed that the preparation is prescribed only for a specific condition occurring in a specific individual.
Children. Use in children under 12 years only on the advice of a doctor. Children are more likely to develop local and systemic adverse reactions of topical corticosteroids and, in general, require shorter courses and less potent agents than adults.
Care should be taken when using Eumovate cream to ensure the amount applied is the minimum that provided therapeutic benefit.
Elderly. Clinical studies have not identified differences in responses between the elderly and younger patients. The greater frequency of decreased hepatic or renal function in the elderly may delay elimination if systemic absorption occurs. Therefore the minimum quantity should be used for the shortest duration to achieve the desired clinical benefit.
Renal/ hepatic impairment. In case of systemic absorption (when application is over a large surface area for a prolonged period) metabolism and elimination may be delayed therefore increasing the risk of systemic toxicity. Therefore the minimum quantity should be used for the shortest duration to achieve the desired clinical benefit.
4.3 Contraindications
Eumovate cream should not be used in patients with a history of hypersensitivity to clobetasone butyrate or to any of the excipients in the product.
The following conditions should not be treated with Eumovate cream:
Rosacea, acne, pruritus without rash, perioral dermatitis.
Untreated bacterial infections such as cellulitis, folliculitis, furunculosis or impetigo.
Fungal infections such as those associated with tinea (e.g. athletes foot, jock itch).
Viral infections including cold sores (herpes simplex), chicken pox or shingles (varicella zoster) or vaccinia.
Parasitic infestations such as scabies.
Do not use on broken or infected skin or on inflamed skin near chronic ulcers.
Topical corticosteroids inhibit wound healing processes and are contraindicated in skin ulcers, cuts and abrasions.
4.4 Special Warnings and Precautions for Use
Manifestations of hypercortisolism (Cushing's syndrome) can occur in some individuals, due to prolonged duration of use, extensive application to the skin, or because of increased systemic absorption due to use of occlusive dressings or application to broken or thin skin.
The management of eczema and dermatitis in adults and children usually requires the supervision of a doctor.
Visual disturbances have been reported with the use of systemic and topical corticosteroids as a result of increased systemic availability and direct contact with the eyes.
Consequently, if a patient presents with symptoms such as blurred vision or other visual disturbances, the patient should be considered for referral to an ophthalmologist for evaluation as possible causes may include cataract, glaucoma or rare diseases such as central serous chorioretinopathy (CSCR).
Eumovate cream should not be used for the treatment of psoriasis as this condition needs to be managed by a doctor.
Eumovate cream should not be used concomitantly with other corticosteroids (by systemic or topical routes), as this may increase the risk of unwanted effects.
For external use only. This and all medication should be kept out of the reach of children. In the case of accidental ingestion, professional assistance should be sought or the Poisons Information Centre contacted immediately (see Section 4.9 Overdose).
Systemic absorption. Eumovate cream treatment for more than a few days may lead to significant systemic absorption.
The systemic absorption of clobetasone would be expected to increase if: large amounts of Eumovate cream are used; large areas of skin are treated; treated skin is damaged or diseased; thin skin (such as on the face) or skin in intertrigenous regions is treated; the treated area is occluded.
In comparison with adults, children and infants may absorb proportionally larger amounts of topical corticosteroids and thus be more susceptible to systemic adverse effects. This is because children have an immature skin barrier and a greater surface area to body weight ratio compared with adults.
Infection. Topical corticosteroid therapy may predispose to local infection. Appropriate antimicrobial therapy should be used whenever treating inflammatory lesions which have become infected. Any spread of infection requires withdrawal of topical corticosteroid therapy and administration of appropriate antimicrobial therapy.
Skin damage. Topical corticosteroids cause atrophy of the epidermis and damage to the dermis. This may produce atrophic striae and discolouration, which are usually permanent. These are more likely to occur with prolonged therapy, occlusive dressings, application to intertrigenous areas, application to the face, and in children.
Eumovate cream should not be used on the face, groin, genitals or between the toes. As with other topical corticosteroids, it should not be used on skin with impaired circulation, such as stasis ulcers, since it may cause prolonged vasoconstriction.
Occlusive dressings. Do not use with occlusive dressings, as occlusion increases the possibility of local and systemic side effects. Infants and children are at greater risk than adults.
Use near eyes. Care should be taken to ensure that the cream does not enter the eye, as cataracts and glaucoma might result from repeated exposure.
Use in renal/hepatic impairment. In case of systemic absorption (when application is over a large surface area for a prolonged period) metabolism and elimination may be delayed therefore increasing the risk of systemic toxicity).
Use in the elderly. See Section 4.2 Dose and Method of Administration.
Paediatric use. In infants and children, long term continuous topical therapy should be avoided, since skin damage and adrenal suppression can occur even without occlusion. The least potent corticosteroid that will control the disease should be selected. In infants, the napkin may act as an occlusive dressing, and increase absorption. Corticosteroids may inhibit linear bone growth and inhibit epiphyseal maturation. Treatment should be minimised and supervised closely.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
None reported. Eumovate cream should not be used concomitantly with other topical or systemic corticosteroids, either prescribed or obtained over the counter (such as hydrocortisone) as this may increase the likelihood of drug interactions.
Coadministered drugs that can inhibit CYP3A4 (e.g. ritonavir, itraconazole) have been shown to inhibit the metabolism of corticosteroids leading to increased systemic exposure. The extent to which this interaction is clinically relevant depends on the dose and route of administration of the corticosteroids and the potency of the CYP3A4.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. There are no data in humans to evaluate the effect of topical corticosteroids on fertility.
Use in pregnancy. (Category B3)
There are limited data from the use of Eumovate cream in pregnant women. Topical administration of corticosteroids to pregnant animals can cause abnormalities of foetal development including cleft palate and intrauterine growth retardation. Studies in mice, rats and rabbits revealed similar findings following administration of clobetasone butyrate. The relevance of this finding to humans has not been established.
Administration of clobetasone during pregnancy should only be considered if the expected benefit to the mother outweighs the risk to the foetus. The minimum quantity should be used for the minimum duration.
Women who are pregnant should consult a doctor before use.
Use in lactation. The safe use of topical corticosteroids during lactation has not been established. It is not known whether the topical administration of corticosteroids could result in sufficient systemic absorption to produce detectable amounts in breast milk. Administration of clobetasone during lactation should only be considered if the expected benefit to the mother outweighs the risk to the infant.
Women who are breastfeeding should seek medical advice before using this product.
If used during lactation, clobetasone should not be applied to the breasts to avoid accidental ingestion by the infant.
4.7 Effects on Ability to Drive and Use Machines
There have been no studies to investigate the effect of clobetasone on driving performance or the ability to operate machinery. A detrimental effect on such activities would not be anticipated from the adverse reaction profile of topical clobetasone.
4.8 Adverse Effects (Undesirable Effects)
Systemic side effects are more likely in children, if large areas of the skin are treated or if large amounts are used, if treatment is prolonged or if treated areas are occluded.
The use of corticosteroids by multiple routes of administration (e.g. topical and oral or inhaled) may increase the likelihood of adverse reactions occurring.
Adverse events drug reactions (ADRs) are listed below by MedDRA system organ class and by frequency. Frequencies are defined as: very common (≥ 1/10), common (≥ 1/100 and < 1/10), uncommon (≥ 1/1000 and < 1/100), rare (≥ 1/10,000 and < 1/1000) and very rare (< 1/10,000) including isolated reports.
Infections and infestations. Very rare: opportunistic infection.
Immune system disorders. Very rare: hypersensitivity; local hypersensitivity reactions such as erythema, rash, pruritus, urticaria, local skin burning and allergic contact dermatitis may occur at the site of application and may resemble symptoms of the condition under treatment.
Patients should be advised to stop treatment if signs of hypersensitivity appear.
Endocrine disorders. Very rare: hypothalamic pituitary adrenal (HPA) axis suppression (cushingoid features (e.g. moon face, central obesity), delayed weight gain/ growth retardation in children, osteoporosis, glaucoma, hyperglycaemia/ glucosuria, cataract, hypertension, increased weight/ obesity, decreased endogenous cortisol levels).
Skin and subcutaneous tissue disorders. Rare: hypersensitivity, allergic contact dermatitis, urticaria, skin atrophy*, pigmentation changes*, exacerbation of underlying symptoms, local skin burning, hypertrichosis, rash, pruritus, erythema, hair disorders, bruising, rosacea; exacerbation of eczema and dermatitis has also been reported.
With prolonged treatment, permanent damage (including development of stria and telangiectases) to the dermis may occur.
Patients should be advised to stop treatment if signs of hypersensitivity appear.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Symptoms and signs. Topically applied clobetasone may be absorbed in sufficient amounts to produce systemic effects. Acute overdosage is very unlikely to occur. However, in the case of chronic overdosage or misuse, the features of hypercortisolism may occur (see Section 4.8 Adverse Effects (Undesirable Effects)). There is also a risk of skin atrophy with the chronic use of topical steroids.
Treatment. In the event of overdose, clobetasone should be withdrawn gradually under medical supervision because of the risk of glucocorticosteroid insufficiency.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Pharmacological studies in man and animals have shown that clobetasone butyrate has a relatively high level of topical activity accompanied by a low level of systemic activity.
The anti-inflammatory properties of clobetasone butyrate reduce the erythema and itchiness associated with eczema and dermatitis.
The cream base in Eumovate cream has moisturising properties.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Data are lacking on the systemic distribution, metabolism and excretion of clobetasone butyrate in humans.
Although pharmacokinetic studies were not carried out with the cream formulation, plasma clobetasone butyrate levels would be expected to be no greater or even lower, with the cream formulation, as creams are less occlusive than ointments.
Absorption. A single application of 30 g clobetasone butyrate 0.05% ointment to eight patients (3 with eczema and 5 with psoriasis) resulted in a small rise in plasma clobetasone butyrate levels during the first three hours not exceeding 0.6 nanogram/mL then the levels gradually decreased. The maximum plasma level reached in the first three hours was 0.6 nanogram/mL. This rise in levels was followed by a more gradual decline with plasma levels of clobetasone butyrate falling below 0.1 nanogram/mL (the lower limit of the assay) after 72 hours. The normal diurnal variation in plasma cortisol levels was not affected by the application of clobetasone butyrate ointment.
5.3 Preclinical Safety Data
In studies conducted in rats and dogs, histological changes induced by clobetasone butyrate were typical of corticosteroids (thymic involution, adrenal cortex atrophy, fatty replacement of bone marrow, lympholysis of the spleen and lymph nodes, and a reduction or disappearance of eosinophils from the endometrium). However, on a dose for dose basis, these findings were of a lesser severity than those associated with other corticosteroids.
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
The excipients are glycerol, glycerol monostearate, cetostearyl alcohol, beeswax substitute 6621, arlacel 165, dimeticone 20, chlorocresol, sodium citrate dihydrate, citric acid monohydrate, water-purified.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C.
6.5 Nature and Contents of Container
Aluminium tubes containing 15 g and 30 g.
A 5 g physician's sample pack is also available.
Not all pack sizes may be distributed in Australia.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Clobetasone butyrate is a corticosteroid used topically for its glucocorticoid effects. Its chemical name is 21-chloro-9α- fluoro-17α-hydroxy-16β- methylpregna-1,4-diene- 3,11,20-trione 17-butyrate.
Molecular formula: C26H32ClFO5.
Molecular weight: 479.0.
Chemical structure.
7 Medicine Schedule (Poisons Standard)
Pharmacist Only Medicine.
Date of First Approval
02 March 2005
Date of Revision
03 May 2023
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.