Fluad
Brand Information
| Brand name | Fluad |
| Active ingredient | Influenza virus geriatric vaccine, surface antigen, trivalent (inactivated) |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Fluad.
Summary CMI
Fluad®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor, nurse or pharmacist.
1. Why am I being given Fluad®?
Fluad® contains the active ingredient influenza virus haemagglutinin. Fluad® is a vaccine used to help prevent certain types of influenza or “flu”. It is for use in people aged 50 years and older only.
For more information, see Section 1. Why am I being given Fluad®? in the full CMI.
2. What should I know before being given Fluad®?
Tell your doctor, nurse or pharmacist if you have ever had an allergic reaction to any influenza vaccine or any of the ingredients listed in Section 7. Product details.
Talk to your doctor, nurse or pharmacist if you have any other medical conditions or take any other medicines.
For more information, see Section 2. What should I know before being given Fluad®? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Fluad® and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How is Fluad® given?
Fluad® is given as an injection, usually into your upper arm muscle by a doctor, nurse or pharmacist.
Fluad® is given once every year, one injection of 0.5 mL.
More instructions can be found in Section 4. How is Fluad® given? in the full CMI.
5. What should I know about being given Fluad®?
| Things you should do | Call your doctor, nurse or pharmacist straight away if you do not feel well after having Fluad®. Keep an updated record of your vaccinations. Keep any follow-up appointments with your doctor or clinic. |
For more information, see Section 5. What should I know about being given Fluad®? in the full CMI.
6. Are there any side effects?
Common side effects may include local reaction around the injection site, fatigue and headache. Mostly these are mild and usually disappear within 3 days without treatment. Serious side effects are rare.
Sudden signs of allergy may be a serious side effect. If you experience this, tell your doctor immediately. Typical symptoms include rash, itching or hives on the skin, swelling of the face, lips, tongue or other parts of the body.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Fluad®
Active ingredient(s): influenza virus haemagglutinin
Consumer Medicine Information (CMI)
This leaflet provides important information about using Fluad®. You should also speak to your doctor, nurse or pharmacist if you would like further information or if you have any concerns or questions about using Fluad®.
Where to find information in this leaflet:
1. Why am I being given Fluad®?
2. What should I know before being given Fluad®?
3. What if I am taking other medicines?
4. How is Fluad® given?
5. What should I know about being given Fluad®?
6. Are there any side effects?
7. Product details
1. Why am I being given Fluad®?
Fluad® contains the active ingredient influenza virus haemagglutinin.
Fluad® is an adjuvanted vaccine used to help prevent certain types of influenza or “flu”. It is for use in people aged 50 years and older only.
Influenza is a sudden respiratory infection caused by the ‘flu’ virus. It is very contagious. It is usually spread from one person to another by small droplets from coughs and sneezes. The virus enters the nose or throat and may spread to the lungs. Influenza infection can lead to serious illness.
Vaccination with Fluad® helps prevent infection with influenza and to control the spread of the disease. Influenza strains can change every year therefore previous influenza vaccinations are unlikely to give you protection against the current strains most common this year. You will therefore need to receive a vaccine against flu every year to lower the chance of catching the ‘flu’, especially if you are at risk. Influenza vaccination is particularly recommended for people aged 65 years and older.
Fluad® works by causing your body to produce its own protection against the three types of influenza virus from which it is made. It does this by making substances called antibodies in the blood that fight the influenza virus. Your body usually takes 2-3 weeks after vaccination to develop protection against influenza. Most people will produce enough antibodies against the influenza virus. However, as with all vaccines, 100% protection cannot be guaranteed.
An influenza vaccine will only protect you against the types of influenza virus used to make it. It will not protect you from other types of influenza virus or from infections with other agents causing flu-like symptoms (such as the common cold).
The virus in Fluad® has been killed therefore the vaccine cannot ‘give’ you the flu.
2. What should I know before being given Fluad®?
Warnings
Do not use Fluad® if:
- You are under 50 years old. Fluad® is for use in persons 50 years and older only.
- You are severely allergic to any of the ingredients listed at the end of this leaflet or any previous influenza vaccine. Symptoms of a severe allergic reaction may include shortness of breath, wheezing or difficulty breathing, swelling of the face, lips, tongue or other parts of the body or skin rash, itching or hives.
- Always check the ingredients to make sure you can use this medicine.
Tell your doctor, nurse or pharmacist if you:
have reacted to previous vaccination with any of the following:
- severe allergic reaction (anaphylaxis),
- difficulty breathing,
- swelling of the throat,
- fainting or collapse,
- fits or convulsions,
- severe skin reaction at the injection site, including severe bruising.
have or have had any other medical conditions, especially the following:
- Guillain-Barré Syndrome (an illness affecting nerves resulting in weakness of muscles),
- lowered immunity due to diseases such as some blood disorders, malaria, kidney disease requiring dialysis, HIV/AIDS or cancer
- lowered immunity due to treatment with medicines such as corticosteroids, ciclosporin or other medicines used to treat cancer (including radiation therapy)
- have an infection or high temperature (greater than 40°C)
- take any medicines for any other condition
have allergy to
- any other medicines,
- eggs, or egg proteins
- or any other substances, such as latex, food, preservative or dyes.
Your doctor, nurse or pharmacist will decide if you can receive Fluad®. A minor illness such as a cold should not be a problem but talk to your doctor about this if being vaccinated.
After vaccination, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Fluad® is indicated for people aged 50 years and older only.
3. What if I am taking other medicines?
Tell your doctor, nurse or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Fluad® and affect how it works. These include medicines which lower the immune system, such as corticosteroids, cyclosporin or other medicines used to treat cancer (including radiation therapy).
Your doctor, nurse or pharmacist will consider whether adjustment of your medication is necessary.
Tell your doctor, nurse or pharmacist if you have had any vaccines in the last 4 weeks. Fluad® can be given at the same time as other vaccines.
Check with your doctor, nurse or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Fluad®.
4. How is Fluad® given?
How much is given
Fluad® is given as a single 0.5 mL dose.
When Fluad® is given
Fluad® is usually given before the start of the influenza season or when recommended by your doctor, nurse or pharmacist.
Vaccination for influenza should be repeated every year as new types of influenza virus can appear each year.
How is Fluad® given
It is given as an injection, usually into your upper arm muscle by a doctor, nurse or pharmacist.
5. What should I know about being given Fluad®?
Things you should do
Call your doctor, nurse or pharmacist straight away if you do not feel well after having Fluad®.
Keep an updated record of your vaccinations.
Keep any follow-up appointments with your doctor or clinic.
Driving or using machines
Do not drive or use machines if you are feeling unwell after vaccination. Wait until any effects of the vaccine have worn off before you drive or use machines.
Looking after your medicine
Fluad® is usually stored in the doctor's surgery or clinic, or at the pharmacy. However, if you need to store Fluad®:
- Keep it where young children cannot reach it.
- Keep Fluad® in the original pack until it is time for it to be given.
- Keep it in the refrigerator, between 2°C and 8°C. Do not freeze Fluad®. Protect from light. Discard if the vaccine has been frozen. Freezing destroys the vaccine.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal. A doctor, nurse or pharmacist will dispose of any unused vaccine.
Do not use Fluad® after the expiry date.
6. Are there any side effects?
All vaccines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor, nurse or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. Mostly these are mild and usually disappear within 3 days without treatment. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. All of these serious side effects are rare. |
Tell your doctor, nurse or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this vaccine.
7. Product details
Fluad® is a prescription only medicine. It is available from your doctor or pharmacist.
What Fluad® contains
| Active ingredient (main ingredient) | Influenza virus haemagglutinin Each 0.5 mL contains 15 micrograms from each of the following types of influenza virus:
These strains have been recommended by the Australian Influenza Vaccine Committee for the 2025 Southern Hemisphere winter. |
| Other ingredients (inactive ingredients) | Adjuvant: MF59C.1: containing squalene (of fish origin), polysorbate 80, sorbitan trioleate, sodium citrate dihydrate, citric acid monohydrate, water for injections. Other: sodium chloride, potassium chloride, monobasic potassium phosphate, dibasic sodium phosphate dihydrate, magnesium chloride hexahydrate, calcium chloride dihydrate, water for injections. May also contain traces of kanamycin sulfate, neomycin sulfate, egg proteins (ovalbumin), formaldehyde, cetrimonium bromide, sucrose, and hydrocortisone as residues of the manufacturing process. |
Tell your doctor nurse, or pharmacist if you have an allergy to any of the above ingredients. Your doctor, nurse or pharmacist will decide if you can receive Fluad®.
Fluad® does not contain lactose, gluten, tartrazine, any azo dyes, or preservative.
What Fluad® looks like
Fluad® is a milky-white liquid, in glass pre-filled syringe with or without a needle for single use only (AUST R 90339 and AUST R 306718).
Who distributes Fluad®
Seqirus Pty Ltd
ABN 26 160 735 035
63 Poplar Road
Parkville, Victoria 3052
AUSTRALIA
This leaflet was prepared in October 2025.
Fluad® is a trademark of Seqirus UK Limited or its affiliates.
Brand Information
| Brand name | Fluad |
| Active ingredient | Influenza virus geriatric vaccine, surface antigen, trivalent (inactivated) |
| Schedule | S4 |
MIMS Revision Date: 01 March 2026
1 Name of Medicine
Fluad. Inactivated influenza vaccine (surface antigen), adjuvanted, suspension for injection; containing Influenza virus haemagglutinin as active ingredient.
2 Qualitative and Quantitative Composition
Each 0.5 mL dose contains influenza virus surface antigens (haemagglutinin and neuraminidase) of each of three strains representative of the influenza virus types expected to circulate in the Southern Hemisphere winter according to WHO recommendations for the 2026 season:
A/Missouri/11/2025 (H1N1)pdm09-like virus (A/Switzerland/6849/2025 IVR-278): 15 micrograms HA*;
A/Singapore/GP20238/2024 (H3N2)-like virus (A/Singapore/GP20238/2024 IVR-277): 15 micrograms HA*;
B/Austria/1359417/2021-like virus (B/Austria/1359417/2021 BVR-26): 15 micrograms HA* per 0.5 mL dose.
*HA = haemagglutinin.
Fluad vaccine is prepared from virus grown in embryonated hens' eggs and inactivated with formaldehyde before purification and combination with MF59C.1, an adjuvant known to increase the immunogenicity of vaccines. MF59C.1 adjuvant is a squalene based oil-in-water emulsion. Squalene is of fish origin. It is also a normal component in the human body and is easily metabolized and excreted. For a full list of excipients, see Section 6.1 List of Excipients.
The type and amount of viral antigens in Fluad conform to the requirements of the Australian Influenza Vaccine Committee for the 2026 Southern Hemisphere Influenza season. The strains chosen for vaccine manufacture are endorsed by the Australian Influenza Vaccine Committee as being antigenically equivalent to the reference virus.
Fluad is manufactured in eggs and trace amounts of kanamycin sulfate, neomycin sulfate, ovalbumin (≤ 1 microgram/0.5 mL dose), formaldehyde (≤ 1 microgram/0.5 mL dose), cetrimonium bromide (≤ 18 micrograms/0.5 mL dose), sucrose and hydrocortisone may be present as residues of the manufacturing process.
3 Pharmaceutical Form
Fluad is a milky-white suspension for injection.
4 Clinical Particulars
4.1 Therapeutic Indications
Active immunisation against influenza in persons 50 years of age and older.
For full details regarding recommendations for influenza vaccination, please refer to the relevant national immunisation guidelines.
4.2 Dose and Method of Administration
Fluad is for use in adults 50 years of age and older only. See Section 4.1 Therapeutic Indications.
A single 0.5 mL dose should be administered by intramuscular injection, preferably into the deltoid muscle of the upper arm.
Gently shake before use. After shaking, the normal appearance of the vaccine is a milky-white suspension.
Visually inspect the contents of each pre-filled syringe for particulate matter and/or variation in appearance prior to administration. If either condition is observed, do not administer the vaccine.
Fluad contains no antimicrobial preservative. Each pre-filled syringe is for use in one patient on one occasion only. Discard any residue.
Annual revaccination is recommended because immunity declines during the year after vaccination and circulating strains of influenza virus change from year to year.
Persons with a history of egg allergy (non-anaphylaxis) can receive a full dose of vaccine in any immunisation setting (also see Section 4.4 Special Warnings and Precautions for Use).
4.3 Contraindications
The vaccine is contraindicated in individuals with known severe allergic reactions (e.g. anaphylaxis) to:
any component of the vaccine (see Section 6.1 List of Excipients), except egg proteins (also see Section 4.4 Special Warnings and Precautions for Use); or
a previous dose of any influenza vaccine.
4.4 Special Warnings and Precautions for Use
As with all injectable vaccines, appropriate medical treatment and supervision should always be readily available in case an anaphylactic event occurs following the administration of the vaccine.
Persons with a history of anaphylaxis to egg should be vaccinated only in medical facilities with staff experienced in recognising and treating anaphylaxis. For full details regarding recommendations for influenza vaccination in individuals with egg allergy, please refer to the relevant national immunisation guidelines.
Immunisation should be postponed in patients with acute febrile illness until the fever is resolved.
Antibody response in patients with endogenous or iatrogenic immunosuppression may be insufficient.
A protective response may not be elicited in all vaccine recipients.
If Guillain-Barré syndrome has occurred within 6 weeks of previous influenza vaccination, the decision to give Fluad should be based on careful consideration of the potential benefits and risks.
The syringe and all associated syringe components for Fluad AUST R 306718 pre-filled syringe needle-free do not contain natural rubber latex.
Fluad AUST R 90339 pre-filled syringe with attached needle cannot be considered to be latex-free as the sheath covering the needle contains natural rubber latex, refer to statement on carton. See Section 6.5 Nature and Contents of Container for further information.
Use in the elderly. Fluad is approved for active immunisation against influenza in persons 50 years of age and older. See Section 4.1 Therapeutic Indications; Section 5 Pharmacological Properties.
Paediatric use. Paediatric data have not been evaluated.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Data from two studies on the concomitant administration of Fluad with an approved 13-valent pneumococcal conjugate vaccine (PCV13) and an approved 23-valent pneumococcal polysaccharide vaccine (PPSV23) in an elderly population are available. These studies indicated that coadministration of Fluad with either PCV-13 or PPSV23 did not show significant interference in antibody response. Although concomitant vaccination induced more frequent local pain, most of the local adverse reactions were mild. Systemic adverse reactions were generally mild, and no serious vaccine-related adverse events occurred.
If Fluad needs to be used at the same time as another vaccine, immunisation should be given at separate injection sites, preferably on different limbs. It should be noted that the adverse reactions may be intensified.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category B2)
In a reproductive and developmental study in rabbits dosed with Fluad twice pre-mating (21 and 7 days before mating) and during gestation (gestation day 7 and 20), there were no significant effects on the does, their fetuses or pups. The HA dose in rabbits was approximately 11-times the recommended clinical dose of a HA dose per body weight basis. Circulating anti-H1N1 antibodies were detected in the does, fetuses and pups.
There are no adequate and well-controlled studies in pregnant women. Fluad is indicated for persons 50 years and over.
Use in lactation. No data available.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
The overall safety profile of Fluad is similar to the adjuvanted quadrivalent influenza vaccine, Fluad Quad. Data for Fluad Quad are relevant to Fluad because both vaccines are manufactured using the same process and have overlapping compositions.
Clinical trials. Because clinical trials are conducted under widely varying conditions, adverse event rates observed in the clinical trial(s) of a vaccine cannot be directly compared to rates in the clinical trial(s) of another vaccine and may not reflect the rates observed in practice.
Adult and elderly population, 50 years of age and older. Adults 50 to less than 65 years of age. The safety of Fluad Quad in adult subjects 50 to less than 65 years of age was evaluated in clinical study V118_23. This was a randomised observer-blind, controlled, multi-centre study conducted during the 2021-2022 Northern Hemisphere influenza season. In this study, subjects received Fluad Quad (N=1027) or a non-adjuvanted comparator quadrivalent influenza vaccine (N=1016).
Solicited local and systemic reactions were collected for 7 days after vaccination. The majority of solicited reactions were reported as mild or moderate in intensity and resolved within 3 days.
Commonly reported (≥ 10%) adverse reactions in adults 50 to less than 65 years of age who received Fluad Quad were injection site pain (47.1%), fatigue (29.5%), headache (22.2%), arthralgia (13.7%) and myalgia (13.0%).
The frequency of all solicited local and systemic adverse reactions reported in Study V118_23 is presented in Table 1.
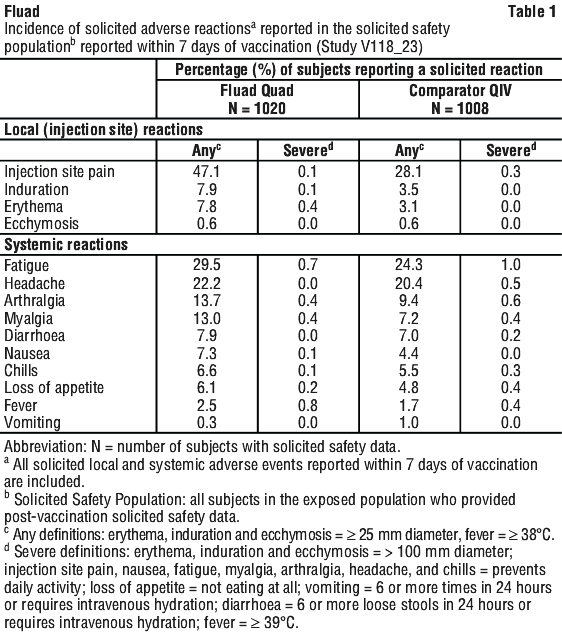
Serious adverse events (SAEs), AEs leading to withdrawal and adverse events of special interest (AESIs) were collected up to Day 271. There were no SAEs, AEs leading to withdrawal, AESIs or deaths in this study that were related to Fluad Quad.
Elderly population 65 years of age and older. The safety of a single dose of Fluad Quad in subjects 65 years of age and older was evaluated in clinical study V118_20. See Section 5.1 Pharmacodynamic Properties for further details.
In study V118_20, adverse events were collected as either solicited or unsolicited AEs. Solicited local and systemic events were collected for 7 days after vaccination (Table 2). Unsolicited AEs were collected for 21 days following vaccination, and for the full duration of study participation for SAEs, AEs leading to withdrawal from the study, new onset of chronic diseases (NOCDs), AESIs.
In the study, 51.8% of subjects reported any solicited AE after Fluad Quad vaccination, compared with 48.7%, and 48.2% in the Fluad and aTIV-2 groups respectively. The most commonly reported (≥ 10%) solicited AE's were injection site pain (31.9%), fatigue (16%) and headache (12.0%) (see Table 2). The majority of the adverse events reported were mild or moderate in intensity and resolved within 3 days. Solicited AEs with severe intensity were uncommon in all study groups.
Overall, the solicited AEs show that the safety profile of Fluad Quad in subjects 65 years of age and older was generally similar compared to the aTIV comparators.
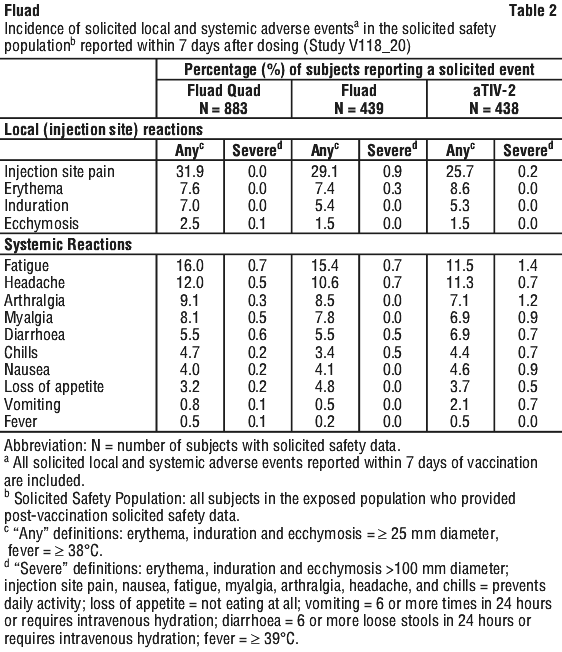
No treatment-related SAE or death were reported in the study.
Two AESIs were reported during the study: one in the Fluad group, and one in the Fluad Quad group. Neither of the AESIs was considered to be related to study vaccine.
The frequency of unsolicited events leading to NOCD was similar across study groups: Fluad Quad (2.6%); Fluad (3.6%) and aTIV-2 (3.2%). NOCDs were heterogeneous in nature and consistent with the clinical conditions spontaneously occurring in subjects 65 years of age and older. No reported NOCDs were considered related to study vaccine.
No unsolicited AEs led to withdrawal from the study.
Post-marketing surveillance. In addition to the adverse reactions observed during clinical trials, the following adverse events were reported from post-marketing surveillance for Fluad Quad or for Fluad, which is relevant because both vaccines are manufactured using the same process and have overlapping compositions.
As these events were reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency or to establish, for all events, a causal relationship to vaccine exposure.
Blood and lymphatic system disorders. Thrombocytopenia (some very rare cases were severe with platelet counts less than 5,000 per mm3), lymphadenopathy.
General disorders and administration site conditions. Extensive swelling of injected limb, injection site swelling, peripheral swelling, injection-site cellulitis-like reaction, asthenia, malaise, pyrexia.
Immune system disorders. Allergic or immediate hypersensitivity reactions, including anaphylactic shock (in rare cases), anaphylaxis.
Musculoskeletal and connective tissue disorders. Muscular weakness, pain in extremity.
Nervous system disorders. Encephalomyelitis, Guillain-Barré Syndrome, convulsions, neuritis, neuralgia, paraesthesia, syncope, presyncope, dizziness.
Skin and subcutaneous tissue disorders. Generalised skin reactions including erythema multiforme, urticaria, pruritus or non-specific rash, erythema, angioedema.
Vascular disorders. Vasculitis which may be associated with transient renal involvement.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems (Australia).
4.9 Overdose
There are no data on overdose with Fluad.
For information on the management of overdose, contact the Poison Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Fluad provides active immunisation against three influenza virus strains (two A subtypes and one B type) contained in the vaccine.
Fluad has been shown to evoke antibody responses to the viral surface glycoproteins, haemagglutinin and neuraminidase. These antibodies provide protection against clinical illness in a high proportion of vaccine recipients.
The antibody response to Fluad Quad is similar to Fluad which is increased when compared to the response to vaccines without adjuvant, and is most pronounced for A/H3N2 influenza antigens. This increased response is even more pronounced in subjects 65 years of age and older.
The adjuvant MF59 broadens the overall immune response allowing the vaccine to offer greater protection against heterologous strains of the virus. This may be important when there is a mismatch between the virus strains included in the vaccine and the strains circulating in the community. The antibody response is increased when compared to the response to non-adjuvanted Inactivated Influenza Vaccine. This increased response is seen particularly in elderly subjects with low pre-immunisation titres and/or with underlying diseases (diabetes, cardiovascular and respiratory diseases) who are at increased risk of complications of influenza infection.
Specific levels of haemagglutination inhibition (HI) antibody titres post-vaccination with inactivated influenza vaccine have not been correlated with protection from influenza virus. In some human studies in adults, antibody titres of 1:40 or greater have been associated with protection from influenza illness in up to 50% of subjects.
Antibody against a specific influenza virus type or subtype confers limited or no protection against another. Furthermore, antibody to a specific antigenic variant of influenza virus might not protect against a new antigenic variant of the same type or subtype.
The addition of the squalene-based MF59 oil-in-water emulsion adjuvant in Fluad leads to enhanced antigen uptake by recruiting immune cells at the injection site and differentiating into antigen presenting cells. This results in an increased magnitude, breadth and persistence of the immune response through the duration of the influenza season compared with non-adjuvanted influenza vaccines.
Annual revaccination is recommended because immunity declines during the year after vaccination and circulating strains of influenza virus change from year to year.
Clinical trials. Data for Fluad Quad are relevant to Fluad because both vaccines are manufactured using the same process and have overlapping compositions.
Immunogenicity. Adults 50 to less than 65 years of age. Immunogenicity of Fluad Quad in adults 50 to less than 65 years of age was evaluated in Study V118_23. This was a randomised, observer-blind, controlled, multi-centre, trial conducted in the US, Germany, and Estonia, during the 2021-22 Northern Hemisphere season. In the study, adults 50 to less than 65 years of age who were healthy or had comorbidities that increased their risk of hospitalisation for influenza-associated complications, were enrolled to receive one dose of either Fluad Quad (N=1027) or a non-adjuvanted quadrivalent comparator influenza vaccine (Comparator QIV; Fluarix Quadrivalent) (N=1017). The mean age of subjects enrolled in the Fluad Quad group was 57.8 years and females represented 62% of subjects.
The primary endpoints to assess noninferiority were haemagglutination inhibition (HI) geometric mean antibody titre (GMT) and seroconversion rates (SCR) 21 days after vaccination. Fluad Quad met the noninferiority criteria for all 4 strains (Table 3).
A superior immune response was assessed based on post-vaccination GMTs with a prespecified superiority criteria that the upper limit of the 95% CI for the inter-group GMT ratio (Comparator QIV/Fluad QIV) be < 1. A/H1N1 and A/H3N2 met the superiority criteria. The B strains did not meet the pre-specified criteria for immunological superiority.
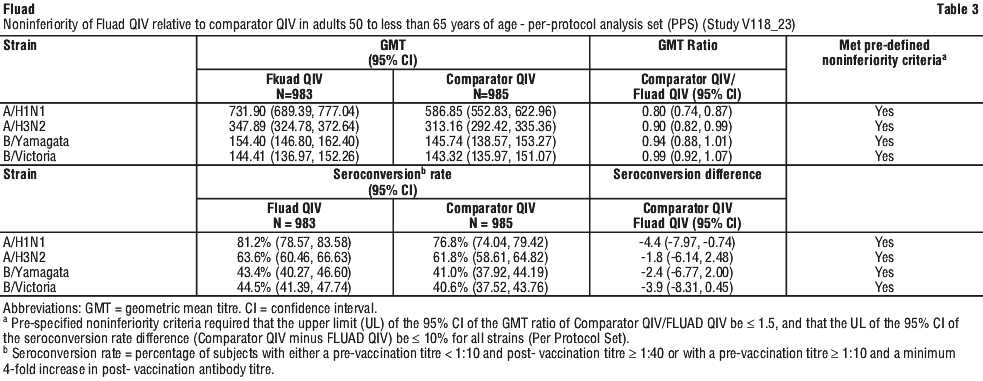
The per protocol immunogenicity set included a total of 1741 subjects: Fluad Quad (N=872), Fluad (N=436) and aTIV-2 (N=433). In the per protocol set, the mean age of subjects at enrolment who received Fluad Quad was 72.4 years.
Non-inferiority of the immune response of Fluad Quad to that of Fluad (aTIV-1) and TIV-2 among adults 65 years of age or older was assessed as a co-primary endpoint.
Adjusted HI Geometric Mean Titre (GMT) ratios and the difference in seroconversion rates for each vaccine strain were assessed 21 days after vaccination. Pre-specified non-inferiority criteria required that the upper bound of the 2-sided 95% CI of the GMT ratio (GMTaTIV/GMTFluad Quad) did not exceed 1.5 and the upper bound of the 2-sided 95% CI of the seroconversion rate difference (SCRaTIV-SCRFluad Quad) did not exceed 10% for each strain.
Fluad Quad was non-inferior for all 4 influenza strains for both HI antibody titres and seroconversion rates (Table 4).
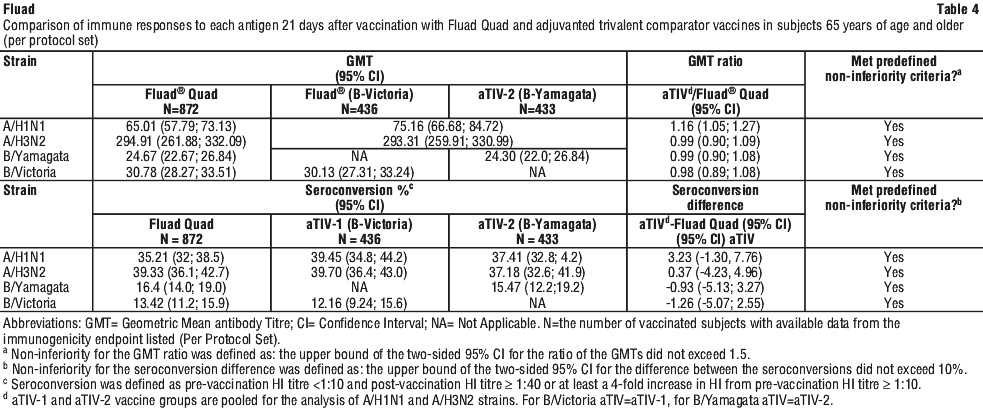
The second co-primary objective was met for A strains (H1N1 and H3N2), but not for B strains (B-Yamagata and B-Victoria). Results for B strains in the Fluad and aTIV-2 groups were similar to those obtained for Fluad Quad.
5.2 Pharmacokinetic Properties
Not applicable.
5.3 Preclinical Safety Data
Non-clinical data reveal no special hazard for humans based on conventional studies of repeated-dose toxicity (60 microgram HA/dose), local tolerance and sensitization (45 microgram HA/dose). For the reproductive and development toxicity see Section 4.6 Fertility, Pregnancy and Lactation.
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Each 0.5 mL dose of Fluad contains MF59C.1 (a proprietary adjuvant): containing squalene (of fish origin) 9.75 mg, polysorbate 80 1.175 mg, sorbitan trioleate 1.175 mg, sodium citrate dihydrate 0.66 mg, citric acid monohydrate 0.04 mg and water for injections and the following excipients. See Table 5.

6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store at 2°C to 8°C (Refrigerate. Do not freeze). Discard if the vaccine has been frozen. Protect from light.
6.5 Nature and Contents of Container
Not all presentations or pack sizes may be marketed.
AUST R 306718. Fluad, inactivated, trivalent influenza vaccine (surface antigen) adjuvanted, suspension for injection, 0.5 mL pre-filled syringe, needle-free (AUST R 306718) is a 0.5 mL suspension for injection in a needle-free pre-filled syringe (type I glass).
The syringe barrel, plunger and rubber stopper are not manufactured with natural rubber latex.
Pack sizes: 1's; 10's.
AUST R 90399. Fluad, inactivated, trivalent influenza vaccine (surface antigen), adjuvanted, suspension for injection, 0.5 mL pre-filled syringe, with attached needle (AUST R 90339) is a 0.5 mL suspension for injection in a pre-filled syringe (type I glass) with attached needle.
The sheath covering the needle contains natural rubber latex, refer to statement on carton (see Section 4.4 Special Warnings and Precautions for Use).
The syringe barrel, plunger and rubber stopper are not manufactured with natural rubber latex.
Pack sizes: 1's; 10's.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Not applicable.
7 Medicine Schedule (Poisons Standard)
Schedule 4 (Prescription Only Medicine).
Date of First Approval
15 October 2002
Date of Revision
16 October 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.