FluMist
Brand Information
| Brand name | FluMist |
| Active ingredient | Influenza virus vaccine, trivalent (live attenuated) |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the FluMist.
Summary CMI
FLUMIST®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this vaccine, speak to your healthcare professional (doctor, nurse or pharmacist).
1. Why am I/my child being given FLUMIST?
FLUMIST contains three different types (strains) of influenza virus – two Type A (H1N1 and H3N2) and one Type B (Victoria lineage) strain. It is a vaccine used to help prevent certain types of influenza (flu).
FLUMIST may be given to a child (including adolescents) aged 24 months (2 years) to under 18 years old.
For more information, see Section 1. Why am I/my child being given FLUMIST? in the full CMI.
2. What should I know before I am/my child is given FLUMIST?
Do not use if you/your child have ever had an allergic reaction to any medicine containing influenza virus, eggs or egg proteins (e.g. ovalbumin), gentamicin (an antibiotic), or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you/your child have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I am/my child is given FLUMIST? in the full CMI.
3. What if I am/my child is taking other medicines?
Some medicines may interfere with FLUMIST and affect how it works. Tell your healthcare professional if you/your child are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription.
A list of these medicines is in Section 3. What if I am/my child is taking other medicines? in the full CMI.
4. How is FLUMIST given?
- FLUMIST is given by a trained healthcare professional
- FLUMIST is a nasal spray not an injection. It must not be injected.
More instructions can be found in Section 4. How is FLUMIST given? in the full CMI.
5. What should I know after having FLUMIST?
| Things you should do |
|
| Looking after your vaccine | FLUMIST is usually stored in the doctor's surgery, clinic or pharmacy. However, if you need to store FLUMIST:
|
For more information, see Section 5. What should I know after having FLUMIST? in the full CMI.
6. Are there any side effects?
The following side effects may occur after you/your child have FLUMIST.
Common side effects include runny or stuffy nose, loss of appetite, generally feeling unwell, headache, fever, muscle tenderness or weakness/aches not caused by exercise, nose bleeds.
Serious side effects include sudden signs of allergy such as rash, itching or hives on the skin, swelling of face, lips, tongue or other parts of the body, shortness of breath and wheezing or difficulty breathing, feeling weak or paralysed, or generally feeling sore or tender, headache and high temperature associated with hallucinations, confusion, paralysis of part or all of the body, disturbances of behaviour, speech and eye movements, stiff neck and sensitivity to light.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
FLUMIST®
Active ingredient(s): influenza virus vaccine
Consumer Medicine Information (CMI)
This leaflet provides important information about using FLUMIST. You should also speak to your healthcare professional if you would like further information or if you have any concerns or questions about using FLUMIST.
Where to find information in this leaflet:
1. Why am I/my child being given FLUMIST?
2. What should I know before I am/my child is given FLUMIST?
3. What if I am/my child is taking other medicines?
4. How is FLUMIST given?
5. What should I know after having FLUMIST?
6. Are there any side effects?
7. Product details
1. Why am I/my child being given FLUMIST?
FLUMIST contains three different types (strains) of influenza virus – two Type A (H1N1 and H3N2) and one Type B (Victoria lineage) strain. It is a vaccine used to help prevent certain types of influenza (flu).
FLUMIST may be given to a child (including adolescents) aged 24 months (2 years) to under 18 years old.
Influenza (flu) is an infectious disease caused by different types of influenza viruses. It can spread very rapidly, particularly in homes and at kindergartens/preschools and schools. The most common symptoms include fever, headache, general aches and pains, sore throat, runny nose, coughing, weakness and tiredness.
Vaccination for the flu helps to prevent infection as well as controlling the spread of flu within the community including to the very young, the elderly and other people at risk.
New types of viruses can appear each year. Every year the World Health Organization (WHO) and the Australian Influenza Vaccine Committee (AIVC) decide which virus types should be in flu vaccines for the next flu season in Australia (typically from May to October). This means that the viruses in the vaccines usually change every year, so previous season flu vaccines are unlikely to give protection from the flu in the next season. It is recommended that flu vaccines be given every year, particularly for people who are at risk or are in contact with others at risk.
Like all vaccines, FLUMIST may not fully protect all people who are vaccinated. It will only help to prevent flu if it is caused by one of the strains in the vaccine or another strain which is closely related to one in the vaccine.
FLUMIST will not help prevent the flu if it is caused by another strain of virus or if you already have the virus prior to vaccination. It also will not help prevent other diseases that have flu-like symptoms (e.g. the common cold).
FLUMIST will not give you/your child the flu.
How flu vaccines work
Flu vaccines (injections and nasal sprays) work by helping the body to produce its own protection against the flu. They do this by making substances called antibodies to fight flu viruses. If a vaccinated person comes into contact with a flu virus the body usually destroys it.
The body usually takes several weeks after vaccination to develop protection against flu viruses.
Your healthcare professional can recommend the best time for you/your child to be vaccinated for the coming flu season.
2. What should I know before I am/my child is given FLUMIST?
Warnings
Do not use FLUMIST if you/your child:
- are allergic to any vaccines containing influenza virus, eggs or egg proteins (e.g. ovalbumin), gentamicin (an antibiotic), or any of the ingredients listed at the end of this leaflet. Always check the ingredients to make sure you can use this vaccine.
- have a blood disorder or a cancer that affects the immune system; or you/your child have been told by your doctor that you have a weakened immune system as a result of a disease, medicine or other treatment.
- have been taking, or have recently been told to take aspirin (a substance in many medicines used to relieve pain and lower fever).
- are less than 24 months (2 years) old.
- have Guillain-Barre Syndrome-a rare condition where the body's immune system attacks the nerves, causing weakness and tingling in your arms and legs that can get worse over time.
Check with your healthcare professional if you/your child:
- have allergies to any other medicines, foods, preservatives or dyes.
- have or have had asthma/wheezing
- may need to be in contact with anyone who has a weakened immune system (e.g. a parent/grandparent undergoing cancer treatment, or someone in isolation following a bone marrow transplant).
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your/your child's healthcare professional if you/your child are pregnant or intend to become pregnant.
Talk to your/your child's healthcare professional if you/your child are breastfeeding or intend to breastfeed.
Your healthcare professional can discuss with you/your child the risks and benefits involved.
3. What if I am/my child is taking other medicines?
Tell your healthcare professional if you/your child are taking any other medicines or vaccines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
You/your child should not be given aspirin (a substance in many medicines used to relieve pain and lower fever) for at least 4 weeks after vaccination with FLUMIST, unless your healthcare professional tells you otherwise.
Some medicines may interfere with FLUMIST and affect how it works. These include:
- medicines which affect the body's immune response such as corticosteroids (e.g. prednisolone), some cancer treatments (chemotherapy) or radiotherapy.
- flu-specific antiviral medicines such as oseltamivir and zanamivir
These medicines may be affected by FLUMIST or may affect how well it works. Your healthcare professional will advise whether or not you/your child should have the vaccine.
Check with your healthcare professional if you are not sure about what medicines, vitamins or supplements you/your child are taking and if these affect FLUMIST.
Your healthcare professional have more information on medicines to be careful with or avoid while taking this vaccine.
Having other vaccines
FLUMIST may be given at the same time as other vaccines (e.g. for measles, mumps, rubella etc).
4. How is FLUMIST given?
FLUMIST is given by a trained healthcare professional (e.g. doctor or nurse). Unlike other flu vaccines, it is a nasal spray not an injection.
FLUMIST must not be injected.
How much is given and how will it be given
Children (including adolescents) aged 24 months to less than 18 years of age are given one single nasal applicator (200 µL) of FLUMIST.
This is administered by the healthcare professional as one spray (half the applicator; 100 µL) in one nostril. A clip is then removed and the other half of the applicator (100 µL) is sprayed into the other nostril.
You/your child can breathe normally when it is given - there is no need to sniff in.
Some children who have not previously had FLUMIST may need a second vaccination a month later. Please ask your healthcare professional if this includes you/your child and when/how to arrange for another visit if required.
If you have any concerns, ask your healthcare professional.
When will it be given
FLUMIST is usually given annually before the start of each flu season.
If you forget to get FLUMIST
If you forget to get your/your child's annual vaccination or your child's second vaccination (if advised by your doctor), talk to your doctor or nurse to arrange a visit as soon as possible.
If you have trouble remembering to arrange for an annual vaccination, ask your healthcare professional for some hints.
If too much FLUMIST is given
Overdose is unlikely as your healthcare professional gives you/your child the nasal spray and these are supplied as single-use nasal applicators.
If you think that you/your child have been given too much FLUMIST, you/your child may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know after having FLUMIST?
Things you/your child should do
Keep an updated record of you/your child's vaccinations.
Keep any follow-up appointments with your/your child's doctor or clinic.
Things to be careful of
Wherever possible, the person who has been vaccinated should avoid close contact with anyone who has a weakened immune system (e.g. parent/grandparent undergoing cancer treatment, or someone in isolation following a bone marrow transplant) for 1-2 weeks following vaccination.
Looking after your vaccine
FLUMIST is usually stored in the doctor's surgery, clinic or pharmacy. However if you need to store FLUMIST:
- Keep FLUMIST in the original pack until it is time for it to be given.
- Keep it in the refrigerator between 2°C and 8°C.
Protect from light. Do not freeze – freezing destroys the vaccine. - Before use, the vaccine may be taken out of the refrigerator once for a maximum period of 12 hours at a temperature not above 25°C. If the vaccine has not been used after this 12 hour period, it should be discarded.
Keep it where young children cannot reach it.
Getting rid of any unwanted vaccine
FLUMIST is usually stored and disposed of at the doctor's surgery, clinic or pharmacy. However if you need to dispose of either full or empty FLUMIST nasal applicators you should return these to the doctor's surgery, clinic or pharmacy for appropriate vaccine disposal. No medicines, including vaccines, should ever be disposed of via wastewater or household waste.
FLUMIST should not be given after the expiry date.
6. Are there any side effects?
All medicines, including vaccines, can have side effects. If you/your child do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
Tell your healthcare professional as soon as possible if you/your child feel unwell after having the vaccine.
See the information below and, if you need to, ask your healthcare professional if you/your child have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you/your child have any of these less serious side effects and they worry you. They are the more common side effects and usually mild and short-lived |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you/your child notice any of these serious side effects. You may need urgent medical attention or hospitalisation. These side effects are rare. |
Tell your healthcare professional if you notice anything else that may be making you/your child feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you/your child experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this vaccine.
Always make sure you speak to your healthcare professional before you decide to stop taking any of your medicines.
7. Product details
This vaccine is only available with a doctor's prescription.
What FLUMIST contains
| Active ingredients (main ingredients) | Three strains of influenza virus
|
| Other ingredients (inactive ingredients) |
|
| Potential allergens | *Egg products (FLUMIST may contain trace amounts of egg protein [ovalbumin <0.24µg/dose]) *Antibiotics (gentamicin sulfate <0.015 µg/dose) |
*from the manufacturing process
**derived from pig
Do not take this vaccine if you are allergic to any of these ingredients.
FLUMIST contains no preservatives (e.g. thiomersal), lactose, gluten, tartrazine or any azo dyes.
What FLUMIST looks like
FLUMIST comes in a single-use glass pre-filled nasal applicator (looks like a small syringe). The nozzle, cap and plunger are made of plastic or rubber. The applicators do not contain latex.
Each applicator contains 200 µL of colourless to pale yellow liquid (vaccine) which may contain small white particles. It may come in boxes of 1 or 10 applicators (AUST R 470065).
Who distributes FLUMIST
AstraZeneca Pty Ltd
ABN 54 009 682 311
66 Talavera Road
MACQUARIE PARK NSW 2113
Telephone:- 1800 805 342
This leaflet was prepared on 31 Oct 2025
® FLUMIST is a registered trade mark of the AstraZeneca group of companies.
© AstraZeneca 2025
VV-RIM-10046708 v2.0
Brand Information
| Brand name | FluMist |
| Active ingredient | Influenza virus vaccine, trivalent (live attenuated) |
| Schedule | S4 |
MIMS Revision Date: 01 April 2026
1 Name of Medicine
FluMist influenza virus vaccine (live attenuated).
2 Qualitative and Quantitative Composition
Each 0.2 mL dose of FluMist contains 107.0 ± 0.5 FFU (fluorescent focus units) of each of three live attenuated influenza virus reassortant strains propagated in specific pathogen-free (SPF) eggs from SPF chicken flocks.
The three strains used for the 2026 season are:
A/Missouri/11/2025 (H1N1) pdm09-like virus (A/Switzerland/6849/2025, MEDI 400592);
A/Singapore/GP20238/2024 (H3N2)-like virus (A/Valladolid/1187/2025, MEDI 400411);
B/Austria/1359417/2021 (B/Victoria lineage)-like virus (B/Austria/1359417/2021, MEDI 355292).
This influenza vaccine complies with the World Health Organization (WHO) and Australian Influenza Vaccine Committee (AIVC) recommendations for the 2026 Southern Hemisphere influenza season.
FluMist contains hydrolysed gelatin (porcine Type A). Residual amounts of ovalbumin (< 0.24 microgram/dose; an egg protein) and also traces of gentamicin sulfate (< 0.015 microgram/dose; a trace residue) from the manufacturing process may be present. For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Nasal spray.
FluMist nasal spray is a colourless to pale yellow, clear to opalescent suspension. Small white particles may be present.
4 Clinical Particulars
4.1 Therapeutic Indications
FluMist is indicated for the prevention of influenza in children and adolescents from 24 months to less than 18 years of age.
4.2 Dose and Method of Administration
See Table 1.

FluMist is for nasal use, administered as a divided dose as described below. Each FluMist nasal applicator is for single use, in one patient only. See Figure 1.
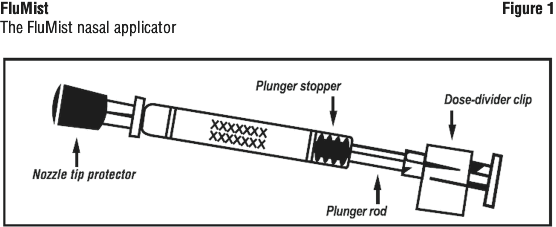
2. Remove rubber tip protector. Do not remove dose-divider clip at the other end of the applicator.
3. With the patient in an upright position, place the tip just inside the nostril to ensure the vaccine is delivered into the nose.
4. With a single motion, depress plunger as rapidly as possible until the dose-divider clip prevents you from going further.
5. Pinch and remove the dose-divider clip from plunger.
6. Place the tip just inside the other nostril and with a single motion, depress plunger as rapidly as possible to deliver remaining vaccine.
Do not inject. Do not use a needle.
Note: active inhalation (i.e. sniffing) is not required by the patient during vaccine administration.
Do not use FluMist if damaged, for example if the plunger is loose or displaced from the sprayer or if there are any signs of leakage.
In the absence of compatibility studies, this vaccine must not be mixed with other medicines/vaccines.
After use, any remaining vaccine and container must be disposed of safely, preferably by heat inactivation or incineration, according to locally agreed procedures.
4.3 Contraindications
Do not administer FluMist to:
children and adolescents who have had a severe allergic reaction (e.g. anaphylaxis) to the active substances, any of the excipients (e.g. gelatin), gentamicin (a trace residue), eggs or egg proteins (e.g. ovalbumin). See Section 2 Qualitative and Quantitative Composition; Section 6.1 List of Excipients;
children and adolescents with clinical immunodeficiency due to conditions or immunosuppressive therapy such as: acute and chronic leukaemias; lymphoma; symptomatic HIV infection; cellular immune deficiencies; and high-dose corticosteroids (also see Section 4.4 Special Warnings and Precautions for Use);
children and adolescents less than 18 years of age receiving salicylate therapy because of the association of Reye's syndrome with salicylates and wild-type influenza infection (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
4.4 Special Warnings and Precautions for Use
FluMist may not protect all individuals receiving the vaccine.
Risk in children < 24 months of age. Do not administer FluMist to children younger than 12 months. In a clinical study, an increase in hospitalisations was observed in children younger than 12 months after vaccination with FluMist. It is not recommended to administer FluMist to children 12-23 months of age. In a clinical study, an increased rate of wheezing was observed in children 12-23 months of age after vaccination with FluMist (see Section 4.8 Adverse Effects (Undesirable Effects)).
Severe asthma or active wheezing. Individuals with severe asthma or active wheezing have not been adequately studied in clinical studies.
Management of acute allergic reactions. As with all vaccines, appropriate medical treatment and supervision should always be readily available in case of an anaphylactic event following the administration of the vaccine.
Altered immunocompetence. FluMist has not been studied in immunocompromised individuals. Data on the safety and shedding of FluMist in immunocompromised individuals are limited (see Section 5.1 Pharmacodynamic Properties, Clinical trials). FluMist administration to immunocompromised individuals should be based on careful consideration of potential benefits and risks (also see Section 4.3 Contraindications). FluMist is not contraindicated for use in individuals with asymptomatic HIV infection (see Section 5.1 Pharmacodynamic Properties, Clinical trials); or individuals who are receiving topical/inhaled corticosteroids or low-dose systemic corticosteroids or those receiving corticosteroids as replacement therapy e.g. for adrenal insufficiency.
Vaccination with a live vaccine. Vaccine recipients/caregivers should be informed that FluMist is an attenuated live virus vaccine and has the potential for transmission to immunocompromised contacts. Vaccine recipients should attempt, whenever possible, to avoid close association with severely immunocompromised individuals (e.g. bone marrow transplant recipients requiring isolation) for 1-2 weeks following vaccination.
Guillain Barre syndrome. If Guillain Barre syndrome (GBS) has occurred within 6 weeks of any prior influenza vaccination, the decision to give FluMist should be based on careful consideration of the potential benefits and potential risks.
Use in the elderly. FluMist is not recommended in adults.
Paediatric use. FluMist is not recommended in children below the age of 24 months.
Effects on laboratory tests. No data available.
Medical conditions predisposing to influenza complications. The safety of FluMist in individuals with underlying medical conditions that may predispose them to complications following wild-type influenza infection has not been established.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Aspirin. Do not administer FluMist to children and adolescents (under 18 years of age) receiving salicylate therapy (see Section 4.3 Contraindications). Do not use salicylates in children and adolescents for 4 weeks after vaccination with FluMist unless medically indicated as Reye's syndrome has been reported following the use of salicylates during wild-type influenza infection.
Other vaccines. FluMist may be given at the same time as other vaccines. Concurrent administration of FluMist with the MMR vaccine (1233 children 11-23 months of age), the MMR and varicella vaccine (1245 children 12-15 months of age), and the orally administered poliovirus vaccine (2503 children 6-35 months of age) has been studied (see Section 5.1 Pharmacodynamic Properties, Clinical trials). Adverse events were similar to those seen in other clinical studies with FluMist. Studies did not show clinically meaningful changes in immune responses to measles, mumps, rubella, varicella, orally administered poliovirus or FluMist.
Antiviral agents. The concurrent use of FluMist with antiviral agents that are active against influenza A and/or B viruses has not been evaluated. However, based upon the potential for influenza antiviral agents to reduce the effectiveness of FluMist, it is recommended:
not to administer FluMist until 48 hours after the cessation of influenza antiviral therapy.
not to administer influenza antiviral agents until two weeks after administration of FluMist unless medically indicated.
If influenza antiviral agents and FluMist are administered concomitantly, revaccination should be considered when appropriate.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. FluMist Quadrivalent has not been evaluated for its potential to impair human fertility. However, a reproductive and developmental toxicity study in which rats were intranasally administered the human dose of FluMist Quadrivalent 3 times in the 2 weeks prior to mating, and 3 times during gestation, showed no adverse effects on female fertility.
Use in pregnancy. (Category B1)
Available data, including post-marketing reports, suggest that FluMist has not been associated with adverse maternal or fetal outcomes. FluMist is not recommended during pregnancy as these reports do not adequately inform the presence or absence of drug-associated risk with the use of FluMist during pregnancy. Refer to National recommendations as per the current Immunisation Handbook.
There are limited data from the use of FluMist in pregnant women. There was no evidence of significant maternal adverse outcomes in 138 pregnant women who had a record of receiving FluMist in a US based health insurance claims database. In more than 300 case reports in the AstraZeneca safety database and over 150 case reports from the US Vaccine Adverse Event Reporting System of trivalent and quadrivalent version of FluMist administration to pregnant women, no unusual patterns of pregnancy complications or fetal outcomes were observed.
The effects of FluMist on embryo-fetal and pre-weaning development were evaluated in developmental toxicity studies in pregnant rats and pregnant ferrets. Rats were intranasally administered the human dose of FluMist Quadrivalent on gestation days 6, 13 and 20 (and in some groups also 3 times in the 2 weeks prior to mating). Ferrets were administered the human dose of FluMist intranasally on gestation days 3, 6, 13 and 22. No adverse effects on pregnancy, parturition, lactation or embryo-fetal development were observed in either study and, in addition no adverse effects on pre-weaning development were observed in the rat study. There were no fetal malformations or other evidence of teratogenesis observed.
Use in lactation. Studies in lactating women have not been conducted with FluMist. Limited available evidence suggests that FluMist is not excreted in human milk. There are some viruses that are excreted in human milk. Caution should be exercised if FluMist is administered to nursing mothers.
Vaccine antigen-specific antibodies were transferred to rat pups via milk from dams administered FluMist Quadrivalent during gestation and lactation, with no adverse effects.
4.7 Effects on Ability to Drive and Use Machines
The vaccine is not expected to have an effect on the ability to drive and use machines.
4.8 Adverse Effects (Undesirable Effects)
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a vaccine cannot be directly compared to rates in the clinical trials of another vaccine and may not reflect the rates observed in practice.
Additional experience has occurred with marketed use of trivalent and quadrivalent versions of FluMist.
Adverse reactions in clinical studies. Children and adolescents. The safety of FluMist was evaluated in an AF-SPG placebo-controlled Study (AV019) conducted in a Health Maintenance Organisation (HMO) in children 1 through 17 years of age (FluMist = 6473, placebo = 3216). An increase in asthma events, captured by review of diagnostic codes, was observed in children younger than 5 years of age who received FluMist compared to those who received placebo (Relative risk 3.53, 90% CI: 1.1, 15.7).
In Study MI-CP111, children 6 through 59 months of age were randomised to receive FluMist or inactivated influenza virus vaccine. Wheezing requiring bronchodilator therapy or accompanied by respiratory distress or hypoxia was prospectively monitored from randomisation through 42 days post last vaccination. Hospitalisation due to all causes was prospectively monitored from randomisation through 180 days post last vaccination. Increases in wheezing and hospitalisation (for any cause) were observed in children 6 months through 23 months of age who received FluMist compared to those who received inactivated influenza virus vaccine, as shown in Table 2.
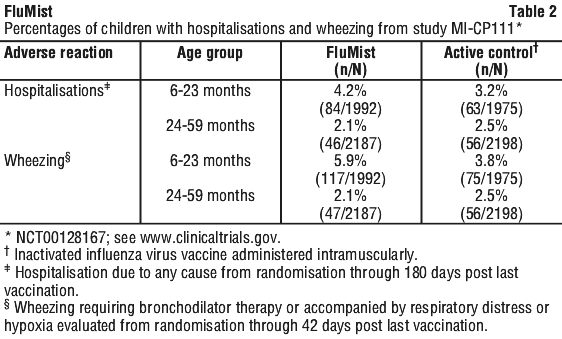
Table 3 shows pooled solicited adverse reactions occurring in at least 1% of FluMist recipients and at a higher rate (≥ 1% rate difference after rounding) compared to placebo post Dose 1 for Studies D153-P501 and AV006, and solicited adverse reactions post Dose 1 for Study MI-CP111. Solicited adverse reactions were those about which parents/guardians were specifically queried after receipt of FluMist, placebo, or control vaccine. In these studies, solicited reactions were documented for 10 days post vaccination. Solicited reactions following the second dose of FluMist were similar to those following the first dose and were generally observed at a lower frequency.
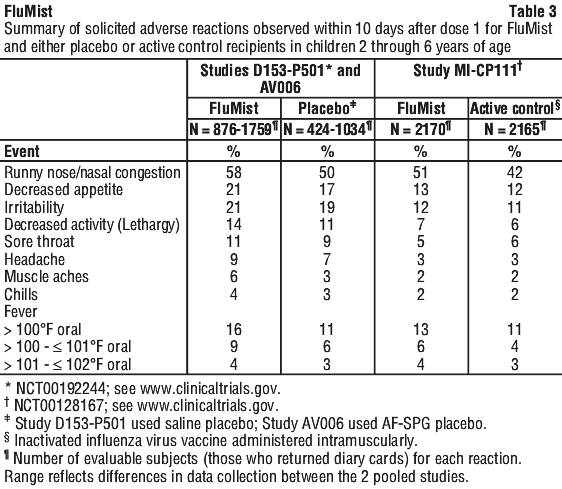
In a separate saline placebo-controlled trial (D153-P526) in a subset of older children and adolescents 9 through 17 years of age who received one dose of FluMist, the solicited adverse reactions as well as unsolicited adverse reactions reported were generally consistent with observations from the trials in Table 3. Abdominal pain was reported in 12% of FluMist recipients compared to 4% of placebo recipients and decreased activity was reported in 6% of FluMist recipients compared to 0% of placebo recipients.
FluMist is not recommended for use in children younger than 24 months, and should not be used in children younger than 12 months (see Section 4.4 Special Warnings and Precautions for Use).
Post marketing experience. The following adverse reactions have been identified during post-approval use of FluMist (see Table 4). Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to vaccine exposure.

4.9 Overdose
There have been occasional reports of administration of twice the recommended dose of FluMist in the post-marketing setting. The adverse reactions reported were similar to those seen with the recommended single dose of FluMist.
For information on the management of overdose, contact the Poison Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
FluMist is a trivalent vaccine that contains antigens for three influenza virus strains, an A/(H1N1) strain, an A/(H3N2) strain, and a B strain (Victoria lineage). The influenza virus strains in FluMist are (a) cold-adapted (ca) (i.e. they replicate efficiently at 25°C, a temperature that is restrictive for replication of many wild-type influenza viruses); (b) temperature-sensitive (ts) (i.e. they are restricted in replication at 37°C (Type B strain) or 39°C (Type A strains), temperatures at which many wildtype influenza viruses grow efficiently); and (c) attenuated (att) (i.e. they do not produce classic influenza-like illness in the ferret model of human influenza infection). The cumulative effect of the antigenic properties and the ca, ts, and att phenotypes is that the attenuated vaccine viruses replicate in the nasopharynx and induce protective immunity.
No evidence of reversion has been observed in the recovered vaccine strains that have been tested (135 of possible 250 recovered isolates). The three viruses contained in FluMist maintain the replication characteristics and phenotypic properties of the master donor virus (MDV) and express the haemagglutinin (HA) and neuraminidase (NA) of wild-type viruses that are related to strains expected to circulate during the influenza season. For each strain, at least five genetic loci contribute to the ca/ts/att phenotype.
Mechanism of action. Immune mechanisms conferring protection against influenza following receipt of FluMist vaccine are not fully understood. Likewise, naturally acquired immunity to wild-type influenza has not been completely elucidated. Serum antibodies, mucosal antibodies and influenza-specific T cells may play a role in prevention of infection and in recovery from infection.
Clinical trials. Studies in children and adolescents. FluMist has been administered to over 20,000 children and adolescents in controlled clinical studies over multiple years, in various regions and using different vaccine strains. The results of these pivotal efficacy and safety studies are discussed in detail below.
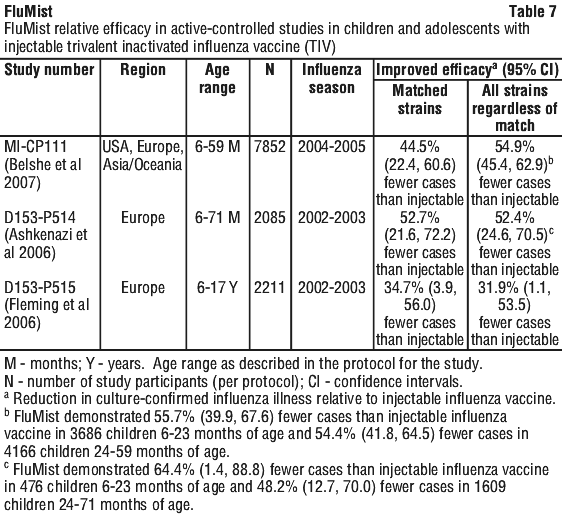
Efficacy data in the paediatric population for FluMist consists of 9 controlled studies conducted during 7 influenza seasons. Four placebo-controlled studies included second season revaccination. FluMist demonstrated superiority in 3 active-controlled studies with injectable trivalent inactivated influenza vaccines (TIV). See Table 5 (placebo controlled) and Table 7 (active controlled) for a summary of FluMist efficacy results in children. The pivotal studies are then detailed further within the text below each table.
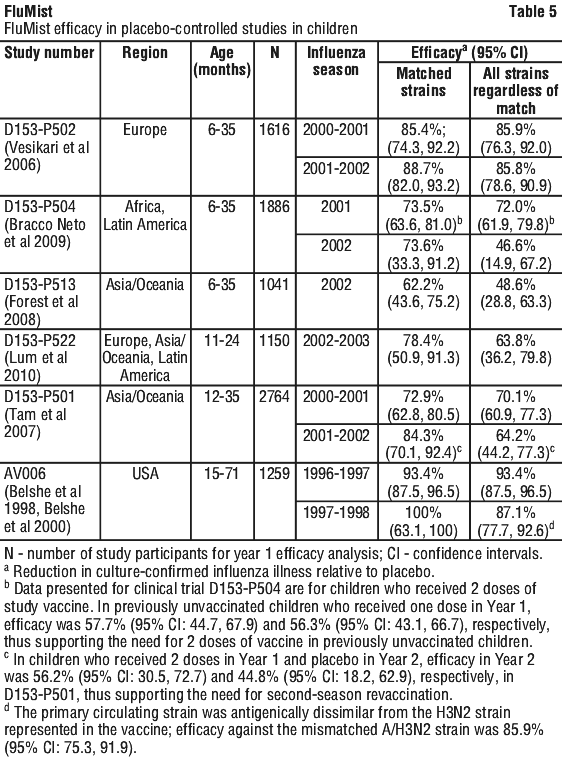
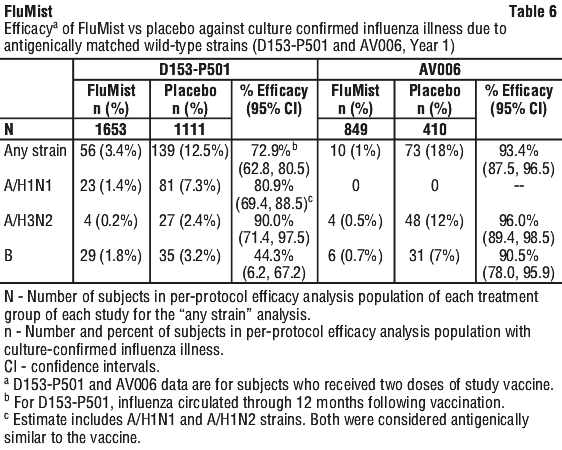
During the second year of Study D153-P501, for children who received two doses in Year 1 and one dose in Year 2, FluMist demonstrated 84.3% (95% CI: 70.1, 92.4) efficacy against culture-confirmed influenza illness due to antigenically matched wild-type influenza.
Study AV006: paediatric study. AV006 was a multi-centre, randomised, double-blind, placebo-controlled trial performed in US children without high-risk medical conditions to evaluate the efficacy FluMist against culture-confirmed influenza over 2 successive seasons. The primary endpoint of the trial was the prevention of culture-confirmed influenza illness due to antigenically matched wild-type influenza in children who received two doses of vaccine in the first year and a single revaccination dose in the second year. During the first year of the study, 1602 children 15-71 months of age were randomised 2:1 (vaccine: placebo). In Year 2, children remained in the same treatment group as in year one and received a single dose of FluMist or placebo. See Table 6 for a description of the results.
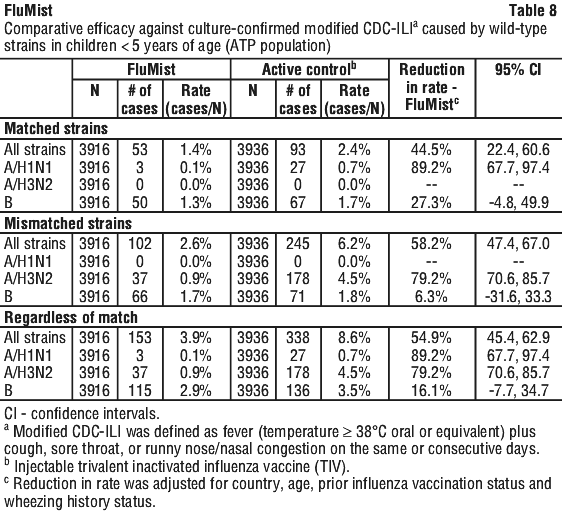
During the 2004-2005 influenza season, a total number of 3916 children < 5 years of age and without severe asthma, without use of bronchodilator or steroids and without wheezing within the prior 6 weeks were randomised to FluMist and 3936 were randomised to active control. Participants were then followed through the influenza season to identify illness caused by influenza virus. As the primary endpoint, culture-confirmed modified CDC-ILI (Centers for Disease Control and Prevention [CDC]-defined influenza-like illness) was defined as a positive culture for a wild-type influenza virus associated within ± 7 days of modified CDC-ILI. Modified CDC-ILI was defined as fever (temperature ≥ 38°C oral or equivalent) plus cough, sore throat or runny nose/nasal congestion on the same or consecutive days.
In the primary efficacy analysis, FluMist demonstrated a 44.5% (95% CI: 22.4, 60.6) reduction in influenza rate compared to active control as measured by culture confirmed modified CDC-ILI caused by wild-type strains antigenically similar to those contained in the vaccine. See Table 7 for a description of the overall results and Table 8 for a description of the results by strain and antigenic similarity.
Twenty mild to moderately immunocompromised children and adolescents 5-17 years of age (receiving chemotherapy and/or radiation therapy or who had received chemotherapy in the 12 weeks prior to enrolment) were randomised 1:1 to receive FluMist or placebo (Study MI-CP114; Halasa et al., 2011). Frequency and duration of vaccine virus shedding in these immunocompromised children and adolescents were comparable to that seen in healthy children and adolescents. The effectiveness of FluMist in preventing influenza illness in immunocompromised individuals has not been evaluated.
Studies with concomitant live vaccines. In Study AV018 (Nolan et al., 2008), concomitant administration of FluMist, measles, mumps and rubella virus vaccine live (MMR) and varicella virus vaccine live was studied in 1245 subjects 12-15 months of age. Subjects were randomised in a 1:1:1 ratio to MMR, varicella vaccine and placebo (group 1); MMR, varicella vaccine and FluMist (group 2); or FluMist alone (group 3). Immune responses to MMR and varicella vaccines were evaluated 6 weeks post-vaccination while the immune responses to FluMist were evaluated 4 weeks after the second dose. Adverse reactions were similar to those seen in other clinical trials with FluMist (see Section 4.8 Adverse Effects (Undesirable Effects)). No evidence of interference with immune response to measles, mumps, rubella, varicella and FluMist vaccines was observed (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
In Study D153-P511 (Breiman et al., 2009) concomitant administration of FluMist and oral poliovirus (OPV) was studied in 2,503 subjects 6-35 months of age. Subjects were randomised in a 1:1:1 ratio to 1 of 3 study groups: FluMist + OPV; placebo + OPV; or FluMist alone. The rate of reactogenicity events reported by vaccine recipients in Study D153-P511 was similar to that observed in previous trials with FluMist. Immune responses after concomitant vaccination were non-inferior to those elicited when the vaccines were administered independently of one another (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Duration of efficacy. Analyses that examined the impact of time on the efficacy of LAIV in young children compared with placebo, demonstrated comparable efficacy through 12 months post-vaccination; two studies have shown that LAIV efficacy can persist at a lower but clinically meaningful level through the following influenza season without revaccination (Study D153-P501, Tam et al., 2007; Study D153-P504, Bracco Neto et al., 2009).
5.2 Pharmacokinetic Properties
Not applicable.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
FluMist contains the following as inactive ingredients: sucrose, dibasic potassium phosphate, monobasic potassium phosphate, hydrolysed gelatin (porcine Type A), arginine hydrochloride, monosodium glutamate monohydrate and water for injections.
Residual amounts of ovalbumin (< 0.24 microgram/dose) and also traces of gentamicin sulfate (< 0.015 microgram/dose) from the manufacturing process may be present.
FluMist contains no preservatives (i.e. no thiomersal).
Also see Section 2 Qualitative and Quantitative Composition.
6.2 Incompatibilities
In the absence of compatibility studies, this vaccine must not be mixed with other medicines/vaccines.
Also see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store at 2°C to 8°C (Refrigerate. Do not freeze).
A single temperature excursion up to 25°C for 12 hours has been shown to have no adverse impact on the vaccine. After a temperature excursion, the vaccine should be returned immediately to the recommended storage condition (2°C to 8°C) and used as soon as feasible. Subsequent excursions are not permitted.
Keep the nasal applicator in the carton in order to protect from light.
6.5 Nature and Contents of Container
FluMist nasal spray is a 0.2 mL suspension in a pre-filled, single-use nasal applicator (Type 1 glass) with a nozzle (polypropylene with polyethylene transfer valve), nozzle tip-protector cap (synthetic rubber), plunger rod, plunger-stopper (butyl rubber) and a dose-divider clip. The nasal applicator contains no latex.
Packs of 1 or 10 nasal applicators are registered. Not all pack sizes may be available in Australia.
6.6 Special Precautions for Disposal
After use, any remaining vaccine and container must be disposed of safely, preferably by heat inactivation or incineration, according to locally agreed procedures.
6.7 Physicochemical Properties
Not applicable for vaccines.
7 Medicine Schedule (Poisons Standard)
Prescription only medicine (Schedule 4).
Date of First Approval
18 November 2025
Date of Revision
02 March 2026
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.