Fruzaqla
Brand Information
| Brand name | Fruzaqla |
| Active ingredient | Fruquintinib, Fruquintinib |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Fruzaqla.
Summary CMI
FRUZAQLA®
Consumer Medicine Information (CMI) summary
The full CMI on next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
▼ This medicine is new or being used differently. Please report side effects. See the full CMI for further details.
1. Why am I using FRUZAQLA?
FRUZAQLA contains the active ingredient fruquintinib. FRUZAQLA is used to treat adult patients with colorectal cancer (CRC) that has spread to other parts of the body (metastatic). It is used when other treatments have not worked or when other treatments are not suitable for you.
For more information, see Section 1. Why am I using FRUZAQLA? in the full CMI.
2. What should I know before I use FRUZAQLA?
Do not use if you have ever had an allergic reaction to FRUZAQLA or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use FRUZAQLA? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with FRUZAQLA and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use FRUZAQLA?
- Recommended dose: 5 mg once daily (approximately same time each day) for 21 days (3 weeks), followed by 7 days (1 week) of no medicine.
- FRUZAQLA can be taken with or without food and should be swallowed as a whole capsule.
- More instructions can be found in Section 4. How do I use FRUZAQLA? in the full CMI.
5. What should I know while using FRUZAQLA?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using FRUZAQLA? in the full CMI.
6. Are there any side effects?
Reduced blood platelets, reduced thyroid activity, weight loss and decreased appetite, high blood pressure, voice change/ hoarseness, diarrhoea, painful dry mouth, mouth sores/ulcers, redness, pain, blisters and swelling of palms/ soles of feet, pain in muscles, bones, joints, chest or neck, protein in urine, weakness, tiredness. Upper respiratory infection, fever, sore throat, cough, pneumonia, low blood cell counts, nosebleeds, throat pain, pancreatic enzyme elevation, hole in stomach, toothache, gum or lip pain, mouth sores, rash. Severely elevated blood pressure, digestive system bleeding (stomach, throat, rectum bowel), severe stomach pain, confusion, seizure, vision change, abnormal liver function test, yellowing skin/white part of eyes, dark coloured urine, burning pain when urinating, right upper stomach pain, nausea, vomiting, bruising, severe cough, trouble breathing, redness/pain in any part body, severe chest pain/ pressure, pain in arm, leg back, neck or jaw, shortness of breath, numbness/weakness of face, arm or leg, confusion, trouble talking or walking, severe headache, dizziness/loss of balance. For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
▼ This medicine is subject to additional monitoring. This will allow quick identification of new safety information. You can help by reporting any side effects you may get. You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems.
FRUZAQLA®
Active ingredient(s): fruquintinib
Consumer Medicine Information (CMI)
This leaflet provides important information about using FRUZAQLA. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using FRUZAQLA.
Where to find information in this leaflet:
1. Why am I using FRUZAQLA?
2. What should I know before I use FRUZAQLA?
3. What if I am taking other medicines?
4. How do I use FRUZAQLA?
5. What should I know while using FRUZAQLA?
6. Are there any side effects?
7. Product details
1. Why am I using FRUZAQLA?
FRUZAQLA contains the active ingredient fruquintinib. FRUZAQLA belongs to a group of medicines called antineoplastic (or anti-cancer) agents which are used to treat cancer.
- FRUZAQLA is used to treat adult patients with colorectal cancer (CRC) that has spread to other parts of the body (metastatic). It is used when other treatments have not worked or when other treatments are not suitable for you.
- FRUZAQLA stops tumours from making new blood vessels and therefore slows down the growth of cancer. Blood vessels would usually provide the tumour with nutrients and oxygen.
2. What should I know before I use FRUZAQLA?
Warnings
Do not use FRUZAQLA if:
- you are allergic to fruquintinib, or any of the ingredients listed at the end of this leaflet.
- Always check the ingredients to make sure you can use this medicine.
Check with your doctor if you:
- take any medicines for any other conditions
- have any other medical conditions including:
- high blood pressure
- bleeding problems or are taking medicines to thin the blood or prevent blood clots
- severe stomach or bowel problems
- liver problems
- kidney problems or have protein in your urine
- skin problems
- recently had severe or persistent headache, trouble with your vision, seizures or fits, or altered mental status such as confusion, memory loss or loss of orientation
- have an infection
- recently had or going to have a surgical procedure or have an unhealed wound
- recently had problems with blood clots in your blood vessels including stroke, heart attack, embolism or thrombosis
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Tell your doctor if you are pregnant or intend to become pregnant.
FRUZAQLA has not been studied in pregnant women. FRUZAQLA should not be used during pregnancy. If you are pregnant, think you may be pregnant or are planning to have a baby, ask your doctor or pharmacist for advice before taking this medicine. Your doctor will discuss with you the potential risks of taking this medicine during pregnancy.
- Women who are able to become pregnant and male patients with female partners who are able to become pregnant should use highly effective contraceptive methods during treatment, and for at least 2 weeks following the last dose of FRUZAQLA. Talk to your doctor about the right methods of contraception for you and your partner.
- Talk to your doctor if you are breastfeeding or intend to breastfeed.
- It is not known whether FRUZAQLA passes into breast milk. You should not breast-feed during treatment with this medicine and for at least 2 weeks following the last dose of FRUZAQLA. Talk to your doctor about the best way to feed your baby during this time.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with FRUZAQLA and affect how it works. These include:
- rifampicin, a medicine used to treat tuberculosis or other infections
- efavirenz, a medicines used to treat HIV-1 infection
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect FRUZAQLA.
4. How do I use FRUZAQLA?
FRUZAQLA should only be prescribed by a doctor experienced in cancer treatment.
How much to take
- The recommended dose of FRUZAQLA is 5 mg once daily at approximately the same time each day for 21 days (3 weeks), followed by 7 days (1 week) of no medicine. This three weeks on, one week off treatment period is 1 cycle of treatment.
- FRUZAQLA can be taken with or without food and should be swallowed as a whole capsule.
- Your doctor may reduce your dose if necessary.
- Follow the instructions provided and use FRUZAQLA until your doctor tells you to stop.
When to take FRUZAQLA
- FRUZAQLA should be taken once a day at about the same time each day.
If you forget to take FRUZAQLA
If you miss your dose at the usual time and it is almost time for your next dose (less than 12 hours), skip the dose you missed and take your next dose when you are meant to.
If there are more than 12 hours until your next dose, take the dose you missed dose and then take your next dose when you are meant to.
Do not take a double dose to make up for the dose you missed. This may increase the chance of getting an unwanted side effect.
If you vomit after taking FRUZAQLA, do not take a replacement capsule. Continue to take your next dose at the usual time.
If you use too much FRUZAQLA
If you think that you have used too much FRUZAQLA, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using FRUZAQLA?
Things you should do
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are taking FRUZAQLA.
Tell any other doctors, dentists and pharmacists who treat you that you are taking this medicine.
Use highly effective contraception to prevent pregnancy while you are being treated with FRUZAQLA.
If you are going to have surgery, tell the surgeon that you are taking this medicine.
Keep all of your doctor's appointments so that your progress can be checked.
Your doctor will do blood tests and other tests from time to time to monitor your progress and check for side effects. If necessary, your doctor may decide to reduce your dose, temporarily interrupt your treatment or stop it altogether.
Call your doctor straight away if you:
experience:
- very high blood pressure
- severe headache
- severe chest pain
- passing blood in the stools or passing black stools
- passing blood in the urine
- severe stomach (abdominal) pain or stomach pain that does not go away
- coughing/vomiting up blood.
- confusion, seizures (fits)
- changes in vision
Remind any doctor, dentist or pharmacist you visit that you are using FRUZAQLA.
Things you should not do
- Do not stop taking medicine unless your doctor tells you to.
- Do not give this medicine to anyone else, even if they have the same condition as you.
- Do not use this medicine to treat any other complaints unless your doctor tells you to.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how FRUZAQLA affects you.
FRUZAQLA may cause vision disturbances or tiredness in some people.
Do not drive or use machines during treatment if such signs occur.
Looking after your medicine
- Keep in a cool, dry place where the temperature stays below 30°C.
- Keep your capsules in the bottle until it is time to take them. If you take the capsules out of the bottle they may not keep well. Keep the bottle tightly closed. Do not remove desiccant cartridge from the bottle.
Store away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What FRUZAQLA contains
| Active ingredient (main ingredient) | 1 mg or 5 mg fruquintinib |
| Other ingredients (inactive ingredients) | FRUZAQLA 1 mg hard capsules:
FRUZAQLA 5 mg hard capsules:
|
Do not take this medicine if you are allergic to any of these ingredients.
What FRUZAQLA looks like
FRUZAQLA 1 mg capsules are an opaque hard gelatin capsule, size 3 (approximate length 16 mm), with a yellow cap and a white body imprinted with “HM013” over “1mg” in black ink (AUST R 422039).
FRUZAQLA 5 mg capsules are an opaque hard gelatin capsule, size 1 (approximate length 19 mm), with a red cap and a white body imprinted with “HM013” over “5mg” in black ink (AUST R 422038).
Who distributes FRUZAQLA
Takeda Pharmaceuticals Australia Pty Ltd
Level 39, 225 George Street
Sydney, NSW 2000
Australia
Telephone: 1800 012 612
This leaflet was prepared in October 2024.
TAKEDA and the TAKEDA Logo are registered trademarks of Takeda Pharmaceutical Company Limited.
FRUZAQLA and the FRUZAQLA Logo are registered trademarks of HUTCHMED Group Enterprises Limited, used under license.
Brand Information
| Brand name | Fruzaqla |
| Active ingredient | Fruquintinib, Fruquintinib |
| Schedule | S4 |
▼ This medicinal product is subject to additional monitoring in Australia. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse events at www.tga.gov.au/reporting-problems.
MIMS Revision Date: 01 May 2026
1 Name of Medicine
Fruquintinib.
2 Qualitative and Quantitative Composition
Fruzaqla 1 mg hard capsules. Fruzaqla 1 mg hard capsules contain 1 mg fruquintinib.
Excipients with known effect. Each 1 mg hard capsule contains tartrazine.
Fruzaqla 5 mg hard capsules. Fruzaqla 5 mg hard capsules contain 5 mg fruquintinib.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Fruzaqla 1 mg hard capsules. Opaque hard gelatin capsule, size 3 (approximate length 16 mm), with a yellow cap and a white body imprinted with "HM013" over "1 mg" in black ink.
Fruzaqla 5 mg hard capsules. Opaque hard gelatin capsule, size 1 (approximate length 19 mm), with a red cap and a white body imprinted with "HM013" over "5 mg" in black ink.
4 Clinical Particulars
4.1 Therapeutic Indications
Fruzaqla is indicated for the treatment of adult patients with metastatic colorectal cancer (mCRC) who have been previously treated with fluoropyrimidine-, oxaliplatin-, and irinotecan-based chemotherapy, an anti-VEGF agent, and an anti-EGFR agent if appropriate.
4.2 Dose and Method of Administration
Fruzaqla should be initiated by a physician experienced in the administration of anticancer therapy.
Posology and method of administration. The recommended dose of Fruzaqla is 5 mg taken orally once daily (at approximately the same time each day) for the first 21 days of each 28-day cycle. Fruzaqla capsules should be swallowed whole, and can be taken with or without food.
Duration of treatment. Treatment with Fruzaqla should be continued until disease progression or unacceptable toxicity occurs.
Missed doses or vomiting. If a dose is missed by less than 12 hours, it should be taken, and the next dose should be taken as scheduled. If a dose is missed by more than 12 hours, it should be skipped, and the next dose should be taken as scheduled.
If a patient vomits after taking a dose, the patient should not repeat the dose on the same day but resume the usual dosing as scheduled on the following day.
Dose adjustments for adverse reactions. The recommended dose reduction schedule for adverse reactions is provided in Table 1.
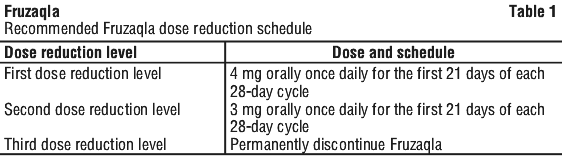

Hepatic impairment. No dose adjustment is required for patients with mild hepatic impairment (elevated AST and normal total bilirubin, or total bilirubin 1 to 1.5 x the upper limit of normal [ULN] with any AST; see Section 5.2). Inadequate data are available to support dose recommendations for moderate hepatic impairment (total bilirubin > 1.5 to 3 x ULN and any AST). Fruzaqla is not recommended for use in patients with severe hepatic impairment (total bilirubin > 3 x ULN and any AST).
Elderly population. No dose adjustment is required for elderly patients.
Paediatric population. The safety and efficacy of Fruzaqla in patients aged 0 to < 18 years have not been established. No data are available.
4.3 Contraindications
Hypersensitivity to the active substance or any of the excipients listed in Section 6.1.
4.4 Special Warnings and Precautions for Use
Hypertension. Fruzaqla can cause hypertension. Amongst 911 patients with mCRC who received Fruzaqla in clinical trials, hypertension occurred in 49%, including Grade 3-4 events in 19%, and hypertensive crisis in three patients (0.3%). The median time to first onset of hypertension was 14 days from first dose of Fruzaqla (also see Section 4.8). Do not initiate Fruzaqla unless blood pressure is adequately controlled. Monitor blood pressure weekly the first month, at least monthly thereafter and as clinically indicated. Initiate or adjust anti-hypertensive therapy as appropriate. Withhold, reduce dose, or permanently discontinue Fruzaqla based on the severity of hypertension (see Section 4.2).
Haemorrhage. Fruzaqla can cause serious haemorrhagic events, which may be fatal. Amongst 911 patients with mCRC who received Fruzaqla in clinical trials, 6% experienced a gastrointestinal haemorrhage, including 13 Grade ≥ 3 events (1% of patients) and 2 fatal events (0.2% of patients). Also see Section 4.8. Monitor haematologic and coagulation profiles more frequently in patients at risk for bleeding, including due to concomitant anticoagulants. Withhold, reduce dose, or permanently discontinue Fruzaqla based on the severity of haemorrhage (see Section 4.2).
Infections. Fruzaqla treatment may cause an increased risk of infections, including fatal infections. Amongst 911 patients with mCRC who received Fruzaqla across these and other clinical trials, the most common infections were urinary tract infections (6.8%), upper respiratory tract infections (3.2%) and pneumonia (2.5%); fatal infections included pneumonia (0.4%), sepsis (0.2%), bacterial infection (0.1%), lower respiratory tract infection (0.1%), and septic shock (0.1%). The most common fatal infection was pneumonia (also see Section 4.8). Withhold Fruzaqla for Grade 3 or 4 infections, or worsening of an infection of any grade. Resume Fruzaqla at the same dose when the infection is resolved.
Gastrointestinal (GI) perforation. Medicines that inhibit the vascular endothelial growth factor (VEGF) signalling pathway can cause GI perforation (see Section 4.8). Amongst 911 patients with mCRC who received Fruzaqla in clinical trials, twelve patients (1.3%) experienced a Grade ≥ 3 gastrointestinal perforation, including one fatal event (0.1%). Permanently discontinue Fruzaqla if gastrointestinal perforation or fistula occurs.
Hepatotoxicity. Fruzaqla can cause liver injury. Amongst 911 patients with mCRC who received Fruzaqla in clinical trials, 48% experienced increased ALT or AST, including Grade ≥ 3 events in 5%, and fatal events in 0.2%. Median time to first onset of elevated liver enzymes was 29 days from first dose of Fruzaqla (also see Section 4.8). Monitor liver function tests (ALT, AST, and bilirubin) before initiation and periodically throughout treatment with Fruzaqla. Withhold, reduce dose or permanently discontinue Fruzaqla depending on the severity and persistence of hepatic enzyme derangement (see Section 4.2).
Proteinuria. Fruzaqla can cause proteinuria. Amongst 911 patients with mCRC who received Fruzaqla in clinical trials, 36% experienced proteinuria and 2.5% of patients experienced Grade ≥ 3 events. Median time to first onset of proteinuria was 22 days from first dose of Fruzaqla (also see Section 4.8). Monitor for proteinuria before initiation and periodically throughout treatment with Fruzaqla. Withhold, reduce dose or permanently discontinue Fruzaqla depending on the severity and persistence of proteinuria, or presence of nephrotic syndrome (see Section 4.2).
Palmar-plantar erythrodysaesthesia syndrome (PPES). Fruzaqla can cause PPES. Amongst 911 patients with mCRC who received Fruzaqla in clinical trials, PPES occurred in 35%, including 8% with Grade 3 events. Median time to first onset of PPES was 19 days from first dose of Fruzaqla (also see Section 4.8). Depending on severity, withhold Fruzaqla for PPES and resume at the same or a reduced dose (see Section 4.2).
Posterior reversible encephalopathy syndrome (PRES). Fruzaqla can cause PRES, a syndrome of subcortical vasogenic oedema diagnosed by characteristic finding on MRI. PRES occurred in one of 911 patients (0.1%) with mCRC who received Fruzaqla in clinical trials. Perform an evaluation for PRES in any patient presenting with seizures, headache, visual disturbances, altered consciousness or cognition, or other evidence of neurological dysfunction. Discontinue Fruzaqla in patients who develop PRES.
Impaired wound healing. Impaired wound healing can occur in patients receiving medicines that inhibit the VEGF signalling pathway. Amongst 911 patients with mCRC who received Fruzaqla in clinical trials, 1 patient (0.1%) experienced a Grade 2 event of wound dehiscence. Do not administer Fruzaqla for at least 2 weeks prior to surgery. Do not administer Fruzaqla until adequate wound healing. The safety of resumption of Fruzaqla after resolution of wound healing complications has not been established.
Arterial thromboembolic events. Fruzaqla may increase the risk of arterial thromboembolic events. Fruzaqla studies excluded patients with clinically significant cardiovascular disease, uncontrolled hypertension, or with thromboembolic events within the prior 6 months. Despite this, amongst 911 patients with mCRC who received Fruzaqla in clinical trials, 7 patients (0.8%) experienced an arterial thromboembolic event. This risk should be carefully considered in deciding whether or not to initiate treatment with Fruzaqla in a patient with a recent history of thromboembolic events. Discontinue Fruzaqla if arterial thromboembolism occurs.
Use in the elderly. No overall differences in pharmacokinetics, safety or efficacy were observed between elderly (aged 65 years and above) and those younger than 65 years.
Paediatric use. The safety and efficacy of Fruzaqla in patients younger than 18 years of age have not been established.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
In vitro studies. Cytochrome P450 enzymes. Fruquintinib is metabolised by CYP and non-CYP enzymes in vitro. CYP3A4 was the main CYP enzyme isoform involved, with minor contributions from CYP2C8, CYP2C9 and CYP2C19. Fruquintinib is not an inhibitor of CYP1A2, CYP2B6, CYP2C8, CYP2C9, CYP2C19, CYP2D6 and CYP3A, or an inducer of CYP1A2, CYP2B6, CYP3A.
Transporter systems. Fruquintinib is not a substrate of P-glycoprotein (P-gp), organic anion transport protein (OATP)1B1, or OATP1B3. Fruquintinib is not an inhibitor of OATP1B1, OATP1B3, organic anion transporter (OAT)1, OAT3, organic cation transporter (OCT)2, multidrug and toxin extrusion protein (MATE)1, or MATE2-K at clinically relevant concentrations.
Effects of other medicinal products on the pharmacokinetics of Fruzaqla. CYP3A inducers. Coadministration of fruquintinib with rifampicin (a strong CYP3A inducer) 600 mg once daily decreased fruquintinib AUC by 65% and decreased Cmax by 12%. Coadministration of fruquintinib with efavirenz (a moderate CYP3A inducer) 600 mg once daily is predicted to decrease fruquintinib AUC by 32% and fruquintinib Cmax by 4%. No clinically meaningful differences in the AUC of fruquintinib are predicted when fruquintinib is coadministered with dexamethasone (a weak CYP3A inducer) 8 mg twice daily. The concomitant use of Fruzaqla with strong and moderate CYP3A inducers should be avoided.
CYP3A inhibitors. Co-administration of fruquintinib with itraconazole (a strong CYP3A inhibitor) 200 mg twice daily did not result in clinically meaningful changes to fruquintinib exposure.
Gastric acid lowering agents. Fruquintinib demonstrated pH-dependent aqueous solubility in vitro. However, co administration of Fruzaqla with rabeprazole (a proton pump inhibitor) 40 mg once daily did not result in clinically meaningful changes to fruquintinib exposure.
Effect of fruquintinib on the pharmacokinetics of other medicinal products. Medicinal products that are substrates of P-glycoprotein (P-gp). Fruquintinib inhibited P-gp in a dose-dependent manner in vitro. Co-administration of a single dose of dabigatran etexilate 150 mg (a P-gp substrate) with a single dose of Fruzaqla 5 mg decreased AUC of dabigatran by 9%. Co-administration of a single dose of digoxin (a P-gp substrate) 0.5 mg with multiple doses of Fruzaqla is predicted to result in a 6% increase in AUC of digoxin. This effect is not likely to be clinically significant, and no dose adjustment is recommended for P-gp substrates during concomitant use with Fruzaqla.
Medicinal products that are substrates of breast cancer resistance protein (BCRP). Fruquintinib inhibited BCRP in a dose-dependent manner in vitro. Co-administration of a single 10 mg dose of rosuvastatin (a BCRP substrate) with a single 5 mg dose of Fruzaqla decreased AUC of rosuvastatin by 19%. Co-administration of a single 20 mg dose of rosuvastatin with multiple doses of Fruzaqla is predicted to result in a 19% increase in AUC of rosuvastatin (a BCRP substrate). This effect is not likely to be clinically significant, and no dose adjustment is recommended for BCRP substrates during concomitant use with fruquintinib.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. There are no data on the effects of Fruzaqla on human fertility. In a fertility and early embryonic development study via oral route in rats, no effect was seen on male and female fertility at 3 and 0.5 mg/kg/day, respectively (1.2 and 0.19 times the exposure at the maximum recommended human dose [MRHD] based on AUC, respectively).
Use in pregnancy. (Category D)
Based on findings in animal studies (see below) and its mechanism of action, Fruzaqla has the potential to cause fetal harm if administered to a pregnant person. Advise all patients of the potential risk to a fetus. Advise patients who could become pregnant or whose partner could become pregnant to use effective contraception during treatment with Fruzaqla and for at least 2 weeks after the last dose.
In an embryo-fetal development study in pregnant rats, following oral administration of fruquintinib during the period of organogenesis, embryotoxic and teratogenic effects were observed at exposures below the clinical exposure (doses ≥ 0.1 mg/kg/day; 0.05 times the clinical exposure at MRHD based on AUC). Observations included fetal external (head and tail malformations and oedema), visceral (malpositioned or absent blood vasculature), and skeletal (lumbar hemi-vertebrae) malformations. Other skeletal anomalies included unossified forelimb metacarpals and phalanx, misaligned or unossified caudal vertebrae, supernumerary lumbar vertebra, bipartite or unilateral ossification of the lumbar centrum, and bipartite, incomplete, unilateral or non-ossification of the thoracic centrum at ≥ 0.1 mg/kg/day. In a fertility and early embryonic developmental study in rats, embryo resorption and post-implantation loss were increased, resulting in decreased viable fetuses, at 0.5 mg/kg/day (0.19 times the clinical exposure at MRHD based on AUC).
Use in lactation. It is unknown whether Fruzaqla or its metabolites are excreted in human milk. A risk to newborns/infants cannot be excluded. Breast-feeding should be discontinued during treatment with Fruzaqla and for at least 2 weeks after the last dose.
4.7 Effects on Ability to Drive and Use Machines
Studies to evaluate the effects of Fruzaqla on the ability to drive or operate machinery have not been conducted. Fatigue is very common amongst patients taking Fruzaqla (see Section 4.8).
4.8 Adverse Effects (Undesirable Effects)
Summary of the safety profile. The pooled safety population described in Section 4.4 reflects exposure to Fruzaqla as a single agent in 911 patients with mCRC who received at least one dose of Fruzaqla at the recommended dosage across multiple studies: 781 patients from three randomised, placebo-controlled studies (FRESCO-2 [2019-013-GLOB1], FRESCO [2013-013-00CH1] and 2012-013-00CH1); 124 patients from three open-label studies (2009-013-00CH1, 2012-013-00CH3 and 2015-013-00US1); and 6 patients from an open-label lead-in cohort of FRESCO-2. Amongst the 911 patients who received Fruzaqla, the median exposure was 3.7 months; 23% were exposed for 6 months or longer and 3.5% were exposed for greater than one year. The median age was 60 years (range: 23 to 82) and 34% were 65 years of age or older.
Tabulated list of adverse reactions. FRESCO-2 study. The safety of Fruzaqla was evaluated in FRESCO-2, a randomised, double-blind, placebo-controlled study (see Section 5.1). Patients received either Fruzaqla 5 mg daily for the first 21 days of each 28-day cycle plus best supportive care (BSC) (n=456) or matching placebo plus BSC (n=230).
The median duration of therapy with Fruzaqla was 3 months (range: 0.3 to 19.1 months).
Serious adverse reactions occurred in 38% of patients treated with Fruzaqla. Serious adverse reactions in ≥ 2% of patients treated with Fruzaqla included haemorrhage (2.2%) and gastrointestinal perforation (2.0%). Fatal adverse reaction(s) occurred in 14 (3.1%) patients who received Fruzaqla. Fatal adverse reactions occurring in ≥ 2 patients include pneumonia (n=3), sepsis/septic shock (n=2), and hepatic failure/encephalopathy (n=2).
Adverse reactions leading to treatment discontinuation occurred in 20% of patients treated with Fruzaqla. Adverse reactions leading to treatment discontinuations of Fruzaqla in ≥ 1% of patients were asthenia and gastrointestinal perforation.
Dose interruptions of Fruzaqla due to an adverse reaction occurred in 47% of patients. Adverse reactions leading to dose interruptions of Fruzaqla in ≥ 2% of patients were PPE, proteinuria, asthenia, abdominal pain, hypertension, vomiting, and diarrhoea.
Dose reductions of Fruzaqla due to an adverse reaction occurred in 24% of patients. Adverse reactions leading to dose reductions of Fruzaqla in ≥ 2% of patients were PPE, hypertension and asthenia.
Table 3 summarises the adverse reactions in FRESCO-2.
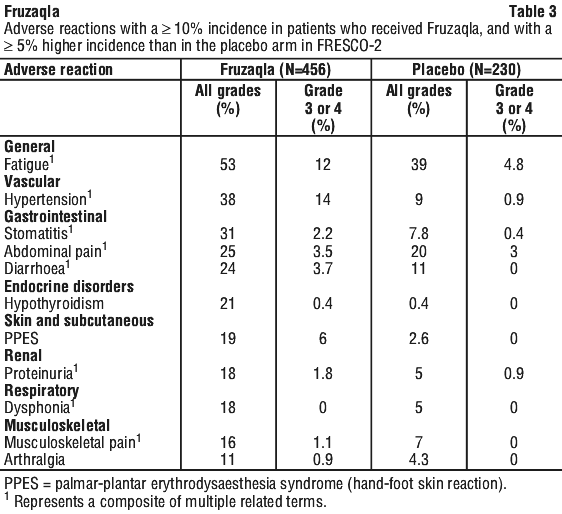
Table 4 describes selected laboratory abnormalities observed in FRESCO-2.
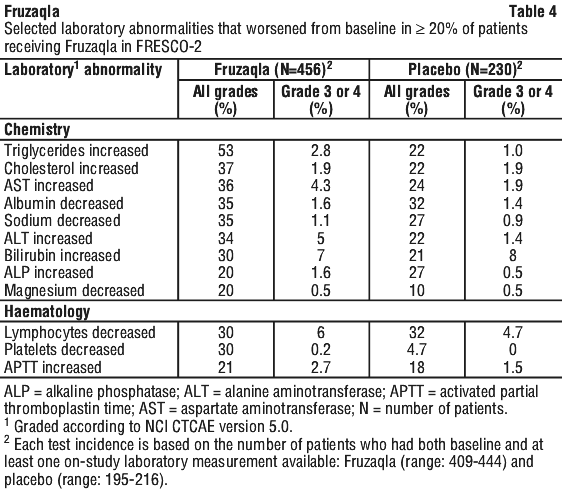
FRESCO study. The safety of Fruzaqla was generally similar in a Chinese population who had not previously received regorafenib or trifluridine/tipiracil who took part in an earlier, randomised, double-blind, placebo-controlled study, FRESCO (see Section 5.1). The median duration of therapy with Fruzaqla in FRESCO was 3.68 months (range: 0.3 to 22.1 months).
Serious adverse reactions occurred in 15% of patients treated with Fruzaqla. Serious adverse reactions in ≥ 2% of patients included intestinal obstruction (2.9%) and haemorrhage (2.2%). Fatal adverse reaction(s) occurred in 7 (2.5%) patients who received Fruzaqla including cerebral infarction (n=1), gastrointestinal haemorrhage (n=1), haemoptysis (n=1), bacterial infection (n=1), lung/lower respiratory infection (n=2), and multiple organ dysfunction (n=1).
Adverse reactions leading to treatment discontinuation occurred in 15% of patients who received Fruzaqla. Adverse reactions leading to treatment discontinuations of Fruzaqla in ≥ 1% were intestinal obstruction, proteinuria and hepatic function abnormalities.
Dose interruptions of Fruzaqla due to an adverse reaction occurred in 35% of patients. Adverse reactions leading to dose interruptions of Fruzaqla in ≥ 2% of patients were PPE, proteinuria, platelet count decreased, ALT increased, hypertension, and diarrhoea.
Dose reductions of Fruzaqla due to an adverse reaction occurred in 24% of patients. Adverse reactions leading to dose reduction of Fruzaqla in ≥ 2% of patients were PPE, proteinuria, and hypertension.
In FRESCO, proteinuria was very common in both the Fruzaqla arm (55%, including 4.7% incidence of grade 3-4 events) and the placebo arm (30%, with no grade 3 or 4 events). Haemorrhages were reported in 28% of the Fruzaqla arm (including Grade 3-4 events in 1%) and 14% of the placebo arm in FRESCO. PPES was also very common in FRESCO, occurring in 49% of patients who received Fruzaqla, including 11% who experienced grade 3-4 PPES, whilst there was an incidence of 2.9% (with no grade 3-4 events) in the placebo arm. Common reactions were otherwise similar in FRESCO to FRESCO-2.
Other clinically important adverse reactions (all grades) that occurred in < 10% of patients treated with Fruzaqla in FRESCO included urinary tract infection (9%), rash (9%), upper respiratory tract infection (4.7%), proctalgia (3.6%), pneumonia (2.9%), and gastrointestinal perforation or fistula (2.2%).
Laboratory abnormalities were similar in FRESCO to FRESCO-2. Creatinine increase from baseline occurred in 87% (0.7% Grade 3-4) of the Fruzaqla arm and 75% (1.5% Grade 3-4) of the placebo arm. Calcium decrease occurred in 25% (0.4% Grade 3-4) of the Fruzaqla arm and 13% (0% Grade 3-4) of the placebo arm. Platelet decrease was notably more common with Fruzaqla than placebo, similarly to in FRESCO-2.
Other important laboratory abnormalities (all grades) that occurred in < 20% of patients treated with Fruzaqla included pancreatic enzymes increased (4.3%).
Description of selected adverse reactions. Data for the following selected adverse reactions are based on patients who received at least 1 dose (5 mg) of fruquintinib (5 mg once daily 3 weeks on/1 week off) across three randomised placebo-controlled studies (2012-013-00CH1; 2013-013-00CH1/ FRESCO; 2019-013-GLOB1/FRESCO-2). The management guidelines for these adverse reactions are described in Section 4.4.
Hypertension. Amongst 781 patients with mCRC who received Fruzaqla in randomised trials, the incidence of hypertension was 47.1% (Grade ≥ 3 incidence: 18.4%) including three cases of hypertensive crisis. The incidence of hypertension was 11.8% (Grade ≥ 3 incidence: 1.3%) amongst 391 patients receiving placebo in the same studies.
Hypertension led to dose interruption in 3.1%; dose reduction in 3.7% and permanent discontinuation in 0.5% of patients receiving Fruzaqla.
Haemorrhage. Amongst 781 patients with mCRC who received Fruzaqla in randomised trials, the incidence of haemorrhage was 16.6% (Grade ≥ 3 incidence: 1.3%) including two fatal cases. The incidence of haemorrhage was 9.5% (Grade ≥ 3 incidence: 1%) amongst 391 patients receiving placebo in the same studies. Haemorrhage led to discontinuation in 0.9% of patients receiving Fruzaqla.
Infections. Amongst 781 patients with mCRC who received Fruzaqla in randomised trials, the incidence of infection was 18.2% (Grade ≥ 3 incidence: 4.6%). The incidence of infection was 12% (Grade ≥ 3 incidence: 3.6%) amongst 391 patients receiving placebo in the same studies. Infection led to discontinuation in 0.6% of patients receiving Fruzaqla.
Gastrointestinal (GI) perforation or fistula. Amongst 781 patients with mCRC who received Fruzaqla in randomised trials, the incidence of GI perforation events was 2.9% (including 1 fatal case in FRESCO-2). The incidence was 0.3% amongst patients receiving placebo in the same studies.
Hepatotoxicity. Amongst 781 patients with mCRC who received Fruzaqla in randomised trials, the incidence of liver function test (LFT) abnormalities was 47.5% (Grade ≥ 3 incidence: 5.2%). The incidence of LFT abnormalities was 32.7% (Grade ≥ 3 incidence: 2.8%) amongst 391 patients receiving placebo in the same studies. Fatal LFT abnormalities were reported in 0.3% of patients in the Fruzaqla arm and 0.8% in the placebo arm.
Proteinuria. Amongst 781 patients with mCRC who received Fruzaqla in randomised trials, the incidence of proteinuria was 32.9% (Grade ≥ 3 incidence: 2.8%). The incidence of proteinuria was 15.1% (Grade ≥ 3 incidence: 0.5%) amongst 391 patients receiving placebo in the same studies. Proteinuria led to dose interruption in 5.9%; dose reduction in 3.2% and permanent treatment discontinuation in 1.8% of patients receiving Fruzaqla.
PPES. Amongst 781 patients with mCRC who received Fruzaqla in randomised trials, the incidence of PPES was 32.7% (Grade ≥ 3 incidence: 8.5%). The incidence of PPES was 3.1% (Grade ≥ 3 incidence: 0.3%) amongst 391 patients receiving placebo in the same studies. PPES led to dose interruption in 6.4%, dose reduction in 6.3%, and permanent treatment discontinuation in 0.5% of patients treated with Fruzaqla.
Hypothyroidism. Amongst 781 patients with mCRC who received Fruzaqla in randomised trials, the incidence of hypothyroidism was 18.3% (Grade ≥ 3 incidence: 0.3%). The incidence of hypothyroidism was 1% (none Grade ≥ 3) amongst 391 patients receiving placebo in the same studies. No events led to dose reduction or discontinuation.
Pancreatic enzyme elevation. Amongst 781 patients with mCRC who received Fruzaqla in randomised trials, the incidence of pancreatic enzyme elevation was 4.1% (Grade ≥ 3 incidence: 1.9%). The incidence of pancreatic enzyme elevation was 1.0% (none Grade ≥ 3) amongst 391 patients receiving placebo in the same studies. Pancreatic enzyme elevation led to dose interruption in 0.8%; dose reduction in 0.1% and permanent discontinuation in 0.3% of patients receiving Fruzaqla.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
The highest dose of Fruzaqla studied in clinical studies was 6 mg per day. The effects of Fruzaqla overdose are unknown, and there is no known antidote for Fruzaqla overdose. In the event of an overdose, interrupt Fruzaqla, general supportive measures should be undertaken and observe until clinical stabilisation.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: Antineoplastic agents, protein kinase inhibitors, ATC code: L01EK04.
Mechanism of action. Fruquintinib is a small molecule tyrosine kinase inhibitor of vascular endothelial growth factor receptors (VEGFR) -1, -2, and -3 (with IC50 values of 33, 35, and 0.5 nanoM, respectively), with antitumor effects resulting from suppression of tumour angiogenesis.
VEGF-mediated endothelial cell proliferation, and tubular formation were inhibited by fruquintinib in vitro. In vitro and in vivo studies showed fruquintinib inhibited VEGF-induced VEGFR-2 phosphorylation. Fruquintinib was shown to inhibit tumour growth in tumour xenograft mouse models.
Cardiac electrophysiology. Significant prolongation of heart rate-corrected QT (QTc) interval (> 20 milliseconds) was not observed at the recommended dosage of fruquintinib (in FRESCO-2). A concentration-QT analysis (N=205) based on data from FRESCO-2 did not detect an association between fruquintinib plasma concentrations and changes in QTc interval from baseline.
Clinical trials. Clinical efficacy and safety. The clinical safety (see Section 4.8) and efficacy of Fruzaqla plus best supportive care (BSC) was evaluated in two randomised, placebo-controlled, double-blind, phase III studies (FRESCO-2 and FRESCO) in patients with mCRC who had previously received oxaliplatin and irinotecan-based chemotherapies.
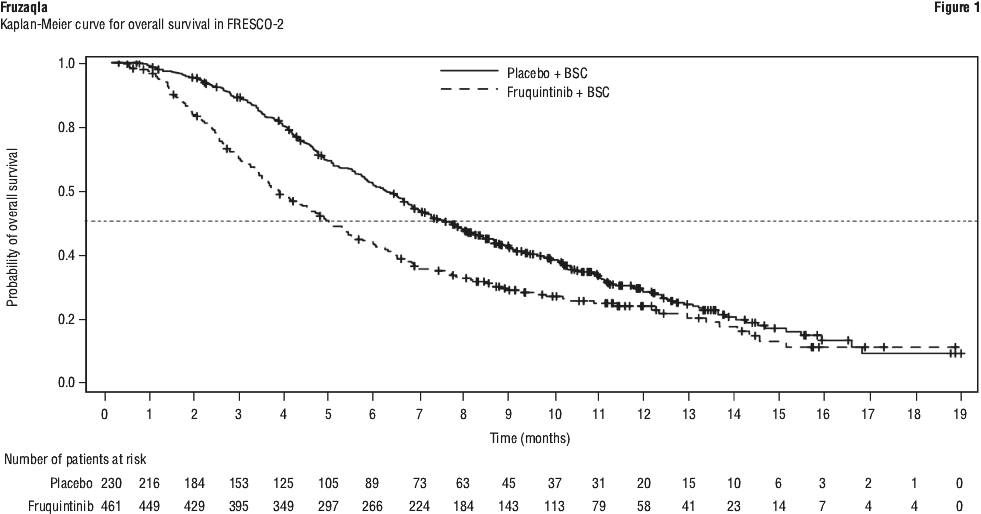
FRESCO-2 study. The efficacy of Fruzaqla was evaluated in FRESCO-2 (NCT04322539): an international, randomised, double-blind, placebo-controlled study that enrolled 691 patients with mCRC who had disease progression during or after prior treatment with fluoropyrimidine-, oxaliplatin-, and irinotecan-based chemotherapy; an anti-VEGF therapy; an anti-EGFR therapy (if RAS wild type); and trifluridine/tipiracil, regorafenib, or both. Patients with MSI-H or dMMR tumours must have previously received an immune checkpoint inhibitor, and patients with BRAF V600E mutant tumours must have previously received a BRAF inhibitor, if locally approved and available. Patients with an Eastern Cooperative Oncology Group (ECOG) performance status (PS) ≥ 2, left ventricular fraction ≤ 50%, systolic blood pressure > 140 mm Hg or diastolic blood pressure > 90 mm Hg, urine protein ≥ 1 g/24 h, or body weight < 40 kg were ineligible. Randomisation was stratified by prior receipt of trifluridine/tipiracil and regorafenib (trifluridine/tipiracil only vs. regorafenib only vs. both), RAS mutational status (wild-type vs. mutant) and duration of metastatic disease (≤ 18 months vs. > 18 months).
In addition to BSC, patients were randomised (2:1) to receive Fruzaqla 5 mg orally once daily (N=461) for the first 21 days of each 28-day cycle or placebo orally once daily (N=230). Treatment continued until disease progression or unacceptable toxicity. The primary efficacy endpoint was overall survival (OS) and an additional efficacy endpoint was progression-free survival (PFS) as determined by investigators according to Response Evaluation Criteria in Solid Tumours version 1.1 (RECIST v1.1).
Among the 691 randomised patients, the median age was 64 years (range: 25 to 86), with 47% ≥ 65 years of age. Male patients comprised 56%, 81% of patients were White, 43% had an ECOG PS of 0, 57% had an ECOG PS of 1, and 63% had RAS-mutant tumours. Eighteen percent of the patients were enrolled in North America, 72% in Europe, and 10% in Asia Pacific (Japan and Australia) region.
The median duration of metastatic disease was 39 months (range: 6 months to 16.1 years). The median number of prior lines of therapy for metastatic disease was 4 (range: 2 to 16). All patients had received prior treatment with fluoropyrimidine-, oxaliplatin-, and irinotecan-based chemotherapy. Additionally, 96% of patients had received prior anti-VEGF therapy, 39% had received prior anti-EGFR therapy, 52% had received trifluridine/tipiracil, 8% had received regorafenib, 39% had received both trifluridine/tipiracil and regorafenib, 4.6% had received immunotherapy, and 2.3% had received a BRAF inhibitor.
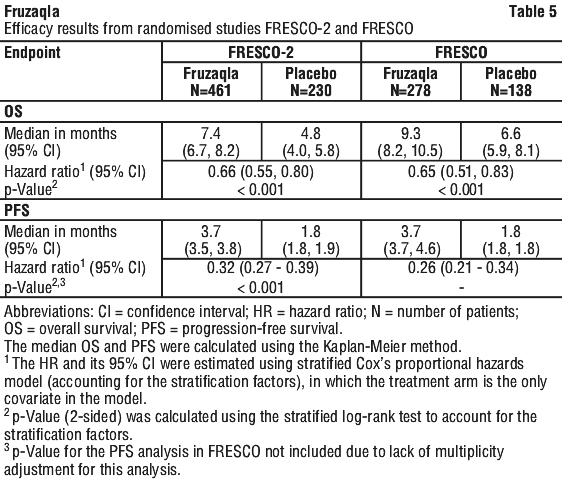
The addition of Fruzaqla to BSC resulted in a statistically significant improvement in OS and PFS compared to placebo plus BSC (see Table 5 and Figure 1).
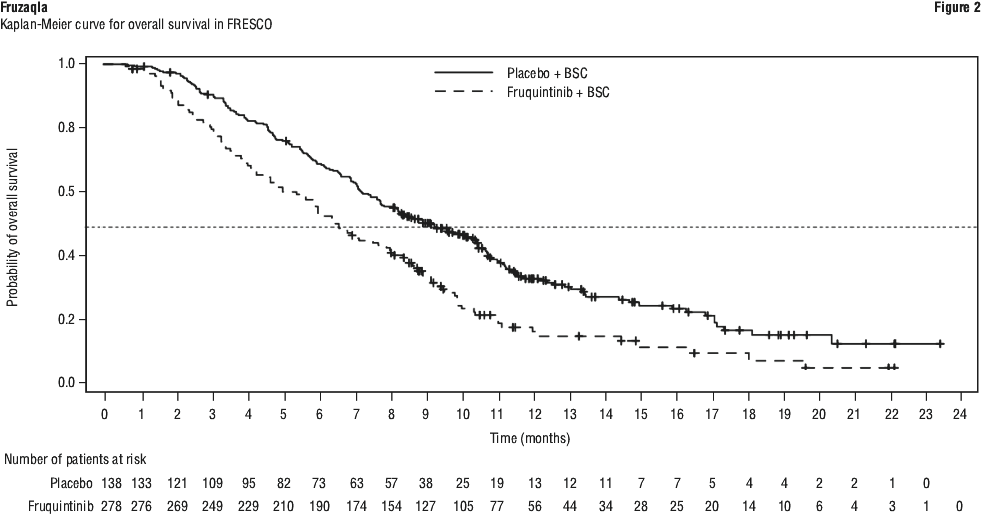
FRESCO study. The efficacy of Fruzaqla was initially evaluated in FRESCO (NCT02314819), a multicentre, randomised, double-blind, placebo-controlled study conducted in China that enrolled 416 patients with mCRC who had disease progression during or after prior treatment with fluoropyrimidine-, oxaliplatin-, or irinotecan-based chemotherapy. Patients with ECOG PS ≥ 2, left ventricular fraction ≤ 50%, systolic blood pressure > 140 mm Hg or diastolic blood pressure > 90 mm Hg, urine protein ≥ 1 g/24 h, and body weight < 40 kg were ineligible. Randomisation was stratified by prior use of VEGF inhibitor (yes vs. no) and KRAS gene status (wild-type vs. mutant).
In addition to BSC, patients were randomised (2:1) to receive Fruzaqla 5 mg orally once daily (N=278) for the first 21 days of each 28-day cycle or placebo (N=138). Treatment continued until disease progression or unacceptable toxicity. The primary efficacy endpoint was OS. Secondary efficacy endpoints included PFS as determined by investigators according to RECIST v1.
Among the 416 randomised patients, the median age was 56 years (range: 23 to 75), with 19% ≥ 65 years of age. Male patients comprised 61%, all patients were Asian, 27% had an ECOG PS of 0, 73% had an ECOG PS of 1, and 44% had KRAS mutant tumours.
The median number of prior lines of therapy for metastatic disease was 2 (range: 2 to 3). All patients had received prior treatment with fluoropyrimidine-, oxaliplatin- and irinotecan-based chemotherapy. Additionally, 30% of patients had received prior anti-VEGF therapy, and 14% had received prior anti-EGFR therapy.
The addition of Fruzaqla to BSC resulted in a statistically significant improvement in OS compared to placebo plus BSC (see Table 5 and Figure 2).
5.2 Pharmacokinetic Properties
Absorption. At the recommended dosage (Fruzaqla 5 mg once daily orally for the first 21 days of each 28-day cycle), fruquintinib exposure (maximum concentration [Cmax] and area under the concentration-time curve for the dosing interval [AUC0-24h]) is dose-proportional across the dose range of 1 to 6 mg (0.2 to 1.2 times the recommended dosage). The median (range) time from oral administration to Cmax (Tmax) is 2 (0-26) hours. Steady state fruquintinib serum concentration is achieved after 14 days, with a mean accumulation ratio (based on AUC0-24h) of 4 fold. At the recommended dose, the geometric mean (%CV) steady-state fruquintinib Cmax and AUC0-24h are 300 nanogram/mL (28%) and 5880 nanogram*h/mL (29%), respectively.
Effect of food. No clinically significant differences in fruquintinib pharmacokinetics were observed following administration of a high-fat meal. Fruzaqla can be administered with or without food.
Distribution. The mean (SD) apparent volume of distribution of fruquintinib is approximately 46 (13) L. Plasma protein binding of fruquintinib is approximately 95%.
Metabolism. Fruquintinib is primarily eliminated by CYP450 and non-CYP450 (i.e. sulfation and glucuronidation) metabolism. CYP3A and to a lesser extent CYP2C8, CYP2C9, and CYP2C19 are the CYP450 enzymes involved in fruquintinib metabolism.
Excretion. The fruquintinib mean (SD) elimination half-life is approximately 42 (11) hours and the apparent clearance is 14.8 (4.4) mL/min.
Following oral administration of a 5 mg radiolabelled fruquintinib dose, approximately 60% of the dose was recovered in urine (0.5% unchanged), and 30% of the dose was recovered in faeces (5% unchanged).
Pharmacokinetics in special populations. No clinically significant differences in the pharmacokinetics of fruquintinib were observed based on age (18 to 82 years), sex, race/ethnicity, body weight (48 to 108 kg), renal impairment (CrCL 15 to 89 mL/min), or mild hepatic impairment (either total bilirubin ≤ ULN and AST > ULN; or total bilirubin > 1 to 1.5 x ULN and any AST).
The effect of moderate to severe hepatic impairment (total bilirubin > 1.5 x ULN and any AST) on fruquintinib pharmacokinetics is unknown.
5.3 Preclinical Safety Data
In repeat dose and reproductive studies, toxicity was observed at fruquintinib average plasma concentrations below the expected human therapeutic concentrations.
Genotoxicity. No evidence of genotoxicity was observed in in vitro (bacterial reverse mutation assay and in vitro chromosomal aberrations assay in Chinese hamster ovary cells) and in vivo (rat micronucleus test with inclusion of an alkaline comet assay) studies.
Carcinogenicity. Carcinogenicity studies have not been conducted with fruquintinib.
Repeat dose toxicity. In repeat-dose toxicity studies in rats and dogs, the main target organ effects were identified in the gastrointestinal tract, hepatobiliary system, immune system, skeletal system, kidneys, hematopoietic system and adrenal gland at subclinical exposure levels or comparable exposure levels at the MRHD. All findings were reversible after a 4-week period without treatment, apart from the skeletal system (broken/lost teeth) and adrenal gland.
6 Pharmaceutical Particulars
6.1 List of Excipients
Fruzaqla 1 mg hard capsules. Inactive ingredients include maize starch, microcrystalline cellulose and purified talc. Capsule shells contain gelatin, titanium dioxide, tartrazine and sunset yellow FCF. The capsules are imprinted with TekPrint SW-9008 Black Ink (ARTG PI No 2328).
Fruzaqla 5 mg hard capsules. Inactive ingredients include maize starch, microcrystalline cellulose and purified talc. Capsule shells contain gelatin, titanium dioxide, brilliant blue FCF and allura red AC. The capsules are imprinted with TekPrint SW-9008 Black Ink (ARTG PI No 2328).
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C. Protect from moisture. Keep bottle tightly closed. Do not remove desiccant cartridge from the bottle.
6.5 Nature and Contents of Container
Fruzaqla 1 mg and 5 mg hard capsules.
High density polyethylene (HDPE) bottle with a polypropylene (PP) child resistant closure, containing 21 capsules of Fruzaqla and desiccant cartridge. The HDPE bottle is packaged in a carton.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Fruquintinib is a highly selective tyrosine kinase inhibitor.
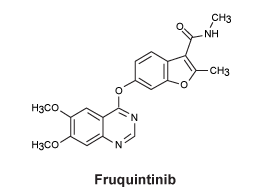
Chemical structure. The chemical name for fruquintinib is 6-[(6,7-dimethoxyquinazolin-4-yl)oxy]-N,2-dimethyl-1-benzofuran-3- carboxamide. The molecular formula is C21H19N3O5, which corresponds to a molecular weight of 393.39 g/mol. The chemical structure is shown:
CAS number. 1194506-26-7.
7 Medicine Schedule (Poisons Standard)
Prescription Only Medicine (Schedule 4).
Date of First Approval
02 October 2024
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.