Ganirelix Lupin
Brand Information
| Brand name | Ganirelix Lupin |
| Active ingredient | Ganirelix |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Ganirelix Lupin.
Summary CMI
Ganirelix Lupin
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Ganirelix Lupin?
Ganirelix Lupin is used together with other medications to regulate hormone response in women undergoing Assisted Reproductive Technology (ART) such as in vitro fertilisation (IVF). It works by preventing women from ovulating (releasing an egg from the ovary) too soon during stimulation of their ovaries to produce a mature egg.
For more information, see Section 1. Why am I using Ganirelix Lupin? in the full CMI.
2. What should I know before I use Ganirelix Lupin?
Do not use if you have ever had an allergic reaction to ganirelix or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Ganirelix Lupin? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Ganirelix Lupin and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Ganirelix Lupin?
Your doctor will decide the dose suitable for you based on your condition and response.
More instructions can be found in Section 4. How do I use Ganirelix Lupin? in the full CMI.
5. What should I know while using Ganirelix Lupin?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Ganirelix Lupin? in the full CMI.
6. Are there any side effects?
The common side effects are: redness; pain or swelling at injection site, headache, nausea, tiredness.
The serious side effects are: shortness of breath, wheezing, difficulty breathing or a tight feeling in your chest; swelling of the face, lips, tongue and/or throat that may cause difficulty in breathing or swallowing (also called as angioedema and/or anaphylaxis), or other parts of the body; rash, itching, hives (urticaria); or flushed, red skin.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Ganirelix Lupin
Active ingredient(s): ganirelix
Consumer Medicine Information (CMI)
This leaflet provides important information about using Ganirelix Lupin. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Ganirelix Lupin.
Where to find information in this leaflet:
1. Why am I using Ganirelix Lupin?
2. What should I know before I use Ganirelix Lupin?
3. What if I am taking other medicines?
4. How do I use Ganirelix Lupin?
5. What should I know while using Ganirelix Lupin?
6. Are there any side effects?
7. Product details
1. Why am I using Ganirelix Lupin?
Ganirelix Lupin contains the active ingredient ganirelix. Ganirelix Lupin works by preventing women from ovulating (releasing an egg from the ovary) too soon during stimulation of their ovaries to produce a mature egg.
Ganirelix Lupin is used together with other medications to regulate hormone response in women undergoing Assisted Reproductive Technology (ART) such as in vitro fertilisation (IVF).
2. What should I know before I use Ganirelix Lupin?
Warnings
Do not use Ganirelix Lupin if:
- you are allergic (hypersensitive) to ganirelix or to any other components of Ganirelix Lupin, including the ingredients listed at the end of this leaflet and/or synthetic rubber.
Always check the ingredients to make sure you can use this medicine. - you are allergic to any other similar medicines.
- you are pregnant.
- you are breastfeeding.
- you have moderate to severe kidney or liver disease.
- the solution is not clear or colourless.
- the expiry date on the pack has passed.
- the package shows any signs of tampering.
Check with your doctor if you:
- have allergies to any other medicines, substances such as foods, preservatives, or dyes.
- are currently experiencing allergic symptoms.
- take any medicines for any other condition.
- have any other medical conditions.
Compared to natural conception, the frequency of multiple pregnancies and births is increased in patients undergoing assisted reproductive techniques. Discuss the risk of multiple pregnancies and births with your doctor.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Ganirelix Lupin is not to be used during pregnancy.
Check with your doctor if you are pregnant or intend to become pregnant.
Ganirelix Lupin should not be used by breastfeeding women. It is not known whether ganirelix is excreted into animal or human breast milk.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Ganirelix Lupin.
4. How do I use Ganirelix Lupin?
How much to use
- The usual dose is the contents of one pre-filled syringe of Ganirelix Lupin once a day on specific days of the menstrual cycle. Your doctor will tell you when to inject Ganirelix Lupin.
- You may notice air bubbles in the pre-filled syringe.
This is expected, and removal of the air bubbles is not needed. - Follow the instructions provided and use Ganirelix
Lupin until your doctor tells you to stop.
When to use Ganirelix Lupin
- Ganirelix Lupin should be injected subcutaneously (under the skin) once daily, starting from day 5 or day 6 of follicle stimulating hormone (FSH)administration depending on the level of ovarian response.
How to use Ganirelix Lupin
- Treatment with Ganirelix Lupin should be started under the supervision of a fertility specialist.
- Ganirelix Lupin is given as a subcutaneous (under the skin) injection in the thigh or stomach/abdomen.
- The injection site should be changed every day to lessen possible injection site reactions.
- If your doctor or nurse decides you can give the injections yourself, they will teach you the injection technique.
Do not attempt self-injection until you are sure of how to do it. Follow all instructions given to you by your doctor or nurse carefully.
Follow these steps for self-injection:
- Prepare the injection site:
- Wash your hands thoroughly with soap and water.
- Swab the injection site with a disinfectant to remove any surface bacteria. Clean about 5 cm around the point where the needle will go in.
- Let the disinfectant dry for at least one minute before proceeding.
- Open the outer pack and plastic container inside:
- While waiting for the disinfectant to dry, open the Ganirelix Lupin pack and remove the plastic container.
- Carefully open the plastic container and remove the Ganirelix Lupin syringe.
- You will see the needle is already attached, covered by a black needle shield.
- Prepare the syringe for injection:
- Remove the needle shield and discard it in a Sharps Container.
- You are now ready to inject Ganirelix Lupin.
- Inserting the needle and injecting
- Ganirelix Lupin is injected in either the thigh or the stomach/abdomen, usually near the navel.
- Pinch up a large bit of skin between your finger and thumb.
- Insert the needle at the base of the pinched-up skin at an angle of 45 degrees to 90 degrees to the skin surface.
- Gently draw back on the plunger to see if the needle is inserted correctly.
- If any blood appears in the syringe, the needle is not inserted correctly so do not inject Ganirelix Lupin. Remove the needle, cover the injection site with a sterile swab and dispose of the syringe in a Sharps Container. Start again with a new syringe.
- If the needle has been inserted correctly, press down on the plunger slowly and steadily until all the solution has been injected.
- Vary the injection site each time to minimise local irritation.
- Removing the needle:
- Pull the needle out of the skin quickly and apply pressure to the site with a swab containing disinfectant.
- Dispose of the syringe (with the attached needle) in a Sharps Container.
- Use the syringe only once and then dispose of it in a Sharps Container.
If you forget to use Ganirelix Lupin
If you forget an injection, contact your doctor or IVF clinic immediately for advice.
Do not inject a double dose to make up for the forgotten dose.
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
If you use too much Ganirelix Lupin
If you think that you have used too much Ganirelix Lupin, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26); or - contact your doctor; or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using Ganirelix Lupin?
Things you should do
- Keep all your doctor's appointments so that your progress can be checked.
Your doctor will want to follow the developing eggs inside the ovaries by doing an ultrasound examination and measuring hormones in your blood. - Remind any doctor, dentist or pharmacist you visit that you are using Ganirelix Lupin.
- Tell the hospital doctor that you are using Ganirelix Lupin if you need to have an operation or go to hospital in an emergency.
If you are about to be started on any new medicine, tell your doctor or pharmacist that you are using Ganirelix Lupin.
Call your doctor straight away if you:
- have severe pelvic pain, nausea, vomiting and weight gain. These are early warning signs of Ovarian Hyperstimulation Syndrome (OHSS). Other symptoms of OHSS can include:
- indigestion;
- diarrhoea;
- shortness of breath;
- reduced amounts of urine;
- painful breasts.
OHSS is a possible complication of hormonal stimulation of the ovaries.
Remind any doctor, dentist or pharmacist [add other health professionals as appropriate] you visit that you are using Ganirelix Lupin.
Things you should not do
- Do not stop using this medicine suddenly without telling your doctor.
- Do not change the dose unless your doctor tells you to. Changing your dose without telling your doctor can increase your risk of unwanted side effects or can prevent the drug from working properly.
- Do not give this medicine to anyone else, even if they have the same condition as you.
- Do not use this medicine to treat any other complaints.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Ganirelix Lupin affects you.
No effects on ability to drive or use of machines have been observed.
Looking after your medicine
- Store below 25°C in the original package.
- Do not put in the freezer as the syringe may break.
- Keep the syringe in the outer carton to protect it from light.
Follow the instructions on the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink; or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Alternatively, dispose of your Ganirelix Lupin syringe and needle safely into a Sharps Container.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Skin-related:
| Speak to your doctor or IVF clinic if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Respiratory-related:
| Call your doctor or IVF clinic straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Other side effects
Other side effects are known to occur with Assisted reproductive Technology (ART) procedures. These may include:
- Ovarian Hyperstimulation Syndrome or OHSS (enlargement of the ovaries). Since overstimulation can occur rapidly you must contact your doctor if you experience any of the following:
- pain in the abdomen or pelvis, indigestion, nausea, vomiting, weight gain, shortness of breath, reduced amounts of urine, diarrhoea and painful breasts. - vaginal bleeding.
- miscarriage.
- ectopic pregnancy. The incidence of ectopic pregnancies (embryo implanted outside the womb) may be increased in women undergoing ART. Your doctor will perform an ultrasound scan early during pregnancy to confirm that a pregnancy is intrauterine (in the womb).
These side effects are probably unrelated to treatment with Ganirelix Lupin.
The incidence of congenital malformations (a physical defect present in a baby at birth) after ART may be slightly higher than after spontaneous conceptions. The slightly higher incidence is thought to be related amongst other factors or characteristics of the patients undergoing fertility treatment (eg. age of the female, sperm characteristics, etc.) and to the higher incidence of multiple gestations after ART. The incidence of congenital malformations after ART using Ganirelix Lupin is not different from that after using other GnRH analogues during ART.
The needle shield of Ganirelix Lupin contains synthetic rubber which comes in contact with the product and may cause allergic reactions.
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. PRODUCT DETAILS
This medicine is only available with a doctor's prescription.
What Ganirelix Lupin contains
| Active ingredient (main ingredient) | Ganirelix |
| Other ingredients (inactive ingredients) | Glacial acetic acid Mannitol Water for injections |
| Potential allergens | Synthetic rubber |
Do not take this medicine if you are allergic to any of these ingredients.
What Ganirelix Lupin looks like
Ganirelix Lupin is supplied as a clear, colourless solution in a single use, pre-filled syringe with a fixed needle closed by a needle shield (AUST R 442691).
Who distributes Ganirelix Lupin
Generic Health Pty Ltd
Suite 2, Level 2
19-23 Prospect Street
Box Hill, VIC, 3128
Australia ghinfo@generichealth.com.au
ghinfo@generichealth.com.au +61 3 9809 7900
+61 3 9809 7900 www.generichealth.com.au
www.generichealth.com.au
This leaflet was prepared in February 2026.
Brand Information
| Brand name | Ganirelix Lupin |
| Active ingredient | Ganirelix |
| Schedule | S4 |
MIMS Revision Date: 01 June 2025
Notes
Distributed by Generic Health Pty Ltd
1 Name of Medicine
Ganirelix acetate.
2 Qualitative and Quantitative Composition
Each pre-filled syringe contains 250 micrograms ganirelix (as acetate) in 0.5 mL.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Solution for injection.
Ganirelix Lupin is presented as a sterile, clear and colourless aqueous solution intended for subcutaneous administration.
4 Clinical Particulars
4.1 Therapeutic Indications
For the prevention of premature luteinisation and ovulation in patients undergoing controlled ovarian stimulation, followed by oocyte pick-up and assisted reproductive techniques.
4.2 Dose and Method of Administration
Ganirelix Lupin should only be prescribed by a specialist experienced in the treatment of fertility.
Dosage. Ganirelix Lupin is used to prevent premature luteinising hormone (LH) surges in patients undergoing controlled ovarian hyperstimulation (COH). COH with a follicle stimulating hormone (FSH) may start at day 2 or 3 of menses. Ganirelix Lupin (0.25 mg) should be injected subcutaneously once daily, starting from day 5 or day 6 of FSH administration depending on the level of ovarian response. Ganirelix Lupin should not be mixed with the FSH but both preparations should be administered approximately at the same time. Daily treatment with Ganirelix Lupin should be continued up to the day that sufficient follicles of adequate size are present. Final maturation of follicles can be induced by administering human chorionic gonadotropin (hCG). Because of the half-life of ganirelix, the time between two Ganirelix Lupin injections and between the last Ganirelix Lupin injection and the hCG injection should not exceed 30 hours, as otherwise a premature LH surge may occur.
Method of administration. Inspect the solution before use. It must only be used if it is clear and without particulate matter.
Ganirelix Lupin should be administered subcutaneously. The injection site should be varied to prevent lipoatrophy. The patient or their partner may perform the injections of Ganirelix Lupin themselves, provided that they are adequately instructed and have access to expert advice. Air bubble(s) may be seen in the pre-filled syringe. This is expected, and removal of the air bubble(s) is not needed.
4.3 Contraindications
Hypersensitivity to the active substance or to any of the components, including synthetic rubber (see Section 6.1 List of Excipients; Section 6.5 Nature and Contents of Container).
Hypersensitivity to gonadotropin releasing hormone (GnRH) or any other GnRH analogue.
Pregnancy or lactation.
Moderate to severe renal impairment and hepatic impairment.
4.4 Special Warnings and Precautions for Use
Special care should be taken in women with signs and symptoms of active allergic conditions. Cases of hypersensitivity reactions (both generalised and local) have been reported with ganirelix as early as with the first dose during post-marketing surveillance. These events have included anaphylaxis (including anaphylactic shock), angioedema, and urticaria (see Section 4.8 Adverse Effects (Undesirable Effects)). If a hypersensitivity reaction is suspected, Ganirelix Lupin should be discontinued and appropriate treatment administered. Use of ganirelix in patients with active allergic symptoms has not been investigated. Administration of Ganirelix Lupin is not advised to patients with currently severe allergic symptoms. Patients should be advised to contact the attending physician before administering the next injection in case a general or an extensive local allergic reaction occurs.
The needle shield of Ganirelix Lupin contains synthetic rubber which comes into contact with this product and may cause allergic reactions (see Section 4.3 Contraindications; Section 6.5 Nature and Contents of Container).
Ovarian hyperstimulation syndrome (OHSS) may occur during or following ovarian stimulation. OHSS must be considered an intrinsic risk of gonadotropin stimulation. OHSS should be treated symptomatically, e.g. with rest, intravenous infusion of electrolyte solutions or colloids and heparin.
Since infertile women undergoing assisted reproduction, and particularly in vitro fertilisation (IVF), often have tubal abnormalities, the incidence of ectopic pregnancies might be increased. Early ultrasound confirmation that a pregnancy is intrauterine is therefore important.
The safety and efficacy of ganirelix have not been established in women weighing less than 50 kg or more than 90 kg.
Congenital abnormalities. The incidence of congenital malformations after assisted reproductive technologies (ART) may be slightly higher than after spontaneous conceptions. This slightly higher incidence is thought to be related amongst other factors, to differences in parental characteristics (e.g. maternal age, sperm characteristics) and by the higher incidence of multiple gestations after ART. In clinical trials investigating more than 1,000 newborns it has been demonstrated that the incidence of congenital malformation in children born after COH treatment using ganirelix is comparable with that reported after COH treatment using a GnRH agonist.
Use in hepatic impairment. See Section 4.3 Contraindications.
Use in renal impairment. See Section 4.3 Contraindications.
Use in the elderly. No data available.
Paediatric use. There is no relevant use of Ganirelix Lupin in the paediatric population.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
In clinical studies, interactions of ganirelix with other medicines have not been investigated. Therefore, interactions with commonly used medicinal products can not be excluded.
In the absence of incompatibility studies, this medicinal product must not be mixed with other medicinal products.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Ganirelix treatment of female rats resulted in reversible impairment of mating and fertility at a subcutaneous dose of 2.5 micrograms/kg/day, and reversible cessation of mating was seen in males treated with a subcutaneous dose of 0.1 mg/kg/day.
Use in pregnancy. (Category D)
Ganirelix Lupin is not intended to be used during pregnancy (see Section 4.3 Contraindications). No clinical data on exposed pregnancies are available.
Studies in animals have indicated that ganirelix increased the incidence of total foetal resorptions when administered to pregnant rats and rabbits during the period of organogenesis, at respective doses of 10 micrograms/kg/day and 30 micrograms/kg/day (approximately 0.4 and 3 times the human dose, based on body surface area). The effects on foetal resorption are logical consequences of the alteration in hormonal levels brought about by the antigonadotrophic properties of ganirelix and could result in foetal loss in humans. There was no increase in foetal abnormalities. No treatment related changes in fertility, physical or behavioural characteristics were observed in the offspring of female rats treated with ganirelix during pregnancy and lactation.
Use in lactation. Ganirelix Lupin should not be used by lactating women (see Section 4.3 Contraindications). It is not known whether ganirelix is excreted into animal or human breast milk. Subcutaneous ganirelix doses of 2.5 micrograms/kg/day given to lactating rats did not result in impairment of postnatal development of the offspring.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
General disorders and administrative site conditions. Ganirelix Lupin may cause a skin reaction at the site of injection (in 10% - 15% of the patients moderate or severe redness with or without swelling was reported), which normally disappears within 4 hours after administration. Malaise was reported in 0.3% of patients.
Immune system disorders. Very rarely, post-marketing cases of hypersensitivity reactions (including rash, facial swelling, dyspnoea, anaphylaxis (including anaphylactic shock), angioedema, and urticaria) have been reported, as early as with the first dose among patients administered ganirelix.
Nervous system disorders. Headache (0.4%).
Gastrointestinal disorders. Nausea (0.5%).
Other. Other reported adverse events are related to the COH treatment for ART than to ganirelix (e.g. pelvic pain, abdominal distension, OHSS, ectopic pregnancy and spontaneous abortion).
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Overdosage in humans may result in a prolonged duration of action. In case of overdose, Ganirelix Lupin treatment should be (temporarily) discontinued.
No data on acute toxicity of ganirelix in humans are available. Clinical studies with subcutaneous administration of ganirelix at single doses up to 12 mg did not show systemic side effects. Clinical signs of systemic toxicity including collapse, laboured respiration and inactivity in rats or facial flushing in monkeys were observed after intravenous administration of ganirelix at 2.0 mg/kg and 3.0 mg/kg, respectively. Blood pressure was also reduced by about 50% in rats treated with an intravenous ganirelix dose of 0.9 mg/kg.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: anti-gonadotropin releasing hormone; ATC code: H01CC01.
Mechanism of action. Ganirelix is a GnRH antagonist, which modulates the hypothalamic-pituitary-gonadal axis by competitively binding to the GnRH receptors in the pituitary gland. As a result a rapid, reversible suppression of endogenous gonadotropins occurs, without initial stimulation as induced by GnRH agonists. The inhibitory effect of ganirelix on the release of LH is more pronounced than on FSH. When LH has started to rise prior to the first administration of ganirelix, a premature LH surge can still be prevented.
Important features of the GnRH-antagonist regimen. Within a few hours suppression of gonadotropin release due to GnRH receptor blockade.
Ganirelix treatment is restricted to those days when a premature LH surge may occur. Therefore, the overall duration of treatment is only several days.
Less suppression of endogenous FSH and therefore less FSH required.
Recovery of pituitary functioning within two days following discontinuation of the treatment.
The competitive mode of action of ganirelix may allow the administration of a GnRH agonist instead of hCG to trigger ovulation, which is especially relevant for patients at risk of developing OHSS.
Generally, estradiol levels are lower (though remaining above natural cycle levels) compared to the relatively high levels in the agonist regimen.
Clinical trials. The efficacy of ganirelix was established in three clinical studies. In these studies, the administration of exogenous recombinant FSH [Puregon (follitropin beta for injection)] was initiated on the morning of day 2 or 3 of a natural menstrual cycle. Ganirelix was administered on the morning of day 7 or 8 (day 6 of recombinant FSH administration). The dose of recombinant FSH administered was adjusted according to individual responses starting on the day of initiation of ganirelix. Both recombinant FSH and ganirelix were continued daily until at least three follicles were 17 mm or greater in diameter at which time hCG [Pregnyl (chorionic gonadotropin for injection)] was administered. Following hCG administration, ganirelix and recombinant FSH administration were discontinued. Oocyte retrieval, followed by IVF or intracytoplasmic sperm injection (ICSI), was subsequently performed.
In a multi-centre, double blind, randomised, dose finding study (protocol 38602), ganirelix doses ranging from 62.5 micrograms to 2000 micrograms and recombinant FSH were administered to 332 patients undergoing COH for IVF. The results of the selected dose (250 micrograms) are summarised in Table 1.
Two multi-centre, open-label, randomised studies (protocol 38607 and 103-001) were conducted in which follicular phase treatment with ganirelix 250 micrograms was studied using a GnRH agonist as a reference treatment. A total of 661 subjects were treated with ganirelix by subcutaneous injection once daily starting on day 6 of recombinant FSH treatment. Recombinant FSH was maintained at 150 IU or 225 IU in the 38607 and 103-001 study respectively for the first 5 days of ovarian stimulation and was then adjusted by the investigator on the sixth day of gonadotropin use according to individual responses. The results for the ganirelix arm are summarised in Table 1.
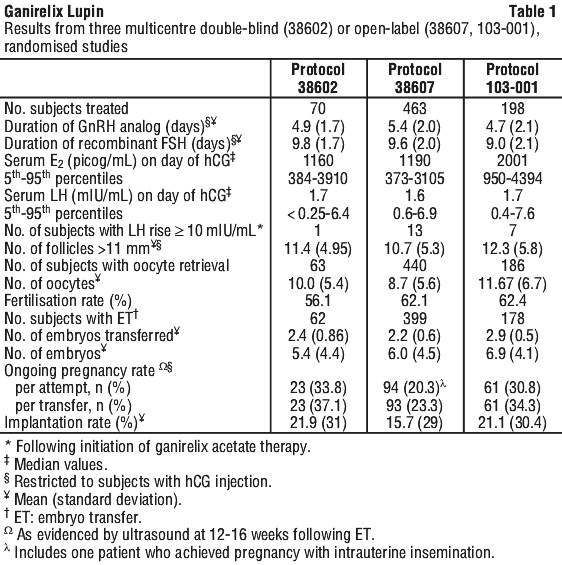
In subjects administered ganirelix 250 micrograms, a premature LH surge prior to hCG administration, (LH rise ≥ 10 mIU/mL with a significant rise in serum progesterone > 2 nanogram/mL, or a significant decline in serum estradiol) occurred in less than 1% of subjects.
In case of high ovarian response, as assessed by the number and size of growing follicles and/or the amount of circulating estradiol, either as a result of a high FSH exposure in the early follicular phase or as a result of high ovarian responsiveness, premature LH rises may occur earlier than day 6 of stimulation. Initiation of ganirelix treatment on day 5 can effectively prevent these premature LH rises without compromising the clinical outcome.
5.2 Pharmacokinetic Properties
Absorption. After a single subcutaneous administration of 250 micrograms, serum levels of ganirelix rise rapidly and reach peak levels (Cmax) of approximately 15 nanogram/mL within 1 to 2 hrs (tmax). The bioavailability of ganirelix following subcutaneous administration is approximately 91%.
Pharmacokinetic parameters after multiple subcutaneous dosing of ganirelix (once daily injection) were similar to those after a single subcutaneous dose. After repeated dosing at 0.25 mg/day, steady-state levels of approximately 0.6 nanogram/mL were reached within 2 to 3 days.
Distribution. The mean (SD) volume of distribution of ganirelix in healthy females following intravenous administration of a single 250 microgram dose is approximately 44 (± 11) litres. In vitro protein binding to human plasma is approximately 82%.
Metabolism. The major circulating component in plasma is ganirelix. Ganirelix is also the main compound found in urine, and faeces only contained metabolites.
Excretion. After a single subcutaneous administration of 250 micrograms, the elimination half-life (t1/2) is approximately 13 hours and clearance is approximately 2.4 L/h.
In a radiolabelled study (n = 3), ganirelix was excreted via faeces (approximately 75%) and urine (approximately 22%).
5.3 Preclinical Safety Data
Genotoxicity. Ganirelix showed no evidence of genotoxicity in assays for gene mutation in bacterial or mammalian cells. Ganirelix was not clastogenic in tests for chromosomal damage in Chinese hamster ovary cells in vitro and for micronucleus formation in mice in vivo.
Carcinogenicity. Long-term carcinogenicity studies with ganirelix have not been carried out.
6 Pharmaceutical Particulars
6.1 List of Excipients
Ganirelix Lupin solution for injection also contains glacial acetic acid, mannitol and water for injections. The pH may have been adjusted with sodium hydroxide and/or glacial acetic acid.
6.2 Incompatibilities
See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C, in the original package. Do not freeze as the syringe may break. Protect from light.
6.5 Nature and Contents of Container
Ganirelix Lupin is supplied in disposable pre-filled syringes (siliconised clear type I glass), containing 250 micrograms of ganirelix acetate per 0.5 mL. Each pre-filled syringe is affixed with a needle closed by a rigid needle shield of rubber (synthetic elastomer) which comes into contact with this product (see Section 4.3 Contraindications; Section 4.4 Special Warnings and Precautions for Use).
Ganirelix Lupin is available in packs of 1 or 5 prefilled syringes.
6.6 Special Precautions for Disposal
This product is for single use in one patient only. Discard any residue.
Any unused product or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Ganirelix contains the synthetic decapeptide ganirelix (INN) as its acetate salt, with high antagonistic activity to the naturally occurring GnRH.
Chemical structure.
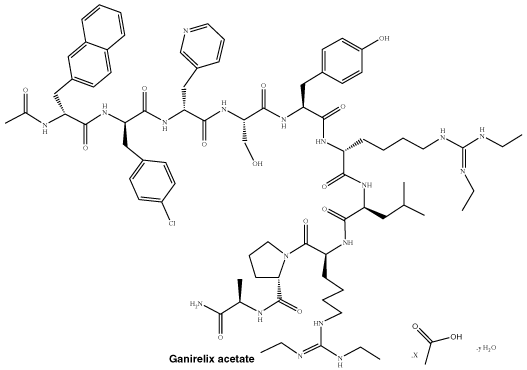
Molecular formula: C80H113N18O13Cl.xCH3CO2H.yH2O, hydrated salt where 2 ≤ x ≤ 3 and y ≤ 10.
Molecular weight: 1570.4 (anhydrous free base).
pH at 25°C ± 2°C: 4.5-5.5.
Osmolality: 250-330.
CAS number. 124904-93-4 (free base).
129311-55-3 (acetate).
7 Medicine Schedule (Poisons Standard)
Prescription Only Medicine (Schedule 4).
Date of First Approval
11 December 2024
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.