Gentamicin Injection BP
Brand Information
| Brand name | Gentamicin Injection BP |
| Active ingredient | Gentamicin |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Gentamicin Injection BP.
Summary CMI
Gentamicin Injection BP
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I being treated with Gentamicin Injection BP?
Gentamicin Injection BP contains the active ingredient gentamicin sulfate. Gentamicin Injection BP is used to treat bacterial infections.
For more information, see Section 1. Why am I being treated with Gentamicin Injection BP? in the full CMI.
2. What should I know before treatment with Gentamicin Injection BP?
You should not be given Gentamicin Injection BP if you have ever had an allergic reaction to gentamicin or any of the ingredients listed at the end of the CMI, or if you have experienced previous toxic reactions (hearing loss, kidney damage) to an antibiotic.
Talk to your doctor if you have: kidney problems, hearing problems, neuromuscular disorders, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before treatment with Gentamicin Injection BP? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Gentamicin Injection BP and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How is Gentamicin Injection BP given?
Gentamicin Injection BP is given as an injection into a muscle or as a slow injection (drip) into a vein.
More instructions can be found in Section 4. How is Gentamicin Injection BP given? in the full CMI.
5. What should I know during treatment with Gentamicin Injection BP?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know during treatment with Gentamicin Injection BP? in the full CMI.
6. Are there any side effects?
Side effects of this medicine may include loss of appetite, weight loss; numbness, skin tingling; skin rash/itching/reddening; vomiting, nausea; increased saliva; sore mouth; muscle/joint pain; pain at injection site; increased temperature; confusion, hallucinations; mental depression; headache; visual disorders; hair loss; kidney problems; shallow breathing, shortness of breath, swelling of throat; diarrhoea; convulsions; dizziness, vertigo, balance problems tinnitus, roaring in ears/hearing loss.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Gentamicin Injection BP
Active ingredient: gentamicin sulfate
Consumer Medicine Information (CMI)
This leaflet provides important information about using Gentamicin Injection BP. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about treatment with Gentamicin Injection BP.
Where to find information in this leaflet:
1. Why am I being treated with Gentamicin Injection BP?
2. What should I know before treatment with Gentamicin Injection BP?
3. What if I am taking other medicines?
4. How is Gentamicin Injection BP given?
5. What should I know during treatment with Gentamicin Injection BP?
6. Are there any side effects?
7. Product details
1. Why am I being treated with Gentamicin Injection BP?
Gentamicin Injection BP contains the active ingredient gentamicin sulfate. It is an antibiotic that belongs to a group of medicines called aminoglycoside antibiotics.
Gentamicin Injection BP is used to treat the bacterial infections in the following areas of the body: bloodstream, lung, urine, skin, bone, liver, stomach, spleen, gallbladder, intestines; and also septic abortion (miscarriage) and burns complicated by the infection.
Gentamicin Injection BP works by stopping protein production in susceptible bacteria which leads to their death.
2. What should I know before treatment with Gentamicin Injection BP?
Warnings
Do not use Gentamicin Injection BP if:
- you are allergic to gentamicin, or any of the ingredients listed at the end of this leaflet.
- you have experienced previous toxic reactions including hearing loss, often with loss of balance or damage to your kidneys to an antibiotic.
If you are not sure if you are allergic to any of the above, ask your doctor.
Some of the symptoms of an allergic reaction may include rash, itching or hives on the skin, swelling of the face, lips, tongue or other parts of the body, shortness of breath, wheezing or troubled breathing.
Tell your doctor if you have allergies to any other medicines, foods, preservatives or dyes.
Check with your doctor if you:
- have problems with your kidneys
- have neuromuscular disorders, such as:
myasthenia gravis, parkinsonism (a disorder affecting movement with trembling, rigid posture, slow movements and/or a shuffling, unbalanced walk) - are significantly overweight
- have diarrhoea following antibiotic use
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Tell your doctor if you are pregnant or intend to become pregnant
Gentamicin Injection BP is potentially harmful to the fetus. Like most aminoglycoside antibiotics, Gentamicin Injection BP is not recommended for use during pregnancy. If there is a need for you to be given Gentamicin Injection BP, your doctor will discuss with you the benefits and risks of using it during your pregnancy.
Tell your doctor if you are breast-feeding or plan to breast-feed
Gentamicin Injection BP passes into breast milk and is not recommended for use during breastfeeding. If there is a need for you to be given Gentamicin Injection BP, your doctor will discuss with you the benefits and risks of being given Gentamicin Injection BP whilst you are breast-feeding.
If you have not told your doctor about any of the above, tell them before you are given Gentamicin Injection BP.
Use in the elderly
There may be a greater chance of loss of hearing if you are over 65 years of age.
Use in children
Gentamicin Injection BP should be used with caution in premature and newborn babies.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Gentamicin Injection BP, these include:
- Digoxin, a medicine used to treat heart problems
- some medicines used during surgery, such as suxamethonium, some anaesthetics, large transfusions with citrated anticoagulated blood
- opioid analgesics, medicine used for strong pain relief
- some other anti-infective medicines, such as aminoglycosides, polymyxin B, colistin, vancomycin, amphotericin B, clindamycin, cephalosporins, ciclosporins, penicillins
- cisplatin, a medicine used in cancer treatment
- some diuretics, medicines used to treat swelling of the ankles, feet or legs, such as etacrynic acid, furosemide
- indometacin, a medicine used to treat arthritis and inflammation
- neostigmine, a medicine used to relax muscles
- Vitamin K
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Gentamicin Injection BP.
4. How is Gentamicin Injection BP given?
How much is given
- Your doctor will decide how much Gentamicin Injection BP you should receive and the length of time for which you should receive it.
How is Gentamicin Injection BP given
- Gentamicin Injection BP is given as an injection into a muscle or as a slow injection (drip) into a vein.
If you are given too much Gentamicin Injection BP
As Gentamicin Injection BP is given under the close supervision of your doctor, it is very unlikely that you will receive too much. If you experience any side effects, tell your doctor or nurse immediately.
5. What should I know during treatment with Gentamicin Injection BP?
Things you should do
Tell your doctor straight away if you:
- the symptoms of your infection do not improve within a few days, or if they become worse
- have stomach cramps or diarrhoea
- have dizziness, vertigo, tinnitus, roaring in the ears or hearing loss
If you are about to start any new medicines, tell your doctor and pharmacist that you are being given Gentamicin Injection BP.
Remind any doctor, dentist or pharmacist who are treating you that you are using Gentamicin Injection BP.
Things you should not do
Do not take any other medicines whether they require a prescription or not without first telling your doctor or consulting with a pharmacist.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Gentamicin Injection BP affects you.
Gentamicin Injection BP may cause dizziness and vertigo in some people
Drinking alcohol
No information is available.
Looking after your medicine
Gentamicin Injection BP is stored in the pharmacy or on the hospital ward under the correct conditions.
Disposal of Gentamicin Injection BP
The hospital staff will dispose of any leftover Gentamicin Injection BP.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
side effects
| side effects | What to do |
| Speak to your doctor if you have any of these side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Alert your nurse and/or doctor straight away if you notice any of these serious side effects. |
Tell your doctor, nurse or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Some of these side effects (for example certain liver conditions, and blood abnormalities) can only be found when your doctor does tests from time to time to check your progress.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Gentamicin Injection BP contains
| Active ingredient (main ingredient) | Gentamicin sulfate |
| Other ingredients (inactive ingredients) | Disodium edetate Water for injections Sodium hydroxide Sulfuric acid |
Do not take this medicine if you are allergic to any of these ingredients.
Each Gentamicin Injection BP contains Gentamicin (as Sulfate) 80 mg/2 mL.
What Gentamicin Injection BP looks like
Gentamicin Injection BP is clear, colorless solution in an ampoule.
Aust R 11376
Who distributes Gentamicin Injection BP
Pfizer Australia Pty Ltd
Sydney NSW
Toll Free Number: 1800 675 229
www.pfizermedicalinformation.com.au
This leaflet was prepared in September 2025.
Brand Information
| Brand name | Gentamicin Injection BP |
| Active ingredient | Gentamicin |
| Schedule | S4 |
MIMS Revision Date: 01 October 2023
1 Name of Medicine
Gentamicin sulfate.
2 Qualitative and Quantitative Composition
Gentamicin Injection BP is a sterile, preservative-free solution containing gentamicin (as sulfate) 80 mg/2 mL.
3 Pharmaceutical Form
Solution for injection.
4 Clinical Particulars
4.1 Therapeutic Indications
For the treatment of infections due to one or more susceptible strains of bacteria, including Pseudomonas aeruginosa, Proteus species (indole positive and indole negative), Escherichia coli, Klebsiella, Enterobacter and Serratia species and Staphylococcus (including strains resistant to other antibiotics).
Gentamicin may also be used for the treatment of the following conditions when caused by susceptible organisms: bacteraemia, respiratory tract infections, urinary tract infections, skin and skin structure infections, bone infections, peritonitis, septic abortion and burns complicated by sepsis. Aminoglycosides, including gentamicin are generally not indicated in uncomplicated initial episodes of urinary tract infection unless the causative organisms are not susceptible to less toxic antibiotics.
In suspected or documented Gram negative sepsis, gentamicin should be considered for initial antimicrobial therapy. Therapy may be instituted before obtaining results of susceptibility tests. The decision to continue therapy is based on results of the susceptibility tests, the severity of the infection and risk of toxicity. If anaerobic organisms are suspected, antimicrobial therapy in addition to the gentamicin regimen should be considered.
4.2 Dose and Method of Administration
Each ampoule is for use in a single patient on one occasion only.
Gentamicin is normally given by intramuscular injection. Intravenous administration may be used for particular indications when the intramuscular route is not appropriate. The dosage is the same for either route of administration. It is desirable to measure both peak and trough serum levels during treatment.
Prior to administration, the patient's bodyweight should be measured for the correct calculation of dosage. In obese patients, the appropriate dose can be calculated by assuming the bodyweight is the patient's estimated lean bodyweight plus 40% of the excess.
Blood specimens for the determination of peak gentamicin concentrations should be obtained approximately one hour following IM administration and 30 minutes after completion of a 30 minute infusion. Blood specimens for the trough gentamicin concentration should be obtained immediately prior to the next IM or IV dose.
Intravenous administration. For IV administration, the prescribed dose of gentamicin may be diluted in 100-200 mL of sterile normal saline or 5% glucose in water.The concentration of gentamicin in the solution should not exceed 1 mg/mL. Infusion periods of 30 minutes to 2 hours have been advocated.
Administration of the dose by bolus injection produces serum levels that are initially in excess of what is regarded as being safe from toxic side effects. The high serum level does however, rapidly fall and the potential danger or safety of this method is yet to be established.
Gentamicin Injection must not be physically mixed with other drugs, but should be administered by separate infusion (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Adults (dosage in patients with normal renal function). For serious infections (systemic and urinary tract infections). 3 mg/kg/day in three equal doses given every eight hours.
Life threatening infections. Up to 5 mg/kg/day in 3 or 4 equal doses with reduction to 3 mg/kg/day as soon as clinically indicated. Doses should never exceed 5 mg/kg/day unless serum levels are monitored. Table 1 should be used as a guide.
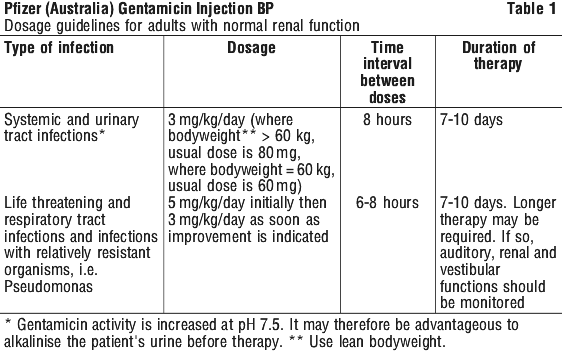
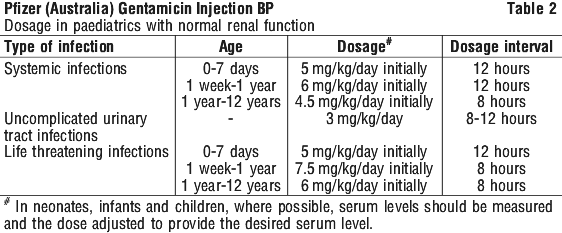
Table 3 should be used as a guide.
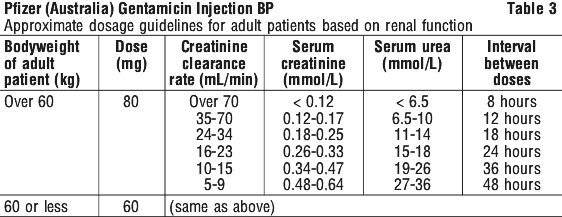
Note. The standard dose of 80 mg three times a day may be inappropriate and a more appropriate dose can be calculated using a nomogram which takes into account the patient's serum creatinine levels, bodyweight and age. This dose can be adjusted if necessary following determination of serum creatinine levels. Desirable serum levels of gentamicin are 5-8 microgram/mL as a peak and 1-2 microgram/mL as a trough.
Note. In children with impaired renal function serum levels should be monitored and frequency of dosage reduced if indicated.
In adults with renal failure undergoing haemodialysis, the amount of gentamicin removed from the blood may vary depending upon several factors including the dialysis method used. An eight hour haemodialysis may reduce serum concentrations of gentamicin by approximately 50%. The recommended dosage at the end of each dialysis period is 1 to 1.7 mg/kg depending upon the severity of infection.
4.3 Contraindications
Known hypersensitivity to gentamicin, any of the excipients (see Section 6.1 List of Excipients), or patients who have experienced previous toxic reactions (ototoxicity, nephrotoxicity) resulting from aminoglycoside therapy.
4.4 Special Warnings and Precautions for Use
Gentamicin, as with other aminoglycosides, is potentially nephrotoxic and ototoxic. As for other aminoglycosides, patients being treated with gentamicin should be under close clinical observation during treatment because of the potential toxicity associated with their use.
Neurotoxicity. Neurotoxicity may be manifested by both vestibular and auditory ototoxicity. These auditory changes are generally irreversible, usually bilateral and may be partial or total. Other manifestations of neurotoxicity may include numbness, skin tingling, muscle twitching and convulsions. The risk of this toxicity is higher in patients receiving high doses, prolonged treatment, or with impaired renal function. Gentamicin should therefore be used with caution in patients with impaired renal function. In such patients the frequency of administration should be reduced and renal function should be monitored. Prolonged concentrations above 10 microgram/mL should be avoided and trough concentrations should not exceed 2 microgram/mL. In neonates, infants and children, dosage reductions may also be necessary to avoid toxicity.
Diabetes, auditory vestibular dysfunctions, previous use of ototoxic drugs and a genetically determined high sensitivity to aminoglycoside-induced ototoxicity, are other main factors that may predispose the patient to ototoxicity.
Patients with mitochondrial DNA mutations, particularly the nucleotide 1555 A to G substitution in the 12S rRNA gene may be at higher risk for ototoxicity, even if the patient's aminoglycoside serum levels were within the recommended range. In case of family history of aminoglycoside induced deafness or known mitochondrial DNA mutations in the 12S rRNA gene, alternative treatments other than aminoglycosides should be considered.
Renal and eighth cranial nerve function should be closely monitored, especially in patients with known or suspected reduced renal function at onset of therapy and also in those whose renal function is initially normal but who develop signs of renal dysfunction during therapy. Urine should be examined for decreased specific gravity, increased excretion of protein, and the presence of cells or casts. Serum urea, serum creatinine, or creatinine clearance should be determined periodically. Where possible, it is recommended that serial audiograms be obtained in patients old enough to be tested, particularly high risk patients. Evidence of ototoxicity (dizziness, vertigo, tinnitus, roaring in the ears or hearing loss) or nephrotoxicity requires dosage adjustment or discontinuance of the drug. As with the other aminoglycosides, on rare occasions changes in renal and eighth cranial nerve function may not become manifest until soon after completion of therapy. Treatment period should not normally exceed 10 to 14 days.
Serum concentrations of aminoglycosides should be monitored to assure adequate levels and to avoid potentially toxic levels. When monitoring gentamicin peak concentrations, dosage should be adjusted so that prolonged levels above 10 microgram/mL are avoided. When monitoring gentamicin trough concentrations, dosage should be adjusted so that levels above 2 microgram/mL are avoided. Excessive peak and/or trough serum concentrations of aminoglycosides may increase the risk of renal and eighth cranial nerve toxicity. In the event of overdose or toxic reactions, haemodialysis may aid in the removal of gentamicin from the blood, especially if renal function is or becomes compromised. The rate of removal of gentamicin is considerably less by peritoneal dialysis than by haemodialysis.
Concurrent and/or sequential systemic or topical use of other potentially neurotoxic and/or nephrotoxic drugs can increase the possibility of gentamicin toxicity, (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions). Co-administration with the following agents should be avoided: neuromuscular blocking agents such as suxamethonium and tubocurarine; other potentially nephrotoxic or ototoxic drugs such as cephalosporins; potent diuretics such as etacrynic acid and furosemide; other aminoglycosides; amphotericin B; cisplatin; ciclosporin.
Other factors which may increase the risk of toxicity are dehydration, advancing age and diabetes mellitus. Patients should be well hydrated during therapy.
Patients treated with aminoglycoside antibiotics, including gentamicin, by injection, irrigation or local application, should be under close clinical observation because these drugs have the inherent potential for causing neurotoxicity and nephrotoxicity, particularly if patients have pre-existing renal damage or if the drug is administered for longer periods or at higher doses than those recommended.
Recent evidence suggests that neurotoxic and nephrotoxic antibiotics may be absorbed in significant quantities from body surfaces after local irrigation or application. The potential toxic effect of antibiotics administered in this fashion should be considered and inadvertent contact with the skin should be removed with water.
Neuromuscular disorders. Aminoglycosides should be used cautiously in patients with neuromuscular disorders such as myasthenia gravis or parkinsonism. In such cases, gentamicin may aggravate muscle weakness because of its curare-like effect on neuro-muscular function. Gentamicin should be used with care in conditions characterised by muscular weakness. Gentamicin induced renal tubular dysfunction including Fanconi syndrome acquired and Pseudo Bartter syndrome, with acid base and electrolyte disturbances has been reported in some infants, children and adults being given gentamicin injections. Muscle weakness, paresthesias, tetany, positive Chvostek and Trousseau signs have been described in patients with hypomagnesemia, hypocalcemia and hypokalemia. All required appropriate corrective electrolyte therapy.
Use during anaesthesia. The possibility of prolonged or secondary apnoea should be considered if the drug is administered to anaesthetised patients who are concurrently receiving neuromuscular blocking agents such as suxamethonium (succinylcholine), tubocurarine or decamethonium. This also applies to patients who are receiving massive transfusions of citrated blood. If neuromuscular blockade occurs, it may be reversed by the administration of calcium salts.
Superinfection. Treatment with gentamicin may lead to an overgrowth of nonsusceptible organisms. If overgrowth of nonsusceptible organisms occurs, appropriate therapy should be initiated.
Allergic reactions. May occur after administration of gentamicin. Cross allergenicity among aminoglycosides has also been known to occur.
Obesity. In cases of significant obesity, gentamicin serum concentrations should be closely monitored and a reduction in dose should be considered (see Section 4.2 Dose and Method of Administration).
Clostridioides difficile-associated diarrhoea. Clostridioides difficile-associated diarrhoea (CDAD) has been reported with the use of nearly all antibacterial agents, including gentamicin, and may range in severity from mild diarrhoea to fatal colitis. Treatment with antibacterial agents alters the normal flora of the colon, leading to overgrowth of C. difficile.
C. difficile produces toxins A and B, which contribute to the development of CDAD. Hypertoxin producing strains of C. difficile cause increased morbidity and mortality, as these infections can be refractory to antimicrobial therapy and may require colectomy. CDAD must be considered in all patients who present with diarrhoea following antibiotic use. Careful medical history is necessary since CDAD has been reported to occur over 2 months after the administration of antibacterial agents.
Use in renal impairment. Gentamicin should be used with caution generally in premature infants because of their renal immaturity, in elderly people and generally in patients with impaired renal function. In some patients with impaired renal function, there has been a transient rise in serum urea, which has usually reverted to normal during or following cessation of therapy. It is important to adjust the frequency of dosage according to the degree of renal function.
Use in the elderly Because of its toxicity, gentamicin should be used with caution in elderly patients only after less toxic alternatives have been considered and/or found ineffective. Elderly patients are more likely to have an age related decrease in renal function which may not be evident in the results of routine screening test such as serum urea or serum creatinine. A creatinine clearance determination may be more useful. Recommended doses should not be exceeded, and the patient's renal function should be carefully monitored during therapy. Elderly patients may require smaller daily doses of gentamicin in accordance with their increased age, decreased renal function, and possibly, decreased weight. In addition, loss of hearing may result even in patients with normal renal function.
Paediatric use Gentamicin should be used with caution in premature and neonatal infants because their renal immaturity may result in the prolongation of the serum half-life of the drug and subsequent gentamicin induced toxicity.
Effects on laboratory tests. Laboratory abnormalities possibly related to gentamicin include increased levels of serum transaminase (ALT, AST), serum LDH and bilirubin; decreased serum calcium, magnesium, sodium and potassium; anaemia, leucopenia, granulocytopenia, transient agranulocytosis, eosinophilia, increased and decreased reticulocyte counts, and thrombocytopenia. While clinical laboratory test abnormalities may be isolated findings, they may also be associated with clinically related signs and symptoms. For example, tetany and muscle weakness may be associated with hypomagnesaemia, hypocalcaemia and hypokalaemia.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Concurrent administration of gentamicin and other potentially ototoxic or nephrotoxic drugs should be avoided. Co-administration with the following agents should be avoided: neuromuscular blocking agents such as suxamethonium and tubocurarine; other potentially nephrotoxic or ototoxic drugs such as cephalosporins; potent diuretics such as etacrynic acid and furosemide; other aminoglycosides; amphotericin B; cisplatin; ciclosporin (see Section 4.4 Special Warnings and Precautions for Use).
Digoxin. Gentamicin has been known to increase serum digoxin levels.
Neuromuscular blocking agents or medications with neuromuscular blocking activity. Concurrent use of gentamicin with agents with neuromuscular blocking activity e.g. suxamethonium (succinylcholine), tubocurarine, decamethonium, halogenated hydrocarbon inhalation anaesthetics, opioid analgesics or massive transfusions with citrated anticoagulated blood, should be carefully monitored; neuromuscular blockade may be enhanced, resulting in skeletal muscle weakness and respiratory depression or paralysis (apnoea); caution is recommended when these medications and gentamicin are used concurrently during surgery or in the postoperative period, especially if there is a possibility of incomplete reversal of neuromuscular blockade postoperatively; treatment with anticholinesterase agents or calcium salts may help reverse the blockade.
Concurrent use of the botulinum toxin and gentamicin may increase the risk of toxicity due to enhanced neuromuscular block.
Other neurotoxic and/or nephrotoxic agents. Since the ototoxic or nephrotoxic effects of gentamicin may be additive, avoid concurrent or sequential use of other neurotoxic and/or nephrotoxic antibiotics, including other aminoglycosides, polymyxin B, colistin, vancomycin, amphotericin B, clindamycin, cephalosporins, cisplatin and ciclosporins.
Any potential nephrotoxicity of cephalosporins may also be increased in the presence of gentamicin. Consequently, monitoring of kidney function is advised if this combination is used.
Potent diuretics. If possible, do not give gentamicin in conjunction with etacrynic acid, furosemide or other potent diuretics that may themselves cause ototoxicity or enhance aminoglycoside toxicity by altering antibiotic concentrations in serum and tissue.
Penicillins. Gentamicin is inactivated by solutions containing beta-lactam antibiotics (penicillins and cephalosporins) so the two drugs should not be administered simultaneously nor should they be combined in the intravenous fluid. The inactivation of gentamicin by penicillins may occur in vivo, especially in patients with renal failure who maintain a higher level of the penicillin for a longer period of time compared to patients with normal renal function. Therefore, when gentamicin and penicillins are used together in patients with renal failure, the time of administration of each drug should be staggered so that several hours separate each infusion.
Although the inactivation of gentamicin and penicillin proceeds on an equimolar basis, in practice the penicillin is present in such an excess that only the decline in activity of gentamicin is of concern. A combination of penicillin and gentamicin is often used in the treatment of enterococcal endocarditis.
Indometacin. Indometacin possibly increases plasma concentrations of gentamicin in neonates.
Neostigmine. Antagonism of effect may occur with concomitant administration of gentamicin with neostigmine.
Vitamin K. Gentamicin may inhibit the action of intravenous vitamin K upon the synthesis of clotting factors.
Potential interactions. In vitro synergism and antagonism have been found between various antineoplastic agents and aminoglycosides.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category D)
Gentamicin and other aminoglycosides are known to cross the placenta. There is evidence of selective uptake of aminoglycosides by the fetal kidney resulting in damage (probably reversible) to immature nephrons. Eighth cranial nerve damage has also been reported following in utero exposure to some of the aminoglycosides. Because of their chemical similarity, all aminoglycosides must be considered potentially nephrotoxic and ototoxic to the fetus. It should also be noted that therapeutic blood concentrations in the mother do not equate with safety for the fetus.
Use in lactation. Small amounts of gentamicin have been detected in breast milk. Because of the potential risk to the newborn, it is recommended that breastfeeding be discontinued during therapy unless the expected benefits outweigh any potential risk.
4.7 Effects on Ability to Drive and Use Machines
The effect of gentamicin sulfate on the ability to drive or use machines has not been systematically evaluated.
4.8 Adverse Effects (Undesirable Effects)
These effects are reported in decreasing order of seriousness within each system organ class (SOC) and absolute frequency, see Table 4.
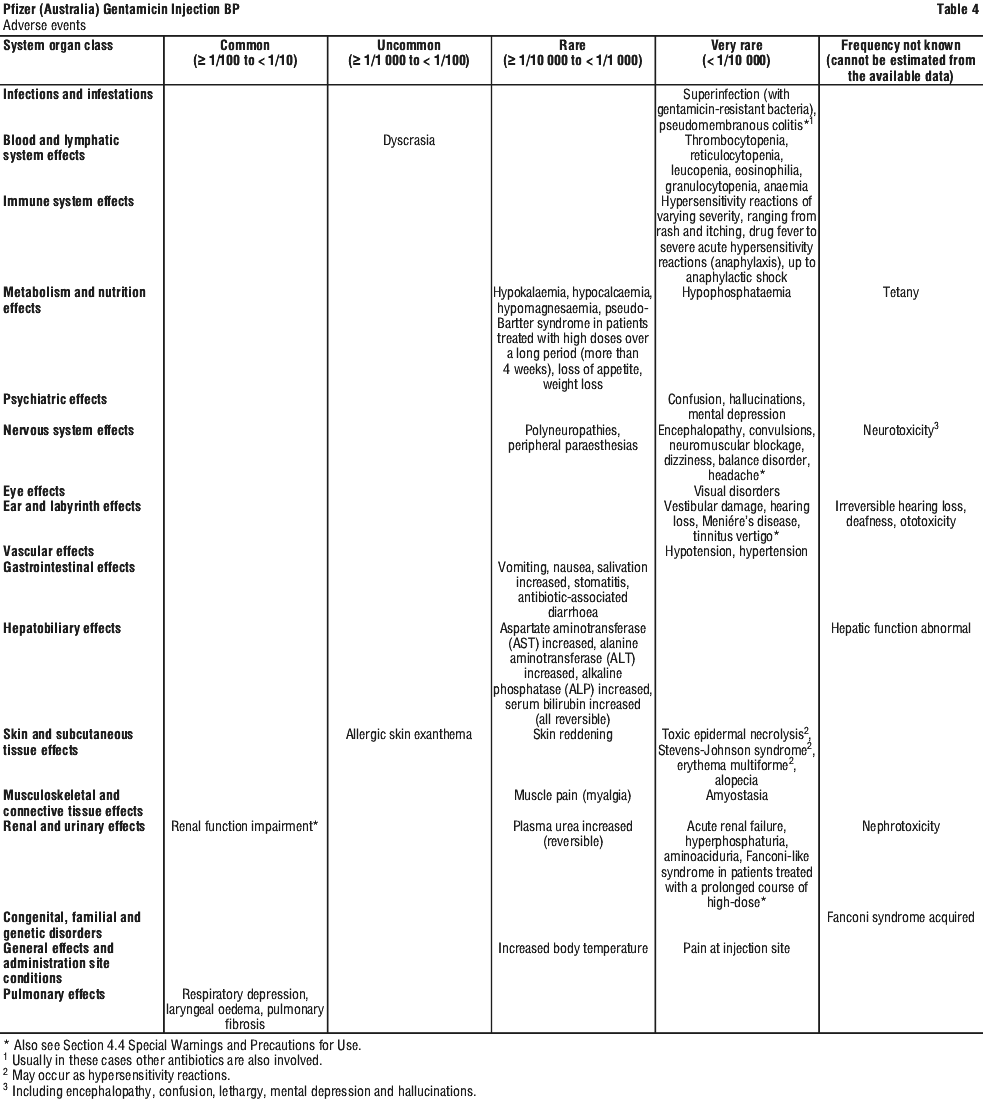
Neurotoxicity. (See Section 4.4 Special Warnings and Precautions for Use). Serious adverse effects on both vestibular and auditory branches of the eighth cranial nerves have been reported, primarily in patients with renal impairment (especially if dialysis is required), and in patients on high doses and/or prolonged therapy. Symptoms reported include dizziness, vertigo, tinnitus, roaring in the ears and hearing loss, which, as with the other aminoglycosides, may be irreversible. Hearing loss is usually manifested initially by diminution of high tone acuity. Other factors that may increase the risk of toxicity include excessive dosage, dehydration and previous exposure to other ototoxic drugs.
More common reactions.Peripheral neuropathy or encephalopathy, including numbness, skin tingling, muscle twitching, convulsions and a myasthenia gravis-like syndrome, have also been reported.
Note. The risk of toxic reactions is low in patients with normal renal function who do not receive gentamicin at higher doses or for longer periods of time than recommended.
Other adverse reactions. Other reported adverse reactions possibly related to gentamicin include: respiratory depression, lethargy, confusion, depression, visual disturbances, decreased appetite, weight loss, hypotension, hypertension, rash, itching, urticaria, generalised burning, laryngeal oedema, anaphylactoid reactions, fever, headache, nausea, vomiting, increased salivation, stomatitis, purpura, pseudotumor cerebri, acute organic brain syndrome, pulmonary fibrosis, alopecia, joint pain, transient hepatomegaly and splenomegaly.
While local tolerance of gentamicin injection is generally excellent, there has been an occasional report of pain at the injection site. Subcutaneous atrophy or fat necrosis suggesting local irritation has been reported rarely.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/safety/reporting-problems.
4.9 Overdose
As the drug is almost entirely eliminated by the kidneys, fluid loading may hasten its elimination following overdosage. Peritoneal dialysis or haemodialysis will also aid in the drug's removal. This is particularly important in patients with renal malfunction.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Class. Aminoglycoside antibiotic.
Microbiology. Gentamicin is bactericidal and acts by inhibiting protein synthesis in susceptible bacteria. Cell death results. It is active against a wide range of pathogenic Gram negative organisms including Escherichia coli, Pseudomonas aeruginosa, Proteus sp. (both indole positive and indole negative), Klebsiella, Enterobacter and Serratia species. It is also active against some Gram positive organisms, e.g. Staphylococcus sp. (including methicillin and penicillin resistant strains). In vitro, gentamicin is also active against Salmonella and Shigella species. Some species have demonstrated resistance to aminoglycosides including Streptococcus pneumoniae and anaerobic organisms such as Bacteroides or Clostridioides species.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. Gentamicin is rapidly absorbed after intramuscular injection and peak serum levels are usually achieved within 30 to 90 minutes and are measurable for 6-8 hours.
Gentamicin is poorly absorbed by the oral route, and only minimal amounts have been found in the blood following oral administration.
In patients with normal renal function, peak serum concentrations of gentamicin, expressed in microgram/mL, are usually about four times the single dose expressed in mg/kg; for example, an injection of gentamicin 1 mg/kg may be expected to result in peak serum concentration of approximately 4 microgram/mL. Gentamicin administered every 8 hours does not accumulate in the serum except in patients with impaired renal function in whom the serum concentration of gentamicin is usually higher, and measurable for longer periods. When gentamicin is administered by intravenous infusion, over 1 to 2 hours, the serum concentrations are similar to those obtained with intramuscular administration. About 25 to 30% of the administered dose of gentamicin is bound by serum protein; it is released as the drug is excreted. Gentamicin is excreted principally in the urine by glomerular filtration.
Distribution. Following parenteral administration gentamicin can be detected in tissues and body fluids. Concentration in bile is low. Gentamicin administered intramuscularly has been found in low concentrations in the cerebrospinal fluid. Gentamicin has also been found in the sputum, pleural, peritoneal, ascitic, pericardial, synovial and abscess fluids. Gentamicin crosses the peritoneal as well as the placental membranes.
Excretion. After initial administration to patients with normal renal function, 30 to 100% of the gentamicin is recoverable in the urine in 24 hours. High urine concentrations (above 100 microgram/mL) may be achieved. After several days of treatment, the amount of gentamicin excreted in the urine approaches the daily dose administered. Renal clearance of gentamicin is similar to that of endogenous creatinine. The serum half-life of gentamicin is approximately 2-3 hours in adults with normal renal function. It is prolonged in patients with impaired renal function and in premature or newborn infants.
Gentamicin is excreted almost entirely by renal glomerular filtration, hence the half-life of the drug is prolonged in the presence of renal failure. Adjustments in the frequency of administration of gentamicin are necessary to allow for the degree of renal failure (see Section 4.2 Dose and Method of Administration). Endogenous creatinine clearance rate and serum creatinine, which have high correlation with serum half-life of gentamicin, may be used as a guide for this purpose (see Section 4.2 Dosage and Method of Administration).
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Disodium edetate, water for injections, sodium hydroxide, sulfuric acid.
6.2 Incompatibilities
When gentamicin is used in combination with any other drug, mixing the drugs before administration should be avoided at all costs.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Protect from light. Single use only. Discard unused portion.
6.5 Nature and Contents of Container
AUST R 11376. Gentamicin Injection BP 80 mg in 2 mL Steriluer (10s).
AUST R 11376. Gentamicin Injection BP 80 mg in 2 mL Steriluer (50s).
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Gentamicin sulfate is a mixture of the sulfates of antimicrobial substances produced by Micromonospora purpurea. The potency is not less than 590 I.U. per milligram, calculated with reference to the anhydrous substance. It is a white or almost white powder, freely soluble in water, practically insoluble in alcohol and in ether.
Chemical structure. The structural formula is represented below.
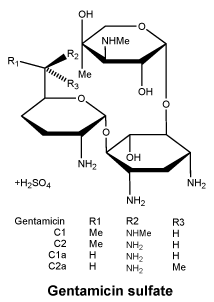
7 Medicine Schedule (Poisons Standard)
Australia - S4.
Date of First Approval
28 February 2001
Date of Revision
14 August 2023
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.