Gliclazide MR Viatris
Brand Information
| Brand name | Gliclazide MR Viatris |
| Active ingredient | Gliclazide |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Gliclazide MR Viatris.
Summary CMI
GLICLAZIDE MR VIATRIS
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about taking this medicine, speak to your doctor or pharmacist.
1. Why am I taking GLICLAZIDE MR VIATRIS?
GLICLAZIDE MR VIATRIS contains the active ingredient gliclazide. GLICLAZIDE MR VIATRIS is used control blood glucose (sugar) in patients with Type II diabetes mellitus. For more information, see Section 1. Why am I taking GLICLAZIDE MR VIATRIS? in the full CMI.
2. What should I know before I take GLICLAZIDE MR VIATRIS?
Do not take if you have ever had an allergic reaction to gliclazide or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, are pregnant or breastfeeding or plan to become pregnant or breastfeed. For more information, see Section 2. What should I know before I take GLICLAZIDE MR VIATRIS? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with GLICLAZIDE MR VIATRIS and affect how it works. A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I take GLICLAZIDE MR VIATRIS?
Your doctor will tell you how many tablets to take; swallow them whole with water at the same time each day, with breakfast.
More instructions can be found in Section 4. How do I take GLICLAZIDE MR VIATRIS? in the full CMI.
5. What should I know while taking GLICLAZIDE MR VIATRIS?
| Things you must do |
|
| Things you must not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while taking GLICLAZIDE MR VIATRIS? in the full CMI.
6. Are there any side effects?
Serious side effects: skin rash, redness itching and/or hives, angioedema (rapid swelling of tissues such as eyelids, face, lips, mouth, tongue or throat that may result in breathing difficulty. Common side effects: low or high blood sugar, cold-like or respiratory symptoms (runny/blocked nose, sneezing, sinus or throat discomfort, coughing, bronchitis, upper respiratory infection), back or joint pain, high blood pressure, chest pain, headache, weakness, viral or urinary infections, dizziness, stomach upset (nausea, pain, vomiting, diarrhoea, constipation), or skin blistering/peeling. For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
GLICLAZIDE MR VIATRIS
Active ingredient(s): gliclazide
Consumer Medicine Information (CMI)
This leaflet provides important information about taking GLICLAZIDE MR VIATRIS. Speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about taking GLICLAZIDE MR VIATRIS.
Where to find information in this leaflet:
1. Why am I taking GLICLAZIDE MR VIATRIS?
2. What should I know before I take GLICLAZIDE MR VIATRIS?
3. What if I am taking other medicines?
4. How do I take GLICLAZIDE MR VIATRIS?
5. What should I know while taking GLICLAZIDE MR VIATRIS?
6. Are there any side effects?
7. Product details
1. Why am I taking GLICLAZIDE MR VIATRIS?
GLICLAZIDE MR VIATRIS contains the active ingredient gliclazide. Gliclazide belongs to a group of medicines called sulphonylureas. The medicine releases the active ingredient gliclazide progressively over 24 hours.
It lowers blood glucose by increasing the amount of insulin (a hormone that controls blood glucose levels) produced by your pancreas.
GLICLAZIDE MR VIATRIS is used to control blood glucose (sugar) in patients with Type II diabetes mellitus. This type of diabetes is also known as non-insulin-dependent diabetes (NIDDM), or maturity-onset diabetes.
GLICLAZIDE MR VIATRIS is used for Type II diabetes mellitus when diet and exercise are not enough to control your blood glucose.
GLICLAZIDE MR VIATRIS can be used alone or together with insulin or other medicines for treating diabetes.
How it works
Glucose is used by the body as fuel, and all people have glucose circulating in their blood. In diabetes, levels of blood glucose are higher than is needed, which is also known as hyperglycaemia. If your blood glucose is not properly controlled, you may experience hypoglycaemia (low blood glucose) or hyperglycaemia (high blood glucose). High blood glucose can lead to serious problems with our heart, circulation and/or kidneys.
It is very important to control high blood glucose whether or not you feel unwell. This really helps to avoid serious long-term health problems, which can involve the heart, eyes, circulation, and/or kidneys.
As with many medicines used for the treatment of diabetes, there is a possibility that blood glucose levels may become very low during treatment with GLICLAZIDE MR VIATRIS. This is known as hypoglycaemia.
Section 5. What should I know while taking GLICLAZIDE MR VIATRIS? of this leaflet contains advice about recognising and treating hypoglycaemia.
Ask your doctor if you have any questions about why GLICLAZIDE MR VIATRIS has been prescribed for you.
GLICLAZIDE MR VIATRIS is not addictive.
2. What should I know before I take GLICLAZIDE MR VIATRIS?
Warnings
Do not take GLICLAZIDE MR VIATRIS if:
- you are allergic to:
- gliclazide, or any of the ingredients listed at the end of this leaflet
- other sulphonylureas,
- GLICLAZIDE MR VIATRIS related medicines such as sulfa antibiotics or to thiazide diuretics (a type of "fluid" or "water" tablet)
Symptoms of an allergic reaction to GLICLAZIDE MR VIATRIS or to these medicines may include:
- skin rash, itchiness or hives,
- shortness of breath, swelling of the face, lips or tongue,
- muscle pain or tenderness or joint pain.
If you are not sure if you have an allergy to GLICLAZIDE MR VIATRIS, check with your doctor.
Always check the ingredients to make sure you can take GLICLAZIDE MR VIATRIS. - you have Type 1 diabetes mellitus (also known as insulin dependent diabetes (IDDM), or juvenile-onset diabetes)
- you have unstable diabetes
- you have diabetic ketoacidosis (a problem which affects the acidity of your blood and can lead to coma - which is mainly associated with Type 1 diabetes)
- you have severe kidney disease
- you have severe liver disease
- you are taking an antibiotic medicine containing the active ingredient miconazole
- the expiry date printed on the pack has passed
If you take it after the expiry date has passed, it may not work as well. - the packaging is torn or shows signs of tampering.
If it is damaged, return it to your pharmacist for disposal.
If you are not sure whether you should start taking GLICLAZIDE MR VIATRIS, talk to your doctor.
Check with your doctor if you:
- are allergic to any of the ingredients listed at the end of this leaflet; to any other medicines; or to any other substances, such as foods, preservatives or dyes
- have a family history of or know you have the hereditary condition glucose-6-phosphate dehydrogenase (G6PD) deficiency (abnormality of red blood cells), lowering of the haemoglobin level and breakdown of red blood cells (haemolytic anaemia) can occur
- have had any of the following medical conditions:
- kidney disease
- liver disease
- a history of diabetic coma
- adrenal, pituitary or thyroid problems
- heart failure - take any medicines for any other condition
- have any medical condition, or do anything, that may increase the risk of hyperglycaemia - for example:
- you are ill or feeling unwell (especially with fever or infection)
- you are injured
- you are having surgery
- you are taking less GLICLAZIDE MR VIATRIS than prescribed
- you are taking on less exercise than normal
- you are eating more carbohydrate than normal.
- drinking alcoholic drinks
- not eating regular meals
- taking on more exercise than usual.
If you have not told your doctor, pharmacist or diabetes educator about any of the above, tell them before you start taking GLICLAZIDE MR VIATRIS.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Do not take GLICLAZIDE MR VIATRIS if are pregnant or trying to become pregnant.
GLICLAZIDE MR VIATRIS may affect your developing baby if you take it during pregnancy. Insulin is more suitable for controlling blood glucose during pregnancy. Your doctor will usually replace GLICLAZIDE MR VIATRIS with insulin while you are pregnant.
Do not take GLICLAZIDE MR VIATRIS are breastfeeding or plan to breastfeed.
GLICLAZIDE MR VIATRIS is not recommended while you are breastfeeding. It is not known whether GLICLAZIDE MR VIATRIS passes into breast milk.
Use in children
Do not give GLICLAZIDE MR VIATRIS to a child.
There is no experience with the use of GLICLAZIDE MR VIATRIS in children.
Use in older people
Elderly people can generally use GLICLAZIDE MR VIATRIS safely.
There are no special instructions for older people taking GLICLAZIDE MR VIATRIS.
3. What if I am taking other medicines?
Tell your doctor, pharmacist or diabetes educator if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Taking GLICLAZIDE MR VIATRIS may change the effect of some medicines, and some medicines may affect how well GLICLAZIDE MR VIATRIS works.
Some medicines may lead to low blood glucose (hypoglycemia) by increasing the blood-glucose-lowering effect of GLICLAZIDE MR VIATRIS. These include:
- some medicines used to treat fungal or yeast infections (miconazole which is contraindicated)
- alcohol
- other medicines used to treat diabetes (such as biguanides and insulin)
- some medicines used to treat high blood pressure and other heart conditions (including angiotensin receptor blocker, beta-blockers)
- some medicines used to treat depression and other mental illness (MAOIs)
- some cholesterol-lowering medicines (clofibrate)
- some medicines used to treat arthritis, pain and inflammation (including high dose aspirin, ibuprofen, phenylbutazone)
- some antibiotics (chloramphenicol; clarithromycin; sulphonamides)
- some medicines used to treat acid reflux and stomach ulcers
- some medicines used to treat gout (probenecid)
- some medicines used to treat tuberculosis.
Some medicines may lead to high blood glucose levels (hyperglycaemia) by weakening the blood glucose-lowering effect of GLICLAZIDE MR VIATRIS. These include:
- alcohol
- some medicines for epilepsy (danazol)
- some medicines used to treat depression and other mental illness (chlorpromazine)
- St John's Wort (Hypericum perforatum) preparations used to treat depression
- some hormones used in hormone replacement therapy and oral contraceptives (oestrogen, progesterone)
- some medicines for asthma (salbutamol, intravenous terbutaline)
- barbiturates, medicines used for sedation
- glucocorticoids
- some medicines for fluid retention (thiazides, furosemide)
- nicotinic acid.
Some medicines may lead to unstable blood glucose (low blood sugar and high blood sugar) when taken at the same time as GLICLAZIDE MR VIATRIS, especially in elderly patients. These include:
- a class of antibiotics called fluoroquinolones.
GLICLAZIDE MR VIATRIS may change the effects of some other medicines. These include:
- some medicines used to prevent blood clots (warfarin).
You may need different amounts of your medicine or you may need to take different medicines. Your doctor, pharmacist or diabetes educator can tell you what to do if you are taking any of these medicines. They also have a more complete list of medicines to be careful with or avoid while taking GLICLAZIDE MR VIATRIS.
Ask your doctor or pharmacist if you are not sure if you are taking any of these medicines.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect GLICLAZIDE MR VIATRIS.
4. How do I take GLICLAZIDE MR VIATRIS?
How much to take
- Follow all directions given to you by your doctor, pharmacist, and diabetes educator carefully.
They may differ from the information contained in this leaflet. - If you do not understand the instructions on the box, ask your doctor or pharmacist for help.
- Your doctor will tell you how many tablets to take each day. They may increase or decrease the dose, depending on your blood glucose levels.
When to take GLICLAZIDE MR VIATRIS
- Swallow the tablets whole with a glass of water. GLICLAZIDE MR VIATRIS tablets should not be crushed or chewed.
Crushing or chewing the tablets may change the effectiveness of the tablet. - GLICLAZIDE MR VIATRIS tablets do not have a break bar and should not be broken in half.
- It is important to take your GLICLAZIDE MR VIATRIS at the same time each day - usually with breakfast. Taking GLICLAZIDE MR VIATRIS with food can help to minimise the risk of hypoglycaemia.
- Do not skip meals while taking GLICLAZIDE MR VIATRIS.
How long to take GLICLAZIDE MR VIATRIS
- Continue taking GLICLAZIDE MR VIATRIS for as long as your doctor recommends. Make sure you keep enough GLICLAZIDE MR VIATRIS to last over weekends and holidays.
- GLICLAZIDE MR VIATRIS will help control your diabetes but will not cure it. Therefore, you may have to take it for a long time.
If you forget to take GLICLAZIDE MR VIATRIS
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Otherwise, take it as you remember (with food), then go back to taking your tablets as you would normally.
Missed doses can cause high blood glucose (hyperglycaemia).
If you are not sure whether to skip the dose, talk to your doctor or pharmacist.
Do not take a double dose to make up for the dose you missed.
If you double a dose, this may cause low blood glucose (hypoglycaemia).
If you take too much GLICLAZIDE MR VIATRIS
If you think that you or anyone else has taken too much GLICLAZIDE MR VIATRIS, urgent medical attention may be needed.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26) for advice, or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
Do this even if there are no signs of discomfort or poisoning.
If you take too much GLICLAZIDE MR VIATRIS together with other medicines for diabetes or alcohol, you may experience symptoms of low blood glucose (hypoglycaemia).
If not treated quickly, these symptoms may progress to loss of co-ordination, slurred speech, confusion, loss of consciousness and fitting.
At the first signs of hypoglycaemia, raise your blood glucose quickly by following the instructions at the end of this leaflet. Section 5. What should I know while taking GLICLAZIDE MR VIATRIS? contains advice about recognising and treating hypoglycaemia.
If you experience any of these symptoms, immediately get medical help.
5. What should I know while taking GLICLAZIDE MR VIATRIS?
Things you must do
- If you become pregnant while you are taking GLICLAZIDE MR VIATRIS, tell your doctor.
- Tell all doctors, dentists, pharmacists and diabetes educators who are involved with your treatment that you are taking GLICLAZIDE MR VIATRIS.
- If you are about to start taking any new medicines, tell you doctor and pharmacist that you are taking GLICLAZIDE MR VIATRIS.
- Take GLICLAZIDE MR VIATRIS exactly as your doctor has prescribed. Otherwise you may not get the full benefits from treatment.
- Make sure you check your blood glucose levels regularly.
This is the best way to tell if your diabetes is being controlled properly. Your doctor or diabetes educator will show you how and when to do this. - Make sure that you, your friends, family and work colleagues can recognise the symptoms of hypoglycaemia and hyperglycaemia and know how to treat them.
Instructions at the end of this leaflet. Section 5. What should I know while taking GLICLAZIDE MR VIATRIS? can help you with this. - Visit your doctor regularly so that they can check on your progress.
- Carefully follow your doctor's and dietician's advice on diet, drinking alcohol and exercise.
- Tell your doctor immediately if you notice the return of any symptoms of hyperglycaemia that you had before starting GLICLAZIDE MR VIATRIS.
These may include lethargy or tiredness, headache, thirst, passing large amounts of urine and blurred vision. These may be signs that GLICLAZIDE MR VIATRIS is no longer working, even though you may have been taking it successfully for some time.
Remind any doctor, dentist or pharmacist you visit that you are taking GLICLAZIDE MR VIATRIS.
Things you must not do
- Do not give GLICLAZIDE MR VIATRIS to anyone else, even if they have the same condition as you.
- Do not take GLICLAZIDE MR VIATRIS to treat other complaints unless your doctor tells you to.
- Do not stop taking GLICLAZIDE MR VIATRIS, or change the dosage, without checking with your doctor.
- Do not skip meals while taking GLICLAZIDE MR VIATRIS.
Things to be careful of
- Protect your skin when you are in the sun, especially between 10am and 3pm.
Sulphonylureas (the group of medicines that GLICLAZIDE MR VIATRIS belongs to) may cause your skin to be more sensitive to sunlight than it is normally. Exposure to sunlight may cause a skin rash, itching, redness, or a severe sunburn. - If outdoors, wear protective clothing and use a SPF 30+ sunscreen. If your skin does appear to be burning, tell your doctor immediately.
- If you are travelling, it is a good idea to:
- wear some form of identification showing you have diabetes
- carry some form of sugar to treat hypoglycaemia if it occurs, for example, sugar sachets or jelly beans
- carry emergency food rations in case of a delay, for example, dried fruit, biscuits or muesli bars
- keep GLICLAZIDE MR VIATRIS readily available - If you become sick with a cold, fever or flu, it is very important to continue taking GLICLAZIDE MR VIATRIS, even if you fell unable to eat your normal meal. If you have trouble eating solid food, use sugar-sweetened drinks as a carbohydrate substitute or eat small amounts of bland food.
Your diabetes educator or dietician can give you a list of foods to use for sick days.
Recognising and treating hyPOglycaemia (very LOW blood sugar levels)
Hypoglycaemia may occur during GLICLAZIDE MR VIATRIS treatment.
The first signs of hypoglycaemia are usually weakness, trembling or shaking, sweating, lightheadedness, dizziness, headache or lack of concentration, irritability, tearfulness, hunger, and/ or numbness around the lips and tongue.
At the first signs of hypoglycaemia take some sugar to raise your blood sugar level quickly.
Do this by eating 5 to 7 jelly beans, 3 teaspoons of sugar or honey, drinking half a can of non-diet soft drink, taking 2-3 glucose tablets or a tube of glucose gel.
Then take some extra carbohydrates -
such as plain biscuits, fruit or milk - unless you are within 10-15 minutes of your next meal. Taking this extra carbohydrate will help to prevent a second drop in your blood glucose level.
If not treated quickly, hypoglycaemia symptoms may progress to loss of co-ordination, slurred speech, confusion, fits or loss of consciousness.
If hypoglycaemia symptoms do not get better straight away after taking sugar then go to the Accident and Emergency department at your nearest hospital - if necessary by calling an ambulance.
Contact your doctor or diabetes educator for advice if you are concerned about hypoglycaemia.
Recognising and treating hyPERglycaemia (HIGH blood sugar levels)
Some people may feel fine when their glucose levels are high. Others notice symptoms of hyperglycaemia like tiredness, lack of energy, thirst, passing large amounts of urine, headache, and/or blurred vision.
If you notice symptoms of hyperglycaemia, or your blood sugar levels are high, tell your doctor immediately. You may need adjustments of the dose or type of medicines you are taking.
It is very important to control high blood glucose whether or not you feel unwell. This really helps to avoid serious long-term health problems, which can involve the heart, eyes, circulation, and/or kidneys.
If you experience any of the signs of hyperglycaemia (high blood glucose) contact your doctor or diabetes educator for advice immediately.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how GLICLAZIDE MR VIATRIS affects you.
Also, be especially careful not to let your blood glucose levels fall too low.
GLICLAZIDE MR VIATRIS may cause dizziness and drowsiness in some people. Low blood glucose levels may also slow your reaction time and affect your ability to drive or operate machinery. Section 5. What should I know while taking GLICLAZIDE MR VIATRIS? contains advice about recognising and treating hypoglycaemia.
Drinking alcohol can make this worse. If either of these occurs, do not drive, operate machinery or do anything else that could be dangerous.
Drinking alcohol
Tell your doctor if you drink alcohol.
If you drink alcohol while taking GLICLAZIDE MR VIATRIS, you may get flushing, sensation of warmth, headache, breathing difficulties, rapid heartbeat, stomach pains or feel sick and vomit.
Looking after your medicine
- Keep them in a cool dry place below 25°C. Protect from moisture.
- Keep your tablets in the pack until it is time to take them.
GLICLAZIDE MR VIATRIS will not keep as well outside its blister packaging.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Heat and dampness can destroy some medicines.
Keep it where young children cannot reach it.
A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Getting rid of any unwanted medicine
If you no longer need to take this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not take this medicine after the expiry date.
6. Are there any side effects?
If you do not feel well while you are taking GLICLAZIDE MR VIATRIS then tell your doctor, pharmacist, or diabetes educator as soon as possible.
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
However if you do - or if you notice anything else that is making you feel unwell - you should consult your doctor or pharmacist.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor, pharmacist or diabetes educator if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What GLICLAZIDE MR VIATRIS contains
| Active ingredient (main ingredient) | gliclazide 30 mg |
| Other ingredients (inactive ingredients) | hypromellose magnesium stearate microcrystalline cellulose |
Do not take this medicine if you are allergic to any of these ingredients.
What GLICLAZIDE MR VIATRIS looks like
GLICLAZIDE MR VIATRIS 30 mg tablets are white to off-white, oblong, biconvex tablet debossed with ‘M’ on one side of the tablet and 'GL 30' on the other side (AUST R 295541).
They are available in blister packs of 100 tablets.
Who distributes GLICLAZIDE MR VIATRIS
Alphapharm Pty Ltd trading as Viatris
Level 1, 30 The Bond
30-34 Hickson Road
Millers Point NSW 2000
www.viatris.com.au
Phone: 1800 274 276
This leaflet was prepared in December 2025.
GLICLAZIDE MR VIATRIS_cmi\Dec25/00
Brand Information
| Brand name | Gliclazide MR Viatris |
| Active ingredient | Gliclazide |
| Schedule | S4 |
MIMS Revision Date: 01 August 2024
1 Name of Medicine
Gliclazide.
2 Qualitative and Quantitative Composition
Each modified release tablet contains 30 mg of gliclazide as the active ingredient.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Gliclazide 30 mg modified release tablets: A white to off-white, oblong, biconvex tablet debossed with 'M' on one side of the tablet and 'GL 30' on the other side.
4 Clinical Particulars
4.1 Therapeutic Indications
Type II diabetes in association with dietary measures when dietary measures alone are inadequate to control blood glucose.
During controlled clinical trials in patients with type II diabetes, a modified release formulation of gliclazide (30 mg - 120 mg), taken as a single daily dose, was shown to be effective long term in controlling blood glucose levels, based on monitoring of HbA1c.
4.2 Dose and Method of Administration
For adult use only.
Gliclazide MR Viatris 30 mg tablets do not have a break bar and therefore should not be broken.
So that the modified release properties of the product can be maintained, Gliclazide MR Viatris tablets should not be chewed or crushed.
Gliclazide MR Viatris should be taken with food because there is an increased risk of hypoglycaemia if a meal is taken late, if an inadequate amount of food is consumed or if the food is low in carbohydrate. It is recommended that the medication be taken at breakfast time. If a dose is forgotten, the dose taken on the next day should not be increased.
A single daily dose provides an effective blood glucose control. The daily dose may vary from 30 mg to 120 mg taken orally, once daily. The initial recommended dose is 30 mg daily, even in elderly patients (≥ 65 years). It should not exceed 120 mg.
As with all hypoglycaemic agents, the dose should be titrated according to the individual patient's response. Titration should be carried out in steps of 30 mg, according to the fasting blood glucose response. Each step should last for at least two weeks.
Previously untreated patients should commence with a dose of 30 mg and will benefit from dose titration until the appropriate dose is reached.
Gliclazide MR Viatris 30 mg tablets can replace gliclazide 80 mg tablets, tablet for tablet, for doses of one to four tablets per day.
Gliclazide MR Viatris tablets may be used to replace other antidiabetic treatments without any transitional period. If a patient is switched from a hypoglycaemic sulfonylurea with a prolonged half-life he/she should be carefully monitored (for one to two weeks) in order to avoid hypoglycaemia due to possible residual effects of the previous therapy.
Gliclazide MR Viatris tablets may be given in combination with biguanides, alpha glucosidase inhibitors or insulin.
Elderly patients. The efficacy and tolerance of the modified release formulation of gliclazide (30 mg - 120 mg) has been confirmed in clinical trials in patients over 65 years who were given the same dosage regimen as the general population. The dosage is therefore identical to that recommended for adults under the age of 65 years.
Renal impairment. The efficacy and tolerance of the modified release formulation of gliclazide (30 mg - 120 mg) has been confirmed in clinical trials of patients with mild to moderate renal failure (creatinine clearance of between 15 and 80 mL/min) who were given the same dosage regimen as the general population. No dosage adjustment is therefore required in patients with mild to moderate renal impairment.
Use of Gliclazide MR Viatris tablets in patients with severe renal impairment is contraindicated (see Section 4.3 Contraindications).
4.3 Contraindications
Hypersensitivity to gliclazide, other sulfonylureas, sulfonamides, or to any of the excipients.
Type I diabetes, diabetic keto-acidosis, diabetic pre-coma and coma.
Severe renal or hepatic impairment: in these cases the use of insulin is recommended.
Treatment with miconazole (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Pregnancy and lactation (see Section 4.6 Fertility, Pregnancy and Lactation, Use in pregnancy, Use in lactation).
4.4 Special Warnings and Precautions for Use
The risks of hypoglycaemia, together with its symptoms, treatment and conditions that predispose to its development, should be explained to the patient and to family members. The patient should be informed of the importance of following dietary advice, of taking regular exercise, and of regular monitoring of blood glucose levels.
Hypoglycaemia. Hypoglycaemia may occur following administration of sulfonylureas. Rarely cases may be severe and prolonged. This may involve hospitalisation and glucose infusion may need to be continued for several days.
Careful selection of patients and of the dose used, as well as provision of adequate information to the patient are necessary to avoid hypoglycaemic episodes.
The following factors may increase the risk of hypoglycaemia:
patient does not follow the doctor's treatment advice (particularly elderly patients);
malnutrition or debilitated patients;
irregular mealtimes, skipping meals, periods of fasting or dietary changes;
imbalance between physical exercise and carbohydrate intake;
renal impairment;
severe hepatic impairment;
overdose of anti-diabetic agents;
certain endocrine disorders: thyroid disorders, hypopituitarism and adrenal impairment, concomitant administration of certain other medicines (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Gliclazide should only be prescribed if the patient is likely to have a regular food intake (including breakfast). It is important to have a regular carbohydrate intake due to the increased risk of hypoglycaemia if a meal is delayed, an inadequate amount of food is consumed or the food is low in carbohydrate. Hypoglycaemia is more likely to occur during periods of low-calorie diet, following prolonged or strenuous exercise, following alcohol intake or during treatment with a combination of hypoglycaemic agents.
Since the effects of oral hypoglycaemic agents on the vascular changes and other long-term sequelae of diabetes mellitus are not fully known, patients receiving such medicinal products must be closely observed for both short and long-term complications. Periodic assessment of cardiovascular, ophthalmic, renal, and hepatic status is advisable.
Poor blood glucose control. Blood glucose control in treated patients may be affected by St. John's wort (Hypericum perforatum) preparations (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions), fever, trauma, infection or surgical intervention. It may be necessary to discontinue treatment and to administer insulin in these cases.
The efficacy of oral antidiabetic agents often decreases in the long term. This may be due to progression in the severity of the diabetes, or to a reduced response to treatment. This phenomenon is known as secondary failure and should be distinguished from primary failure, when the drug is ineffective as first-line treatment. However, before classifying the patient as a secondary failure, dose adjustment and reinforcement of dietary measures should be considered.
If a loss of adequate blood glucose-lowering response to gliclazide is detected, the medicine should be discontinued.
Unstable blood glucose level (dysglycaemia). Disturbances in blood glucose, including hypoglycaemia and hyperglycaemia have been reported, in diabetic patients receiving concomitant treatment with fluoroquinolones, especially in elderly patients. Indeed, careful monitoring of blood glucose is recommended in all patients receiving gliclazide and a fluoroquinolone at the same time.
Glucose-6-phosphate dehydrogenase deficiency (G6PD). Treatment of patients with G6PD-deficiency with sulfonylurea agents can lead to haemolytic anaemia. Since gliclazide belongs to the chemical class of sulfonylurea drugs, caution should be used in patients with G6PD-deficiency and a non-sulfonylurea alternative should be considered.
Use in renal and hepatic impairment. Severe renal or hepatic impairment may affect the distribution of gliclazide and hepatic impairment may also reduce the capacity for neoglucogenesis. These two effects increase the risk of severe hypoglycaemic reactions. A hypoglycaemic episode in these patients may be prolonged and appropriate management should be initiated.
Hepatic and renal functions should be assessed before initiating therapy and periodically in patients with mild to moderately impaired hepatic and renal function.
Patients with porphyria. Cases of acute porphyria (which can cause severe abdominal pain, gastrointestinal symptoms, unspecified neurologic symptoms along with chronic, blistering lesions on sun-exposed skin) have been described with the class of sulfonylurea drugs, in patients who have porphyria. Therefore, caution should be taken in the administration of gliclazide, as it may precipitate attacks of acute porphyria.
Use in the elderly. See Section 4.2 Dose and Method of Administration; Section 5.2 Pharmacokinetic Properties.
Paediatric use. Not recommended for paediatric use, see Section 4.2 Dose and Method of Administration.
Effects on laboratory tests. Glycated haemoglobin should be monitored regularly. Blood glucose measurement may also be useful.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Blood glucose monitoring during and after treatment is necessary when Gliclazide MR Viatris is used with medicines which can interact with gliclazide. It may also be necessary to adjust the dose of Gliclazide MR Viatris during and after treatment with such medicines.
1. The following medications are likely to increase the risk of hypoglycaemia. Concomitant use which is contraindicated. Miconazole (systemic route, oromucosal gel). Increases the hypoglycaemic effect with possible onset of hypoglycaemia symptoms, or even coma.
Concomitant use which is not recommended. Phenylbutazone (systemic route). Increases the hypoglycaemic effect of sulfonylureas (displaces their binding to plasma proteins and/or reduces their elimination).
It is preferable to use a different anti-inflammatory agent, or else to warn the patient and emphasise the importance of self-monitoring. Where necessary, adjust the dose during and after treatment with the anti-inflammatory agent.
Alcohol. Acute alcohol intoxication potentiates the hypoglycaemic action of all sulfonylurea agents by inhibiting compensatory reactions. This can lead to the onset of hypoglycaemic coma. Ingestion of alcohol may also cause a disulfiram-like reaction with characteristic flushing of the face, sensation of warmth, throbbing headache, giddiness, nausea, tachypnoea, tachycardia or angina pectoris. Chronic alcohol abuse may, as a result of liver enzyme induction, increase the metabolism of sulfonylurea drugs, shortening the plasma half-life and duration of action.
Avoid alcohol or medicines containing alcohol.
Concomitant use which requires special care. Potentiation of the blood glucose lowering effect and therefore in some instances, hypoglycaemia may occur when one of the following medications is taken:
Other antidiabetic agents (insulins, acarbose, biguanides, metformin, thiazolidinediones, dipeptidyl peptidase-4 inhibitors, GLP-1 receptor agonists), sulfonamides, clarithromycin, clofibrate, salicylates (high doses), fibrates, chloramphenicol, MAOIs, probenecid, beta-blockers, azole antifungal agents (oral and parenteral preparations), H2-receptor antagonists, ACE inhibitors, fluconazole, tuberculostatics and nonsteroidal anti-inflammatory agents.
2. The following medications may cause an increase in blood glucose levels. Advise the patient and emphasise the importance of glucose monitoring.
Concomitant use which is not recommended. Danazol. Combination is not recommended because of diabetogenic effect of danazol. If the use of danazol cannot be avoided, warn the patient and emphasise the importance of urine and blood glucose monitoring. It may be necessary to adjust the dose of Gliclazide MR Viatris during and after treatment with danazol.
Concomitant use which requires special care. Chlorpromazine. High doses (> 100 mg per day of chlorpromazine) can increase blood glucose levels (reduced insulin release). Advise the patient and emphasise the importance of glucose monitoring. It may be necessary to adjust the dose of Gliclazide MR Viatris during and after treatment with chlorpromazine.
Glucocorticoids (systemic and local route: intra-articular, cutaneous and rectal preparations) and tetracosactide. Concomitant use may increase blood glucose levels with possible ketosis (glucocorticoids cause reduced tolerance to carbohydrates). Emphasise the importance of blood glucose monitoring, particularly at the start of treatment. It may be necessary to adjust the dose of Gliclazide MR Viatris during and after treatment with glucocorticoids.
Salbutamol, terbutaline (intravenous). May cause increased blood glucose levels due to beta-2 agonist effects. If necessary, switch to insulin.
Barbiturates, oestrogens and progestogens (oral contraceptives). May adversely affect blood sugar control with hypoglycaemic agents in some patients by causing increased blood glucose levels.
St John's wort (Hypericum perforatum) preparations. Gliclazide exposure is decreased by St John's wort (Hypericum perforatum).
Certain drugs tend to induce hyperglycaemia and may lead to loss of blood sugar control. These include diuretics (thiazides, furosemide), ritodrine and nicotinic acid in pharmacological doses.
3. The following products may cause unstable blood glucose. Concomitant use which requires special care. Fluoroquinolones. In case of a concomitant use of gliclazide and a fluoroquinolone, the patient should be warned of the risk of unstable blood glucose, and the importance of blood glucose monitoring should be emphasised.
Concomitant use to be taken into consideration. Anticoagulant therapy (warfarin). Sulfonylureas may lead to potentiation of anticoagulation during concurrent treatment. Adjustment of warfarin may be necessary.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category C)
In animal studies embryo-toxicity and/or birth defects have been demonstrated with some sulfonylureas.
Gliclazide should not be used in pregnant women. From a clinical point of view, there are limited data (less than 300 pregnancies) to allow evaluation of the possible malformative or foetotoxic effects of gliclazide, when administered during pregnancy. Animal studies of gliclazide have not shown any teratogenic effect.
Gliclazide is contraindicated during pregnancy and insulin is the drug of first choice for treatment of diabetes during pregnancy. Treatment should be changed from gliclazide to insulin therapy before pregnancy is attempted, or as soon as pregnancy is discovered. Control of diabetes should be achieved before the time of conception to reduce the risk of congenital abnormalities linked to uncontrolled diabetes.
Use in lactation. It is not known whether gliclazide or its metabolites are excreted in breast milk. Given the risk of neonatal hypoglycaemia, Gliclazide MR Viatris is contraindicated in women who are breastfeeding. A risk to newborns/infants cannot be excluded.
4.7 Effects on Ability to Drive and Use Machines
Gliclazide can have effects on ability to drive and use machines. Patients should be made aware of the symptoms of hypoglycaemia and should be careful if driving or operating machinery, especially at the beginning of treatment.
4.8 Adverse Effects (Undesirable Effects)
Good clinical acceptability of gliclazide, has been established in many studies as well as in medical practice.
The safety of a modified release formulation of gliclazide (30 mg - 120 mg) has been evaluated in controlled clinical trials in 955 patients, of which 728 patients were treated in long-term comparative trials, against a gliclazide immediate release formulation (80 mg - 320 mg), for up to ten months. In these comparative trials, the overall incidence and type of adverse events were similar in both groups. Adverse events were generally mild and transient, not requiring discontinuation of therapy.
However, where patients did discontinue due to adverse events, the percentage was lower in the modified release group (2.9%) than in the immediate release group (4.5%).
Hypoglycaemia (see Section 4.3 Contraindications; Section 4.4 Special Warnings and Precautions for Use). The most frequent adverse reaction with gliclazide is hypoglycaemia.
As is the case with all sulfonylurea drugs, hypoglycaemic reactions have been reported following gliclazide administration. However, a number of studies have shown that hypoglycaemia is less common with gliclazide than with glibenclamide.
Possible symptoms of hypoglycaemia are: headache, intense hunger, nausea, vomiting, lassitude, sleep disorders, agitation, aggression, poor concentration, reduced awareness and slowed reactions, depression, confusion, visual and speech disorders, aphasia, tremor, paraesthesia, flushing or pallor, numbness, chilliness, paresis, sensory disorders, dizziness, feeling of powerlessness, loss of self-control, delirium, convulsions, increased pulse rate, increased blood pressure, shallow respiration, bradycardia, drowsiness and loss of consciousness, apprehensiveness in mild cases possibly resulting in coma and/or death. In addition, signs of adrenergic counter-regulation may be observed: sweating, clammy skin, anxiety, tachycardia, hypertension, palpitations, angina pectoris and cardiac arrhythmia.
Usually, symptoms disappear after intake of carbohydrate such as sugar (artificial sweeteners have no effect). Experience with other sulfonylureas shows that hypoglycaemia can recur even when these measures are initially effective. If a hypoglycaemic episode is severe or prolonged, and even if it is temporarily controlled by intake of sugar, immediate medical treatment or even hospitalisation is required.
In long-term comparative studies, the percentage of patients experiencing hypoglycaemic episodes was similar between patients treated with the modified release formulation of gliclazide (11.6%) and those treated with the immediate release formulation of gliclazide (11.1%). However, the number of hypoglycaemic episodes per 100 patient months was lower in the modified release group (3.5) than in the immediate release group (4.8).
Analysis of elderly patients (over 65 years old) showed less hypoglycaemia than in the general population, with a prevalence of hypoglycaemic episodes lower in the modified release group (2.6 hypoglycaemic episodes for 100 patient months) than in the immediate release group (4.1).
The percentage of patients experiencing hypoglycaemic episodes in the sub-population with renal failure, was similar to that observed in the general population.
Adverse events reported during controlled clinical trials with the modified release formulation of gliclazide were those expected in an ageing population with diabetes.
Adverse events that were reported in at least 2.0% of patients, in long-term controlled clinical studies, are presented in Table 1. The most frequent adverse events were not specifically related to the disease (such as respiratory infections or back pain).
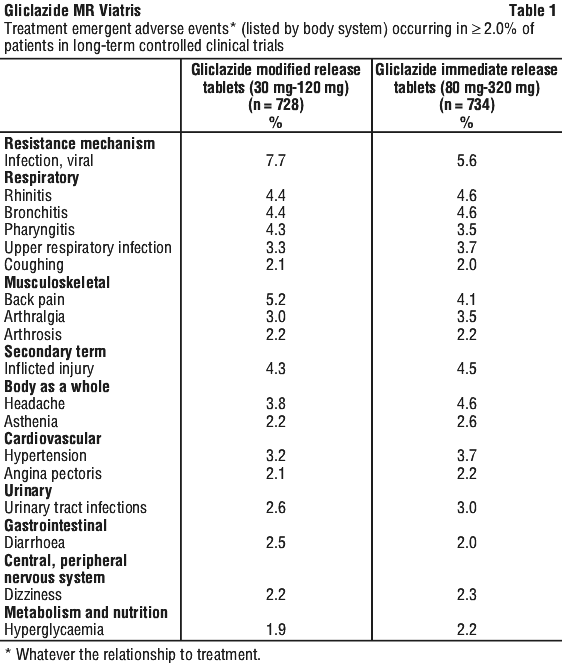
Other adverse effects. Gastrointestinal disturbances (reported with gliclazide), including nausea, dyspepsia, diarrhoea, abdominal pain, vomiting and constipation may be avoided or minimised if gliclazide is taken with breakfast.
The following adverse effects have been rarely reported:
Skin and subcutaneous tissue disorders. Pruritus, urticaria, maculopapular rashes, rash, angioedema, erythema and bullous reactions (such as Stevens-Johnson syndrome (SJS) and toxic epidermal necrolysis (TEN) (as with other sulfur-containing medications) and autoimmune bullous disorders and exceptionally, drug rash with eosinophilia and systemic symptoms (DRESS).
Blood and lymphatic system disorders (as with other sulfonylurea medications). Anaemia, leucopenia, thrombocytopenia and agranulocytosis. These are in general reversible upon discontinuation of medication.
Hepatobiliary disorders. Elevations of serum bilirubin and hepatic enzymes (AST, ALT, alkaline phosphatase) levels, pancreatitis acute and exceptionally, hepatitis (isolated reports). Treatment should be discontinued if cholestatic jaundice appears. These symptoms usually disappear after discontinuation of treatment.
Investigations. Occasional elevations of serum creatinine, blood urea nitrogen.
Eye disorders. Transient visual disturbances may occur due to changes in blood glucose levels, particularly on initiation of treatment. As with any glucose-lowering medication, transient visual disturbances may occur on initiation of treatment due to changes in blood glucose levels.
Class effects. The following adverse events have been observed with sulfonylureas: cases of erythrocytopenia, agranulocytosis, haemolytic anaemia, pancytopenia and allergic vasculitis, hyponatremia, elevated liver enzyme levels and even impairment of liver function (e.g. with cholestasis and jaundice) and hepatitis which regressed after withdrawal of the sulphonylurea or led to life-threatening liver failure in isolated cases.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Overdose of sulfonylureas may cause hypoglycaemia. It should be noted that the dosage which causes such hypoglycaemia varies widely and may be within the accepted therapeutic range in sensitive individuals.
Moderate symptoms of hypoglycaemia (without loss of consciousness or neurological signs), should be corrected by carbohydrate intake, dose adjustment and/or modification of diet. Strict monitoring should be continued until the doctor is sure that the patient is out of danger.
Severe hypoglycaemic reactions are possible (with coma, convulsions or other neurological disorders) and must be treated as a medical emergency, requiring immediate hospitalisation.
If hypoglycaemic coma is diagnosed or suspected, the patient should be given a rapid I.V. injection of 50 mL of concentrated glucose solution (20 to 30%). This should be followed by continuous infusion of a more dilute glucose solution (10%) at a rate necessary to maintain blood glucose levels above 5 mmol/L. It is recommended that patients should be monitored closely for a 48 hour period at least. Some sulfonylurea-induced hypoglycaemias may be refractory to treatment and susceptible to relapse especially in elderly or malnourished patients.
Plasma clearance of gliclazide may be prolonged in patients with hepatic disease. However, due to the strong binding of gliclazide to proteins, dialysis is not effective in these patients.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Gliclazide reduces blood glucose levels by stimulating insulin secretion from the β-cells of the islets of Langerhans. Gliclazide shows high affinity, strong selectivity and reversible binding to the β-cell KATP channels with a low affinity for cardiac and vascular KATP channels. Increased postprandial insulin and C-peptide secretion persists after two years of treatment.
In type II diabetes, gliclazide restores the first peak of insulin secretion in response to glucose and increases the second phase of insulin secretion. A significant increase in insulin release is seen in response to stimulation induced by a meal or glucose.
Gliclazide also has extra-pancreatic effects and haemovascular properties. It has been shown to increase peripheral insulin sensitivity.
In muscle, euglycaemic hyperinsulinaemic clamp studies with gliclazide have demonstrated significantly increased (35%) insulin mediated glucose uptake which may improve diabetes control. Gliclazide potentiates insulin action on muscle glycogen synthase. These effects are consistent with a post-transcriptional action of gliclazide on GLUT4 glucose transporters.
Studies on glucose turnover have further shown that gliclazide decreases hepatic glucose production, leading to an improvement in fasting blood glucose levels.
Gliclazide has been shown in some studies to have actions independent of that on glucose levels. These haemovascular effects of gliclazide include:
Partial inhibition of platelet aggregation and adhesion with a decrease in markers of platelet activation (beta thromboglobulin, thromboxane B2).
Increased vascular endothelial fibrinolytic activity (increased tPA activity).
Antioxidant properties, notably a reduction in plasma lipid peroxides and increased erythrocyte superoxide dismutase activity.
Inhibition of the increased adhesiveness of type II diabetic patient's monocytes to endothelial cells in vitro.
The antioxidant, platelet inhibiting and fibrinolytic actions of gliclazide involve processes which have been implicated in the pathogenesis of vascular complications of type II diabetes. There is no clinical evidence that the haemovascular effects of gliclazide are of therapeutic benefit in type II diabetes patients.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. Hydration of the tablets induces formation of a gel to activate drug release. Plasma levels increase progressively, resulting in a plateau-shaped curve from the sixth to the twelfth hour after administration. Intra-individual variability is low. Gliclazide is completely absorbed and food intake does not affect the rate or degree of absorption.
Distribution. Plasma protein binding is approximately 95%. The relationship between the dose administered and the area under the concentration curve as a function of time is linear for doses of gliclazide up to 90 mg/day. At the highest evaluated dose (135 mg/day), the AUC increases slightly more than proportionally to the dose.
Metabolism. Gliclazide is mainly metabolised in the liver, the products of which are extensively excreted in the urine.
Excretion. Less than 1% of unchanged drug is recovered in the urine. No active metabolites have been detected in plasma. The clearance of gliclazide has been found to be slightly reduced as a function of age. This reduction, however, is not considered to be clinically significant. The elimination half-life of gliclazide is approximately 16 hours.
No clinically significant modifications in the pharmacokinetic parameters have been observed in elderly patients.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Gliclazide MR Viatris includes the following excipients: hypromellose, microcrystalline cellulose, magnesium stearate.
6.2 Incompatibilities
See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Store in original container. Protect from moisture.
6.5 Nature and Contents of Container
Gliclazide MR Viatris 30 mg: HDPE bottle with CR cap or PVC/Al or PVC/PVDC/Al blister pack in packs of 100 tablets.
Some strengths, pack sizes and/or pack types may not be marketed.
Australian register of therapeutic goods (ARTG). AUST R 295541 - Gliclazide MR Viatris gliclazide 30 mg modified release tablet blister pack.
AUST R 295540 - Gliclazide MR Viatris gliclazide 30 mg modified release tablet bottle.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking it to your local pharmacy.
6.7 Physicochemical Properties
Gliclazide is an oral hypoglycaemic sulfonylurea which differs from other related compounds by an N-containing heterocyclic ring with an endocyclic bond.
It is a white or almost white powder, practically insoluble in water, freely soluble in dichloromethane, sparingly soluble in acetone and slightly soluble in ethanol 96%. The melting point of gliclazide is approximately 168°C.
Chemical structure.
Molecular formula: C15H21N3O3S.
Molecular weight: 323.4.
CAS number. 21187-98-4.
7 Medicine Schedule (Poisons Standard)
S4 (Prescription Only Medicine).
Date of First Approval
31 March 2023
Date of Revision
09 July 2024
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.