Glimepiride Sandoz
Brand Information
| Brand name | Glimepiride Sandoz |
| Active ingredient | Glimepiride |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Glimepiride Sandoz.
Summary CMI
GLIMEPIRIDE SANDOZ®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Glimepiride Sandoz?
Glimepiride Sandoz contains the active ingredient glimepiride. Glimepiride Sandoz is used to control blood glucose (sugar) in patients with type 2 diabetes mellitus.
For more information, see Section 1. Why am I using Glimepiride Sandoz? in the full CMI.
2. What should I know before I use Glimepiride Sandoz?
Do not use if you have ever had an allergic reaction to glimepiride or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Glimepiride Sandoz? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Glimepiride Sandoz and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Glimepiride Sandoz?
Your doctor will tell you how many Glimepiride Sandoz tablets to take each day. Depending on your blood glucose levels, your doctor may increase or decrease the dose.
More instructions can be found in Section 4. How do I use Glimepiride Sandoz? in the full CMI.
5. What should I know while using Glimepiride Sandoz?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Glimepiride Sandoz? in the full CMI.
6. Are there any side effects?
Common side effects: stomach upset (nausea, vomiting), abdominal pain, discomfort, diarrhoea, eye problems.
Serious side effects: yellowing of skin or eyes, shortness of breath, pale, sore throat, mouth ulcers, bleeding or bruising more easily, itching, paleness, wheezing, swelling of face/lips/tongue/other parts of the body.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
GLIMEPIRIDE SANDOZ®
Active ingredient(s): glimepiride
Consumer Medicine Information (CMI)
This leaflet provides important information about using Glimepiride Sandoz. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Glimepiride Sandoz.
Where to find information in this leaflet:
1. Why am I using Glimepiride Sandoz?
2. What should I know before I use Glimepiride Sandoz?
3. What if I am taking other medicines?
4. How do I use Glimepiride Sandoz?
5. What should I know while using Glimepiride Sandoz?
6. Are there any side effects?
7. Product details
1. Why am I using Glimepiride Sandoz?
Glimepiride Sandoz contains the active ingredient glimepiride. Glimepiride Sandoz belongs to a group of medicines called sulfonylureas. It works by increasing the amount of insulin produced by your pancreas.
Glimepiride Sandoz is used to control blood glucose (sugar) in patients with type 2 diabetes mellitus.
Your doctor may have prescribed it for another reason.
Ask your doctor if you have any questions about why this medicine has been prescribed for you.
2. What should I know before I use Glimepiride Sandoz?
Warnings
Do not use Glimepiride Sandoz if:
- you are allergic to glimepiride, other sulfonylureas, other similar medicines such as sulfonamides (e.g. sulfur antibiotics or thiazide diuretics) or any of the ingredients listed at the end of this leaflet. Always check the ingredients to make sure you can use this medicine. Some symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin - the package is torn or shows signs of tampering
Do not take this medicine if you have or have any of the following medical conditions:
- a previous reaction to glimepiride, other sulfonylureas or sulfonamides
- type 1 diabetes mellitus
- diabetic ketoacidosis
- diabetic coma or pre-coma
- severe kidney disease or if you require dialysis
- severe liver failure
- hereditary problems of galactose intolerance, Lapp lactase deficiency or glucose-galactose malabsorption.
Check with your doctor if you:
- have any other medical conditions
- kidney problems
- liver problems
- unstable diabetes
- adrenal, pituitary or thyroid problems
- heart failure
- glucose-6-phosphate dehydrogenase (G6PD) deficiency
- diabetic coma - take any medicines for any other condition
- are allergic to any other medicines, foods, dyes or preservatives
- drink alcohol in any amount
- do not eat regular meals
- do a lot of exercise or heavy exercise or work
- are ill or feeling unwell.
Alcohol, diet, exercise and your general health all strongly affect the control of your diabetes.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Do not take this medicine if you are pregnant or intend to become pregnant.
It may affect your developing baby if you take it during pregnancy.
Insulin is more suitable for controlling blood glucose during pregnancy. Your doctor will replace Glimepiride Sandoz with insulin.
Do not take this medicine if you are breastfeeding or intend to breastfeed.
The active ingredient in Glimepiride Sandoz passes into breast milk and there is a possibility that your baby may be affected.
Use in children
Do not give this medicine to children.
Safety and effectiveness in children have not been established.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Glimepiride Sandoz and affect how it works.
These medicines include:
- alcohol
- other medicines used to treat diabetes such as metformin or insulin
- some medicines used to treat reflux and stomach ulcers
- some anti-inflammatory medicines, such as aspirin, ibuprofen, naproxen or mesalazine
- some medicines used to treat arthritis and gout
- some antibiotics, such as clarithromycin or rifampicin
- anabolic steroids
- oral contraceptives
- corticosteroids, glucagon, thyroid hormones and other hormonal medications
- some cancer and organ transplant treatments
- some cholesterol-lowering and weight reduction medicines
- some antidepressants, psychiatric and sedating medicines
- some medicines used to treat epilepsy
- some medicines used to treat high blood pressure and some heart conditions, such as ACE inhibitors and beta-blockers
- some medicines used to treat blood clots, blood vessel problems and irregular heart rhythms such as coumarin derivatives
- some fluid and glaucoma medications
- large doses of laxatives.
Certain medicines such as beta-blockers and clonidine may hide the warning symptoms of hypoglycaemia (low blood glucose).
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Glimepiride Sandoz.
4. How do I use Glimepiride Sandoz?
How much to take
- Your doctor will tell you how many tablets to take each day. Depending on your blood glucose levels, your doctor may increase or decrease the dose.
- If you take the wrong dose, Glimepiride Sandoz may not work as well and your condition may not improve.
When to take Glimepiride Sandoz
- Take your medicine at about the same time each day. Taking it at the same time each day will have the best effect. It will also help you remember when to take it.
- Take your medicine during or immediately after a meal, at about the same time each day. Glimepiride Sandoz tablets are usually taken once a day, immediately before breakfast. If your breakfast is very light, you may wait until lunch to take the tablet.
- Do not skip meals while taking Glimepiride Sandoz.
How to take Glimepiride Sandoz
- Swallow the tablets whole with a full glass of water.
How long to take Glimepiride Sandoz
Continue taking your medicine for as long as your doctor tells you.
This medicine helps to control your condition, but does not cure it. It is important to keep taking your medicine even if you feel well.
If you forget to use Glimepiride Sandoz
Take your dose as soon as you remember, and continue to take it as you would normally.
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Tell your doctor if you have missed several doses. Missed doses can cause hyperglycaemia (high blood glucose).
Do not take a double dose to make up for the dose that you missed. This may increase the risk of hypoglycaemia (low blood glucose).
If you are not sure what to do, ask your doctor or pharmacist.
If you use too much Glimepiride Sandoz
If you think that you have used too much Glimepiride Sandoz, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
Symptoms of an overdose may include symptoms of hypoglycaemia (low blood glucose), such as:
- weakness, trembling or shaking
- sweating
- lightheadedness, dizziness, headache or lack of concentration
- tearfulness, crying, irritability
- hunger
- numbness around the lips and tongue
- loss of co-ordination or confusion
- slurred speech
- loss of consciousness or seizures.
5. What should I know while using Glimepiride Sandoz?
Things you should do
- If you are about to be started on any new medicine, remind your doctor and pharmacist that you are taking Glimepiride Sandoz.
- If you are going to have surgery, tell the surgeon or anaesthetist that you are taking this medicine
- If you are about to have any blood tests, tell your doctor that you are taking this medicine. It may interfere with the results of some tests.
- Keep all of your doctor's appointments so that your progress can be checked. Your doctor may do some regular tests to make sure the medicine is working and to prevent unwanted side effects. These include:
- blood glucose testing
- checks of your eyes, feet, kidneys, heart, circulation, blood and blood pressure. - Make sure all your friends, relatives, work colleagues or carers know that you have diabetes. Make sure they can recognise the symptoms of hypoglycaemia and know how to treat them. Provide them with the telephone number for your doctor, the Poisons Information Centre (13 11 26) and Emergency Services.
Symptoms of hypoglycaemia can occur suddenly. These may include:
- weakness, trembling or shaking
- sweating
- lightheadedness, dizziness, headache or lack of concentration
- tearfulness, crying, irritability
- hunger
- numbness around the lips and tongue. - Always carry some sugary food or drink with you.
If you experience any of the symptoms of hypoglycaemia, raise your blood glucose quickly by taking one of the following:
- 5-7 jelly beans
- 3 teaspoons of sugar or honey
- 1/2 can of sugar-containing soft drink
- 2-3 concentrated glucose tablets.
Unless you are within 10-15 minutes of your next meal or snack, follow up with extra carbohydrates such as plain biscuits, fruit or milk. Taking this extra carbohydrate will prevent a second drop in your blood glucose level.
Call your doctor straight away if you:
- become pregnant while taking Glimepiride Sandoz.
- become ill or experience extra stress, injury, fever, infection or need surgery.
- experience hypoglycaemia that is severe; it can result in seizures or a loss of consciousness. In this case, an ambulance should be called immediately. The risk of hypoglycaemia is increased in the following situations:
- too much Glimepiride Sandoz
- certain other medicines
- too much or unexpected exercise
- delayed meal or snack
- too little food. - notice the return of any symptoms you had before starting Glimepiride Sandoz (signs of hyperglycaemia (high blood glucose)). Signs of hyperglycaemia may include:
- lethargy or tiredness
- headache
- thirst
- passing large amounts of urine
- blurred vision.
The risk of hyperglycaemia is increased in the following situations:
- uncontrolled diabetes
- illness, infection or stress
- taking less Glimepiride Sandoz than prescribed
- certain other medicines
- too little exercise or sudden immobilisation, e.g. after an accident
- eating more carbohydrates than normal.
Remind any doctor, dentist or pharmacist you visit that you are using Glimepiride Sandoz.
Things you should not do
- Do not stop taking Glimepiride Sandoz or change the dose without first checking with your doctor.
- Do not give Glimepiride Sandoz to anyone else even if they have the same condition as you.
- Do not skip meals while taking Glimepiride Sandoz.
Things to be careful of
Protect your skin when you are in the sun, especially between 10am and 3pm
- Glimepiride Sandoz may cause your skin to be more sensitive to sunlight than it is normally. Exposure to sunlight may cause a skin rash, itching, redness or a severe sunburn.
If you become sick with a cold, fever or flu, it is very important to continue taking Glimepiride Sandoz, even if you feel unable to eat your normal meal.
- If you have trouble eating solid foods, use sugar-sweetened drinks as a carbohydrate substitute or eat small amounts of bland food. Your doctor, pharmacist or diabetes educator can give you a list of food to eat for sick days
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Glimepiride Sandoz affects you.
This medicine may cause dizziness, light-headedness and tiredness if your blood glucose levels fall too low. If you have any of these symptoms, do not drive, operate machinery or do anything else that could be dangerous.
Drinking alcohol
Be careful when drinking alcohol while you are taking this medicine.
If you drink alcohol, your blood glucose levels may fall and you may experience symptoms of hypoglycaemia.
Hypoglycaemia may slow your reaction time and affect your ability to drive or operate machinery. Drinking alcohol can also make this worse.
Looking after your medicine
- Keep your tablets in their container until it is time to take them.
If you take the tablets out of their container they may not keep well.
Store it in a cool dry place away from moisture, heat or sunlight below 25°C; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. They are generally mild and do not cause patients to stop taking Glimepiride Sandoz. |
Serious side effects
| Serious side effects | What to do |
Liver related:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Some side effects (for example changes in liver function or low blood cell counts) can only be found when your doctor does tests from time to time to check your progress
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Glimepiride Sandoz contains
| Active ingredient (main ingredient) | glimepiride |
| Other ingredients (inactive ingredients) |
Each strength tablet also contains the following colouring agents:
|
| Potential allergens | lactose |
Do not take this medicine if you are allergic to any of these ingredients.
What Glimepiride Sandoz looks like
Glimepiride Sandoz comes in four types of tablets:
Glimepiride Sandoz 1 mg– Light red, flat, modified oblong, scored tablet encoded G|1 on one side (Aust R 121781).
Glimepiride Sandoz 2 mg– Light green, flat, modified oblong, scored tablet encoded G|2 on one side
(Aust R 121786).
Glimepiride Sandoz 3 mg– Light yellow, flat, modified oblong, scored tablet encoded G|3 on one side
(Aust R 121788).
Glimepiride Sandoz 4 mg– Light blue, flat, modified oblong, scored tablet encoded G|4 on one side
(Aust R 121790).
Each tablet strength comes in blisters packs of 30 tablets.
Who distributes Glimepiride Sandoz
Sandoz Pty Ltd
100 Pacific Highway
North Sydney, NSW 2060
Australia
Tel 1800 726 369
This leaflet was prepared in December 2025.
® Registered Trade Mark. The trade marks mentioned in this material are the property of their respective owners.
Brand Information
| Brand name | Glimepiride Sandoz |
| Active ingredient | Glimepiride |
| Schedule | S4 |
MIMS Revision Date: 01 January 2025
1 Name of Medicine
Glimepiride.
2 Qualitative and Quantitative Composition
Glimepiride is a white odourless, crystalline powder, practically insoluble in methanol and water, slightly soluble in ethanol and sparingly soluble in methylene chloride.
Active. Glimepiride 1 mg / 2 mg / 3 mg / 4 mg.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Glimepiride Sandoz 1 mg tablets. A light red, flat, modified oblong, scored tablet encoded G/1 on one side: 30's.
Glimepiride Sandoz 2 mg tablets. A light green, flat, modified oblong, scored tablet encoded G/2 on one side: 30's.
Glimepiride Sandoz 3 mg tablets. A light yellow, flat, modified oblong, scored tablet encoded G/3 on one side: 30's.
Glimepiride Sandoz 4 mg tablets. A light blue, flat, modified oblong, scored tablet encoded G/4 on one side: 30's.
4 Clinical Particulars
4.1 Therapeutic Indications
As an adjunct to diet, exercise and weight loss, to lower the blood glucose in patients with noninsulin dependent diabetes mellitus (type 2 diabetes).
4.2 Dose and Method of Administration
Dosage. In the management of type 2 diabetes mellitus, administration of an oral antidiabetic agent is not a substitute for appropriate dietary control.
In initiating treatment for non-insulin dependent diabetes mellitus (NIDDM), diet should be emphasised as the primary form of treatment. Caloric restriction and weight loss are essential in the obese diabetic patient. Proper dietary management alone may be effective in controlling the blood glucose and symptoms of hyperglycaemia. The importance of regular physical activity should also be stressed, and cardiovascular risk factors should be identified and corrective measures taken where possible.
If this treatment program fails to reduce symptoms and/or blood glucose, the use of an oral sulfonylurea should be considered. Use of Glimepiride Sandoz must be viewed by both the doctor and patient as a treatment in addition to diet, and not as a substitute for diet or as a convenient mechanism for avoiding dietary restraint. Furthermore, loss of blood glucose control on diet alone may be transient, thus requiring only short-term administration of Glimepiride Sandoz. During maintenance programs, Glimepiride Sandoz should be discontinued and insulin therapy initiated if satisfactory lowering of blood glucose is no longer achieved. Judgments should be based on regular clinical and laboratory evaluations.
The dosage of Glimepiride Sandoz must be the lowest, which is sufficient to achieve the desired metabolic control. Dosage must be based on regular blood and urine glucose determinations, and must be carefully individualised to obtain optimum therapeutic effect. Periodic measurement of glycosylated haemoglobin is also recommended to monitor the patient's response to treatment. If appropriate glimepiride dosage regimens are not followed, hypoglycaemia may be precipitated.
Measures for dealing with errors in dosage such as forgetting to take a dose, skipping a meal or inability to take a dose at the prescribed time should be discussed with the patient at the time of initiating therapy. A missed dose must never be corrected by subsequently taking a larger dose.
Short-term administration of Glimepiride Sandoz may be sufficient during periods of transient loss of metabolic control in patients usually well controlled on diet.
Food does not alter the bioavailability or other pharmacokinetic parameters of glimepiride.
Initial dose and dose titration. The initial dose of Glimepiride Sandoz is one 1 mg tablet once daily.
If good metabolic control is achieved within the first week of treatment (as determined by blood and urine glucose), continue the daily dose of one 1 mg tablet as maintenance therapy.
If metabolic control is unsatisfactory after one to two weeks of treatment, increase the daily dose in increments of 1 mg at one to two week intervals, until satisfactory metabolic control is achieved. Most patients will achieve optimum control at doses of 1 to 4 mg once daily. Only in exceptional cases will doses of more than glimepiride 4 mg/day give better results. Normally, a single daily dose will maintain good blood glucose control for 24 hours.
Secondary dosage adjustment. Glimepiride Sandoz requirements may fall as treatment proceeds because an improvement in diabetes control results in greater insulin sensitivity. To avoid hypoglycaemia, timely dose reduction or cessation of therapy should be considered.
Correction of Glimepiride Sandoz dosage must also be considered whenever the patient's weight or lifestyle changes or other factors arise which affect glycaemic control.
Secondary failures should be treated by discontinuing Glimepiride Sandoz and starting insulin.
Changeover from other antidiabetic agents to Glimepiride Sandoz. There is no exact dosage relationship between Glimepiride Sandoz and other oral antidiabetic agents. When transferring patients from another oral antidiabetic drug to Glimepiride Sandoz, it is recommended to begin with the usual starting dose of 1 mg once daily. This recommendation applies even in cases where the patient is being switched from the maximum dose of another antidiabetic agent.
Depending on the pharmacokinetic and pharmacodynamic characteristics of the previous medication, a drug free transition period may be necessary in order to avoid overlapping drug effects possibly resulting in hypoglycaemia.
Method of administration. The tablet should be swallowed whole without chewing with adequate liquid (e.g. half a glass of water) immediately before breakfast. Patients who eat only a light breakfast should defer the first dose of the day until the first main meal of the day (e.g. lunch). It is very important that meals are not skipped after the tablet has been taken.
Dosage adjustment. Renal impairment. There is limited information available on the use of Glimepiride Sandoz in renal insufficiency. Patients with impaired renal function may be more sensitive to the glucose-lowering effect of Glimepiride Sandoz (see Section 5.2 Pharmacokinetic Properties, Renal impairment). In patients with mild (CrCl > 50 mL/min) to moderate (CrCl 30-50 mL/min) renal impairment, a starting dose of 1 mg once daily must not be exceeded. The dose may then be carefully titrated upwards if necessary based on fasting blood glucose levels according to the protocol mentioned above (i.e. in increments of 1 mg at intervals of one to two weeks).
No experience has been gained in the use of glimepiride in dialysis patients or patients with severe renal impairment. These patients should be changed over to insulin therapy to achieve optimum metabolic control.
Hepatic impairment. No experience has been gained in the use of glimepiride in patients with severe hepatic impairment. These patients should be changed over to insulin therapy to achieve optimum metabolic control.
4.3 Contraindications
Glimepiride is contraindicated in patients with the following conditions:
Hypersensitivity to glimepiride, other sulfonylureas, other sulfonamides or any excipient.
Severe impairment of renal function (CrCl < 30 mL/min).
Dialysis patients.
Severe hepatic dysfunction.
Pregnancy (see Section 4.6 Fertility, Pregnancy and Lactation, Use in pregnancy).
Lactation (see Section 4.6 Fertility, Pregnancy and Lactation, Use in lactation).
Glimepiride Sandoz is not suitable for the treatment of insulin dependent (type 1) diabetes mellitus (e.g. for the treatment of patients with a history of ketoacidosis), nor for the treatment of diabetic ketoacidosis, nor for the treatment of diabetic precoma or coma.
In patients with severe impairment of hepatic function, changeover to insulin is indicated to achieve optimal metabolic control.
4.4 Special Warnings and Precautions for Use
Patients receiving glimepiride should be monitored with regular clinical and laboratory evaluations, including blood and urine glucose determinations, to determine the minimum effective dosage and to detect primary failure (inadequate lowering of blood glucose concentration at the maximum recommended dosage) or secondary failure (loss of control of blood glucose concentration following an initial period of effectiveness) to the drug. Glycosylated haemoglobin measurements may also be useful for monitoring the patient's response to glimepiride therapy.
Some improvement in glucose tolerance may take place after a few weeks of treatment with glimepiride. The clinical status should be checked within the first four to eight weeks and at regular intervals thereafter to ascertain whether it is possible to reduce the dose.
The treatment of diabetes requires regular checks. Alertness and reactions may be impaired due to hypoglycaemia or hyperglycaemia, especially when beginning or after altering treatment, or when glimepiride is not taken regularly. This may, for example, affect the ability to drive or to operate machinery.
In cases of unusual stress (e.g. trauma, emergency or elective surgery, febrile infection), blood glucose regulation may deteriorate and a temporary change to insulin may become necessary to maintain good metabolic control.
Glimepiride Sandoz tablets must not be used beyond the expiry date marked on the pack and must be stored out of the reach of children.
This medicinal product contains lactose monohydrate. Patients with rare hereditary problems of galactose intolerance, the Lapp lactase deficiency or glucose-galactose malabsorption should not take this medicine.
Hypoglycaemic reactions. Hypoglycaemia is a potential risk from treatment with any sulfonylurea, particularly in the first month of treatment or when dosage is increased.
Debilitated patients, malnourished patients and patients with adrenal, pituitary, renal or hepatic insufficiency are particularly susceptible to the hypoglycaemic action of sulfonylureas and should therefore be carefully monitored. The dosage of glimepiride should be carefully adjusted in these patients.
Hepatic insufficiency may cause increased serum concentrations of glimepiride and may diminish gluconeogenic capacity, both of which increase the risk of severe hypoglycaemic reactions.
Alcohol ingestion, severe or prolonged exercise, deficient caloric intake or use of more than one antidiabetic agent may predispose patients to the development of hypoglycaemia.
If risk factors for hypoglycaemia are present, it may be necessary to adjust the dosage of glimepiride or the entire therapy. This also applies whenever illness occurs during therapy or the patient's lifestyle changes.
Hypoglycaemia may be difficult to recognise in the elderly and in people who are taking beta-adrenergic blocking drugs or other sympatholytic agents.
Patients and responsible family members should be made aware of the signs and symptoms of hyperglycaemia (severe thirst, dry mouth, frequent micturition, dry skin) and hypoglycaemia (intense hunger, sweating, tremor, restlessness, irritability, depression, headache, disturbed sleep or transient neurological disorders) and the prompt action to be taken if either event should occur.
The potential for primary and secondary failure should also be explained.
Hypoglycaemia can almost always be promptly controlled by the intake of carbohydrates (glucose or sugar). It is known from other sulfonylureas that, despite initial successful countermeasures, hypoglycaemia may recur. Patients must therefore remain under close observation. Severe hypoglycaemia requires immediate treatment and follow-up by a doctor and, in some circumstances, in-patient hospital care.
Haemolytic anaemia. Treatment of patients with glucose-6-phosphate dehydrogenase (G6PD) deficiency with sulfonylurea agents can lead to haemolytic anaemia. Since glimepiride belongs to the class of sulfonylurea agents, caution should be used in patients with G6PD-deficiency and a nonsulfonylurea alternative should be considered.
Use in the elderly. No data available.
Paediatric use. The safety and efficacy of glimepiride in children have not been established. Glimepiride is not recommended for use in this age group.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Glimepiride is metabolised by cytochrome P450 2C9 (CYP2C9). This should be taken into account when glimepiride is co-administered with inducers, inhibitors or substrates of CYP2C9 (e.g. rifampicin, fluconazole, amiodarone, tolbutamide, diclofenac, ibuprofen, naproxen).
Based on experience with glimepiride and known interactions for other sulfonylureas, the following interactions must be considered.
In addition to insulin and other oral antidiabetic agents, drugs which may potentiate the hypoglycaemic action of glimepiride include ACE inhibitors, aminosalicylic acid, anabolic steroids and male sex hormones, allopurinol, azapropazone, chloramphenicol, clarithromycin, clofibrate, coumarin derivatives, cyclophosphamide, disopyramide, fenfluramine, fenyramidol, fibrates, fluconazole, fluoxetine, guanethidine, ifosphamides, monoamine oxidase inhibitors (MAOIs), miconazole, oxpentifylline (high dose parenteral), oxyphenbutazone, para-aminosalicylic acid, pentoxifylline (high dose parenteral), phenylbutazone, probenecid, quinolone antibiotic, salicylates, sulfinpyrazone, sulfonamide antibiotics, tetracyclines, tritoqualine, trofosphamide.
Drugs which may attenuate the hypoglycaemic action of glimepiride include acetazolamide, barbiturates, calcium channel blockers, corticosteroids, diazoxide, diuretics, glucagon, isoniazid, laxatives (protracted use), nicotinic acid (high doses), oestrogens, phenothiazines, chlorpromazine, phenytoin, progestogens, rifampicin, adrenaline and other sympathomimetic agents, thyroid hormones.
H2-receptor antagonists, beta-blockers, clonidine and reserpine may lead to either potentiation or weakening of the blood-glucose-lowering effect.
Concomitant treatment with a beta-receptor blocker, clonidine, guanethidine or reserpine may mask the warning symptoms of a hypoglycaemic attack.
Acute and chronic alcohol intake may either potentiate or attenuate the activity of glimepiride in an unpredictable fashion.
The effect of coumarin derivatives may be potentiated or weakened.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Glimepiride had no recognisable effects on the rearing, physical development, functional and learning behaviour, memory or fertility of the progeny.
Use in pregnancy. (Category C)
Category C. Drugs, which, owing to their pharmacological effects, have caused or may be suspected of causing, harmful effects on the human fetus or neonate without causing malformations. These effects may be reversible. Accompanying texts should be consulted for further details.
It is important to achieve strict normoglycaemia during pregnancy. Glimepiride must not be taken during pregnancy. Otherwise, there is risk of harm to the child. The patient must change over to insulin during pregnancy. Patients planning a pregnancy must inform their doctor. It is recommended that such patients change over to insulin.
The sulfonylureas may enter the foetal circulation and cause neonatal hypoglycaemia. In rats, dietary glimepiride at high doses (approximately 82 mg/kg) during gestation caused limb deformations. In rabbits, effects on pregnancy were characterised by increased incidences of abortions/ total resorptions and malformations. Similar foetal wastage was not seen in rats although the finding of anophthalmia in a proportion of foetuses may be indicative of a treatment-related effect as eye malformations were found in the rabbit study. Adverse pregnancy outcomes in the rat and rabbit are probably due to the pharmacodynamic effects of glimepiride at excessive doses and are not substance-specific.
Use in lactation. Studies in rats showed that glimepiride is excreted in milk. High doses caused hypoglycaemia in suckling young rats. Dietary administration of glimepiride (120 to 206 mg/kg) during lactation caused limb deformations in adolescent pups from day 4 of lactation onwards. To prevent possible ingestion of glimepiride with the breast milk and possible harm to the child, glimepiride must not be taken by breastfeeding women. Nursing mothers must either be changed over to insulin or cease breastfeeding.
4.7 Effects on Ability to Drive and Use Machines
Alertness and reactions may be impaired due to hypo- or hyperglycaemia, especially when beginning or after altering treatment or when glimepiride is not taken regularly. This may affect the ability to drive or to operate machinery.
4.8 Adverse Effects (Undesirable Effects)
Glimepiride is generally well tolerated. Clinical experience has shown that adverse reactions serious enough to compel discontinuation of therapy are uncommon, even during long-term treatment.
Hypoglycaemia. Hypoglycaemia is the greatest potential risk with all sulfonylureas. These hypoglycaemic reactions mostly occur immediately, may be severe and are not always easy to correct.
Based on what is known of other sulfonylureas, hypoglycaemia may be prolonged. Possible symptoms of hypoglycaemia include headache, ravenous hunger, nausea, vomiting, lassitude, sleepiness, disordered sleep, restlessness, aggressiveness, impaired concentration, impaired alertness and reactions, depression, confusion, speech disorders, aphasia, visual disorders, tremor, pareses, sensory disturbances, dizziness, helplessness, loss of self control, delirium, cerebral convulsions, somnolence and loss of consciousness up to and including coma, shallow respiration and bradycardia.
In addition, signs of adrenergic counter-regulation may be present, including sweating, clammy skin, anxiety, tachycardia, hypertension, palpitations, angina pectoris and cardiac arrhythmias.
The clinical picture of a severe hypoglycaemic attack may resemble that of a stroke.
The symptoms nearly always subside when hypoglycaemia is corrected.
Visual reactions. Especially at the start of treatment, there may be temporary visual impairment (e.g. changes in accommodation and/or blurred vision) due to the change in blood glucose levels. The cause is a temporary alteration in the turgidity and hence the refractive index of the lens, this being dependent on blood glucose level.
Gastrointestinal reactions. Occasionally (0.1 to 1% of patients), gastrointestinal symptoms such as nausea, vomiting, sensations of pressure or fullness in the epigastrium, abdominal distension, abdominal discomfort, abdominal pain and diarrhoea may occur. These symptoms seldomly lead to discontinuation of therapy.
Dysgeusia, taste disturbance or loss of taste has been reported.
Haematological reactions. Changes in the blood picture may occur: thrombocytopenia, leucopoenia, haemolytic anaemia, erythrocytopenia, granulocytopenia, agranulocytosis or pancytopenia may develop. Cases of severe thrombocytopenia with platelet count less than 10,000/microlitre and thrombocytopenic purpura have been reported in post-marketing experience (frequency not known). Anaemia, eosinophilia and aplastic anaemia have been reported with sulfonylureas.
Dermatological reactions. Allergic or pseudo-allergic skin reactions (e.g. itching, pruritus, erythema, urticaria, rashes, erythematous and maculopapular and bullous skin eruptions or psoriasiform drug eruption) may occur in patients treated with sulfonylureas. If skin reactions persist, the drug should be discontinued. Mild reactions in the form of urticaria may develop into serious and even life threatening reactions with dyspnoea and hypotension, sometimes progressing to shock. In the event of urticaria, the doctor must be notified immediately. In isolated cases, a decrease in serum sodium concentration and allergic vasculitis or hypersensitivity of the skin to light may occur. Porphyria cutanea tarda and pellagra-like changes and photosensitivity reactions have been reported with sulfonylureas. It should be noted that cross reactivity exists between sulfonylureas and sulfonamides.
Hepatic reactions. In isolated cases, increased liver enzymes (AST, ALT), abnormal liver function, cholestasis, cholestatic hepatitis, granulomatous hepatitis, bilirubinaemia and liver failure have been reported with sulfonylureas. In isolated cases, there may be hepatitis, elevation of liver enzymes and/or cholestasis and jaundice which may progress to life-threatening liver failure but can regress after withdrawal of glimepiride.
Electrolyte disturbance. In isolated cases, hyponatraemia has been reported in patients receiving glimepiride and other sulfonylureas, most often in patients who are on other medications or have medical conditions known to cause hyponatraemia or to increase release of antidiuretic hormone.
Other. Isolated cases of leukocytoclastic vasculitis have been reported with sulfonylureas.
Occasionally, allergic or pseudoallergic reactions may occur, e.g. in the form of itching, urticaria or rashes. Such mild reactions may develop into serious reactions with dyspnoea and a fall in blood pressure, sometimes progressing to shock. In the event of urticaria a doctor must therefore be notified immediately. Cases of alopecia and weight gain have also been reported.
In isolated cases, a decrease in serum sodium concentration or hypersensitivity of the skin to light may occur.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Symptoms. Accidental or intentional overdose may cause severe and prolonged hypoglycaemia, lasting from 12 to 72 hours and may be life threatening. Symptoms may not be present for up to 24 hours after ingestion.
Nausea, vomiting and epigastric pain may occur. The hypoglycaemia may in general be accompanied by neurological symptoms like restlessness, tremor, visual disturbances, coordination problems, sleepiness, coma and convulsions.
Treatment. In case of overdosage with glimepiride, a doctor must be notified immediately. At the first signs of hypoglycaemia, the patient must immediately take sugar, preferably glucose, unless a doctor has already started care.
Treatment primarily consists of preventing absorption by inducing vomiting and then drinking water or lemonade with activated charcoal (adsorbent) and sodium-sulphate (laxative). If large quantities have been ingested, gastric lavage is indicated, followed by activated charcoal and sodium-sulphate.
Since hypoglycaemia and its clinical symptoms may recur after apparent clinical recovery (even after several days), close and continued medical supervision and possibly referral to a hospital are indicated. In particular, significant overdosage and severe reactions, e.g. with unconsciousness or other neurological dysfunctions, are emergency cases and require immediate care and hospitalisation.
If hypoglycaemic coma is diagnosed or suspected intravenous infusion of a glucose 20% solution (adults: 40 to 100 mL) is indicated. Alternatively, intravenous, subcutaneous or intramuscular administration of glucagon (adults: 0.5 to 1 mg) may be considered. In infants, glucose must be dosed very carefully and close monitoring of blood glucose is required to minimise the risk of potentially severe hyperglycaemia. Other symptomatic therapy (e.g. anticonvulsants) should be administered as necessary.
After acute glucose replacement has been completed, it is usually necessary to give an intravenous glucose infusion in lower concentration so as to ensure that hypoglycaemia does not recur. The patient's blood glucose level should be carefully monitored for at least 24 hours. In severe cases with a protracted course, hypoglycaemia, or the danger of slipping back into hypoglycaemia, may persist for several days.
In cases of acute intake of large amounts of glimepiride, detoxification (e.g. by gastric lavage and administration of medicinal charcoal) is indicated.
For information on the management of overdose, contact the Poison Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Glimepiride is a member of the sulfonylurea group of oral antidiabetic agents.
Glimepiride is a sulfonylurea antidiabetic agent which decreases blood glucose concentrations. The primary mechanism of action of glimepiride appears to be dependent on stimulating the release of insulin from functioning pancreatic beta cells. Glimepiride acts in concert with glucose by improving the sensitivity of beta cells to physiological glucose stimulus, resulting in insulin secretion in the rhythm of meals. In addition, extrapancreatic effects (e.g. reduction of basal hepatic glucose production and increased peripheral tissue sensitivity to insulin and glucose uptake) may also play a limited role in the activity of glimepiride.
In non-fasting diabetic patients, the hypoglycaemic action of a single dose of glimepiride persists for 24 hours.
Evidence from in vitro and animal studies suggests that there is lower glucagon secretion with glimepiride than glibenclamide and this may give rise to a prolonged reduction of blood glucose levels without increased plasma insulin levels. The clinical significance of these findings is yet to be clarified. A long-term, randomised, placebo-controlled clinical trial demonstrated that glimepiride therapy improves postprandial insulin/ C-peptide responses and overall glycaemic control without producing clinically meaningful increases in fasting insulin/ C-peptide levels.
The efficacy of glimepiride is not affected by age, gender or weight. Glimepiride therapy is effective in controlling blood glucose without deleterious changes in the plasma lipoprotein profile of patients. The physiological response to acute exercise (i.e. reduction of insulin secretion) is still present during glimepiride therapy.
Clinical trials. A placebo controlled study using fixed daily doses of glimepiride 1 mg, 4 mg and 8 mg found that all three doses were effective at reducing blood glucose levels. However, there was no significant difference in the reduction in fasting plasma glucose (FPG) between the 4 mg and 8 mg doses at any time point throughout the study.
In another placebo-controlled dose-ranging study of glimepiride (1, 2, 3, 4, 6 and 8 mg/day), the majority of patients were controlled in the dose range of 1 to 4 mg daily. There was only a very small difference in the reduction in median FPG levels between the 4 mg and 8 mg doses (-3.08 mmol/L versus -3.16 mmol/L). The greatest change of -3.27 mmol/L was seen with the 2 mg dose. This supports the results of the aforementioned clinical study.
Two large multicentre studies involving approximately 1,900 patients were conducted to examine the dose-response effect of glimepiride on blood glucose and HbA1c levels. In both these studies, a large proportion of patients achieved a FPG level below 8.32 mmol/L at the 1 mg/day dose, with a further 10% of patients achieving this level at the 2 mg/day dose. Some patients benefited by an increase in dose to 4 mg/day, but only a few patients, mainly those with very high baseline FPG levels, required higher doses. Based on the results of these studies, the World Health Organization (WHO) has set the defined daily dose (DDD) of glimepiride to be 2 mg.
An additional 161 patient, randomised, double blind crossover study, including four weeks active treatment each with 3 mg bd (twice daily) or 6 mg daily of glimepiride, indicated that some patients may have improved results when glimepiride is given twice daily. However, for the majority of patients, once daily dosing provided adequate control. It is important to note that the treatment period in this study was only four weeks and, as such, the long-term safety benefit of twice daily dosing has not been established.
Bioequivalence. Bioequivalence between Glimepiride Sandoz tablets and Amaryl tablets was demonstrated in a comparative, randomised, two-way cross-over study in 24 healthy volunteers (13 males, 11 females, 23 completed) receiving a single dose of 4 mg glimepiride of either formulation orally. The pharmacokinetic parameters are summarised in Table 1.

5.2 Pharmacokinetic Properties
The pharmacokinetics of glimepiride are similar in males and females and also in young and elderly (> 65 years) patients. Intra-individual variability is low.
Absorption. Glimepiride is completely absorbed after oral administration. The peak serum concentration (Cmax) is reached in about 2.5 hours. There is a linear relationship between dose and both Cmax and AUC (area under the plasma concentration-time curve). Food does not significantly affect the rate or extent of absorption of glimepiride.
Distribution. After intravenous dosing in normal subjects, the volume of distribution was 8.8 L (113 mL/kg) and the total body clearance was 48 mL/minute. Protein binding was greater than 99%.
Glimepiride is likely to be only minimally removed by haemodialysis due to its high protein binding.
Multiple-dose studies with glimepiride in diabetic patients demonstrated plasma concentration-time curves similar to single dose studies, indicating that there is no accumulation of drug in tissue depots.
Metabolism. The elimination half-life of glimepiride at steady state is about five to eight hours after oral administration. However, results of a pharmacokinetic study on (type 2 diabetes mellitus) noninsulin dependent diabetes mellitus (NIDDM) patients indicated that higher doses may be associated with a longer half-life.
Glimepiride is completely metabolised by oxidative biotransformation. The major metabolites are the cyclohexyl hydroxy methyl derivative (M1) and the carboxyl derivative (M2). In vitro studies indicate that cytochrome P450 2C9 is the principal enzyme involved in the biotransformation of glimepiride to M1. M1 has been found to have about 40% of the pharmacological activity of glimepiride. It is eliminated via the urine and also by further metabolism to M2 via one or several cytosolic enzymes. M1 has a terminal elimination half-life of three to six hours after an oral dose. The formation of M1 is linear up to a dose of glimepiride 16 mg. The kinetics of M2 have not been fully elucidated due to low plasma levels. Its terminal elimination half-life after an oral dose is about five to six hours.
Excretion. Following an oral dose of glimepiride, 35% of the dose is excreted in faeces and 58% in urine.
Renal impairment. In a single-dose, open-label study conducted in 15 patients with renal impairment, glimepiride (3 mg) was administered to three groups of patients with different levels of mean creatinine clearance (CrCl); (group I, CrCl = 77.7 mL/minute, n = 5), (group II, CrCl = 27.4 mL/minute, n = 3), and (group III, CrCl = 9.4 mL/minute, n = 7). Glimepiride was found to be well tolerated in all three groups. The results showed that glimepiride serum levels decreased as renal function decreased. However, M1 and M2 serum levels (mean AUC (area under the curve) values) increased 2.3 and 8.6 times from group I to group III. The apparent terminal half-life (t1/2) for glimepiride did not change, while the half-lives for M1 and M2 increased as renal function decreased. Mean urinary excretion of M1 plus M2 as percent of dose, however, decreased (44.4%, 21.9% and 9.3% for group I to III).
Results from a multiple-dose titration study conducted in 16 patients with renal impairment using doses ranging from 1 to 8 mg daily for three months were consistent with the results after a single dose. All patients with a CrCl < 22 mL/minute had adequate control of their glucose levels with a dosage regimen of only 1 mg daily. The results from this study suggested that a starting dose of glimepiride 1 mg may be given to a patient with type 2 diabetes mellitus with renal disease, and the dose may be titrated based on fasting blood glucose levels (see Section 4.3 Contraindications; Section 4.2 Dose and Method of Administration).
It is not known if glimepiride is dialysable.
Hepatic failure. The effects of hepatic failure on the clearance of glimepiride have not been systematically examined.
5.3 Preclinical Safety Data
Genotoxicity. A standard battery of laboratory tests did not reveal any genotoxic or mutagenic potential for glimepiride.
Carcinogenicity. In a two year carcinogenicity study in mice receiving glimepiride in the diet up to 813 mg/kg/day, there was an increase in the incidence of pancreatic islet cell hyperplasia and islet cell adenomas; these are regarded to be the result of chronic stimulation of the pancreatic beta cells. In a 30 month carcinogenicity study in rats receiving glimepiride in the diet up to 345 mg/kg/day, there was an increased incidence of pancreatic islet cell adenomas, however these were considered incidental as there was no dose relationship in either sex. There were no malignant tumours in rats or mice.
6 Pharmaceutical Particulars
6.1 List of Excipients
Lactose, cellulose microcrystalline, sodium starch glycollate, povidone and magnesium stearate. Additionally, each strength contains the following colouring agents: 1 mg tablet: iron oxide red; 2 mg tablet: iron oxide yellow and indigo carmine; 3 mg tablet: iron oxide yellow; 4 mg tablet: indigo carmine.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
For information on interactions with other medicines and other forms of interactions, see Section 4.5.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C.
6.5 Nature and Contents of Container
Glimepiride Sandoz tablets are available in PVC-Al blister packs and HDPE bottles*.
* Not currently marketed in Australia.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
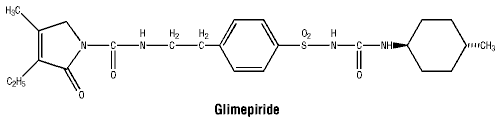
Chemical structure. The chemical name of glimepiride is trans-1-{4-[2- (3-ethyl-4-methyl- 2-oxo-3-pyrroline-1 carboxamido)ethyl]-phenylsulfonyl}-3-(4-methylcyclohexyl) urea. Its empirical formula is C24H34N4O5S (MW: 491) and its chemical structure is:
7 Medicine Schedule (Poisons Standard)
S4 - Prescription Only Medicine.
Date of First Approval
10 July 2006
Date of Revision
22 November 2024
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.