Baxter Heparin Sodium in 0.9% Sodium Chloride Intravenous Infusion
Brand Information
| Brand name | Baxter Heparin Sodium in 0.9% Sodium Chloride Intravenous Infusion |
| Active ingredient | Heparin sodium + Sodium chloride |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Baxter Heparin Sodium in 0.9% Sodium Chloride Intravenous Infusion.
Summary CMI
BAXTER HEPARIN SODIUM in 0.9% SODIUM CHLORIDE Intravenous Infusion
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I receiving Heparin Sodium?
BAXTER HEPARIN SODIUM in 0.9% SODIUM CHLORIDE Intravenous Infusion (‘Heparin Sodium’) contains the active ingredient heparin sodium derived from porcine (pig) intestinal mucosa. Heparin Sodium is used to prevent your blood from clotting when it is removed from the body, for example during dialysis or to ensure that catheters do not become blocked.
For more information, see Section 1. Why am I receiving Heparin Sodium? in the full CMI.
2. What should I know before I receive Heparin Sodium?
Do not use if you have ever had an allergic reaction to heparin, pork products or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I receive Heparin Sodium? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Heparin Sodium and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How is Heparin Sodium given?
- Your doctor will determine the dose of Heparin Sodium you require and the duration of use, depending on your need and condition.
More instructions can be found in Section 4. How is Heparin Sodium given? in the full CMI.
5. What should I know while receiving Heparin Sodium?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while receiving Heparin Sodium? in the full CMI.
6. Are there any side effects?
Some of the common and less serious side effects include local irritation at the injection site, or mild pain around the injection site. Serious side effects that may occur include: allergic reaction, swelling or changing skin colour at the injection site, easy bruising and increased bleeding.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
1. Why am I receiving Heparin Sodium?
Heparin Sodium contains the active ingredient heparin sodium. Heparin Sodium is a medicine that prevents blood from clotting (anticoagulant). It is given to you by a healthcare professional.
Heparin Sodium can be used to:
- prevent your blood from clotting when it is removed from the body, for example during dialysis
- ensure that a catheter (a thin tube used to drain fluids from or deliver fluids into the body) does not become blocked.
2. What should I know before I receive Heparin Sodium?
Warnings
Do not use Heparin Sodium if:
- you are allergic to heparin, pork products or any of the ingredients listed at the end of this leaflet.
- Always check the ingredients to make sure you can use this medicine.
- you have a bleeding condition or problems with your blood vessels
- you have low blood platelet count (thrombocytopenia), or are bruising or bleeding more easily than normal
- you have or have had major bleeding disorders, injury to the brain, stomach or bowel problems, or bacterial infections of the heart.
Check with your doctor if you:
- have asthma
- have heart problems or high blood pressure
- have liver or kidney problems
- have a stomach ulcer or other digestive issues
- have bleeding or clotting problems
- have heavy or irregular periods
- have recently had a surgical, medical or dental procedure, especially surgery on the eyes, brain or spinal cord
- have recently had a spinal tap, or local anaesthetic around the spinal cord
- have diabetes
- have had a reaction to heparin in the past
- have any other medical conditions
- take any medicines for any other condition
- smoke or use vapes
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Tell your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Your doctor can discuss with you the risks and benefits involved.
Use in Elderly
Check with your doctor about the risks of using this medicine if you are over 60 years of age. Your doctor may adjust the dose to prevent bleeding side effects.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Heparin Sodium and affect how it works.
- pain-relieving and non-steroidal anti-inflammatory medicines (aspirin, ibuprofen, indomethacin and phenylbutazone)
- medicines for rheumatoid arthritis (hydroxychloroquine)
- medicines for heart and circulation problems (digitalis, nitroglycerine, dipyridamole and epoprostenol)
- medicines for hay fever, insomnia or travel sickness (antihistamines)
- smoking cessation products containing nicotine such as gum or patches
- anticlotting medicines (dicumarol, clopidogrel, thienopyridines, glycoprotein IIb/IIIa antagonists, antithrombin III and warfarin)
- medicine for boosting blood volume (dextran)
- medicines for treating infections – antibiotics (tetracyclines)
- medicines used in the treatment of high blood pressure (angiotensin converting enzyme inhibitors or angiotensin II receptor antagonist)
Heparin may lead to an increase of potassium in your blood. Therefore, you should tell your doctor if you are taking any medicines that raise your potassium level.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Heparin Sodium.
4. How is Heparin Sodium given?
How it is given
Your doctor will decide the best way to give you Heparin Sodium, depending on your need and condition.
- Your doctor or trained nurse may give Heparin Sodium by an infusion into a vein (intravenously). A cannula (administration needle) is placed in a vein prior to the infusion.
- If Heparin Sodium is being used to prevent blood clotting during dialysis, your doctor or nurse will add it to the dialysis machine. They will do this before and sometimes during the cycle.
- If it is being used to ensure that your catheter does not become blocked, your doctor or nurse will inject a small amount into the catheter from time to time.
Heparin Sodium is usually given in a hospital, clinic or nursing home. The infusion requires special equipment and medical training. You may receive it at home if your doctor has arranged for it.
How much is given
- Your doctor will decide how much Heparin Sodium to give. The amount depends on your need and condition.
- If it is being used to clear your catheter, the dose will depend on the type of catheter you have.
If you use too much Heparin Sodium
Overdose is unlikely as treatment will be given by trained medical personnel. Your doctor or pharmacist has information on how to recognise and treat an overdose.
If you think that you may have been given too much Heparin you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while receiving Heparin Sodium?
Things you should do
During and after treatment, discuss your progress with your doctor. Tell them if you've had any side effects or concerns, especially during the first few days of therapy.
Your doctor and nurse will closely monitor you during treatment, and may use laboratory tests to make sure you are safely receiving the right amount of Heparin Sodium.
Discuss with your doctor straight away if you:
Have had or are having an epidural anaesthetic or spinal puncture, and you notice any of the following symptoms:
- numbness, tingling or muscle weakness
- abnormal function or loss of control of bowel or urine
- any other symptom that worries you.
Remind any doctor, nurse, dentist or pharmacist you visit that you are using Heparin Sodium.
Things you should not do
- Do not stop using this medicine suddenly.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Heparin Sodium affects you.
Looking after your medicine
This medicine is stored in the pharmacy or the hospital ward, kept below 30°C.
Getting rid of any unwanted medicine
The infusion is for single use, and for one person only. Any unused portion must be discarded and not used later, either for you or anyone else.
If you have this medicine at home, and no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Injection site-related
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Allergy-related
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people. Some side effects may only be seen by your doctor.
If you are over 60 years of age you may have an increased chance of getting side effects, especially if you are female.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Heparin Sodium contains
| Active ingredient (main ingredient) | heparin sodium (from porcine mucosal membrane) |
| Other ingredients (inactive ingredients) | sodium chloride, citric acid monohydrate, dibasic sodium phosphate dodecahydrate, water for injections |
Do not take this medicine if you are allergic to any of these ingredients.
What Heparin Sodium looks like
Heparin Sodium is a clear, colourless solution filled in plastic bags:
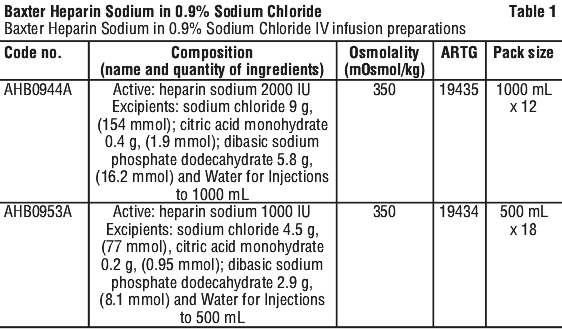
- Baxter Heparin Sodium 1000IU 0.9% Sodium Chloride Injection 500mL Injection Bag (Aust R 19434)
- Baxter Heparin Sodium 2000IU 0.9% Sodium Chloride Injection 1000mL Injection Bag (Aust R 19435)
Who distributes Heparin Sodium
Baxter Healthcare Pty Ltd
1 Baxter Drive
Old Toongabbie
NSW 2146
Australia
This leaflet was prepared in September 2025
Brand Information
| Brand name | Baxter Heparin Sodium in 0.9% Sodium Chloride Intravenous Infusion |
| Active ingredient | Heparin sodium + Sodium chloride |
| Schedule | S4 |
MIMS Revision Date: 01 February 2022
1 Name of Medicine
Heparin sodium.
2 Qualitative and Quantitative Composition
Baxter Heparin Sodium in 0.9% Sodium Chloride Intravenous (IV) Infusion is a sterile, nonpyrogenic solution of heparin sodium standardised for use as an anticoagulant in 0.9% Sodium Chloride IV Infusion buffered with 0.4 mg citric acid monohydrate and 5.8 mg dibasic sodium phosphate dodecahydrate (Na2HPO4.12H2O) per mL to pH range of 5.5 - 8.0. It is supplied in single dose Viaflex plastic bag containers for intravenous administration.
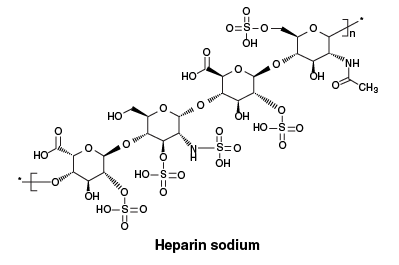
Heparin is a heterogenous mixture of variably sulfated polysaccharide chains composed of repeating units of disaccharides, D-glucosamine and L-iduronic acid or D-glucosamine and D-glucuronic acids. It is extracted from porcine intestinal mucosa. Upon complete hydrolysis, it yields a mixture of D-glucosamine, D-glucuronic acid, L-iduronic acid, acetic acid and sulphuric acid. Heparin is strongly acidic because of its content of covalently linked sulfate and carboxylic acid groups. In heparin sodium, the acidic protons of the sulfate units are partially replaced by sodium ions.
Although others may be present, the main sugars occurring in heparin are: (1) α-L-iduronic acid 2-sulfate, (2) 2-deoxy-2-sulfamino-α-D-glucose 6-sulfate, (3) β-D-glucuronic acid, (4) 2-acetamido-2-deoxy-α-D-glucose, and (5) α-L-iduronic acid. These sugars are present in decreasing amounts, usually in the order (2) > (1) > (4) > (3) > (5), and are joined by glycosidic linkages, forming polymers of varying sizes.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Clear solution for intravenous infusion.
4 Clinical Particulars
4.1 Therapeutic Indications
Baxter Heparin Sodium in 0.9% Sodium Chloride Intravenous IV Infusion is indicated as an anticoagulant in extracorporeal circulation, dialysis procedures and as an aid in the maintenance of catheter patency.
4.2 Dose and Method of Administration
Heparin sodium is not effective by oral administration and Heparin Sodium in 0.9% Sodium Chloride Injection should not be given orally.
Dosage, rate, and duration of administration are to be individualised and depend upon the indication for use, the patient's age, weight, clinical condition and concomitant treatment, and on the patient's clinical and laboratory response to the treatment.
Carefully examine all presentations of heparin to confirm the correct formulation prior to administration of the drug.
Parenteral drug products should be inspected visually for particulate matter and discolouration prior to administration. Do not administer unless solution is clear and seal is intact. Use of a final filter is recommended during administration of all parenteral solutions, where possible.
Do not use plastic containers in series connections. Such use could result in air embolism due to residual air being drawn from the primary container before administration of the fluid from the secondary container is completed.
Pressurising intravenous solutions contained in flexible plastic containers to increase flow rates can result in air embolism if the residual air in the container is not fully evacuated prior to administration.
Use of a vented intravenous administration set with the vent in the open position could result in air embolism. Vented intravenous administration sets with the vent in the open position should not be used with flexible plastic containers.
To open. Tear overwrap down side at slit and remove solution container. Some opacity of the plastic due to moisture absorption during the sterilization process may be observed. This is normal and does not affect the solution quality or safety. The opacity will diminish gradually. Check for minute leaks by squeezing inner bag firmly. If leaks are found, discard solution as sterility may be impaired. Do not add supplementary medication.
Because dosages of this drug are titrated to response, no additives should be made to Heparin Sodium and 0.9% Sodium Chloride Injection.
Preparation for administration. Baxter Heparin Sodium in 0.9% Sodium Chloride IV Infusion is a sterile preparation. Thus, aseptic technique must be applied throughout the administration.
1. Suspend container from eyelet support.
2. Remove plastic protector from outlet port at bottom of container.
3. Attach administration set. Refer to complete directions accompanying set.
For single use only. Discard any unused portion.
Do not reconnect partially used bags.
Use in extracorporeal therapy. The concentration of 2 units/mL will suffice to prevent clotting on initiation of extracorporeal therapy by priming the devices with this solution.
Note. A proper and effective heparinisation schedule must be initiated in the patient before and maintained throughout the procedures to prevent subsequent clotting and blood path obstruction. The particular manufacturer's directions for use of the dialyser or other extracorporeal apparatus must be referred to. The direction sheets for the use of therapeutic dosage forms of heparin must equally be referred to and adjusted to a given patients' condition and response to achieve sustained, effective anticoagulation within clinically safe parameters. Both intermittent and continuous infusion of 5,000, 10,000 or 20,000 units are employed for this purpose and are unrelated to the low dose Heparin in this preparation that is directed at preparing the apparatus for initial use.
Maintenance of catheter patency. Although the rate for infusion of the 2 units/mL formulation is dependent upon age, weight, clinical condition of the patient and the procedure being employed, an infusion rate of 3 mL/hour has been found to be satisfactory.
Periodic platelet counts. Haematocrits and tests for occult blood in stool are recommended during the entire course of heparin therapy, regardless of the route of administration.
All injections in Viaflex plastic containers are intended for administration using sterile equipment. It is recommended that the intravenous administration apparatus be replaced at least every 24 hours.
4.3 Contraindications
Heparin sodium should not be used in patients:
who have had a diagnosis of heparin-induced thrombocytopaenia (HIT) (with or without thrombosis) within the previous 6 months, and while they test positive for HIT antibodies;
with severe thrombocytopaenia;
in whom suitable blood coagulation tests - e.g. the whole-blood clotting time, partial thromboplastin time etc. - cannot be performed at appropriate intervals (this contraindication refers to full-dose heparin; there is usually no need to monitor coagulation parameters in patients receiving low-dose heparin);
with an uncontrollable active bleeding state (see Section 4.4 Special Warnings and Precautions for Use), except when this is due to disseminated intravascular coagulation;
with known hypersensitivity to heparin or porcine derivatives (see Section 4.8 Adverse Effects (Undesirable Effects)) or to any ingredient in the formulation.
4.4 Special Warnings and Precautions for Use
Hypersensitivity reactions. Hypersensitivity reactions with chills, fever and urticaria as the most usual manifestations and also asthma, rhinitis, lacrimation, and anaphylactoid reactions have been reported.
Vasospastic reactions. Vasospastic reactions may develop independent of the origin of heparin, after the initiation of the therapy. The affected limb is painful, ischaemic and cyanosed. An artery to this limb may have been recently catheterised. After repeat injections, the reaction may gradually increase to include generalised vasospasm, with cyanosis, tachypnoea, feeling of oppression and headache.
Hyperkalaemia. Heparin can suppress adrenal secretion of aldosterone leading to hyperkalaemia, particularly in patients with diabetes mellitus, chronic renal failure, pre-existing metabolic acidosis, a raised plasma potassium, or taking potassium sparing drugs. The risk of hyperkalaemia appears to increase with duration of therapy but is usually reversible upon discontinuation of heparin.
Plasma potassium should be measured in patients at risk of hyperkalaemia before starting heparin therapy and periodically in all patients on prolonged duration of treatment.
White clot syndrome. It has been reported that patients on heparin may develop new thrombus formation in association with thrombocytopaenia resulting from irreversible aggregation of platelets induced by heparin, the so-called "white clot syndrome". The process may lead to severe thromboembolic complications like skin necrosis, gangrene of the extremities that may lead to amputation, myocardial infarction, pulmonary embolism, stroke and possible death. Therefore, heparin administration should be promptly discontinued if a patient develops new thrombosis in association with thrombocytopaenia.
Spinal/epidural haematomas. When neuraxial anaesthesia (epidural/spinal anaesthesia) or spinal puncture is employed, patients anticoagulated or scheduled to be anticoagulated with unfractionated heparin or low molecular weight heparins/heparinoids for prevention of thromboembolic complications are at risk of developing an epidural or spinal haematoma which can result in long-term or permanent paralysis.
The risk of these events is increased by the use of indwelling epidural catheters for administration of analgesia or by the concomitant use of drugs affecting haemostasis such as non-steroidal anti-inflammatory drugs (NSAIDs), platelet inhibitors, or other anticoagulants. The risk also appears to be increased by traumatic or repeated epidural or spinal puncture.
Patients should be frequently monitored for signs and symptoms of neurological impairment. If neurological compromise is noted, urgent treatment is necessary. The physician should consider the potential benefit versus risk before neuraxial intervention in patient's anticoagulated or to be anticoagulated for thromboprophylaxis.
Heparin resistance. Increased resistance to heparin is frequently encountered in patients with fever, thrombosis, thrombophlebitis, infections with thrombosing tendencies, myocardial infarction, cancer and in post-surgical patients. Monitor coagulation tests closely in such patients. It may be necessary to adjust the dose of heparin based on anti-Factor Xa levels (see Section 4.8 Adverse Effects (Undesirable Effects)).
Haemorrhage. Haemorrhage can occur at virtually any site in patients receiving heparin (e.g. gastrointestinal bleeding with haematemesis and melaena, or haematuria). Fatal haemorrhages have occurred. An unexplained fall in blood pressure, anaemia and fall in haematocrit or any other unexplained symptom should lead to serious consideration of haemorrhagic event. Haematocrit testing and tests for occult blood in stools should be performed periodically during heparin administration.
Heparin sodium should be used with extreme caution in disease states in which there is increased danger of haemorrhage. Some of the conditions in which increased danger of haemorrhage exists are:
Cardiovascular. Subacute bacterial endocarditis; severe hypertension.
Surgical. During and immediately following (a) spinal tap or spinal anaesthesia or (b) major surgery, especially involving the brain, spinal chord or eye.
Haematologic. Conditions associated with increased bleeding tendencies, such as haemophilia, thrombocytopaenia, and some vascular purpuras.
Gastrointestinal. Ulcerative lesions and continuous tube drainage of the stomach or small intestine. It should be appreciated that gastrointestinal or urinary tract bleeding during anticoagulant therapy may indicate the presence of an underlying occult lesion.
Patients with hereditary antithrombin III deficiency receiving concurrent antithrombin III therapy (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Hepatic. Liver disease with impaired haemostasis.
Other. Menstruation, and in patients with indwelling catheters.
Coagulation testing. When heparin sodium is administered in therapeutic amounts, its dosage should be regulated by frequent blood coagulation tests. If the coagulation test is unduly prolonged or if haemorrhage occurs, heparin sodium should be discontinued promptly (see Section 4.9 Overdose).
Thrombocytopaenia. Thrombocytopaenia has been reported to occur in patients receiving heparin with a reported incidence of up to 30%. Platelet counts should be obtained at baseline and periodically during heparin administration. Mild thrombocytopaenia (count greater than 100,000/mm3) may remain stable or reverse even if heparin is continued. However, thrombocytopaenia of any degree should be monitored closely. If the count falls below 100,000/mm3 or if recurrent thrombosis develops (see HIT and HITT below), the heparin product should be discontinued and, if necessary, an alternative anticoagulant administered. If continued heparin therapy is essential, administration of heparin from a different organ source can be reinstituted with caution.
Heparin-induced thrombocytopaenia (HIT) and heparin-induced thrombocytopaenia and thrombosis (HITT). Heparin-induced Thrombocytopenia (HIT) is a serious antibody-mediated reaction resulting from irreversible aggregation of platelets. HIT may progress to the development of venous and arterial thromboses, a condition referred to as Heparin-induced Thrombocytopaenia and Thrombosis (HITT). Thrombotic events may also be the initial presentation of HITT. These serious thromboembolic events include deep vein thrombosis, pulmonary embolism, cerebral vein thrombosis, limb ischaemia, stroke, myocardial infarction, mesenteric thrombosis, renal arterial thrombosis, skin necrosis, gangrene of the extremities that may lead to amputation, and possibly death.
Once HIT or HITT is diagnosed or strongly suspected, all heparin sodium sources (including heparin flushes) should be discontinued and an alternative anticoagulant used. Future use of heparin sodium, especially within 3 to 6 months following the diagnosis of HIT or HITTS, and while patients test positive for HIT antibodies, should be avoided.
Immune-mediated HIT is diagnosed based on clinical findings supplemented by laboratory test confirming the presence of antibodies to heparin sodium, or platelet activation induced by heparin sodium. Platelet counts should be obtained at baseline and periodically during heparin administration. A drop in platelet count greater than 50% from baseline is considered indicative of HIT. Platelet counts begin to fall 5 to 10 days after exposure to heparin sodium in heparin-naïve individuals, and reach a threshold by days 7 to 14. In contrast, 'rapid onset' HIT can occur very quickly (within 24 hours following heparin sodium initiation), especially in patients with a recent exposure to heparin sodium (i.e. previous 3 months). Thrombosis development shortly after documenting thrombocytopaenia is a characteristic finding in almost half of all patients with HIT.
Thrombocytopaenia of any degree should be monitored closely. If the platelet counts fall below 100,000/mm3 or if recurrent thrombosis develops, the heparin product should be promptly discontinued and alternative anticoagulants considered if patients require continued anticoagulation.
Delayed onset of HIT and HITT. HIT and HITT can occur up to several weeks after the discontinuation of heparin therapy. Patients presenting with thrombocytopaenia or thrombosis after discontinuation of heparin should be evaluated for HIT and HITT.
Other. Solutions containing sodium ions should be used with great care in patients with congestive heart failure, severe renal insufficiency, and in clinical states in which there exists oedema with sodium retention. These solutions should also be used with caution in patients receiving corticosteroids or corticotropin.
The intravenous administration of solutions can cause fluid and/or solute overloading resulting in dilution of serum electrolyte concentrations, over-hydration, congested states or pulmonary oedema. The risk of dilutional states is inversely proportional to the electrolyte concentrations and volume of the infusion. The risk of solute overload causing congested states with peripheral and pulmonary oedema is directly proportional to the electrolyte concentrations and volume of the infusion.
Clinical evaluation and periodic laboratory determinations are necessary to monitor changes in fluid balance and electrolyte concentration and acid base balance during prolonged parenteral therapy or whenever the condition of the patient warrants such an evaluation.
Excessive administration of potassium free solutions may result in significant hypokalaemia.
Use in renal impairment. Heparin sodium should be used with caution in the patients with renal disease. In patients with diminished renal function, administration of heparin may result in sodium retention.
Use in hepatic impairment. Heparin sodium should be used with caution in the patients with hepatic disease.
Use in the elderly. A higher incidence of bleeding has been reported in patients, particularly women, over 60 years of age (see Section 4.4 Special Warnings and Precautions for Use, Haemorrhage). Clinical studies indicate that lower doses of heparin may be indicated in these patients.
Paediatric use. Safety and effectiveness in paediatric patients has not been established.
Effects on laboratory tests. Periodic platelet counts; haematocrits, and tests for occult blood in stool are recommended during the entire course of heparin therapy, regardless of the route of administration (see Section 4.2 Dose and Method of Administration).
4.5 Interactions with Other Medicines and Other Forms of Interactions
Oral anticoagulants. Heparin sodium may prolong the one-stage prothrombin time. Therefore when heparin sodium is given with dicumarol or warfarin sodium, a period of at least 5 hours after the last intravenous dose, or 24 hours after the last subcutaneous dose should elapse before blood is drawn if a valid prothrombin time is to be obtained.
Platelet inhibitors. Drugs such as NSAIDs (e.g. acetylsalicylic acid, ibuprofen, indometacin), dextran, phenylbutazone, dipyridamole, hydroxychloroquine, epoprostenol, clopidogrel, thienopyridines, glycoprotein IIb/IIIa antagonists, and others that interfere with platelet aggregation reactions (the main haemostatic defence of heparinised patients) may induce bleeding and should be used with caution in patients receiving heparin sodium. To reduce the risk of bleeding, a reduction in the dose of antiplatelet agent or heparin is recommended.
Antithrombin III (human). The anticoagulant effect of heparin is enhanced by concurrent treatment with antithrombin III (human) in patients with hereditary antithrombin III deficiency. To reduce the risk of bleeding, a reduction in the dose of heparin is recommended during treatment with antithrombin III (human).
Other interactions. Digitalis, tetracyclines, nicotine or antihistamines may partially counteract the anticoagulant action of heparin sodium.
Tobacco smoke and nicotine may decrease the anticoagulant effects of heparin. Increased doses of heparin may be required in smokers.
Intravenous nitroglycerin administered to patients receiving heparin may result in a decrease of the partial thromboplastin time with subsequent rebound effect upon discontinuation of nitroglycerin. Careful monitoring of partial thromboplastin time and adjustment of heparin dosage are recommended during coadministration of heparin and intravenous nitroglycerin.
The use of ACE inhibitors and angiotensin-II antagonists in conjunction with heparin increase the risk of hyperkalaemia.
When administering Baxter Heparin Sodium in 0.9% Sodium Chloride IV Infusion concomitantly with the drugs listed above monitor coagulation tests frequently and adjust dose as necessary.
Drug/laboratory tests interactions. Hyperaminotransferasaemia. Significant elevations of aminotransferase (SGOT [S-AST] and SGPT [S-ALT]) levels have occurred in a high percentage of patients (and healthy subjects) who have received heparin. Since aminotransferase determinations are important in the differential diagnosis of myocardial infarction, liver disease and pulmonary emboli, elevation of these enzymes in patients that might be caused by drugs (like heparin) should be interpreted with caution.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Animal reproduction studies have not been conducted with heparin sodium. No reproduction studies in animals have been performed concerning mutagenesis or impairment of fertility. It is not known whether heparin sodium can affect reproduction capacity.
Use in pregnancy. (Category C)
Heparin does not cross the placental barrier. There are no adequate data from the use of Baxter Heparin Sodium in 0.9% Sodium Chloride IV Infusion in pregnant women. It is not known whether heparin sodium can cause foetal harm when administered to a pregnant woman. Physicians should carefully consider the potential risks and benefits for each patient before prescribing Baxter Heparin Sodium in 0.9% Sodium Chloride IV Infusion.
Heparin sodium should be given to pregnant women only if clearly needed.
Use in lactation. Heparin is not excreted in human milk. There are no adequate data from the use of Baxter Heparin Sodium in 0.9% Sodium Chloride IV Infusion in lactating women. Physicians should carefully consider the potential risks and benefits for each patient before prescribing Baxter Heparin Sodium in 0.9% Sodium Chloride IV Infusion.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
The following adverse reactions have been reported in the post-marketing experience, listed by MedDRA System Organ Class (SOC), then, where feasible, by Preferred Term in order of severity:
Vascular disorders. Haemorrhage, gastrointestinal haemorrhage, adrenal haemorrage, retroperitoneal haemorrhage, epistaxis, contusion, vasospastic reactions.
Haemorrhage is the chief complication that may result from heparin therapy (see Section 4.4 Special Warnings and Precautions for Use). An overly prolonged clotting time or minor bleeding during therapy can usually be controlled by withdrawing the drug (see Section 4.9 Overdose). It should be appreciated that gastrointestinal or urinary tract bleeding during anticoagulant therapy may indicate the presence of an underlying occult lesion. Bleeding can occur at any site but certain specific haemorrhage complications may be difficult to detect:
a. Adrenal haemorrhage, with resultant acute adrenal insufficiency, has occurred during anticoagulant therapy. Therefore, such treatment should be discontinued in patients who develop signs and symptoms of acute adrenal haemorrhage and insufficiency. Initiation of corrective therapy should not depend on laboratory confirmation of the diagnosis, since any delay in an acute situation may result in a patient's death.
b. Ovarian (corpus luteum) haemorrhage developed in a number of women of reproductive age receiving short or long-term anticoagulant therapy. This complication, if unrecognised, may be fatal.
c. Retroperitoneal haemorrhage.
Blood and lymphatic system disorders. Thrombocytopenia, heparin-induced thrombocytopenia (with or without thrombosis).
Thrombocytopenia has been reported to occur in patients receiving heparin with a reported incidence of up to 30%. While often mild and of no obvious clinical significance, such thrombocytopenia can be accompanied by severe thromboembolic complications such as skin necrosis, gangrene of the extremities that may lead to amputation, myocardial infarction, pulmonary embolism, stroke and possible death (see Section 4.4 Special Warnings and Precautions for Use, Thrombocytopaenia, Heparin-induced thrombocytopaenia (HIT) and heparin-induced thrombocytopaenia and thrombosis (HITT)).
Certain episodes of painful, ischaemic, and cyanosed limbs have in the past been attributed to allergic vasospastic reactions. Whether these are in fact identical to the thrombocytopaenia-associated complications remains to be determined.
Renal and urinary disorders. Haematuria.
Endocrine disorders. Hypoaldosteronism, suppression of aldosterone synthesis.
Skin and subcutaneous tissue disorders. Skin/cutaneous necrosis, delayed transient alopecia.
Musculoskeletal, connective tissue and bone disorders. Osteoporosis (following long term administration of high doses of heparin).
Immune system disorders. General hypersensitivity reactions have been reported, with chills, fever and urticaria as the most usual manifestations, and asthma, rhinitis, lacrimation, headache, nausea and vomiting, and anaphylactoid reactions, including shock, occurring more rarely. Itching and burning, especially on the plantar site of the feet, may occur.
Metabolism and nutrition disorders. Hyperkalaemia, hyperlipidaemia.
Rebound hyperlipidaemia on discontinuation of heparin sodium have also been reported.
Reproductive system and breast disorders. Priapism, ovarian cyst.
General disorders and administration site conditions. Injection site reaction, local irritation, erythema, mild pain, haematoma or ulceration may follow deep subcutaneous (intrafat) injection of heparin sodium. These complications are much more common after intramuscular use, and such use is not recommended.
Investigations. Significant elevations of aspartate aminotransferase (SGOT [S-AST]) and alanine aminotransferase (SGPT [S-ALT]) levels have occurred in a high percentage of patients (and healthy subjects) who have received heparin.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Symptoms. An overdose requires immediate medical attention and treatment. Bleeding is the primary sign of heparin overdose. Easy bruising, petechial formations, nosebleeds, blood in urine or tarry stools may be the first signs or symptoms of a heparin overdose.
Treatment. Neutralization of heparin effect.
When clinical circumstances (bleeding) require reversal of heparinisation, protamine sulfate (1% solution) by slow infusion will neutralise heparin sodium. No more than 50 mg should be administered, very slowly in any 10-minute period. Each mg of protamine sulfate neutralises approximately 100 USP heparin units. Ideally, the dose required to neutralise the action of heparin should be guided by blood coagulation tests or calculated from a protamine neutralisation test. The amount of protamine required decreases over time as heparin is metabolised. Although the metabolism of heparin is complex, it may, for the purpose of choosing a protamine dose, be assumed to have a half-life of about 30 minutes after intravenous injection.
Administration of protamine sulfate can cause severe hypotensive and anaphylactoid reactions. Because fatal reactions often resembling anaphylaxis have been reported, the drug should be given only when resuscitation techniques and treatment of anaphylactoid shock are readily available. For additional information the labeling of protamine sulfate products should be consulted.
Blood or plasma transfusions may be necessary; these dilute but do not neutralise heparin.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Heparin inhibits reactions that lead to the clotting of blood and the formation of fibrin clots both in vitro and in vivo. Heparin acts at multiple sites in the normal coagulation system. Small amounts of heparin in combination with Antithrombin III (heparin cofactor) can inhibit thrombosis by inactivating activated Factor X and inhibiting the conversion of prothrombin to thrombin. Once active thrombosis has developed, larger amounts of heparin can inhibit further coagulation by inactivating thrombin and preventing conversion of fibrinogen to fibrin. Heparin also prevents the formation of a stable fibrin clot by inhibiting the activation of the fibrin stabilising factor.
Bleeding time is usually unaffected by heparin. Clotting time is prolonged by full therapeutic doses of heparin; in most cases it is not measurably affected by low doses of heparin.
Heparin does not have fibrinolytic activity, therefore it will not lyse existing clots.
Clinical trials have not demonstrated the superiority of heparin in the maintenance of catheter patency over fluid not containing anticoagulant medication (e.g. normal saline).
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
No data available.
5.3 Preclinical Safety Data
Genotoxicity. No reproduction studies in animals have been performed concerning mutagenesis or impairment of fertility.
Carcinogenicity. No long-term studies in animals have been performed to evaluate carcinogenic potential of heparin.
6 Pharmaceutical Particulars
6.1 List of Excipients
Sodium chloride, citric acid monohydrate, dibasic sodium phosphate dodecahydrate and water for injections.
See Section 2 Qualitative and Quantitative Composition.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
It is recommended that the product is stored below 30°C.
6.5 Nature and Contents of Container
Baxter Heparin Sodium in 0.9% Sodium Chloride IV Infusion is supplied in Viaflex Plus plastic containers in the following sizes and concentrations (Table 1).
6.6 Special Precautions for Disposal
Any unused product or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure.
Molecular Weight: 1039.83.
Appearance: white or almost off-white powder, odorless, hygroscopic.
Solubility: soluble in water; insoluble in ethanol, acetone and other organic solvents.
CAS number. CAS No.: 9041-08-1.
7 Medicine Schedule (Poisons Standard)
Schedule 4 (Prescription Medicine).
Date of First Approval
30 September 1991
Date of Revision
15 December 2021
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.