Hexaxim
Brand Information
| Brand name | Hexaxim |
| Active ingredient | Diphtheria toxoid + Haemophilus B conjugate vaccine + Tetanus toxoid + Hepatitis B child vaccine + Pertussis vaccine + Poliomyelitis vaccine (inactivated) |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Hexaxim.
Summary CMI
HEXAXIM®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about your child receiving this vaccine, speak to your doctor or pharmacist.
1. Why is my child receiving Hexaxim?
Hexaxim contains the active ingredients diphtheria, tetanus, pertussis (acellular, component), hepatitis B (rDNA), poliomyelitis (inactivated) and haemophilus influenzae type b conjugate vaccine (adsorbed). Hexaxim is a vaccine used to help protect your child against diphtheria, tetanus, pertussis, hepatitis B, poliomyelitis and serious diseases caused by Haemophilus influenzae type b.
For more information, see Section 1. Why is my child receiving Hexaxim? in the full CMI.
2. What should I know before my child receives Hexaxim?
Your child should not receive Hexaxim if they have ever had an allergic reaction to Hexaxim or any of the ingredients listed at the end of the CMI.
Talk to your doctor your child has any other medical conditions or takes any other medicines.
For more information, see Section 2. What should I know before my child receives Hexaxim? in the full CMI.
3. What if my child is taking other medicines?
Some medicines may interfere with Hexaxim and affect how it works.
A list of these medicines is in Section 3. What if my child is taking other medicines? in the full CMI.
4. How does my child receive Hexaxim?
- Hexaxim is injected into the muscle in the upper part of your child's leg or upper arm, by a doctor or nurse.
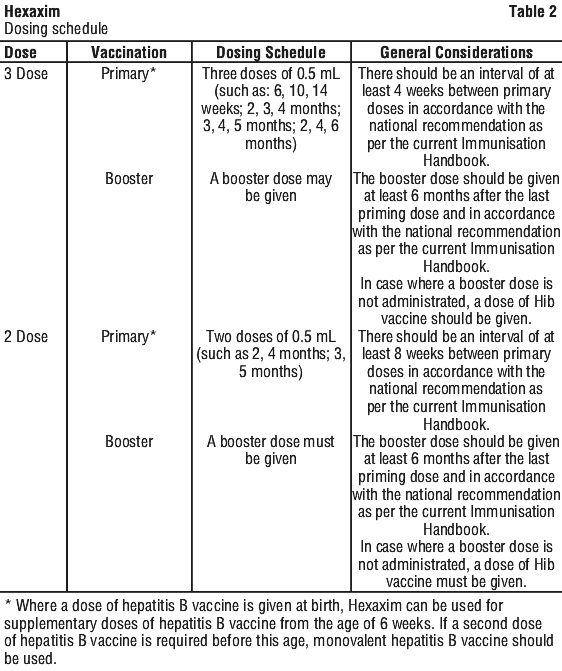
- Your child will receive two injections given at an interval of at least eight weeks apart, or three injections given at an interval of at least four weeks apart. This vaccine should be used according to the local vaccination program.
More instructions can be found in Section 4. How does my child receive Hexaxim? in the full CMI.
5. What should I know after my child receives Hexaxim?
| Things you should do |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know after my child receives Hexaxim? in the full CMI.
6. Are there any side effects?
The most common side effects of Hexaxim are pain, redness or swelling at the injection site, loss of appetite, crying, sleepiness, vomiting, irritability, fever (temperature 38°C or higher), abnormal crying (prolonged crying), diarrhoea and injection site hardness.
Serious side effects can include episodes when your child goes into a shock-like state or is pale, floppy and unresponsive for a period of time (hypotonic reactions or hypotonic hyporesponsive episodes HHE), serious allergic reaction (anaphylactic reaction) and fits (convulsions) with or without fever.
For more information, including what to do if your child has any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
1. Why is my child receiving Hexaxim?
Hexaxim contains the active ingredients diphtheria, tetanus, pertussis (acellular, component), hepatitis B (rDNA), poliomyelitis (inactivated) and haemophilus influenzae type b conjugate vaccine (adsorbed). Hexaxim is a vaccine used to protect against infectious diseases.
Hexaxim is used to help protect infants against diphtheria, tetanus, pertussis, hepatitis B, poliomyelitis and serious diseases caused by haemophilus influenzae type b. Hexaxim can be given to children from six weeks of age.
The vaccine works by causing the body to produce its own protection (antibodies) against the bacteria and viruses that cause these different infections:
- Diphtheria is an infectious disease that usually first affects the throat. In the throat, the infection causes pain and swelling which can lead to suffocation. The bacteria that cause the disease also make a toxin (poison) that can damage the heart, kidneys and nerves.
- Tetanus (often called lock jaw) is usually caused by the tetanus bacteria entering a deep wound. The bacteria make a toxin (poison) that causes spasms of the muscles, leading to inability to breathe and the possibility of suffocation.
- Pertussis (often called whooping cough) is a highly infectious illness that affects the airways. It causes severe coughing that may lead to problems with breathing. The coughing often has a “whooping” sound. The cough may last for one to two months or longer. Whooping cough can also cause ear infections, chest infections (bronchitis) which may last a long time, lung infections (pneumonia), fits, brain damage and even death.
- Hepatitis B is caused by the hepatitis B virus. It causes the liver to become swollen (inflamed). In some people, the virus can stay in the body for a long time, and can eventually lead to serious liver problems, including liver cancer
- Poliomyelitis (often just called polio) is caused by viruses that affect the nerves. It can lead to paralysis or muscle weakness most commonly of the legs. Paralysis of the muscles that control breathing and swallowing can be fatal.
- Haemophilus influenzae type b infections (often just called Hib) are serious bacterial infections and can cause meningitis (inflammation of the outer covering of the brain), which can lead to brain damage, deafness, epilepsy, or partial blindness. Infection can also cause inflammation and swelling of the throat, leading to difficulties in swallowing and breathing, and infection can affect other parts of the body such as the blood, lungs, skin, bones, and joints.
Important information about the protection provided
Hexaxim will only help to prevent these diseases if they are caused by the bacteria or viruses targeted by the vaccine. Your child could get diseases with similar symptoms if they are caused by other bacteria or viruses.
The vaccine does not contain any live bacteria or viruses and it cannot cause any of the infectious diseases against which it protects.
This vaccine does not protect against infections caused by other types of Haemophilus influenzae nor against meningitis due to other micro-organisms.
Hexaxim will not protect against hepatitis infection caused by other agents such as hepatitis A, hepatitis C and hepatitis E.
Because symptoms of hepatitis B take a long time to develop, it is possible for unrecognised hepatitis B infection to be present at the time of vaccination. The vaccine may not prevent hepatitis B infection in such cases.
Remember that no vaccine can provide complete, lifelong protection in all people who are vaccinated
2. What should I know before my child receives Hexaxim?
Warnings
Your child should not receive Hexaxim if:
- they have had a previous allergic reaction to Hexaxim, or any of the ingredients listed at the end of this leaflet.
- Always check the ingredients to make sure you can use this medicine.
- has had respiratory disorder or swelling of the face (anaphylactic reaction) after administration of Hexaxim.
- has had an allergic reaction to any other diphtheria, tetanus, pertussis, poliomyelitis, hepatitis B or Hib containing vaccines.
- suffered from a severe reaction affecting the brain (encephalopathy) within 7 days of a prior dose of a pertussis vaccine (acellular or whole cell pertussis).
- has an uncontrolled condition or severe illness affecting the brain and nervous system (uncontrolled neurologic disorder) or uncontrolled epilepsy.
Check with your doctor if your child:
- has any other medical conditions.
- has a moderate or high temperature or an acute illness (e.g. fever, sore throat, cough, cold or flu). Vaccination with Hexaxim may need to be delayed until your child is better.
- has had any of the following events after receiving a pertussis vaccine, as the decision to give further doses of pertussis containing vaccine will need to be carefully considered:
- fever of 40°C or above within 48 hours not due to another identifiable cause.
- collapse or shock-like state with hypotonic-hyporesponsive episode (drop in energy) within 48 hours of vaccination.
- persistent, inconsolable crying lasting 3 hours or more, occurring within 48 hours of vaccination.
- fits (convulsions) with or without fever, occurring within 3 days of vaccination. - previously had Guillain-Barre syndrome (temporary inflammation of nerves causing pain, paralysis and sensitivity disorders) or brachial neuritis (severe pain and decreased mobility of arm and shoulder) after being given a vaccine containing tetanus toxoid (an inactivated form of tetanus toxin). In this case, the decision to give any further vaccine containing tetanus toxoid should be evaluated by your doctor.
- is having a treatment that suppresses her/his immune system (the body's natural defences) or has any disease that causes the weakness of the immune system. In these cases, the immune response to the vaccine may be decreased. It is normally recommended to wait until the end of the treatment or disease before vaccinating. However, children with long standing problems with their immune system such as HIV infection (AIDS) may still be given Hexaxim but the protection may not be as good as in children whose immune system is healthy.
- is born prematurely. Lower responses to the vaccine may be observed in relation with immaturity of the immune system. However, according to national recommendations, vaccination should not be delayed. In addition, longer gaps than normal between breaths may occur for 2 -3 days after vaccination.
- suffers from an acute or chronic illness including chronic renal insufficiency or failure (inability of the kidneys to work properly).
- suffers from any undiagnosed illness of the brain or epilepsy which is not controlled. Your doctor will assess the potential benefit offered by vaccination.
- has any problems with the blood that cause easy bruising or bleeding for a long time after minor cuts. Your doctor will advise you whether your child should have Hexaxim.
- has fainted when having a previous injection. Fainting can occur before or following needle injection.
- take any medicines for any other condition
During treatment, your child may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
3. What if my child is taking other medicines?
Tell your doctor or pharmacist if your child is taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Hexaxim and affect how it works.
Having other vaccines
Your doctor will advise you if Hexaxim is to be given with another vaccine.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements your child is taking and if these affect Hexaxim.
4. How does my child receive Hexaxim?
How is Hexaxim given
- Hexaxim is administered to your child by your doctor or nurse
- Hexaxim is injected into the muscle in the upper part of your child's leg or upper arm
When is Hexaxim given
First course of vaccination (primary vaccination)
- Your child will receive two injections given at an interval of at least eight weeks apart, or three injections given at an interval of at least four weeks apart.
- This vaccine should be used according to the local vaccination programme.
Additional injections (booster)
- After the first course of injections, your child may require a booster dose, in accordance with local recommendations.
- Your doctor will advise you if your child requires a booster dose.
If you forget a dose of Hexaxim
If your child misses a scheduled injection, it is important that you discuss with your doctor or nurse who will decide when to give the missed dose.
It is important to follow the instructions from the doctor or nurse so that your child completes the course of injections. If not, your child may not be fully protected against the diseases.
If you have any further questions on the use of this vaccine, ask your doctor, pharmacist or nurse.
If you use too much Hexaxim
Overdose is unlikely as your doctor is giving you the injection.
If you think that your child has been given too much Hexaxim, your child may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre (by calling 13 11 26), or
- contact your doctor or nurse, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know after my child receives Hexaxim?
Things you should do
- Keep an updated record of your child's vaccination history.
- Attend any appointments made by your doctor or nurse.
- Report any side effects to your doctor.
Looking after your medicine
Follow the instructions in the carton on how to take care of your medicine properly.
Hexaxim is usually stored in the doctor's surgery or clinic, or at the pharmacy. However, if you need to store Hexaxim:
- Keep out of reach and sight of children.
- Keep Hexaxim in the original pack until it is time for it to be given.
- Keep it in the refrigerator, store at 2°C to 8°C. Do not freeze Hexaxim.
- Do not use Hexaxim if the packaging is torn or shows signs of tampering.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Medicines should not be disposed of via wastewater or household waste. Ask your pharmacist how to dispose of medicines no longer required. These measures will help to protect the environment.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Gastrointestinal-related:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Allergy-related:
Signs and symptoms of serious allergic reactions usually develop quickly after the injection is given and while the child is still in the clinic or doctor's surgery. Nervous system-related:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Other side effects not listed above have been reported occasionally with other diphtheria, tetanus, pertussis, poliomyelitis, hepatitis B or Hib containing vaccines and not directly with Hexaxim:
- temporary inflammation of nerves causing pain, paralysis and sensitivity disorders (Guillain-Barré syndrome) and severe pain and decreased mobility of arm and shoulder (brachial neuritis) have been reported after administration of a tetanus containing vaccine.
- inflammation of several nerves causing sensory disorders or weakness of limbs (polyradiculoneuritis), facial paralysis, visual disturbances, sudden dimming or loss of vision (optic neuritis), inflammatory disease of brain and spinal cord (central nervous system demyelination, multiple sclerosis) have been reported after administration of a hepatitis B antigen containing vaccine.
- swelling or inflammation of the brain (encephalopathy/encephalitis).
- in babies born very prematurely (at or before 28 weeks of gestation) longer gaps than normal between breaths may occur for 2 -3 days after vaccination.
- swelling of one or both feet and lower limbs which may occur along with bluish discolouration of the skin, redness, small areas of bleeding under the skin and severe crying following vaccination with Haemophilus influenzae type b containing vaccines. If this reaction occurs, it is mainly after first injections and within the first few hours following vaccination. All symptoms should disappear completely within 24 hours without the need for treatment.
If your child gets any side effects, talk to your doctor, pharmacist or nurse. This includes any possible side effects not listed in this leaflet.
Reporting side effects
After you have received medical advice for any side effects your child experiences, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems in Australia or to the Medsafe online at pophealth.my.site.com/carmreportnz/s/ in New Zealand. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Hexaxim contains
| Active ingredients (main ingredients) | Each 0.5 ml dose of Hexaxim contains: At least 20 IU (30 Lf) of diphtheria toxoid At least 40 IU (10 Lf) of tetanus toxoid 25 micrograms of pertussis toxoid and 25 micrograms of pertussis filamentous haemagglutinin 10 micrograms of hepatitis B surface antigen 29 D antigen Units of poliovirus Type 1, 7 D antigen Units of poliovirus Type 2, 26 D antigen Units of poliovirus Type 3* 12 micrograms of Haemophilus type B polysaccharide conjugated to 22-36 micrograms of tetanus protein *These quantities are the same as those previously expressed when measured by a suitable method. |
| Other ingredients (inactive ingredients) | dibasic sodium phosphate mono basic potassium phosphate trometamol sucrose essential amino acids (cystine, tyrosine, arginine hydrochloride, histidine, isoleucine, leucine, lysine hydrochloride, methionine, phenylalanine, threonine, tryptophan and valine) water for injections |
Do not take this medicine if you are allergic to any of these ingredients.
The vaccine may contain traces of glutaral, formaldehyde, neomycin, streptomycin and polymyxin B. The manufacture of this product includes exposure to bovine materials. No evidence exists that any case of vCJD (considered to be the human form of bovine spongiform encephalopathy) has resulted from the administration of any vaccine product.
What Hexaxim looks like
Hexaxim is provided as a fully liquid suspension for injection in pre-filled syringe (0.5 mL).
Hexaxim is available in a pack containing 1 pre-filled syringe with 1 or 2 needles.
Hexaxim is available in a pack containing 10 pre-filled syringes with 10 or 20 needles.
Not all pack sizes may be marketed.
After shaking, the normal appearance of the vaccine is a whitish cloudy suspension (Aust R 215536.)
Who distributes Hexaxim
Distributed in Australia by:
sanofi-aventis australia pty ltd
12-24 Talavera Road
Macquarie Park NSW 2113
Freecall: 1800 818 806
Email: medinfo.australia@sanofi.com
Distributed in New Zealand by:
Pharmacy Retailing (NZ) Ltd t/a Healthcare Logistics
PO Box 62027
Sylvia Park Auckland 1644
Freecall: 0800 283 684
Email: medinfo.australia@sanofi.com
This leaflet was prepared in October 2025.
hexa-ccdsv12-14-cmiv5-07oct25
Brand Information
| Brand name | Hexaxim |
| Active ingredient | Diphtheria toxoid + Haemophilus B conjugate vaccine + Tetanus toxoid + Hepatitis B child vaccine + Pertussis vaccine + Poliomyelitis vaccine (inactivated) |
| Schedule | S4 |
MIMS Revision Date: 01 December 2025
1 Name of Medicine
Hexaxim.
DTPa-hepB-IPV-Hib - Diphtheria, tetanus, pertussis (acellular, component), hepatitis B (rDNA), poliomyelitis (inactivated) and Haemophilus influenzae type b conjugate vaccine (adsorbed).
2 Qualitative and Quantitative Composition
Hexaxim is a preservative free liquid formulation for intramuscular administration which combines: diphtheria and tetanus toxoids, acellular pertussis (2-component), recombinant hepatitis B surface antigen, inactivated poliomyelitis virus and Haemophilus influenzae type b polysaccharide conjugated to tetanus protein.
Each 0.5 mL, adsorbed to aluminium hydroxide hydrate (0.6 mg, expressed as Al3+), contains (see Table 1):

Contains phenylalanine. For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Hexaxim is a whitish, cloudy suspension for injection.
4 Clinical Particulars
4.1 Therapeutic Indications
Hexaxim is indicated for vaccination of infants from six weeks of age against diphtheria, tetanus, pertussis, hepatitis B, poliomyelitis and invasive infections caused by Haemophilus influenzae type b.
Use of this vaccine should be in accordance with the national recommendation as per the current immunisation handbook.
4.2 Dose and Method of Administration
Hexaxim is for intramuscular injection according to the dosing schedule in Table 2.
Administration. Before use, the vaccine should be shaken in order to obtain a homogeneous whitish cloudy suspension.
Hexaxim should be administered intramuscularly. The recommended injection sites are generally the antero-lateral aspect of the upper thigh in infants and toddlers and the deltoid muscle in older children.
Do not administer via intravascular route: ensure that the needle does not penetrate a blood vessel.
Do not administer by intradermal or subcutaneous injection.
Separate syringes, separate injection sites and preferably separate limbs must be used in case of concomitant administration with other vaccines.
Hexaxim is for single use only and must not be used in more than one individual. Discard any remaining unused contents.
4.3 Contraindications
Hexaxim should not be administered to anyone with a history of severe allergic reaction to any component of the vaccine or to any pertussis vaccine, after previous administration of the vaccine or a vaccine containing the same components or constituents.
Vaccination with Hexaxim is contraindicated if the individual has experienced an encephalopathy of unknown aetiology within 7 days of administration of a previous dose of any vaccine containing pertussis antigens (whole cell or acellular pertussis vaccines). In these circumstances pertussis vaccination should be discontinued and the vaccination course should be continued with diphtheria, tetanus, hepatitis B, poliomyelitis and Hib vaccines.
Progressive neurological disorder, uncontrolled epilepsy, progressive encephalopathy. Pertussis vaccine should not be administered to individuals with these common conditions until the treatment regimen has been established, the condition has stabilised and the benefit clearly outweighs the risk.
Generally vaccination must be postponed in cases of moderate or severe febrile and/or acute disease and low-grade fever does not constitute a contraindication.
4.4 Special Warnings and Precautions for Use
Do not administer intravenously, intradermally or subcutaneously.
Prior to vaccination. Anaphylaxis. As with all injectable vaccines, appropriate medical treatment and supervision should always be readily available in case of a rare anaphylactic event following administration of the vaccine.
Hypersensitivity. As each dose may contain undetectable traces of glutaraldehyde, formaldehyde, neomycin, streptomycin and polymyxin B, caution should be exercised when the vaccine is administered to individuals with hypersensitivity to these substances.
Bleeding disorder. As with all injectable vaccines, the vaccine must be administered with caution to individuals with thrombocytopenia or a bleeding disorder since bleeding may occur following an intramuscular administration.
Previous pertussis vaccination. If any of the following events are known to have occurred in temporal relation to receipt of pertussis-containing vaccine, the decision to give further doses of pertussis-containing vaccine should be carefully considered:
temperature of ≥ 40°C within 48 hours not due to another identifiable cause;
collapse or shock-like state (hypotonic-hyporesponsive episode) within 48 hours of vaccination;
persistent, inconsolable crying lasting ≥ 3 hours, occurring within 48 hours of vaccination;
convulsions with or without fever, occurring within 3 days of vaccination.
Family and individual history. A history of febrile convulsions, a family history of convulsions or sudden infant death syndrome (SIDS) do not constitute a contraindication for the use of Hexaxim. Individuals with a history of febrile convulsions should be closely followed up as such adverse events may occur within 2 to 3 days post vaccination.
Protection. Hexaxim will not prevent disease caused by pathogens other than Corynebacterium diphtheriae, Clostridium tetani, Bordetella pertussis, hepatitis B virus, poliovirus or Haemophilus influenzae type b. However, it can be expected that hepatitis D will be prevented by immunisation as hepatitis D (caused by the delta agent) does not occur in the absence of hepatitis B infection.
Hexaxim will not protect against hepatitis infection caused by other agents such as hepatitis A, hepatitis C and hepatitis E or by other liver pathogens.
Because of the long incubation period of hepatitis B, it is possible for unrecognised hepatitis B infection to be present at the time of vaccination. The vaccine may not prevent hepatitis B infection in such cases.
Hexaxim does not protect against infectious diseases caused by other types of Haemophilus influenzae or against meningitis of other origins.
As with any vaccine, vaccination with Hexaxim may not protect 100% of susceptible individuals.
Special patient groups. Premature and low birth weight infants. No data are available for premature infants and infants of low birth weight < 2.5 kg. Lower immune response may be observed in this population in relation with immaturity of the immune system. However, according to several national recommendations, vaccination should not be delayed.
The potential risk of apnoea and the need for respiratory monitoring for 48-72 hours should be considered when administering the primary immunisation series to very premature infants (born ≤ 28 weeks of gestation) and particularly for those with a previous history of respiratory immaturity. As the benefit of vaccination is high in this group of infants, vaccination should not be withheld or delayed.
Immunocompromised individuals. The immunogenicity of the vaccine may be reduced by immunosuppressive treatment or immunodeficiency. It is recommended to postpone vaccination until the end of such treatment or disease. Nevertheless, vaccination of individuals with chronic immunodeficiency such as HIV infection is recommended even if the antibody response may be limited.
Neurological disorder. If Guillain-Barré syndrome or brachial neuritis has occurred following receipt of prior vaccine containing tetanus toxoid, the decision to give any vaccine containing tetanus toxoid should be based on careful consideration of the potential benefits and possible risks, such as whether or not the primary immunisation schedule has been completed. Vaccination is usually justified for infants whose primary immunisation schedules are incomplete (i.e. fewer than three doses have been received).
Some case reports of multiple sclerosis have been reported after administration of hepatitis B vaccine. To date a causal relationship has not been demonstrated with hepatitis B vaccine.
Chronic renal failure. In individuals with chronic renal failure, an impaired hepatitis B response is observed and administration of additional doses of hepatitis B vaccine should be considered according to the antibody level against hepatitis B virus surface antigen (anti-HBsAg).
Genetic polymorphism. Immune responses to the vaccine have not been studied in the context of genetic polymorphism.
The immunogenicity of Hexaxim has not been studied in the Australian indigenous populations.
Use in the elderly. Not applicable.
Paediatric use. The safety and efficacy of Hexaxim in children over 24 months of age have not been established.
Effects on laboratory tests. Interference of Hexaxim with laboratory and/or diagnostic tests has not been studied.
However, antigenuria (PRP antigen) has been detected in some instances following receipt of Haemophilus influenzae type b conjugate vaccine. Therefore, urine antigen detection may not have definite diagnostic value in suspected Haemophilus influenzae type b disease within two weeks of immunisation.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Hexaxim must not be mixed with other vaccines or other parenterally administered drugs.
Separate injection sites must be used in case of concomitant administration.
Data on concomitant administration of Hexaxim with 7-valent or 13-valent pneumococcal polysaccharide conjugate vaccines have shown no clinically relevant interference in the antibody response to each of the individual antigens.
Data on concomitant administration of Hexaxim with measles-mumps-rubella vaccine and with varicella vaccine have shown no clinically relevant interference in the antibody response to each of the antigens when given as a booster vaccination.
Data on concomitant administration of rotavirus vaccines have shown no clinically relevant interference in the antibody response to each of the antigens.
Data on concomitant administration of Hexaxim with a meningococcal serogroup C-tetanus toxoid conjugate vaccine or a meningococcal serogroups A, C, W-135, Y-tetanus toxoid conjugate vaccine have shown no clinically relevant interference in the antibody response to each of the antigens.
Except in the case of immunosuppressive therapy (see Section 4.4 Special Warnings and Precautions for Use), no significant clinical interaction with other treatments or biological products has been reported.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Animal studies have not been conducted to determine the effects of Hexaxim on fertility.
Use in pregnancy. (Category B2)
Hexaxim is not indicated for use during pregnancy and has not been evaluated for potential harmful effects during pregnancy in animals or humans.
Use in lactation. Hexaxim is not indicated for use in breastfeeding women and it is not known whether Hexaxim components are excreted in human milk.
4.7 Effects on Ability to Drive and Use Machines
Not applicable.
4.8 Adverse Effects (Undesirable Effects)
The adverse events are ranked under headings of frequency per dose, using the following convention: very common ≥ 1/10 (≥ 10%); common ≥ 1/100 to < 1/10 (≥ 1% and < 10%); uncommon ≥ 1/1,000 to < 1/100 (≥ 0.1% and < 1%); rare ≥ 1/10,000 to < 1/1000 (≥ 0.01% and < 0.1%); very rare < 1/10,000 (< 0.01%); not known: cannot be estimated from available data.
Clinical trials experience. In clinical studies in individuals who received Hexaxim, the most frequently reported reactions (expressed per dose) include injection site pain, irritability, crying and injection site erythema. Slightly higher solicited reactogenicity was observed after the first dose compared to subsequent doses.
Immune system disorders. Uncommon: hypersensitivity reaction.
Metabolism and nutrition disorders. Very common: anorexia.
Nervous system disorders. Very common: crying, somnolence.
Common: abnormal crying (prolonged crying).
Very rare: hypotonic reactions or hypotonic-hyporesponsive episodes (HHE).
Gastrointestinal disorders. Very common: vomiting.
Common: diarrhoea.
Skin and subcutaneous tissue disorders. Rare: rash.
General disorders and administration site conditions. Very common: injection site pain, injection site erythema, injection site swelling, irritability, pyrexia (body temperature ≥ 38.0°C).
Common: injection site induration.
Uncommon: injection site nodule, pyrexia (body temperature ≥ 39.6°C).
Rare: extensive limb swelling.
Large injection site reactions (> 50 mm), including extensive limb swelling from the injection site beyond one or both joints, have been reported in children. These reactions start within 24-72 hours after vaccination, may be associated with erythema, warmth, tenderness or pain at the injection site and resolve spontaneously within 3-5 days. The risk appears to be dependent on the number of prior doses of acellular pertussis containing vaccine, with a greater risk following the 4th and 5th doses.
Adverse reactions from post-marketing surveillance. Immune system disorders. Very rare: anaphylactic reactions.
Nervous system disorders. Very rare: convulsions with or without fever.
Potential adverse events. (i.e. adverse events which have been reported with other vaccines containing one or more of the components or constituents of Hexaxim and not directly with Hexaxim).
Brachial neuritis and Guillain-Barré syndrome have been reported after administration of a tetanus toxoid containing vaccine.
Oedematous reaction affecting one or both lower limbs may occur following vaccination with Haemophilus influenzae type b containing vaccines. If this reaction occurs, it is mainly after primary injections and within the first few hours following vaccination. Associated symptoms may include cyanosis, redness, transient purpura and severe crying. All events resolve spontaneously without sequel within 24 hours.
Peripheral neuropathy (polyradiculoneuritis, facial paralysis), optic neuritis, central nervous system demyelination (multiple sclerosis) have been reported after administration of a hepatitis B antigen containing vaccine.
Encephalopathy/encephalitis.
Apnoea in very premature infants (≤ 28 weeks of gestation) (see Section 4.4 Special Warnings and Precautions for Use).
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Not documented.
For general advice on overdose management, contact the Poisons Information Centre, telephone number 13 11 26.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: vaccines, bacterial and viral combined, ATC code: J07CA09.
Mechanism of action. Hexaxim induces the production of antibodies against diphtheria, tetanus, pertussis, hepatitis B, poliomyelitis and invasive infections caused by Haemophilus influenzae type b.
Clinical trials. The primary vaccination schedules that have been used are: 3, 5 months without hepatitis B vaccination at birth; 6, 10, 14 weeks with and without hepatitis B vaccination at birth; 2, 3, 4 months without hepatitis B vaccination at birth; 2, 4, 6 months with and without hepatitis B vaccination at birth.
Results obtained in the clinical studies for each of the components are summarised in Tables 3 and 4.
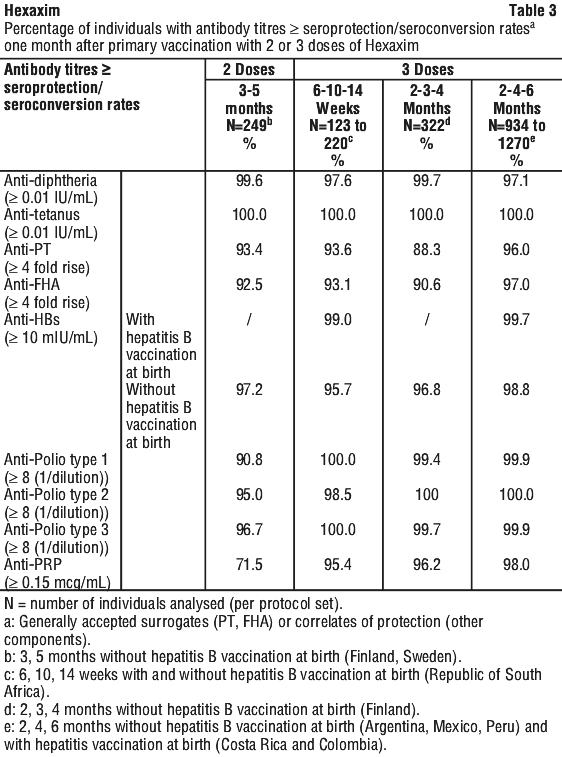
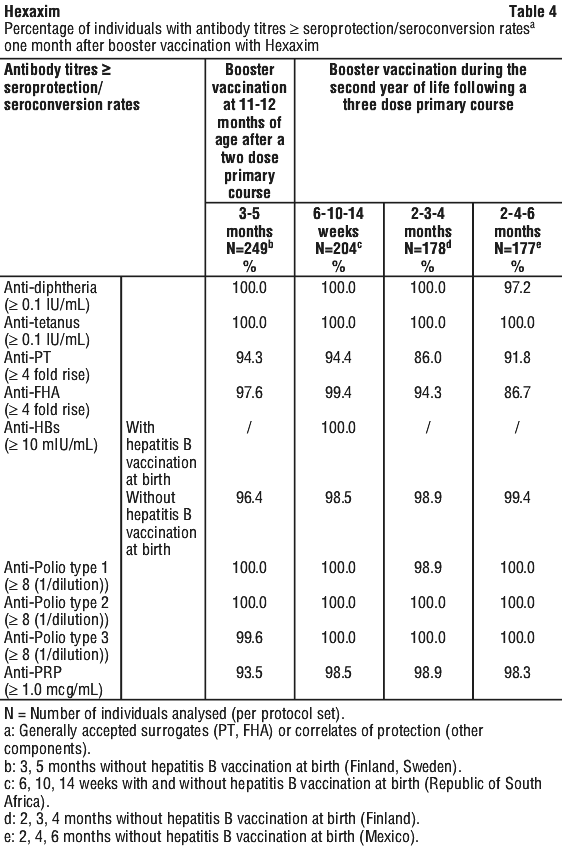
The vaccine effectiveness against Hib invasive disease of DTPa and Hib combination vaccines (pentavalent and hexavalent including vaccines containing the Hib antigen from Hexaxim) has been demonstrated in Germany via an extensive (over five years follow-up period) post-marketing surveillance study. The vaccine effectiveness was 96.7% for the full primary series, and 98.5% for booster dose (irrespective of priming) (2) (3).
5.2 Pharmacokinetic Properties
No pharmacokinetic studies have been performed.
5.3 Preclinical Safety Data
Genotoxicity. Hexaxim has not been evaluated for genotoxic potential.
Carcinogenicity. Hexaxim has not been evaluated for carcinogenic potential.
6 Pharmaceutical Particulars
6.1 List of Excipients
Hexaxim contains the excipients; dibasic sodium phosphate, monobasic potassium phosphate, trometamol, sucrose, essential amino acids (cystine, tyrosine, arginine hydrochloride, histidine, isoleucine, leucine, lysine hydrochloride, methionine, phenylalanine, threonine, tryptophan and valine) and water for injections.
The manufacture of this product includes exposure to bovine materials. No evidence exists that any case of vCJD (considered to be the human form of bovine spongiform encephalopathy) has resulted from the administration of any vaccine product.
6.2 Incompatibilities
Hexaxim must not be mixed with other vaccines or other parenterally administered drugs.
Separate injection sites must be used in case of concomitant administration.
6.3 Shelf Life
48 months.
6.4 Special Precautions for Storage
Store in a refrigerator (2°C-8°C). Do not freeze. Discard if vaccine has been frozen.
Protect from light.
Stability data indicate that the vaccine components are stable at temperatures up to 25°C for 72 hours. At the end of this period, Hexaxim should be used or discarded. These data are intended to guide healthcare professionals in case of temporary temperature excursion only.
6.5 Nature and Contents of Container
Hexaxim is supplied in:
0.5 mL single dose in pre-filled syringe without attached needle and one separate needle in a pack;
0.5 mL single dose in pre-filled syringe without attached needle and two separate needles in a pack.
Pack size of 1 or 10. Not all pack sizes may be marketed.
6.6 Special Precautions for Disposal
After use, any remaining vaccine and container must be disposed of safely according to locally agreed procedures.
6.7 Physicochemical Properties
Not applicable.
7 Medicine Schedule (Poisons Standard)
S4 Prescription Only Medicine.
Date of First Approval
11 September 2014
Date of Revision
07 October 2025
Summary Table of Changes

References
1. The Public Health Agency of Sweden, Pertussis Surveillance in Sweden - Fifteen Year Report, 2013 www.folkhalsomyndigheten.se/pagefiles/17379/pertussis-surveillance%20in-sweden-fifteen-year-report%282%29.pdf.
2. Kalies H et al, Four and one-half year follow-up of the effectiveness of diphtheria-tetanus toxoids-acellular pertussis/Haemophilus influenzae type b and diphtheria-tetanus toxoids-acellular pertussis-inactivated poliovirus/H. influenzae type b combination vaccines in Germany. Pediatr Infect Dis J 2004;23(10):944-950.
3. Schmitt HJ et al. Haemophilus influenzae type b disease: impact and effectiveness of diphtheria-tetanus toxoids-acellular pertussis (-inactivated poliovirus)/H. influenzae type b combination vaccines. Pediatr Infect Dis J 2001;20(8):767-774.
Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.