Hiprex
Brand Information
| Brand name | Hiprex |
| Active ingredient | Methenamine hippurate |
| Schedule | S3 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Hiprex.
Full CMI
Hiprex®
Active ingredient: Methenamine hippurate
Consumer Medicine Information (CMI)
This leaflet provides important information about using Hiprex. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Hiprex.
Where to find information in this leaflet:
1. Why am I using Hiprex?
2. What should I know before I use Hiprex?
3. What if I am taking other medicines?
4. How do I use Hiprex?
5. What should I know while using Hiprex?
6. Are there any side effects?
7. Product details
1. Why am I using Hiprex?
Hiprex contains the active ingredient methenamine hippurate. Hiprex is a urinary antibacterial agent.
Hiprex is used for prevention or suppression of bacteria in the urine that can cause chronic or recurrent urinary tract infections (UTIs).
2. What should I know before I use Hiprex?
Warnings
Do not use Hiprex if:
- you are allergic to methenamine hippurate, formaldehyde, or any of the ingredients listed at the end of this leaflet.
Always check the ingredients to make sure you can use this medicine. - you have severe (kidney failure, a kidney infection, severe dehydration or gout.
- you have neurogenic bladder, renal tract abnormalities or are using long-term catheters.
- you have severe liver impairment or metabolic acidosis.
Check with your doctor or pharmacist if you:
- have any other medical conditions
- take any medicines for any other condition
- are experiencing frequent or recurring urinary tract infections.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor or pharmacist if you are pregnant or intend to become pregnant.
Talk to your doctor or pharmacist if you are breastfeeding or intend to breastfeed.
Methenamine is found in breast milk in very small amounts.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Hiprex and affect how it works.
Medicines that may reduce the effect of Hiprex include:
- Alkalising agents and antacids.
As Hiprex breaks down to formaldehyde under acidic conditions in urine, using urinary alkalising agents such as potassium citrate, or antacid medicines may reduce its effectiveness and should be avoided.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Hiprex.
4. How do I use Hiprex?
How much to take
- Adults and Children 12 years or older: Take 1 tablet twice daily, with or without food.
- Swallow the tablet whole with a glass of water.
- Not recommended for use in children under 12 years.
- Follow the instructions provided with the medicine.
- Do not exceed the recommended dosage.
If you use too much Hiprex
If you think that you have used too much Hiprex, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using Hiprex?
Things you should do
Remind any doctor, dentist or pharmacist you visit that you are using Hiprex.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Hiprex affects you.
Hiprex is not expected to have any effect on your ability to drive or operate machinery.
Looking after your medicine
- Store below 30°C. Keep bottle tightly closed and airtight. This medicine contains a desiccant cannister to protect the product from moisture. The desiccant is not intended for consumption.
Follow the instructions on the bottle on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor or pharmacist if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
7. Product details
This medicine is available over-the-counter without a doctor's prescription.
What Hiprex contains
| Active ingredient (main ingredient) | Methenamine hippurate |
| Other ingredients (inactive ingredients) | Colloidal anhydrous silica Povidone Magnesium stearate |
Do not take this medicine if you are allergic to any of these ingredients.
What Hiprex looks like
Hiprex is a white, oblong-shaped tablet, coded HX with a score line on one side and plain on the other side.
Registration number AUST R 10558
Who distributes Hiprex
iNova Pharmaceuticals (Australia) Pty Limited
Level 10, 12 Help Street
Chatswood NSW 2067
Tel: 1800 630 056
This leaflet was prepared in August 2025.
Brand Information
| Brand name | Hiprex |
| Active ingredient | Methenamine hippurate |
| Schedule | S3 |
MIMS Revision Date: 01 November 2025
1 Name of Medicine
Methenamine hippurate.
2 Qualitative and Quantitative Composition
Each tablet contains 1 g of methenamine hippurate.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
White, oblong-shaped tablet, coded HX with score line on one surface and plain on the other face.
4 Clinical Particulars
4.1 Therapeutic Indications
For prophylaxis or suppression of bacteriuria associated with chronic or recurrent infection of the urinary tract.
4.2 Dose and Method of Administration
Adults and children 12 years or older. 1 tablet twice daily, with or without food.
Not recommended for use in children under 12 years.
Method of administration. For oral administration.
The tablets may be halved or crushed and taken with water if the patient is unable to swallow whole tablets.
Special populations. Renal impairment. No dose adjustments for patients with mild or moderate renal insufficiency.
Methenamine hippurate is not recommended for use by patients with neurogenic bladder, renal tract abnormalities or using long-term catheters. (see Section 4.3 Contraindications).
4.3 Contraindications
Hypersensitivity or allergy to methenamine hippurate, formaldehyde or to any of the excipients listed in Section 6.1 List of Excipients.
Severe renal failure (eGFR < 10 mL/min/1.73 m2), kidney infection, severe dehydration, or gout.
Severe hepatic impairment.
Metabolic acidosis.
4.4 Special Warnings and Precautions for Use
The underlying causes and risk factors for urinary tract infections should be investigated, and changes to perineal hygiene, sexual practices, urinary voiding and diet to maintain the normal urinary tract flora may be indicated before pharmacological interventions.
The effectivity of methenamine hippurate as a prophylactic agent depends on the acidity of urine, the bacterial species and counts in urine, their susceptibility to methenamine, and the exposure interval to formaldehyde that is hydrolysed from methenamine (see Section 5.1 Pharmacodynamic Properties, Mechanism of action). Methenamine may be ineffective when the urinary tract is colonised by new species, when bacteria multiply quickly under certain conditions, or when bacteria migrate from the lower urinary tract to the kidneys, therefore patients should be encouraged to consult their doctor at the onset of signs and symptoms of infection.
Bacteriological analysis of a urine sample is recommended to confirm the clinical diagnosis. When antibiotic treatment of bacteriuria or urinary tract infection is indicated, prophylaxis with methenamine hippurate should be stopped until infection is cleared, and urine becomes sterile (< 104 counts per mL).
Use in the elderly. The elderly are at higher risk of urinary tract infections because of changes in oestrogen levels (women), prostate problems (men), incontinence, increased use of medications, surgical or medical interventions including catheters, or decreased mobility and personal hygienic practices. No difference in the safety of methenamine hippurate in the elderly compared to the younger population has been observed.
Paediatric use. Hiprex is not recommended for children under 12 years of age (see Section 4.2 Dose and Method of Administration).
Effects on laboratory tests. In laboratory tests using acid hydrolysis of urine during pregnancy, the presence of methenamine or formaldehyde can result in unmeasurably low oestriol results. Enzymatic hydrolysis will provide more accurate results.
Methenamine can affect the determination of steroids, catecholamines and 5-hydroxyindole acetic acid in urine and give false results depending on the analytical method used.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Sulphonamides. Some sulphonamides may form an insoluble crystalline precipitate with formaldehyde in urine, therefore concurrent administration with methenamine should be avoided.
Alkalising agents. Methenamine hydrolyses into the therapeutically active formaldehyde under acidic conditions in urine, therefore concomitant use with agents that make urine alkaline or urinary alkalising agents such as potassium citrate or acetazolamide reduce the effectivity of methenamine and should be avoided.
Antacids might cause an increase of urine pH and hence decrease the effect of methenamine.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. There are no human data available on fertility. Data from studies in rats do not indicate any effects on female fertility, while effects on male fertility have not been adequately tested (see Section 5.3 Preclinical Safety Data).
Use in pregnancy. (Category A)
There is inadequate evidence of safety of methenamine hippurate in human pregnancy, but it has been in wide use for many years without apparent ill consequence. Animal studies are insufficient with respect to reproductive toxicity.
In limited studies in pregnant rabbits with methenamine hippurate at approximately 3 times the clinical dose based on body surface area, there was increased post-implantation loss resulting in lower litter sizes and a limited occurrence of foetal deformities including shortness of tail and malrotation of limbs. No effects on development were noted at doses equivalent to the clinical dose. Methenamine hippurate, administered at approximately 3 times the clinical dose, based on body surface area, did not adversely affect the fertility of female rats. Effects on male fertility have not been adequately studied.
As a precautionary measure, it is preferable to avoid the use of methenamine hippurate during pregnancy.
Use in lactation. Methenamine hippurate is excreted in human milk, but at therapeutic doses of Hiprex no effects on the breastfed newborn or infant are anticipated.
4.7 Effects on Ability to Drive and Use Machines
Hiprex has no, or negligible, effects on the ability to drive and use machines.
4.8 Adverse Effects (Undesirable Effects)
The safety of methenamine hippurate is estimated from its well-established use and published literature on studies involving small numbers of patients. Adverse effect frequencies are defined as: very common (≥ 1/l0); common (≥ 1/100 to < 1/10); uncommon (≥ 1/1000 to < 1/100); rare (≥ 1/10,000 to < 1/1000); very rare (< 1/10,000); not known (cannot be estimated from the available data). See Table 1.

Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: urinary antibacterial agent, ATC code: J01XX05.
Mechanism of action. Hiprex contains methenamine hippurate, a salt of methenamine and hippuric acid, which is absorbed and excreted rapidly.
Methenamine hippurate is a urinary antibacterial agent with a wide spectrum covering both gram-positive and gram-negative organisms. Urinary antibacterial activity can be shown within 30 minutes of administration.
The antibacterial action of methenamine hippurate arises from the slow release of formaldehyde when methenamine is hydrolysed in acidic urine.
Formaldehyde denatures the proteins and nucleic acid of bacteria, in particular E. coli, enterococci and staphylococci. Enterobacter aerogenes is generally resistant while urea-splitting bacteria such as Proteus and Pseudomonas species are inhibited by methenamine only when urine is sufficiently acidic.
A formaldehyde concentration above 25 micrograms per mL of urine maintained for about 2 hours is effective as a bactericidal. Higher urinary pH, flow rates and frequency decrease the formation, concentration and exposure time of formaldehyde and therefore its effectivity.
Clinical trials. The authors of a Cochrane review of methenamine hippurate for prevention of urinary tract infections (Lee et al, 2012) concluded that methenamine hippurate may be effective for preventing UTI in patients without renal tract abnormalities, particularly when used for short term prophylaxis, but that it does not appear to work in patients with neuropathic bladder or in patients who have renal tract abnormalities.
In a randomised, double blind, long term, crossover study (Cronberg et al, 1987), 1 g twice daily of methenamine hippurate was compared with placebo for its preventive effect on recurrent attacks of acute cystitis. Methenamine hippurate and placebo were interchanged every six months for two years. Out of 21 enrolled patients, 14 completed the first year and 13 both years of treatment, which permitted the evaluation of 27 patient years. There were 52 episodes of acute cystitis caused by reinfection: 41 occurred during placebo treatment and only 11 during the methenamine hippurate regimen (p < 0.01).
5.2 Pharmacokinetic Properties
Absorption. Following oral administration, methenamine is rapidly absorbed from the gastrointestinal tract, with a peak plasma concentration of about 30 mg/L occurring approximately 1-2 hours after ingestion of a single dose. It then declines with a mean elimination half-life of about 4 hours.
The peak plasma concentration from twice daily dosing is about 35 mg/L at steady-state, indicating that no drug accumulation takes place.
Distribution. The average distribution volume was about 0.56 L/kg, which is similar to the total body water in adults.
Metabolism. A small proportion of methenamine is degraded by stomach acid to formaldehyde, which is absorbed and quickly converted to formic acid - which in turn is oxidised to carbon dioxide and water or eliminated quickly through the kidneys. The remaining methenamine is unchanged until excreted by the kidneys.
Excretion. Excretion is by both tubular secretion and glomerular filtration. About 82% of a single methenamine dose is recovered intact in urine within 24 hours, while about 88% is recovered from 1 g twice daily dosing after a 12-hour interval.
5.3 Preclinical Safety Data
Genotoxicity. Limited in vitro tests indicate that methenamine is non-mutagenic to Salmonella typhimurium strains in the presence of S9, but some mutagenic effects with metabolic activation were observed after nitrosation.
Carcinogenicity. In limited studies, no evidence of carcinogenic activity was found in mice and rats, and methenamine did not induce neoplasms.
6 Pharmaceutical Particulars
6.1 List of Excipients
Magnesium stearate, colloidal anhydrous silica, povidone.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine (also see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C. Keep container tightly closed/airtight.
6.5 Nature and Contents of Container
Bottle. 20 or 100 tablets.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure.
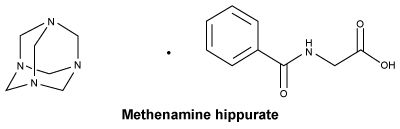
Chemical name. 2-benzamidoacetic acid; 1,3,5,7-tetrazatricyclo[3.3.1.13,7] decane.
Molecular formula. C15H12N5O31.
Molecular weight. 319.36 g/mol.
CAS number. 5714-73-8.
7 Medicine Schedule (Poisons Standard)
(S3) Pharmacist Only Medicine.
Date of First Approval
04 July 1991
Date of Revision
07 April 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.