Hydrea
Brand Information
| Brand name | Hydrea |
| Active ingredient | Hydroxycarbamide (hydroxyurea) |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Hydrea.
Summary CMI
HYDREA®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using HYDREA?
HYDREA contains the active ingredient hydroxycarbamide. HYDREA is used to treat various types of cancers such as, leukemia and cancer of the ovary.
For more information, see Section 1. Why am I using HYDREA? in the full CMI.
2. What should I know before I use HYDREA?
Do not use if you have ever had an allergic reaction to HYDREA or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use HYDREA? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with HYDREA and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use HYDREA?
- HYDREA capsules may be given to you at the hospital or surgery, or you may take them yourself at home
- Swallow HYDREA capsules with a glass of water
- HYDREA is taken as a single dose and can be taken each day, or once every three days, at about the same time each day.
More instructions can be found in Section 4. How do I use HYDREA? in the full CMI.
5. What should I know while using HYDREA?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using HYDREA? in the full CMI.
6. Are there any side effects?
If you get any of the following side effects, tell your doctor immediately or go to Accident and Emergency at your nearest hospital: chills, fever, fast heart beat, wheezing or coughing, difficulty breathing, dizziness, flushing, sweating and swelling of the face, tongue or other parts of the body.
Tell your doctor or nurse as soon as possible if you notice any of the following: nausea, vomiting, diarrhoea, stomach pain or discomfort, constipation. High fever (greater than 39°C) that requires hospitalization, feeling tired or sleepy, fever, high temperature, chills. Discolouration or ulcers on the skin, decay of the skin or nails, nail discolouration. Fever, severe chills. Pain in lungs or difficulty breathing, coughing. Sore mouth, tiredness, headaches, being short of breath, dizziness, convulsions. Joint pain, and/or swelling especially in the hands or feet, skin rashes, made worse by being in the sun, sores in the mouth or nose. Itching of the skin, joint aches, blisters that look like hives on the upper body, legs, arms, palms, hands, or feet and may involve the face or lips.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
HYDREA®
Active ingredient: hydroxycarbamide (hi-DROX-eee-Car-bah-myd)
Consumer Medicine Information (CMI)
This leaflet answers some common questions about HYDREA. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using HYDREA.
Where to find information in this leaflet:
1. Why am I using HYDREA?
2. What should I know before I use HYDREA?
3. What if I am taking other medicines?
4. How do I use HYDREA?
5. What should I know while using HYDREA?
6. Are there any side effects?
7. Product details
1. Why am I using HYDREA?
HYDREA contains the active ingredient hydroxycarbamide, an anti-cancer medicine. It interferes with the replication of cells and causes cell death, particularly in cancer cells.
HYDREA is used to treat various types of cancers such as, leukemia and cancer of the ovary.
HYDREA may be used alone or in combination with other treatments for cancer. Your doctor will inform you if this is necessary.
HYDREA is not recommended for use in children as its safety and effectiveness in patients under 18 years of age has not been established.
2. What should I know before I use HYDREA?
Warnings
Do not use HYDREA if:
- you are allergic to hydroxycarbamide, or any of the ingredients listed at the end of this leaflet
- always check the ingredients to make sure you can use this medicine
- you have marked depression of white blood cells (bone marrow depression) or red blood cells (severe anaemia). Your doctor will be able to advise whether HYDREA is appropriate in this situation.
- you are pregnant or intend to become pregnant
- the packaging shows signs of tampering
Check with your doctor if you:
have or have had any medical conditions, especially the following:
- liver or kidney problems
- any condition which reduces your blood cell “counts” such as white cells (leukopenia), neutrophils (neutropenia) or platelets (thrombocytopenia).
- infection or high temperature. Your doctor may decide to delay your treatment until the infection has gone.
Talk to your doctor if you are using a continuous glucose monitor (CGM) to test your blood glucose. Hydroxycarbamide may affect your sensor glucose results and may lead to low blood sugar (hypoglycaemia). Talk to the healthcare provider that prescribed your CGM about whether it is safe to use while you are taking HYDREA.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Tell your doctor and do not take HYDREA if you are pregnant or intend to become pregnant.
Tell your doctor if you are breastfeeding or plan to breastfeed.
HYDREA passes into breast milk. Therefore there is a possibility that the breast-fed baby may be affected.
Tell your doctor if you are a sexually active man.
HYDREA may cause birth defects if either the male or female is using it at the time of conception. Men who take HYDREA are advised to use a condom to avoid pregnancy in their partner.
Women who take Hydrea are advised to use birth control during therapy and for at least 6 months after therapy.
If you have not told your doctor about any of the above, or not sure whether you should have HYDREA, talk to your doctor.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including vaccinations, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and HYDREA may interfere with each other. These include:
- Vaccinations. HYDREA may suppress the body's normal defence mechanisms and result in a more severe than normal reaction with the virus
- Some other medicines that are used to treat other forms of cancer
- Medicines used to treat gout
- Medicines used to treat HIV infection including Videx® (didanosine) and Zerit® (stavudine)
These medicines may be affected by HYDREA, or affect how well it works. You may need different amounts of your medicine, or you may need to have different medicines. Your doctor will advise you.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect HYDREA.
4. How do I use HYDREA?
How much to take
Your doctor will decide what dose you will receive. Your dose of HYDREA capsules is worked out based on your body weight and height, the type of cancer you have, and other medicines you are taking. The dose worked out for you may be different to the dose for another patient.
HYDREA may be given alone or together with other anti-cancer treatments or drugs.
Ask your doctor if you want to know more about the dose of HYDREA you receive.
How to take HYDREA
HYDREA capsules may be given to you at the hospital or surgery, or you may take them yourself at home.
Swallow HYDREA capsules with a glass of water.
When to take HYDREA
HYDREA is taken as a single dose and can be taken each day, or once every three days. Your doctor will decide how you should take the capsules and will explain this to you.
Take HYDREA at about the same time each day that you are to take it.
HYDREA may or may not be given at the same time as other anti-cancer treatments or agents.
How long it is given
Several courses of HYDREA therapy may be needed depending on your response to treatment.
Additional treatment may not be repeated until your blood cell numbers return to acceptable levels and any side effects have been controlled.
Do not stop taking HYDREA unless your doctor tells you to - even if you feel better.
If you forget to take HYDREA
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Otherwise, take it as soon as you remember, and then go back to taking your medicine as you would normally.
Do not take a double dose to make up for the dose you missed.
This may increase the chance of you getting an unwanted side effect.
If you use too much HYDREA
If you are in hospital HYDREA will be given to you by the hospital staff, so it is most unlikely that too many capsules would be given. However, if an overdose should be given, your doctor will give you the appropriate treatment.
If you are taking HYDREA at home and think that you or someone else may have taken too many HYDREA capsules, you should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
Symptoms of overdose may include acute skin inflammation, soreness, discolouration of the skin, fluid retention associated with the hands & feet followed by scaling of the skin in these areas, soreness & inflammation of the mouth.
5. What should I know while using HYDREA?
Things you must do
Be sure to keep all your doctor's appointments and follow up appointments in order to get the best effect from your treatments and so your progress can be checked.
Remind any doctor, dentist, nurse or pharmacist you visit that you are having HYDREA.
Avoid exposing your skin to sunlight. Use appropriate clothing, a hat, and sunscreen if you must go in the sun. Check your skin regularly for any changes. If you notice any changes, tell your doctor.
You must maintain an adequate fluid intake.
Tell your doctor, dentist or pharmacist straight away that you are taking HYDREA if you:
- are about to be started on any new medicine
- plan to have surgery that needs a general anaesthetic
HYDREA can lower the number of white blood cells and platelets in your blood. This means that you have an increased chance of getting an infection or bleeding. The following precautions should be taken to reduce your risk if infection or bleeding:
- avoid people who have infections. Check with your doctor immediately if you think you may be getting an infection, or if you get a fever, chills, cough, hoarse throat, lower back or side pain or find it painful or difficult to urinate.
- be careful when using a toothbrush, toothpick or dental floss. Your doctor, dentist, nurse or pharmacist may recommend other ways to clean your teeth and gums. Check with your doctor before having any dental work.
- be careful not to cut yourself when you are using sharp objects such as a razor or nail cutters.
- avoid contact sports or other situations where you may bruise or get injured.
Things you must not do
- give HYDREA to anyone else, even if they have the same condition as you.
- take HYDREA to treat any other complaints unless your doctor tells you to.
- stop taking HYDREA, or lower the dose, without checking with your doctor.
If the powder is accidentally spilled, you should wipe up the powder using a damp paper towel. The broken capsule and the paper towel should be disposed of immediately in a closed container. Do not allow the powder to come into contact with your skin or moist areas of the body such as the eyes, mouth or nose. Also avoid inhaling the contents.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how HYDREA affects you.
As with other medicines used to treat cancer, HYDREA may cause dizziness, light-headedness or tiredness in some people. Make sure you know how you react to HYDREA before you drive a car, operate machinery, or do anything else that could be dangerous if you are dizzy or light-headed. If this occurs do not drive.
Looking after your medicine
- HYDREA capsules should be stored below 25°C
- The container must be tightly closed
Follow the instructions on the label on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight where the temperature stays below 25°C; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it. Pay particular attention to this when you are opening the capsules to dissolve the contents in water.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date. If you take this medicine after the expiry date has passed, it may not work as well.
6. Are there any side effects?
Tell your doctor or nurse as soon as possible if you do not feel well while you are having HYDREA.
Like other medicines that treat cancer, HYDREA may have unwanted side effects, some of which may be serious. You may need medical treatment if you get some of the side effects.
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Gut and digestion-related:
| Tell your doctor if you have any of these side effects and they worry you. |
Signs of frequent infections such as:
These could be a sign of serious lung disease Other:
| Tell your doctor or nurse as soon as possible if you notice any of these side effects. |
Serious side effects
| Serious side effects | What to do |
Signs of an allergic reaction such as:
| Tell your doctor or nurse straight away if you notice any of these serious side effects. |
Signs of a sudden life-threatening allergic reaction:
| Tell your doctor immediately or go straight to the Emergency Department at your nearest hospital if you notice any of these life-threatening side effects. |
If you are receiving radiation treatment with HYDREA, or are taking other medications as well as HYDREA, the side effects listed above may occur more frequently or be more severe.
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
The benefits and side effects of HYDREA may take some time to occur. Therefore even after you have finished your HYDREA treatment you should tell your doctor immediately if you notice any of the side effects listed above.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What HYDREA contains
| Active ingredient (main ingredient) | hydroxycarbamide |
| Other ingredients (inactive ingredients) |
|
Do not take this medicine if you are allergic to any of these ingredients.
What HYDREA looks like
HYDREA 500 mg capsules have an opaque green and opaque pink capsule shell, with "CHP 500" marked on each capsule. They are supplied in bottles of 100 capsules. (Aust R 47486).
Who distributes HYDREA
Link Medical Products Pty Ltd
5 Apollo Street
Warriewood, NSW, 2102
Ph: 1800 181 060
linkhealthcare.com.au
This leaflet was prepared in October 2025.
Brand Information
| Brand name | Hydrea |
| Active ingredient | Hydroxycarbamide (hydroxyurea) |
| Schedule | S4 |
MIMS Revision Date: 01 December 2025
1 Name of Medicine
Hydroxycarbamide.
2 Qualitative and Quantitative Composition
Hydrea hydroxycarbamide is an antineoplastic agent, available for oral use as capsules containing 500 mg hydroxycarbamide.
Excipients with known effect. Sugars as lactose.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Capsules.
Opaque green cap and opaque pink body imprinted CHP 500 in black ink.
4 Clinical Particulars
4.1 Therapeutic Indications
Significant tumour response to Hydrea has been demonstrated in chronic myelocytic leukaemia (pretreatment phase and palliative care), and recurrent, metastatic, or inoperable carcinoma of the ovary.
4.2 Dose and Method of Administration
Because of the rarity in children of chronic myelocytic leukaemia, carcinoma of the ovary and carcinomas of the head and neck, dosage regimens have not been established.
All dosage should be based on the patient's actual or ideal weight, whichever is less.
Elderly patients may require a lower dose regimen.
Patients should be reminded that this is a potent medication that must be handled with care (see Section 6.6 Special Precautions for Disposal).
Concurrent use of hydroxycarbamide with other myelosuppressive agents may require adjustments of dosages.
Solid tumours. Intermittent therapy. 80 mg/kg administered orally as a single dose every third day.
Continuous therapy. 20 to 30 mg/kg administered orally as a single dose daily.
The intermittent dosage schedule offers the advantage of reduced toxicity since patients on this dosage regimen have rarely required complete discontinuance of therapy because of toxicity.
Concomitant therapy with irradiation (carcinoma of the head and neck). 80 mg/kg administered orally as a single dose every third day.
Administration of hydroxycarbamide should be begun at least seven days before initiation of irradiation and continued during radiotherapy, as well as indefinitely afterwards, provided that the patient may be kept under adequate observation and evidences no unusual or severe reactions.
Irradiation should be given at the maximum dose considered appropriate for the particular therapeutic situation; adjustment of irradiation dosage is not usually necessary when hydroxycarbamide is used concomitantly.
Resistant chronic myelocytic leukaemia. Continuous therapy. Until the intermittent therapy regimen has been evaluated, continuous therapy (20 to 30 mg/kg administered orally as a single dose daily) is recommended.
An adequate trial period for determining the antineoplastic effectiveness of hydroxycarbamide is six weeks of therapy. When there is regression in tumour size or arrest in tumour growth, therapy should be continued indefinitely. Therapy should be interrupted if the white blood cell count drops below 2500/mm3, or the platelet count below 100,000/mm3. In these cases, the counts should be rechecked after three days, and therapy resumed when the counts rise significantly toward normal values. Since the haematopoietic rebound is prompt, it is usually necessary to omit only a few doses. If prompt rebound has not occurred during combination Hydrea and irradiation therapy, irradiation may also be interrupted. However, the need for postponement of irradiation has been rare; radiotherapy has usually been continued using the recommended dosage and technique. Anaemia, if it occurs, should be corrected with whole blood replacement, without interrupting hydroxycarbamide therapy. Because haematopoiesis may be compromised by extensive irradiation or by other antineoplastic agents, it is recommended that hydroxycarbamide be administered cautiously to patients who have recently received extensive radiation therapy or chemotherapy with other cytotoxic drugs.
Pain or discomfort from inflammation of the mucous membranes at the irradiated site (mucositis) is usually controlled by measures such as topical anaesthetics and orally administered analgesics. If the reaction is severe, hydroxycarbamide therapy may be temporarily interrupted; if it is extremely severe, irradiation therapy may, in addition, be temporarily postponed. However, it has rarely been necessary to terminate these therapies.
Severe gastric distress, such as nausea, vomiting, and anorexia, resulting from combined therapy may usually be controlled by temporary interruption of hydroxycarbamide administration; rarely has the additional interruption of irradiation been necessary.
Renal insufficiency. There are no data that support specific guidance for dosage adjustment in patients with impaired renal function. Since renal excretion is a pathway of elimination, consideration should be given to decreasing the dosage in this population. Close monitoring of haematologic parameters is advised.
Hepatic insufficiency. There are no data that support specific guidance for dosage adjustment in patients with impaired hepatic function. Close monitoring of haematologic parameters is advised.
4.3 Contraindications
Hydroxycarbamide is contraindicated in patients with marked bone marrow depression, i.e. leucopoenia (< 2500 WBC/mm3), thrombocytopenia (< 100,000/mm3), or severe anaemia.
A previous hypersensitivity to hydroxycarbamide or any other component of its formulation.
4.4 Special Warnings and Precautions for Use
Concurrent use of hydroxycarbamide and other myelosuppressive agents or radiation therapy may increase the likelihood of bone marrow depression as other adverse events.
Treatment with hydroxycarbamide should not be initiated if bone marrow function is markedly depressed (see Section 4.3 Contraindications). Bone marrow suppression may occur, and leucopoenia is generally its first and most common manifestation. Thrombocytopenia and anaemia occur less often, and are seldom seen without a preceding leucopoenia. However, the recovery from myelosuppression is rapid when therapy is interrupted. It should be borne in mind that bone marrow depression is more likely in patients who have previously received radiotherapy or cytotoxic cancer chemotherapeutic agents; hydroxycarbamide should be used cautiously in such patients.
Patients who have received irradiation therapy in the past may have an exacerbation of postirradiation erythema.
Cases of haemolytic anaemia in patients treated with hydroxycarbamide for myeloproliferative diseases have been reported (see Section 4.8 Adverse Effects (Undesirable Effects)). Patients who develop persistent anaemia should have laboratory tests evaluated for haemolysis. In the setting of confirmed diagnosis of haemolytic anaemia, hydroxycarbamide should be discontinued.
Fatal and nonfatal pancreatitis have occurred in HIV-infected patients during therapy with hydroxycarbamide and didanosine, with or without stavudine. Hepatotoxicity and hepatic failure resulting in death have been reported during post-marketing surveillance in HIV-infected patients treated with hydroxycarbamide and other antiretroviral agents. Fatal hepatic events were reported most often in patients treated with the combination of hydroxycarbamide, didanosine, and stavudine. This combination should be avoided. Peripheral neuropathy, which was severe in some cases, has been reported in HIV-infected patients receiving hydroxycarbamide in combination with antiretroviral agents, including didanosine, with or without stavudine.
Interstitial lung disease including pulmonary fibrosis, lung infiltration, pneumonitis, and alveolitis/allergic alveolitis have been reported in patients treated for myeloproliferative neoplasm and may be associated with fatal outcome. Patients developing pyrexia, cough, dyspnea, or other respiratory symptoms should be closely monitored, investigated and treated. Promptly discontinue hydroxycarbamide and treat with corticosteroids to resolve the pulmonary events (see Section 4.8 Adverse Effects (Undesirable Effects)).
Severe anaemia must be corrected with whole blood replacement before initiating therapy with hydroxycarbamide.
Erythrocytic abnormalities: megaloblastic erythropoiesis, which is self-limiting, is often seen early in the course of hydroxycarbamide therapy. The morphologic change resembles pernicious anaemia, but it is not related to vitamin B12 or folic acid deficiency. The macrocytosis may mask the incidental development of folic acid deficiency; regular determinations of serum folic acid are recommended. Hydroxycarbamide may also delay plasma iron clearance and reduce the rate of iron utilisation by erythrocytes, but it does not appear to alter the erythrocyte survival time.
Elderly patients may be more sensitive to the effects of hydroxycarbamide, and may require a lower dosage regimen.
In patients receiving long-term therapy with hydroxycarbamide for myeloproliferative disorders, such as polycythemia vera and thrombocythemia, secondary leukemia has been reported. It is unknown whether this leukemogenic effect is secondary to hydroxycarbamide or associated with the patients' underlying disease.
Therapy with hydroxycarbamide requires close supervision. The complete status of the blood, including bone marrow examination, if indicated, as well as kidney function and liver function, should be determined prior to, and repeatedly during, treatment. The determination of the haemoglobin level, total leucocyte counts, and platelet counts should be performed at least once a week throughout the course of hydroxycarbamide therapy. If the white blood cell count decreases to less than 2500/mm3, or the platelet count to less than 100,000/mm3, therapy should be interrupted until the values rise significantly toward normal levels. Anaemia, if it occurs, should be managed with whole blood replacement, without interrupting hydroxycarbamide therapy.
Since hydroxycarbamide may cause drowsiness and other neurologic effects, alertness may be impaired in driving or in operating machinery.
Patients should be advised to maintain adequate fluid intake. Patients should consult with their physician or pharmacist regarding missed doses.
Hydroxycarbamide is not indicated for the treatment of HIV-infection; however, if HIV-infected patients are treated with hydroxycarbamide, and in particular, in combination with didanosine and/or stavudine, close monitoring for signs and symptoms of pancreatitis and hepatotoxicity is recommended. Patients who develop signs and symptoms of pancreatitis or hepatotoxicity should permanently discontinue therapy with hydroxycarbamide. (See Section 4.4 Special Warnings and Precautions for Use; Section 4.8 Adverse Effects (Undesirable Effects)).
Cutaneous vasculitic toxicities including vasculitic ulcerations and gangrene have occurred in patients with myeloproliferative disorders during therapy with hydroxycarbamide. These vasculitic toxicities were reported most often in patients with a history of or currently receiving interferon therapy. Due to potentially severe clinical outcomes for the cutaneous vasculitic ulcers reported in patients with myeloproliferative disease, hydroxycarbamide should be discontinued if cutaneous vasculitic ulcerations develop and alternative cytoreductive agents should be initiated as indicated.
Interference with continuous glucose monitoring systems. Hydroxycarbamide may falsely elevate sensor glucose results from certain continuous glucose monitoring (CGM) systems and may lead to hypoglycaemia if sensor glucose results are relied upon to dose insulin.
If a patient using a CGM is to be prescribed hydroxycarbamide, consult with the CGM prescriber about alternative glucose monitoring methods.
Use in hepatic impairment. No information available.
Use in renal impairment. Hydroxycarbamide should be used with caution in patients with marked renal dysfunction.
Use in the elderly. Elderly patients may be more sensitive to the effects of hydroxycarbamide, and may require a lower dosage regimen.
Paediatric use. Safety and efficacy have not been established in children.
Effects on laboratory tests. Studies have shown that there is an analytical interference of hydroxycarbamide with the enzymes (urease, uricase, and lactic dehydrogenase) used in the determination of urea, uric acid and lactic acid, rendering falsely elevated results of these in patients treated with hydroxycarbamide. (See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.)
4.5 Interactions with Other Medicines and Other Forms of Interactions
Concurrent use of hydroxycarbamide and other myelosuppressive agents or radiation therapy may increase the likelihood of bone marrow depression or other adverse events (see Section 4.4 Special Warnings and Precautions for Use).
Vaccinations. Concomitant use of Hydrea with a live virus vaccine may potentiate the replication of the vaccine virus and/or may increase the adverse reaction of the vaccine virus because normal defense mechanisms may be suppressed by Hydrea. Vaccination with a live vaccine in a patient taking Hydrea may result in severe infection. Patient's antibody response to vaccines may be decreased. The use of live vaccines should be avoided and individual specialist advice sought.
Other interactions. Studies have shown that there is an analytical interference of hydroxycarbamide with the enzymes (urease, uricase, and lactic dehydrogenase) used in the determination of urea, uric acid and lactic acid, rendering falsely elevated results of these in patients treated with hydroxycarbamide. Since hydroxycarbamide may raise the serum uric acid level, dosage adjustment of uricosuric medication may be necessary.
There is increased risk of fatal systemic vaccine disease with the concomitant use of live vaccines. Live vaccines are not recommended in immunosuppressed patients.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Azoospermia or oligospermia, sometimes reversible, have been observed in men. Male patients should be informed about the possibility of sperm conservation before the start of the therapy.
Hydroxycarbamide may be genotoxic. Men under therapy are advised to use safe contraceptive measures during and at least 1 year after therapy. (See Section 4.8 Adverse Effects (Undesirable Effects)).
Female patients of reproductive potential should be counselled to use effective contraception during therapy and for at least 6 months after therapy.
Use in pregnancy. (Category D)
Drugs which affect DNA synthesis, such as hydroxycarbamide, may be potential mutagenic agents. The physician should carefully consider this possibility before administering this drug to male or female patients who may contemplate conception.
Hydroxycarbamide has been demonstrated to be a potent teratogenic agent in animals and can cause foetal harm when administered to a pregnant woman. Therefore, hydroxycarbamide should not be used in women who are or may become pregnant unless in the judgement of the physician the potential benefits outweigh the possible hazards. Women of childbearing potential should be advised to avoid becoming pregnant while taking hydroxycarbamide.
Use in lactation. Hydroxycarbamide is excreted in human milk. Because of the potential for serious adverse reactions in nursing infants from hydroxycarbamide, a decision should be made whether to discontinue nursing or to discontinue the drug, taking into account the importance of the drug to the mother.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Adverse reactions have been primarily bone marrow depression (leucopoenia, anaemia, and occasionally thrombocytopenia), haemolytic anaemia and less frequently gastrointestinal symptoms (stomatitis, anorexia, nausea, vomiting, diarrhoea, and constipation), and dermatological reactions such as maculopapular rash, facial erythema, peripheral erythema, hyperpigmentation, erythema, atrophy of skin and nails, nail pigmentation, skin ulceration, dermatomyositis-like skin changes, scaling, cutaneous lupus erythematosus and violet papules have been observed. Skin cancer has also been reported rarely. Dysuria and alopecia occur very rarely.
Large doses may produce moderate drowsiness. Neurological disturbances have occurred extremely rarely and were limited to headache, dizziness, disorientation, hallucinations and convulsions.
Hydroxycarbamide occasionally may cause temporary impairment of renal tubular function accompanied by elevations in serum uric acid, BUN, and creatinine levels. Abnormal BSP retention has been reported.
Fever, chills, malaise, asthenia, azoospermia, oligospermia, cholestasis, hepatitis, tumour lysis syndrome, and elevation of hepatic enzymes have also been reported.
Adverse reactions observed with combined hydroxycarbamide and irradiation therapy are similar to those reported with the use of hydroxycarbamide alone. These effects primarily include bone marrow depression (anaemia and leucopoenia) and gastric irritation. Almost all patients receiving an adequate course of combined hydroxycarbamide and irradiation therapy will demonstrate concurrent leucopoenia. Platelet depression (less than 100,000 cells/mm3) has occurred rarely and only in the presence of marked leucopoenia. Gastric distress has also been reported with irradiation alone and in combination with hydroxycarbamide therapy.
Musculoskeletal and connective tissue disorders. Systemic lupus erythematosus.
Hypersensitivity. Drug-induced fever. High fever (> 39°C) requiring hospitalisation in some cases has been reported concurrently with gastrointestinal, pulmonary, musculoskeletal, hepatobiliary, dermatological or cardiovascular manifestations. Onset typically occurred within 6 weeks of initiation and resolved promptly after discontinuation of hydroxycarbamide. Upon re-administration fever re-occurred within 24 hours.
It should be borne in mind that therapeutic doses of irradiation alone produce the same adverse reactions as hydroxycarbamide; combined therapy may cause an increase in the incidence and severity of these side effects.
Although inflammation of the mucous membranes at the irradiated site (mucositis) is attributed to irradiation alone, some investigators believe that the more severe cases are due to combination therapy.
Respiratory. The association of hydroxycarbamide with the development of acute pulmonary reactions consisting of diffuse pulmonary infiltrates, fibrosis, fever and dyspnoea has been rarely reported.
Pulmonary oedema, lung infiltration, interstitial lung disease, pneumonitis, alveolitis, allergic alveolitis and cough have also been reported.
Fatal and nonfatal pancreatitis and hepatotoxicity, and severe peripheral neuropathy have been reported in HIV-infected patients who received hydroxycarbamide in combination with anti-retroviral agents, in particular, didanosine plus stavudine. Patients treated with hydroxycarbamide in combination with didanosine, stavudine, and indinavir showed a median decline in CD4 cells of approximately 100/mm3 (see Section 4.4 Special Warnings and Precautions for Use).
Cutaneous vasculitic toxicities including vasculitic ulcerations and gangrene have occurred in patients with myeloproliferative disorders during therapy with hydroxycarbamide. These vasculitic toxicities were reported most often in patients with a history of or currently receiving interferon therapy. (See Section 4.4 Special Warnings and Precautions for Use.)
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Acute mucocutaneous toxicity has been reported in patients receiving hydroxycarbamide at dosages several times the therapeutic dose. Soreness, violet erythema, edema on palms and foot soles followed by scaling of hands and feet, severe generalized hyperpigmentation of skin, and stomatitis have also been observed.
For information on the management of overdose, contact the Poison Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. The precise mechanism by which hydroxycarbamide produces its cytotoxic effects cannot, at present, be described. However, the reports of various studies in tissue culture, rats and man lend support to the hypothesis that hydroxycarbamide causes an immediate inhibition of DNA synthesis without interfering with the synthesis of ribonucleic acid or of protein. This hypothesis explains why, under certain conditions, hydroxycarbamide may induce teratogenic effects.
Three mechanisms of action have been postulated for the increased effectiveness of concomitant use of hydroxycarbamide therapy with irradiation on squamous cell (epidermoid) carcinomas of the head and neck. In vitro studies utilising Chinese hamster cells suggest that hydroxycarbamide (1) is lethal to normally radio resistant S-stage cells and (2) holds other cells of the cell cycle in the G1 or pre-DNA synthesis stage where they are most susceptible to the effects of irradiation. The third mechanism of action has been theorised on the basis of in vitro studies of HeLa cells; it appears that hydroxycarbamide, by inhibition of DNA synthesis, hinders the normal repair process of cells damaged but not killed by irradiation, thereby decreasing their survival rate; RNA and protein synthesis have shown no alteration.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. After oral administration in man, hydroxycarbamide is readily absorbed from the gastrointestinal tract.
Distribution. The drug reaches peak serum concentrations within 2 hours; by 24 hours the concentration in the serum is essentially zero.
Metabolism. No information available.
Excretion. Approximately 80% of an oral or intravenous dose of 7 to 30 mg/kg may be recovered in the urine within 12 hours.
5.3 Preclinical Safety Data
Genotoxicity. Hydroxycarbamide is unequivocally genotoxic and a presumed transpecies carcinogen which implies a carcinogenic risk to humans.
Carcinogenicity. In patients receiving long-term hydroxycarbamide for myeloproliferative disorders, such as polycythemia vera and thrombocythemia, secondary leukemia has been reported; it is unknown whether this leukemogenic effect is secondary to hydroxycarbamide or the patients' underlying disease. Skin cancer has also been reported in patients receiving long-term hydroxycarbamide. Patients should be advised to protect skin from sun exposure, conduct self-inspection of the skin and be screened from secondary malignancies during routine follow-up visits.
6 Pharmaceutical Particulars
6.1 List of Excipients
The capsules contain the inactive ingredients: citric acid, gelatin, lactose monohydrate, magnesium stearate, dibasic sodium phosphate, titanium dioxide and capsule colourants (erythrosine, indigo carmine and iron oxide yellow) and Opacode monogramming ink S-1-277002 Black (ARTG PI No: 107581).
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C.
6.5 Nature and Contents of Container
Hydrea hydroxycarbamide in HDPE bottles with PP child resistant caps containing 100 capsules.
6.6 Special Precautions for Disposal
Guidelines for proper handling and disposal of anticancer drugs. Cytostatics must always be handled with care. Procedures for proper handling and disposal of anticancer drugs should be considered.
To minimize the risk of dermal exposure, always wear impervious gloves and wash your hands with soap and water before and after handling Hydrea or bottles containing Hydrea capsules. This includes all handling activities in clinical settings, pharmacies, storerooms, and home healthcare settings, including during unpacking and inspection, transport within a facility and dose preparation and administration. Pregnant women should not handle this medicinal product. Keep Hydrea out of the sight and reach of children and pets. People who are not taking Hydrea should not be exposed to the product. The contents of capsules should not be inhaled or allowed to come into contact with the skin or mucous membranes. Spilled powder must be immediately wiped up using a damp paper towel and discarded in a closed container, as should the broken capsules. In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure.
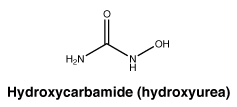
Molecular weight: 76.05 g/mole.
Hydroxycarbamide is an essentially tasteless, white to off-white crystalline powder. It is hygroscopic and freely soluble in water, but practically insoluble in alcohol.
CAS number. 127-07-1.
7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Date of First Approval
18 January 1994
Date of Revision
27 October 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.