Hyoscine Butylbromide SXP Injection
Brand Information
| Brand name | Hyoscine Butylbromide SXP Injection |
| Active ingredient | Hyoscine butylbromide |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Hyoscine Butylbromide SXP Injection.
Summary CMI
Hyoscine Butylbromide SXP
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I given Hyoscine Butylbromide SXP?
Hyoscine Butylbromide SXP contains the active ingredient hyoscine butylbromide. Hyoscine butylbromide is an anticholinergic medicine. Hyoscine Butylbromide SXP is used to relieve the pain of stomach and bowel cramps by helping your digestive system to relax. It is alsoused to help relax gallbladder spasms and kidney spasms, and as a diagnostic aid in radiology.
For more information, see Section 1. Why am I given Hyoscine Butylbromide SXP? in the full CMI.
2. What should I know before I receive Hyoscine Butylbromide SXP?
Do not use if you have ever had an allergic reaction to hyoscine butylbromide or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I receive Hyoscine Butylbromide SXP? in the full CMI.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking or using any other medicines including any that you get without a prescription from your pharmacy, supermarket, or health food shop. Hyoscine Butylbromide SXP must not be given in the muscle if you are being treated withanticoagulant medicines.
For more information see Section 3. What if I am taking or using other medicines? in the full CMI.
4. How do I receive Hyoscine Butylbromide SXP?
- Your doctor will decide what dose you will receive and the duration of treatment.
- Hyoscine Butylbromide SXP is administered by a doctor or nurse. It is given by deep injection into a muscle (e.g. the buttock, upper leg or upperarm) or slow injection into a vein.
More instructions can be found in Section 4. How do I receive Hyoscine Butylbromide SXP? in the full CMI.
5. What should I know after being given Hyoscine Butylbromide SXP?
| Things you should do |
|
| Things you should not do |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know after being given Hyoscine Butylbromide SXP? in the full CMI.
6. Are there any side effects?
Less serious side effects include redness of the skin, itching, a skin condition called dyshidrosis, rash, abnormal sweating, reduced sweating, fast heart rate, drop in blood pressure, dry mouth, flushing, difficulty with passing urine, shortness of breath,dizziness, and temporary blurred vision due to reduce eye focusing. Serious side effects are allergic reactions (such as skin rashes, or swelling of the face and difficulty in breathing) and sudden life-threatening allergic reactions (anaphylaxis with episodes of shortness of breath and shock).
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Hyoscine Butylbromide SXP
Active ingredient: hyoscine butylbromide
Consumer Medicine Information (CMI)
This leaflet provides important information about using Hyoscine Butylbromide SXP. You should also speak to your doctor, or pharmacist if you would like further information or if you have any concerns or questions about using Hyoscine Butylbromide SXP.
Where to find information in this leaflet:
1. Why am I given Hyoscine Butylbromide SXP?
2. What should I know before I am given Hyoscine Butylbromide SXP?
3. What if I am taking other medicines?
4. How do I receive Hyoscine Butylbromide SXP?
5. What should I know after being given Hyoscine Butylbromide SXP?
6. Are there any side effects?
7. Product details
1. Why am I given Hyoscine Butylbromide SXP?
Hyoscine Butylbromide SXP contains the active ingredient hyoscine butylbromide. Hyoscine butylbromide is an anticholinergic medicine. Hyoscine Butylbromide SXP is used to relieve the pain of stomach and bowel cramps by helping your digestive system to relax. It is also used to help relax gallbladder spasms and kidney spasms, and as a diagnostic aid in radiology.
Hyoscine Butylbromide SXP reduces the peristalsis (wave-like contractions) of smooth muscle resulting in relief from spasms in certain organs in the digestive system.
2. What should I know before I receive Hyoscine Butylbromide SXP?
Warnings
Do not receive Hyoscine Butylbromide SXP if:
- you are allergic to hyoscine butylbromide, or any of theingredients listed at the end of this leaflet.
- Some of the symptoms of an allergic reaction may include:
- shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin - Always check the ingredients to make sure you can usethis medicine.
- you have myasthenia gravis - a condition in which the muscles become weak and tire easily.
- you have glaucoma - high pressure in the eye
- you have porphyria - a rare blood pigment disorder
- you have a suspected or confirmed blockage of the bowel or stomach
- you have a weakened intestinal muscles (intestinal atony)
- you have a condition where the bowel is blocked and does not work properly (paralytic or obstructive ileus). Symptoms include severe abdominal pain with lack of stools and/or nausea/vomiting.
- you have a very enlarged bowel (megacolon)
- you have a fast heart rate (tachyarrhythmia)
- you have a condition that causes difficulty in swallowing food (achalasia)
- you have prostate problems - prostatic hypertrophy
- you are taking anticoagulants medicines and Hyoscine Butylbromide SXP will be given in the muscle
Tell your doctor immediately if your abdominal pain continues or worsens or occurs with symptoms like:
- fever
- nausea
- vomiting
- changes in bowel movements
- fainting
- blood in faeces
Check with your doctor if you:
- you have any other medical conditions
- you take any medicines for any other condition
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or plan to breastfeed.
3. What if I am taking other medicines?
Tell your doctor, or pharmacist if you are taking or using any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Hyoscine Butylbromide SXP must not be given in the muscle if you are being treated with anticoagulant medicines.
Some medicines and Hyoscine Butylbromide SXP may interfere with each other:
- medicines used to treat or prevent nausea and vomiting such as metoclopramide
- medicines used to treat malaria such as quinine
- medicines used to treat the symptoms of Parkinson's disease such as amantadine
- medicines used to treat some mental conditions such as tri and tetracyclic antidepressants and antipsychotics
- medicines used to treat allergies such as antihistamines
- medicines for the treatment of depression, heart disease or respiratory disease such as tiotropium, ipratropium, or atropine-like compounds
- any other medicine for the treatment of stomach or bowel conditions
- medicine used to treat certain conditions, such as disopyramide
Check with your doctor, or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Hyoscine Butylbromide SXP.
4. How do I receive Hyoscine Butylbromide SXP?
How much to be given
- Your doctor will decide what dose you will receive. This depends on your condition and other factors.
- The recommended dose is one or two ampoules (20 - 40 milligrams)
The maximum dose per day is 100 milligrams (equivalent to five ampoules).
Your doctor might prescribe a different dose or duration of treatment. If you need further information, ask your doctor or pharmacist.
Do not use it for extended periods.
Worsening or persistent stomach pain should be investigated by your doctor to determine the cause of the pain.
How it is given
This medicine should only be given by a doctor or nurse and in a setting where appropriate equipment is readily available for diagnosis and patient monitoring.
The injections can be given deeply into a muscle (e.g. the buttock, upper leg, or upper arm) or can be given as a slow injection into the vein.
If you receive too much Hyoscine Butylbromide SXP
These injections are given under medical supervision and it is very unlikely that you will be given too much.
Symptoms of an overdose may include drowsiness, dry mouth, difficulty passing urine, reddening of the skin, decreased gastrointestinal tract movement, fast heart rate and sight disturbances.
If you think that you have been given too much Hyoscine Butylbromide SXP, you may need urgent medical attention.
Tell your doctor or medical staff immediately if you feel you have been given too much Hyoscine Butylbromide SXP.
You should do this even if there are no signs of discomfort or poisoning.
If you experience any signs of overdose after you leave the doctor or nurse, you should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26 if you are in Australia), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
5. What should I know after being given Hyoscine Butylbromide SXP?
Things you should do
Before you take any other medicine, talk to your doctor or pharmacist.
Call your doctor straight away if:
- the pain is severe or does not improve within 48 hours of being given Hyoscine Butylbromide SXP. Cramps in the stomach or bowel may be temporary or may signal the presence of a more serious problem.
- you develop painful, red eye/s with loss of vision.
Remind any doctor, dentist, or pharmacist you visit that you have received Hyoscine Butylbromide SXP.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Hyoscine Butylbromide SXP affects you.
Hyoscine Butylbromide SXP may cause dizziness, tiredness, drowsiness in some people. If you have any of these symptoms, do not drive, operate machinery, or do anything else that could be dangerous.
Drinking Alcohol
Do not drink alcohol after receiving Hyoscine Butylbromide SXP.
Alcohol may increase the chance of side effects such as drowsiness.
Looking after your medicine
- Store below 25°C.
Store it in a cool dry place away from moisture, heat or sunlight. Do not store it in the bathroom, near a sink, in the car or on windowsills.
Keep it where young children cannot reach it.
Each ampoule is for single use only.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor, or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serous side effects | What to do |
Skin-related
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Very rarely there have also been isolated reports of:
- coma,
- hallucinations (seeing, feeling, or hearing things that are not there),
- unusual muscle tone causing distortion of the body (dystonia),
- confusion,
- agitation, and
- dizziness.
These side effects were relieved when the patients stopped Buscopan therapy and received appropriate medical treatment.
Serious side effects
| Serious side effects | What to do |
Allergy-related
| Call your doctor straight away or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Hyoscine Butylbromide SXP contains
| Active ingredient (main ingredient) | 20 mg of hyoscine butylbromide per ampoule |
| Other ingredients (inactive ingredients) |
|
| Potential allergens | Not applicable |
Do not take this medicine if you are allergic to any of these ingredients.
What Hyoscine Butylbromide SXP looks like
Hyoscine Butylbromide SXP 1 mL glass ampoules contain a clear, colourless solution.
Hyoscine Butylbromide SXP ampoules are available in packs of 5 or 10 ampoules.
Who distributes Hyoscine Butylbromide SXP?
Southern XP Pty Ltd
Unit 5/118 Church Street
Hawthorn, 3122, Victoria
Australia
Sponsor:
Southern XP IP Pty Ltd
Unit 5/118 Church Street
Hawthorn, 3122, Victoria
Australia
This leaflet was prepared in January 2022.
Australian Registration Number
AUST R 278817
Brand Information
| Brand name | Hyoscine Butylbromide SXP Injection |
| Active ingredient | Hyoscine butylbromide |
| Schedule | S4 |
MIMS Revision Date: 01 May 2022
Notes
Distributed by Southern XP Pty Ltd
1 Name of Medicine
Hyoscine butylbromide.
2 Qualitative and Quantitative Composition
Ampoule containing hyoscine butylbromide 20 mg in 1 mL.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Clear, colourless or almost colourless solution.
4 Clinical Particulars
4.1 Therapeutic Indications
Spasm of the gastrointestinal tract, biliary spasm, renal spasm, diagnostic aid in radiology.
4.2 Dose and Method of Administration
1 or 2 ampoules (20 or 40 mg) by intramuscular or slow intravenous injection. A maximum daily dose of 100 mg should not be exceeded.
Hyoscine butylbromide ampoules should not be administered on a continuous daily basis or for extended periods without investigating the cause of abdominal pain.
Product is for single use in one patient only. Discard any residue.
4.3 Contraindications
Hyoscine butylbromide is contraindicated in patients with:
known hypersensitivity to hyoscine butylbromide or to any excipients of the product (excipients are listed under List of Excipients);
mechanical stenoses of the gastrointestinal tract;
achalasia;
paralytic or obstructive ileus;
intestinal atony;
prostatic hypertrophy with urinary retention;
myasthenia gravis;
glaucoma;
pathological tachyarrhythmias;
megacolon.
Hyoscine butylbromide should not be given to patients with porphyria as, according to a single report, it has been said to exacerbate the disease.
By intramuscular injection, hyoscine butylbromide ampoules are contraindicated in patients being treated with anticoagulant drugs as intramuscular haematoma may occur. In these patients, intravenous routes may be used.
4.4 Special Warnings and Precautions for Use
In case severe, unexplained abdominal pain persists or worsens, or occurs together with symptoms like fever, nausea, vomiting, changes in bowel movements, abdominal tenderness, decreased blood pressure, fainting or blood in stool, medical advice should immediately be sought.
Hyoscine may cause drowsiness: patients so affected should not drive or operate machinery. Patients should abstain from alcohol. However, as a quaternary ammonium compound with low lipid solubility, hyoscine butylbromide cannot cross the blood/brain barrier easily and only rarely causes the central nervous system side effects associated with atropine and hyoscine.
After parenteral administration of hyoscine butylbromide, patients with visual accommodation disturbances should not drive or operate machinery until vision has normalised.
Elevation of intraocular pressure may be produced by the administration of anticholinergic agents such as hyoscine butylbromide in patients with undiagnosed and therefore untreated narrow-angle glaucoma. Patients should be advised to seek urgent ophthalmological advice if they develop a painful, red eye with loss of vision after an injection of hyoscine butylbromide. Because of the potential risk of anticholinergic complications, caution should be used in patients prone to narrow angle glaucoma as well as in patients susceptible to intestinal or urinary outlet obstructions and in those inclined to tachyarrhythmia.
After parenteral administration, cases of anaphylaxis including episodes of shock have been observed. As with all drugs causing such reactions, patients receiving hyoscine butylbromide by injection should be kept under observation.
Patients with cardiac conditions who are undergoing parenteral treatment with hyoscine butylbromide ampoules should be monitored.
Hyoscine butylbromide ampoules can cause tachycardia, hypotension and anaphylaxis, therefore use with caution in patients with cardiac conditions such as cardiac failure, coronary heart disease, cardiac arrhythmia or hypertension, and in cardiac surgery. Monitoring of these patients is advised. Emergency equipment and personnel trained in its use must be readily available.
Use in the elderly. No data available.
Paediatric use. No data available.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
The anticholinergic effects of drugs such as amantadine, tri- and tetracyclic antidepressants, quinidine, antihistamines, antipsychotics, disopyramide, phenothiazines, belladonna alkaloids, other anticholinergics (e.g. tiotropium, ipratropium, atropine-like compounds) and MAO inhibitors etc, together with the tachycardia induced by beta-sympathomimetics, can be potentiated by hyoscine butylbromide.
The concomitant administration of dopamine antagonists, such as metoclopramide, can reciprocally antagonise the effect on gastrointestinal tract motility.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category B2)
There is limited data from the use of hyoscine butylbromide in pregnant women. As a precautionary measure, it is preferable to avoid the use of hyoscine butylbromide during pregnancy.
Use in lactation. There is insufficient information on the excretion of hyoscine butylbromide and its metabolites in human milk. As a precautionary measure, it is preferable to avoid the use of hyoscine butylbromide during lactation.
4.7 Effects on Ability to Drive and Use Machines
No studies on the effects on the ability to drive and use machines have been performed.
However, patients should be advised that they may experience undesirable effects such as accommodation disorder or dizziness during treatment with hyoscine butylbromide ampoules.
Therefore, caution should be recommended when driving a car or operating machinery. If patients experience accommodation disorder or dizziness, they should avoid potentially hazardous tasks such as driving or operating machinery.
4.8 Adverse Effects (Undesirable Effects)
Many of the listed adverse effects of hyoscine butylbromide can be attributed to its anticholinergic properties. Anticholinergic side effects of hyoscine butylbromide are generally mild and self limited.
Immune system disorders. Anaphylactic reactions including fatal outcomes, anaphylactic reactions, dyspnoea, skin reactions (e.g. urticarial rash, erythema, pruritus), facial and periorbital swelling and other hypersensitivity reactions.
Eye disorders. Accommodation disorders, mydriasis, increased intraocular pressure.
Cardiac disorders. Tachycardia.
Vascular disorders. Decreased blood pressure, dizziness and flushing.
Gastrointestinal disorders. Dry mouth.
Skin and subcutaneous tissue disorders. Dyshidrosis, abnormal sweating.
Renal and urinary disorders. Impaired micturition, urinary retention.
Nervous system disorders. Very rarely in the national post marketing surveillance data base, there have been isolated reports following parenteral administration of coma, hallucinations, dystonia, confusion, agitation and dizziness from which the patient recovered after drug withdrawal and appropriate treatment. In very rare cases, dyspnoea has been reported.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
Symptoms. Serious signs of poisoning have not been observed in man. In case of overdose, anticholinergic symptoms such as urinary retention, dry mouth, reddening of the skin, inhibition of gastrointestinal motility, tachycardia, drowsiness and transient visual disorders may occur.
Toxicity data from studies in animals after parenteral administration suggest that the following may be possible: shock, Cheyne-Stokes respiration, respiratory paralysis, clonic spasms, paralysis of striated muscle, coma, paralytic ileus, bladder atony.
Management. Supportive measures if necessary should be instituted. Symptoms of overdosage may respond to parasympathomimetics. Ophthalmological advice should be sought urgently in cases of glaucoma. Pilocarpine may be administered locally in patients with glaucoma. Sympathomimetics may be used for circulatory support. For mental excitation, diazepam.
Cardiovascular complications as a result of using this medicine should be treated according to usual therapeutic principles. In case of respiratory paralysis, intubation and assisted respiration.
Catheterisation may be required for urinary retention. In addition, appropriate supportive measures should be used as required.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Hyoscine butylbromide is a quaternary ammonium compound which, as an anticholinergic agent, has a ganglion blocking component. Due to its anticholinergic action, hyoscine butylbromide reduces the tone and peristalsis of smooth muscle in hollow organs with parasympathetic innervation. As a quaternary ammonium compound with low lipid solubility, it cannot pass the blood/brain barrier easily and only rarely causes the central nervous system side effects associated with atropine and hyoscine.
Hyoscine butylbromide is a spasmolytic. The anticholinergic spasmolytic effect is based both on competitive inhibition of the parasympathetic activation of smooth muscle mediated through muscarinic receptors and, more markedly, through ganglionic blockade of neural transmission.
Hyoscine butylbromide is a powerful smooth muscle relaxant, effective when given by mouth or by injection. In the recommended dosages, hyoscine butylbromide relieves smooth muscle spasm rapidly. Undesirable 'atropine-like' side effects such as blurred vision, palpitation or dry mouth are rare.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. Hyoscine butylbromide is poorly absorbed from the gastrointestinal tract.
Distribution. Hyoscine butylbromide is rapidly distributed. The bioavailability of oral hyoscine butylbromide, as calculated from plasma levels, is reported to be 0.13%. Upon oral administration hyoscine butylbromide concentrates especially in the tissue of the gastrointestinal tract, liver and kidneys.
Metabolism. The high affinity of this agent to the tissue is reflected by the very short half-life, t-alpha of 3 minutes (distribution phase) of the blood levels.
Excretion. Despite the very short half life, the excretion rates are slow. Thus, in spite of the extremely low blood levels measurable over a short period of time, hyoscine butylbromide remains available at the site of action in the tissue in high concentrations.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Hyoscine butylbromide ampoules also contain sodium chloride and water for injections, sodium hydroxide and hydrochloric acid (for pH adjustment).
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Protect from light.
6.5 Nature and Contents of Container
Packs of 5 and 10 clear Type I glass ampoules.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure.
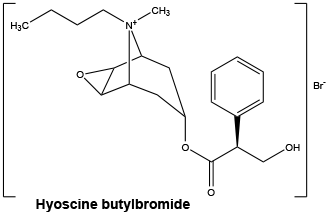
Molecular weight: 440.4.
CAS number. 149-64-4.
7 Medicine Schedule (Poisons Standard)
S4 (Prescription Only Medicine).
Date of First Approval
03 July 2017
Date of Revision
17 March 2022
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.