Icatibant Lupin
Brand Information
| Brand name | Icatibant Lupin |
| Active ingredient | Icatibant |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Icatibant Lupin.
Summary CMI
Icatibant Lupin
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
▼ This medicine is new or being used differently. Please report side effects. See the full CMI for further details.
1. Why am I using Icatibant Lupin?
Icatibant Lupin contains the active ingredient Icatibant acetate. Icatibant Lupin is used for treating the symptoms of an acute attack of hereditary angioedema (HAE) in adults, adolescents and children aged 2 years and older.
For more information, see Section 1. Why am I using Icatibant Lupin? in the full CMI.
2. What should I know before I use Icatibant Lupin?
Do not use if you have ever had an allergic reaction to icatibant acetate or any of the ingredients listed at the end of the CMI.
Do not give Icatibant Lupin to a child under 2 years of age or weighing less than 12 kg.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Icatibant Lupin? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Icatibant Lupin and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Icatibant Lupin?
- Your doctor will determine the exact dose of Icatibant Lupin and will tell you how often it should be used.
- The recommended dose of Icatibant Lupin for adults is one subcutaneous (under the skin) injection of 30 mg.
More instructions can be found in Section 4. How do I use Icatibant Lupin? in the full CMI.
5. What should I know while using Icatibant Lupin?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Icatibant Lupin? in the full CMI.
6. Are there any side effects?
Nausea, pain in the abdomen (tummy), weakness, dizziness, headache, blocked nose, rash, vomiting, fatigue, fever, sore throat, weight gain, asthma, cough, itching, redness of the skin, hot flushes, muscle spasm, hives, and abnormal liver function test. Symptoms may include yellowing of the skin and eyes (jaundice) are commonly seen side effects.
Almost all patients receiving Icatibant Lupin will experience a reaction at the site of the injection. The symptoms may include burning sensations, reddening of the skin, pain, swelling, feeling of warmth, and itching. These effects are usually mild and clear up by themselves without the need for any additional treatment.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
▼ This medicine is subject to additional monitoring. This will allow quick identification of new safety information. You can help by reporting any side effects you may get. You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems.
Icatibant Lupin
Active ingredient: icatibant acetate
Consumer Medicine Information (CMI)
This leaflet provides important information about using Icatibant Lupin. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Icatibant Lupin.
Where to find information in this leaflet:
1. Why am I using Icatibant Lupin?
2. What should I know before I use Icatibant Lupin?
3. What if I am taking other medicines?
4. How do I use Icatibant Lupin?
5. What should I know while using Icatibant Lupin?
6. Are there any side effects?
7. Product details
1. Why am I using Icatibant Lupin?
Icatibant Lupin contains the active ingredient icatibant acetate. Icatibant Lupin is used for treating the symptoms of an acute attack of hereditary angioedema (HAE) in adults, adolescents and children aged 2 years and older.
During attacks of HAE, levels of a substance in your bloodstream called bradykinin are increased and this leads to symptoms like swelling, pain, nausea, and diarrhoea.
Icatibant Lupin blocks the activity of bradykinin and therefore, helps reduce the symptoms of an HAE attack.
2. What should I know before I use Icatibant Lupin?
Warnings
Do not use Icatibant Lupin if:
- you are allergic to icatibant acetate, or any of the ingredients listed at the end of this leaflet.
Some of the symptoms of an allergic response may include:
- shortness of breath, wheezing or difficulty breathing;
- swelling of the face, lips, tongue or other parts of the body, skin rash, itching or hives.
It is important to be able to tell when you might be having an allergic reaction as the symptoms are very similar to those of an attack of HAE, so you should discuss this with your doctor.
Always check the ingredients to make sure you can use this medicine. - the expiry date printed on the pack has passed or if the packaging is torn or shows signs of tampering.
- there are any visible signs of deterioration (eg. if the solution is cloudy, if it has floating particles, or if the solution is not colourless).
Do not give Icatibant Lupin to a child under 2 years of age or weighing less than 12 kg.
If you are not sure whether you should use Icatibant Lupin, talk to your doctor.
Check with your doctor if you:
- have any other medical conditions such as:
- you are suffering from angina (reduced blood flow to the heart muscle);.
- you have recently suffered a stroke. - take any medicines for any other condition.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Icatibant Lupin may interact with medicines known as Angiotensin Converting Enzyme (ACE) inhibitors (eg. captopril, enalapril, ramipril, quinapril, lisinopril), which are used to lower your blood pressure or for any other reason.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Icatibant Lupin.
4. How do I use Icatibant Lupin?
Icatibant Lupin comes in a ready-to-use syringe. A needle is packed separately which you need to attach before use. Each syringe should only be used once. The medicine inside your Icatibant Lupin pre-filled syringe should be clear and colourless.
How much and how frequently to use Icatibant Lupin
- Follow the instructions provided and use Icatibant Lupin until your doctor tells you to stop.
- Your doctor will determine the exact dose of Icatibant Lupin and will tell you how often it should be used.
Adults:
- The recommended dose of Icatibant Lupin is one injection (30 mg in 3 mL) given subcutaneously (under the skin) as soon as you develop symptoms of an angioedema attack (eg. increased skin swelling, particularly affecting the face and neck, increasing tummy pain).
- If the HAE symptoms are still present, or return after initial relief, an additional injection of Icatibant Lupin (3 mL) may be given after 6 hours.
- If after a further 6 hours you still experience symptoms you may need a third injection of Icatibant Lupin (3 mL).
- You should have no more than 3 injections in a 24-hour period and no more than 8 injections in a month.
Children and adolescents aged 2 to 17 years:
- The recommended dose of Icatibant Lupin is one injection of 1 mL up to a maximum of 3 mL based on body weight given subcutaneously (under the skin) as soon as you develop symptoms of an angioedema attack.
- The dose will be determined by your doctor based on your child's body weight.
- The dose will increase over time as your child grows and the doctor will need to periodically review that the dose is appropriate.
The decision to initiate caregiver or self-administration of Icatibant Lupin should be made by a physician experienced in the diagnosis and treatment of HAE. Icatibant Lupin may be administered by a healthcare professional.
Please see step-by-step instructions for the dose to inject in children and adolescents.
If you are not sure which dose to inject, ask your doctor, pharmacist or nurse.
You must seek immediate medical help if your symptoms get worse or do not improve.
How to inject Icatibant Lupin
Icatibant Lupin is intended for subcutaneous injection (under the skin). Icatibant Lupin is injected into the fatty tissue under the skin in the abdomen (tummy).
Please see step-by-step instructions for injecting Icatibant Lupin at the end of this leaflet.
Adults:
- Icatibant Lupin injections are usually administered by healthcare professionals. If you wish to inject Icatibant Lupin yourself and the doctor agrees it is appropriate (eg. if you have a lot of HAE attacks or if you live far from a hospital or doctor), you will be trained on how to give yourself an injection.
- You or your caregiver must be trained on subcutaneous injection technique before you self-inject, or your caregiver injects you with Icatibant Lupin.
- Immediately after you self-inject Icatibant Lupin or your caregiver injects you with Icatibant Lupin while you are experiencing a laryngeal attack (obstruction of the upper airway), you must seek medical care in a medical institution.
The following information is for patients who have been trained to inject themselves:
- As HAE attacks can often be serious, it is best to contact your doctor or hospital when you experience an attack.
- If the HAE attack involves your face, lips, throat, or voice box, or if you have any difficulty breathing, you should always contact your doctor or hospital.
- If your HAE attack has not shown signs of improvement within 2 hours of the injection of Icatibant Lupin, or if the attack spreads to your face, lips, throat, or voice box, or you develop any difficulty breathing, you should contact your doctor or hospital immediately.
- It is possible to have further injections of Icatibant Lupin if you do not have relief from the HAE symptoms following the first injection; however, this should be done by your doctor or in hospital (see below) or following the advice of your doctor.
Children and adolescents aged 2 to 17 years:
- Icatibant Lupin injections are usually administered by healthcare professionals, or by the caregiver of the child.
- The caregiver must be trained on subcutaneous injection technique before administering Icatibant Lupin to the child.
- Immediately after administering Icatibant Lupin to the child while they are experiencing a laryngeal attack (obstruction of the upper airway), the child must seek medical care in a medical institution.
- As HAE attacks can often be serious, it is best to contact your doctor or hospital when the child experiences an attack.
- If the HAE attack involves the face, lips, throat, or voice box, or if the child has any difficulty breathing, contact the doctor or hospital.
- If the HAE attack has not shown signs of improvement within 2 hours of the injection of Icatibant Lupin, or if the attack spreads to the face, lips, throat, or voice box, or the child develops any difficulty breathing, contact the doctor or hospital immediately.
- No more than one injection was given to a child for each HAE attack in clinical studies.
If you use too much Icatibant Lupin
If you think that you have used too much Icatibant Lupin, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26); or - contact your doctor; or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
When high doses have been given, patients have experienced a drop in blood pressure.
5. What should I know while using Icatibant Lupin?
Things you should do
If you are about to be started on any new medicine, remind your doctor and pharmacist that you are using Icatibant Lupin.
Call your doctor straight away if you:
- notice that your symptoms of the attack get worse after you use Icatibant Lupin. Some of the side effects connected with Icatibant Lupin are similar to the symptoms of your disease.
- become pregnant while using this medicine.
Remind any doctor, dentist, or pharmacist you visit that you are using Icatibant Lupin.
Driving or using machines
Do not drive or operate any machinery if you feel tired or dizzy as a result of your HAE attack or after using Icatibant Lupin.
Be careful before you drive or use any machines or tools until you know how Icatibant Lupin affects you.
Looking after your medicine
Keep your medicine in the pack until it is time to use it. If you take the medicine out of the pack it may not keep well.
Keep the medicine in a cool dry place where the temperature stays below 25°C. Do not freeze it.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat, or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Very common side effects: Almost all patients receiving Icatibant Lupin will experience a reaction at the site of the injection. These effects are usually mild and clear up by themselves without the need for any additional treatment. The reaction may include:
Mostly, these are mild and short-lived. These are:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Icatibant Lupin contains
| Active ingredient (main ingredient) | Icatibant acetate |
| Other ingredients (inactive ingredients) | Sodium chloride Glacial acetic acid Sodium hydroxide Water for injections |
Do not take this medicine if you are allergic to any of these ingredients.
What Icatibant Lupin looks like
Icatibant Lupin is supplied as a sterile solution for injection in single use pre-filled syringes. The solution should be clear and colourless and free from visible particles (AUST R 364111).
Who distributes Icatibant Lupin
Generic Health Pty Ltd
Suite 2, Level 2
19-23 Prospect Street
Box Hill, VIC, 3128
Australia
 +61 3 9809 7900
+61 3 9809 7900
This leaflet was prepared in June 2022.
Step-by-step instructions for injecting Icatibant Lupin
1. General Information
- Clean the work area (surface) to be used before beginning the process.
- Wash your hands with soap and water.
- Remove the pre-filled syringe from the tray.
- Remove the cap from the end of the pre-filled syringe by unscrewing the cap.
- Put down the pre-filled syringe after unscrewing the cap.
2a. Preparing the syringe for children and adolescents (2-17 years) weighing 65 kg or less
For patients who have not previously received Icatibant Lupin, initial treatment should be given in a medical institution or under the guidance of a physician. The decision to initiate caregiver or self-administration of Icatibant Lupin should only be made by a physician experienced in the diagnosis and treatment of HAE.
Icatibant Lupin may be administered by a caregiver only after training in subcutaneous injection technique by a healthcare professional.
Important information for healthcare professionals and caregivers
Where the dose is less than 30 mg (3 mL), the following equipment is required to extract the appropriate dose (see below):
a) Icatibant Lupin pre-filled syringe (containing icatibant solution)
b) Adapter (connector)
c) 3 mL graduated syringe
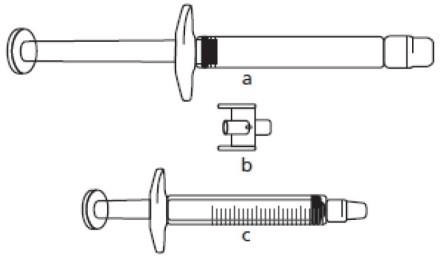
The required injection volume in mL should be drawn up in an empty 3 mL graduated syringe (see table below).
Table 1: Dosage regimen for children and adolescents
| Body Weight | Injection Volume |
| 12 kg to 25 kg | 1.0 mL |
| 26 kg to 40 kg | 1.5 mL |
| 41 kg to 50 kg | 2.0 mL |
| 51 kg to 65 kg | 2.5 mL |
Patients weighing more than 65 kg will use the full contents of the pre-filled syringe (3 mL).
If you are not sure which volume of solution to extract, ask your doctor, pharmacist or nurse.
Preparing to extract dose less than 3 mL:
- Remove the caps on each end of the adapter.
Avoid touching the ends of the adapter and syringe tips, to prevent contamination. - Screw the adapter onto the pre-filled syringe.
- Attach the graduated syringe to the other end of the adapter ensuring that both connections fit securely.
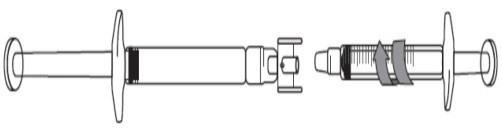
Transferring Icatibant Lupin solution to the graduated syringe:
- To start transfer of Icatibant Lupin solution, push the pre-filled syringe plunger (on far left of the below image).
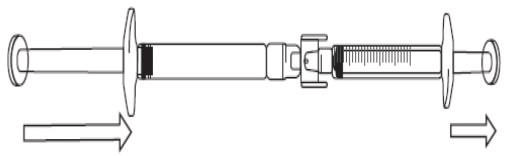
- If the Icatibant Lupin solution does not begin to transfer to the graduated syringe, pull slightly on the graduated syringe plunger until the Icatibant Lupin solution starts to flow into the graduated syringe (see below image).
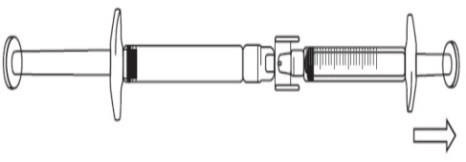
- Continue to push on the pre-filled syringe plunger until the required injection volume (dose) is transferred to the graduated syringe. See Table 1 for dosage information.
If there is air in the graduated syringe:
- Turn the connected syringes so that the pre-filled syringe is on top (see below image).
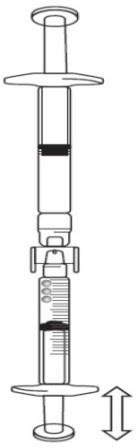
- Push the plunger of the graduated syringe so that any air is transferred back into the pre-filled syringe (this step may need to be repeated several times).
- Withdraw the required volume of Icatibant Lupin solution.
After transfer of Icatibant Lupin solution to the graduated syringe:
- Remove the pre-filled syringe and adapter from the graduated syringe.
- Discard the pre-filled syringe and adapter into the sharps container
2b. Preparing the syringe and needle for injection: All patients (adults, adolescents and children)
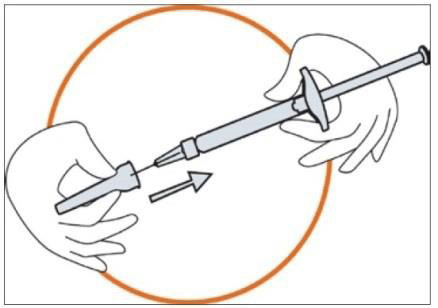
- Remove the needle cap from the blister tray.
- Remove the seal from the needle cap (the needle should be still in the needle cap).
- Grip the syringe firmly. Carefully attach the needle to the syringe containing the colourless solution.
- Screw the syringe on the needle still fixed in the needle cap.
- Remove the needle from the needle cap by pulling the syringe. Do not pull up on the plunger.
- The syringe is now ready for injection.
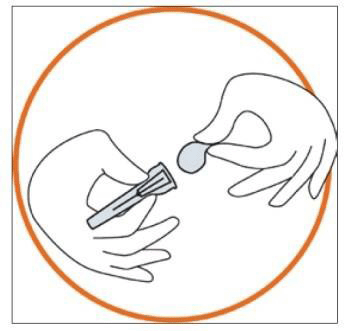
3. Preparing the injection site
- Choose the injection site. The injection site should be a skin fold on your abdomen (tummy), approximately 5 to 10 cm below your navel (belly button) on either side. This area should be at least 5 cm away from any scars. Do not choose an area that is bruised, swollen, or painful.
- Clean the injection site with an alcohol wipe and allow it to dry.
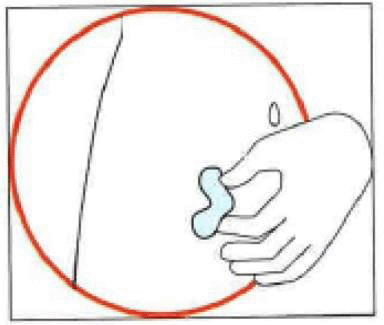
4. Injecting the solution
- Hold the syringe in one hand between two fingers with your thumb at the bottom of the plunger.
- Make sure that there is no air bubble in the syringe by pressing the plunger until the first drop appears on the tip of the needle.
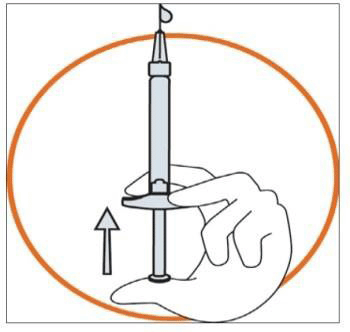
- Hold the syringe between a 45 to 90 degrees angle to your skin with the needle facing the skin.
- Keeping the syringe in one hand, use your other hand to gently hold a fold of skin between your thumb and fingers at the previously disinfected injection site.
- Hold the fold of the skin, bring the syringe to the skin and quickly insert the needle into the skin fold.
- Slowly push the plunger of the syringe with a steady hand until all the solution is injected into the skin and no liquid remains in the syringe.
- Press slowly so that this takes approximately 30 seconds.
- Release the skin fold and gently pull the needle out.
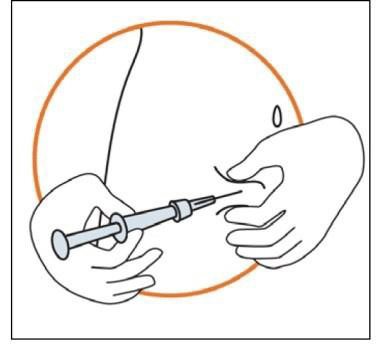
5. Disposal of the injection material
- Discard the syringe, needle and needle cap into the sharps container.
- Ask your pharmacist if you are not sure about the right way to throw away used syringes and needles.
Brand Information
| Brand name | Icatibant Lupin |
| Active ingredient | Icatibant |
| Schedule | S4 |
▼ This medicinal product is subject to additional monitoring in Australia. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse events at www.tga.gov.au/reporting-problems.
MIMS Revision Date: 01 May 2023
1 Name of Medicine
Icatibant acetate.
2 Qualitative and Quantitative Composition
Each pre-filled syringe of 3 mL contains icatibant acetate equivalent to 30 mg icatibant.
Each mL of the solution contains 10 mg of icatibant.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Solution for injection.
Icatibant Lupin is supplied as a sterile solution for injection in single use pre-filled syringes. The solution should be clear and colourless and free from visible particles. The pH of the injection is approximately 5.5.
4 Clinical Particulars
4.1 Therapeutic Indications
Icatibant Lupin is indicated for symptomatic treatment of acute attacks of hereditary angioedema (HAE) in adults, adolescents and children aged 2 years and older with C1-esterase-inhibitor deficiency.
4.2 Dose and Method of Administration
Dosage. Adults. The recommended dose of Icatibant Lupin for adults is one subcutaneous injection of 30 mg.
In the majority of cases a single injection of Icatibant Lupin is sufficient to treat an attack. In case of insufficient relief or recurrence of symptoms, a second injection of Icatibant Lupin can be administered after 6 hours. If the second injection produces insufficient relief or a recurrence of symptoms is observed, a third injection of Icatibant Lupin can be administered after a further 6 hours. No more than 3 injections of Icatibant Lupin should be administered in a 24-hour period.
In clinical trials, not more than 8 injections of Icatibant Lupin per month have been administered.
Adolescents and children (aged 2 to 17 years). The recommended dose of Icatibant Lupin based on body weight in children and adolescents (aged 2 to 17 years) is provided in Table 1. Studies in children aged less than 3.4 years or weighing less than 12 kg have not been performed.

Administration of more than one dose per HAE attack has not been studied (see Section 5.1 Pharmacodynamic Properties, Clinical trials).
No patient younger than 3.4 years was treated in study HGT-FIR-086. Dosages are based on population pharmacokinetic modelling (see Section 5.2 Pharmacokinetic Properties, Special populations).
Method of administration. Icatibant Lupin is intended for subcutaneous injection preferably in the abdominal area. Injection should be given slowly due to the volume to be administered (3 mL).
Adult patients may self-administer Icatibant Lupin upon recognition of symptoms of an HAE attack after training under the guidance of a healthcare professional. Patients who self-inject should be advised to seek urgent medical attention if there is no evidence of resolution of the HAE attack within 2 hours of self-injection, or immediately should the HAE attack progress to involve the face, lips or pharyngolaryngeal area. Patients whose initial HAE attack involves the face, lips or pharyngolaryngeal area should seek urgent medical attention, regardless of their response to Icatibant Lupin following self-injection.
For patients who have not previously received icatibant, initial treatment should be given in a medical institution or under the guidance of a physician. The decision to initiate caregiver or self-administration of icatibant should be made by a physician experienced in the diagnosis and treatment of HAE.
Icatibant Lupin may be administered to children and adolescents (2-17 years) by a healthcare professional or caregiver only after training in subcutaneous injection technique by a healthcare professional.
For instructions for use of the delivery system, refer to the consumer medicine information leaflet which can be found at www.ebs.tga.gov.au/ or www.generichealth.com.au/.
No dosage regimen can be recommended in children aged less than 2 years or weighing less than 12 kg, as the safety and efficacy in this paediatric group has not been established.
Patients with laryngeal symptoms should seek medical attention immediately after administration of Icatibant Lupin and need to be managed in an appropriate medical institution after injection until the physician considers discharge to be safe.
Icatibant Lupin contains no antimicrobial agent and should be used immediately. Each syringe is intended for single use in one patient only. Any residue should be discarded.
Hepatic impairment. No dosage adjustment is required in patients with hepatic impairment.
Renal impairment. No dosage adjustment is required in patients with renal impairment.
4.3 Contraindications
Hypersensitivity to the active substance or to any of the excipients.
4.4 Special Warnings and Precautions for Use
Ischaemic heart disease. Icatibant did not elicit any cardiac conduction change in vitro (hERG channel) or in vivo in normal dogs or in dogs undergoing physical exertion. Icatibant has been shown to aggravate induced cardiac ischaemia in several non-clinical models, including a study in dogs involving coronary ligation, probably as a result of left ventricular failure. Bradykinin and the B2 receptors have been shown to have cardioprotective properties in animals, which were attenuated by icatibant.
Under ischaemic conditions, a deterioration of cardiac function and a decrease in coronary blood flow could theoretically arise from antagonism of the B2 receptor.
Caution should therefore be observed in the administration of icatibant to patients with acute ischaemic heart disease or unstable angina pectoris.
Stroke. There is a theoretical possibility that icatibant may attenuate the positive late phase neuroprotective effects of bradykinin. Accordingly, caution should be observed in the administration of icatibant to patients in the weeks following a stroke.
Use in hepatic impairment. Data from subjects with a wide range of hepatic insufficiency suggest that icatibant exposure is not influenced by hepatic impairment. No dosage adjustment is required in patients with hepatic impairment.
Use in renal impairment. Limited data from subjects with renal insufficiency suggest that icatibant exposure is not influenced by renal impairment. No dosage adjustment is required in patients with renal impairment.
Use in the elderly. Limited information is available for icatibant in patients older than 65 years of age.
Elderly patients have been shown to have increased systemic exposure to icatibant. The relevance of this to the safety of icatibant is unknown (see Section 5.2 Pharmacokinetic Properties).
Paediatric use. Icatibant is not indicated for use in children less than 2 years or weighing less than 12 kg.
Studies in children aged less than 3.4 years or weighing less than 12 kg have not been performed. In children and adolescents, administration of more than one dose per HAE attack has not been studied (see Section 5.1 Pharmacodynamic Properties, Clinical trials). There is limited experience with treatment of more than one HAE attack with icatibant in the paediatric population.
In immature animals, repeated dosing of icatibant reversibly delayed sexual maturation in males and females (see Section 4.6 Fertility, Pregnancy and Lactation, Effects on fertility).
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Pharmacokinetic drug interactions involving CYP450 are not expected (see Section 5.2 Pharmacokinetic Properties).
Co-administration of icatibant with angiotensin-converting enzyme (ACE) inhibitors has not been studied. There is a theoretical risk that icatibant may antagonise the effects of ACE inhibitors. Patients with HAE should not be taking these drugs as they can induce and exacerbate HAE attacks.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. In a study of 39 healthy adult men and women (confined to the follicular phase of the menstrual cycle) treated with either placebo or 30 mg every 6 hours for 3 doses every 3 days for a total of 9 doses with GnRH-stimulation, no clinically significant changes were found between placebo and treatment groups for female and male reproductive hormones, the concentration of luteal phase progesterone and luteal function, menstrual cycle length in females, and sperm count, motility and morphology in males. The dosing regimen used for this study is very unlikely to be sustained in the clinical setting. However, due to the fairly small study size and confinement of women to the follicular phase of their menstrual cycles, it is unclear how fully these results can be generalized to the broader population.
Intermittent icatibant treatment (twice weekly) subcutaneously for 39 weeks in dogs did not elicit toxicity in the testes, prostate, ovary, uterus or mammary gland (30-fold the anticipated clinical exposure in patients administered 240 mg icatibant per month, based on monthly AUC).
Daily subcutaneous administration of icatibant in mature rats and dogs for 26 and 39 weeks, respectively, caused atrophy of the testes, prostate, and uterus, and masculinization of the mammary glands. In rats, atrophy of the testes and prostate, testes germinal epithelial degeneration, hypospermia, and decreased testosterone levels occurred at ≥ 19-fold the anticipated clinical exposure, based on monthly AUC, and atrophy of the uterus, mammary gland masculinization, and decreased luteinizing hormone levels occurred at ≥ 69-fold the anticipated clinical exposure, based on monthly AUC. In dogs, reduced sperm counts and atrophy of the uterus and ovaries occurred at 9-fold the anticipated clinical exposure, based on monthly AUC, and atrophy of the testes, prostate, uterus, ovaries and mammary glands, and decreased testosterone and follicle stimulating hormone levels occurred at 134-fold the anticipated clinical exposure, based on monthly AUC.
Daily subcutaneous administration of icatibant to juvenile rats caused atrophy of the testes and prostate at ≥ 10-fold the anticipated clinical exposure, based on monthly AUC, delayed male sexual maturation, decreased sperm counts and slight atrophy of the uterus at ≥ 9 mg/kg/day, and impaired male fertility at 109-fold the anticipated clinical exposure, based on monthly AUC. These effects were partly/fully reversible. Sexual maturation was also reversibly delayed in immature dogs and appeared to be secondary to changes in gonadotropin levels. Most of these effects were fully reversible over 4 weeks recovery.
Reproductive toxicity studies in adult male mice and rats with daily icatibant subcutaneous administration showed no effects on fertility at doses up to 53-fold the anticipated clinical exposure, based on monthly AUC.
Use in pregnancy. (Category C)
For icatibant, no clinical data on exposed pregnancies are available.
Bradykinin B2 receptors have been shown to be present in tissues of the female reproductive system in animals and humans, and are likely to be involved in implantation and parturition.
There was an increase in pre-implantation loss in female rats treated with 10 mg/kg/day and post-implantation loss in rabbits treated with 10 mg/kg/day icatibant subcutaneously (respectively 30- and 50-fold the anticipated clinical exposure in patients administered 240 mg icatibant per month, based on monthly AUC).
Icatibant and/or its metabolites crossed the placenta in rats. Icatibant was not teratogenic when administered by subcutaneous injection during embryonic and foetal development in rats or rabbits (up to 5-fold the anticipated clinical exposure, based on monthly AUC). In rats, icatibant was associated with delayed parturition, increased foetal distress and perinatal death at 10 mg/kg/day (30-fold the anticipated clinical exposure, based on monthly AUC) and a prolonged gestation period at doses 3-fold the anticipated clinical exposure, based on monthly AUC. There were no observed adverse effects of icatibant administration during pregnancy and lactation on pup development in rats.
Therefore, icatibant should be used during pregnancy only if the potential benefit justifies the potential risk for the foetus (e.g. for treatment of potentially life-threatening laryngeal attacks).
Use in lactation. Icatibant is excreted in the milk of lactating rats at concentrations similar to those in maternal blood. No adverse effects were detected in the post-natal development of rat pups.
It is unknown whether icatibant is excreted in human breast milk, but it is recommended that breastfeeding women who take icatibant should not breastfeed for 12 hours after treatment. If breastfeeding is to be resumed, then milk should be expressed and discarded for the first 12 hours after treatment.
4.7 Effects on Ability to Drive and Use Machines
No studies of icatibant on the effects on the ability to drive and use machines have been performed. Dizziness has been reported in patients using icatibant, therefore patients should be advised not to drive or use machines if they feel dizzy. Symptoms of an HAE attack (for example, somnolence, fatigue, lethargy and tiredness) may also influence the ability to drive or use machines. Patients who experience any of the above should be advised not to drive or use machines.
4.8 Adverse Effects (Undesirable Effects)
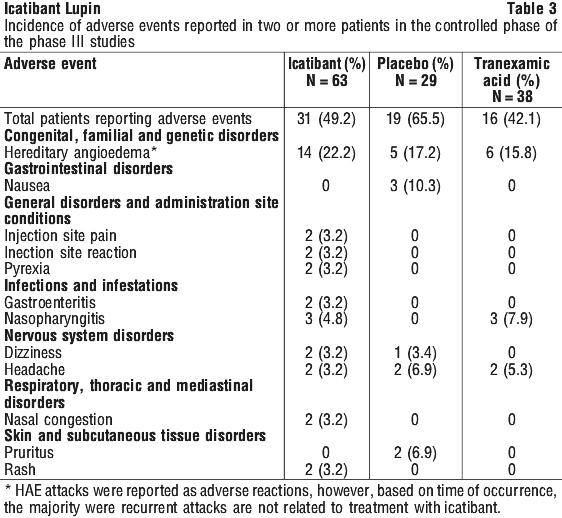
Clinical study experience. The safety of icatibant has been established in 1,273 subjects treated with various doses, regimens and routes of administration during Phase I-III studies in various indications.
Sixty-three HAE patients received icatibant in two Phase III trials for treatment of an attack in the controlled phase and 126 patients were treated in the open-label phase.
Almost all subjects who were treated with subcutaneous icatibant in clinical trials developed reactions at the site of injection including erythema, swelling, warm sensation, burning, itching and/or cutaneous pain. These reactions were generally mild in severity, transient, and resolved without further intervention.
Table 2 lists treatment related adverse reactions reported with icatibant during the Phase III trials. Frequency is defined as: very common (≥ 1/10), common (≥ 1/100, < 1/10) and uncommon (≥ 1/1,000 to < 1/100).
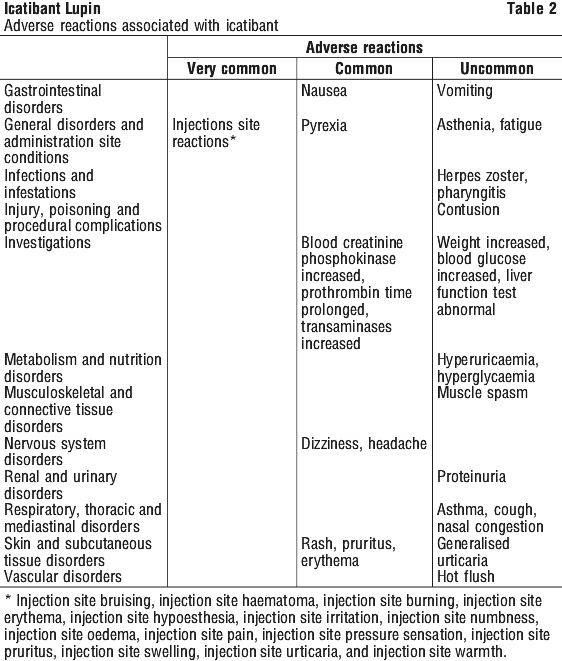
The majority of paediatric patients who were treated with subcutaneous icatibant experienced injection site reactions such as erythema, swelling, burning sensation, skin pain and itching/pruritus. These were found to be mild to moderate in severity and consistent with reactions that have been reported in adults. Two paediatric patients experienced injection site reactions which were assessed as severe and which were completely resolved within 6 hours. These reactions were erythema, swelling, burning and warm sensation.
Changes in some of the reproductive hormones were observed in study HGT-FIR-086. These changes are not expected to be clinically significant, based on limited clinical trial data of single administration of icatibant.
Post-marketing experience. See Table 4.

No anti-icatibant antibodies were detected in paediatric patients after treatment with icatibant in study HGT-FIR-086 (see Section 5.1 Pharmacodynamic Properties, Clinical trials).
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
No clinical information on overdose is available.
A dose of 3.2 mg/kg intravenously (approximately 8 times the therapeutic dose) caused transient erythema, itching, flushing, or hypotension in healthy subjects. No therapeutic intervention was necessary.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 in Australia.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: Drugs used to treat hereditary angioedema. ATC Code: B06AC02.
Mechanism of action. Hereditary angioedema (HAE), an autosomal dominant disease, is caused by an absence or dysfunction of C1-esterase-inhibitor. HAE attacks are accompanied by an increased release of bradykinin, which is the key mediator in the development of the clinical symptoms.
HAE manifests as intermittent attacks of subcutaneous and/or submucosal oedema involving the upper respiratory tract, the skin and the gastrointestinal tract. An attack usually lasts between 2 to 5 days.
Icatibant is a selective competitive antagonist at the bradykinin type 2 (B2) receptor. It is a synthetic decapeptide with a structure similar to bradykinin, but with 5 non-proteinogenic amino acids. Bradykinin has been shown to be elevated during hereditary angioedema attacks and is responsible for oedema formation and related clinical symptoms of swelling and pain.
Pharmacodynamic effects. In healthy young subjects, icatibant administered in doses of 0.8 mg/kg over 4 hours; 1.5 mg/kg/day or 0.15 mg/kg/day for 3 days, development of bradykinin-induced hypotension, vasodilatation and reflex tachycardia was prevented. Icatibant was shown to be a competitive antagonist when the bradykinin challenge dose was increased 4-fold.
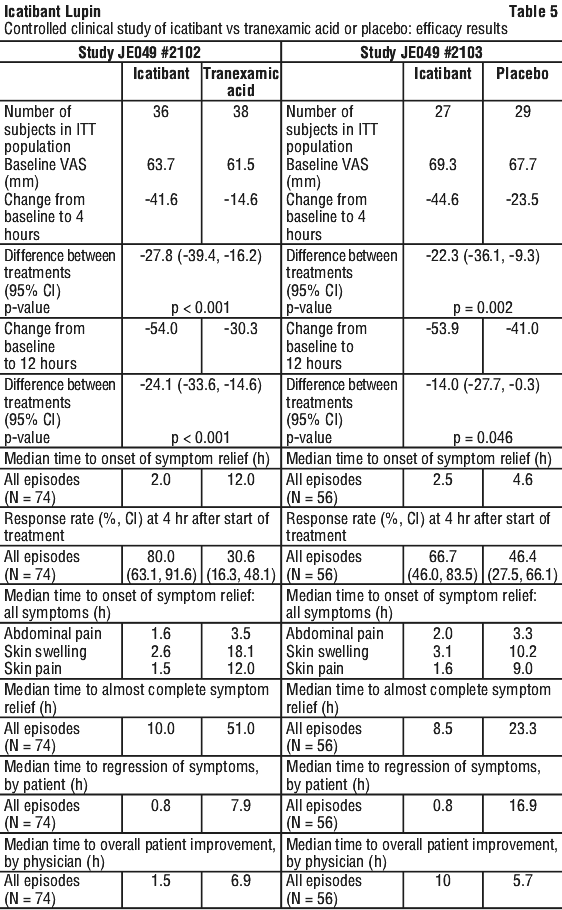
Clinical trials. Efficacy data were obtained from an initial open-label Phase II study and from two randomised, double-blind controlled multi-centre Phase III studies (one with oral tranexamic acid as the comparator and one placebo controlled). The pivotal Phase III studies were otherwise identical in design. A total of 130 patients were randomised to receive either a 30 mg dose of icatibant (63 patients) or comparator (either tranexamic acid - 38 patients, or placebo - 29 patients). Subsequent episodes of HAE were treated in an open-label extension (OLE). Patients with symptoms of laryngeal angioedema received open-label treatment with icatibant.
In the Phase III trials, the primary efficacy endpoint was median time to onset of symptom relief using a visual analogue scale (VAS) defined as absolute reduction from pre-treatment VAS of ≥ 20 mm if the baseline VAS was 30-50 mm or ≥ 30 mm if the baseline VAS was > 50 mm. The FAST-2 study (JE049 #2102) demonstrated that the median time to onset of symptom relief was significantly shorter in the icatibant group than in the tranexamic acid group (2.0 hours compared to 12.0 hours), while in the FAST-1 study (JE049 #2103) comparing icatibant with placebo, the median time to onset of symptom relief was shorter with icatibant than placebo (2.5 hours compared to 4.6 hours) but a statistically significant difference was not achieved.
Additional analyses were carried out with regard to changes from baseline to 4 hours and 12 hours in VAS scores. These direct evaluations of the VAS represent a more accurate clinical picture of the course of the HAE attack. The results show that for both studies, there was a substantial and consistent reduction in the score at 4 hours and 12 hours post-dose in the icatibant groups compared to the comparator groups, and the treatment differences in VAS changes from baseline to 4 hours and 12 hours were statistically significant (p = 0.002 and p = 0.046 for 4 hours and 12 hours in study JE049 #2103 and p < 0.001 for 4 hours and 12 hours in study JE049 #2102).
Table 5 shows the results for the two pivotal trials.
A total of 36 patients were treated for a total of 61 attacks of HAE affecting the larynx. The results were again similar to patients with non-laryngeal attacks of HAE with a median time to start of regression of symptoms of 0.6-1.0 hours (controlled phase).
Paediatric population. An open-label, non-randomised single-arm study (HGT-FIR-086) was performed with a total of 32 patients. All patients received at least one dose of icatibant (0.4 mg/kg body weight up to a maximum dose of 30 mg) and the majority of patients were followed up for a minimum of 6 months. Eleven patients were of pre-pubertal status and 21 patients were either pubertal or post-pubertal.
The efficacy population consisted of 22 patients who had been treated with icatibant (11 pre-pubertal and 11 pubertal/post-pubertal) for a first HAE attack.
The primary efficacy endpoint was the time to onset of symptom relief measured using a composite investigator-reported symptom score. Time to symptom relief was defined as the duration of time (in hours) taken for improvement of symptoms to occur by a magnitude of 20%. Table 6 shows the efficacy results for HGT-FIR-086.

Overall, the median time to minimal symptoms (earliest time post treatment when all symptoms were either mild or absent) was 1.1 hours (95% CI: 1.0-2.0 hours).
5.2 Pharmacokinetic Properties
The pharmacokinetics of icatibant has been extensively characterised by studies using both intravenous and subcutaneous administration to healthy volunteers and patients. The pharmacokinetic profile of icatibant in patients with HAE is similar to that in healthy volunteers.
Absorption. Following subcutaneous administration, the absolute bioavailability of icatibant is 97%. The time to maximum concentration (tmax) is approximately 0.5 hours.
Distribution. Icatibant volume of distribution (Vss) is about 20-25 L. Plasma protein binding is 44%.
Metabolism. Icatibant is extensively metabolised by proteolytic enzymes to inactive metabolites that are primarily excreted in the urine.
In vitro studies have confirmed that icatibant is not degraded by oxidative metabolic pathways and is not an inhibitor of major cytochrome P450 (CYP) isoenzymes (CYP 1A2, 2A6, 2B6, 2C8, 2C9, 2C19, 2D6, 2E1, and 3A4) and is not an inducer of CYP 1A2 and 3A4.
Excretion. Icatibant is mainly eliminated by metabolism with less than 10% of the dose eliminated in the urine as unchanged drug. Clearance is about 15-20 L/h and independent of dose. The terminal half-life (t1/2) is about 1-2 hours.
Special populations. Elderly. Data suggest an age-related decline in clearance resulting in about 50-60% higher exposure in older people (75-80 years) compared to patients aged 40 years.
Gender. Data suggest that there is no difference in the clearance between females and males after correcting for body weight.
Hepatic and renal impairment. Limited data suggest that icatibant exposure is not influenced by hepatic or renal impairment.
Race. Information on individual race effect is limited. Available exposure data suggest no difference in the clearance between non-White (n = 40) and White (n = 132) subjects.
Paediatric population. The pharmacokinetics of icatibant were characterised in paediatric HAE patients in study HGT-FIR-086 (see Section 5.1 Pharmacodynamic Properties). Following a single subcutaneous administration (0.4 mg/kg up to a maximum of 30 mg), the tmax is approximately 30 minutes and the t1/2 is about 2 hours. There are no observed differences in the exposure to icatibant between HAE patients with and without an attack. Population pharmacokinetic modelling using both adult and paediatric data showed that clearance of icatibant is related to body weight with lower clearance values noted for lower body weights in the paediatric HAE population. Based on modelling for weight banded dosing, the predicted exposure to icatibant in the paediatric HAE population (see Section 4.2 Dose and Method of Administration) is lower than the observed exposure in studies conducted with adult HAE patients.
5.3 Preclinical Safety Data
Genotoxicity. In a standard battery of in vitro and in vivo tests icatibant was not genotoxic.
Carcinogenicity. In a 2 year study to evaluate the carcinogenic potential of icatibant in rats, daily subcutaneous doses up to 6 mg/kg/day (11-fold the anticipated clinical exposure in patients administered 240 mg icatibant per month, based on monthly AUC) had no effect on the incidence or morphology of tumours. Results do not indicate a carcinogenic potential for icatibant.
6 Pharmaceutical Particulars
6.1 List of Excipients
Sodium chloride, glacial acetic acid, sodium hydroxide, water for injections.
6.2 Incompatibilities
Not applicable.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Do not freeze.
6.5 Nature and Contents of Container
Icatibant Lupin is supplied as 30 mg icatibant (as acetate) in 3 mL in one pre-filled syringe (Type-I clear glass) with plunger stopper (bromobutyl-coated with fluorocarbon polymer). A hypodermic needle (25 G; 16 mm) is included in the package.
Pack size is one pre-filled syringe with one needle.
The following equipment is required to extract and administer the appropriate dose when the prescribed dose is less than 30 mg (3 mL): adapter; 3 mL graduated syringe.
6.6 Special Precautions for Disposal
The pre-filled icatibant syringe and all other components are for single use only. All needles and syringes should be disposed of in a sharps container.
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Icatibant is isolated as the acetate salt, containing approximately 1-4 equivalents of acetic acid.
Chemical structure.
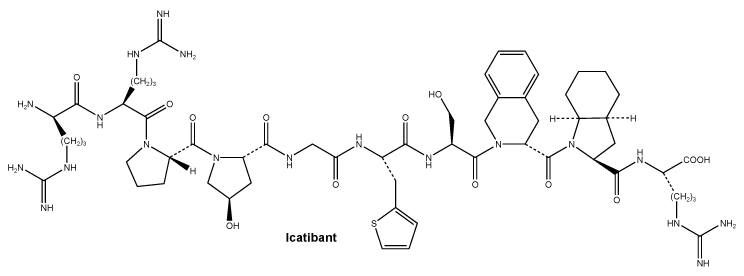
Molecular formula: C59H89N19O13S.
Molecular weight: 1304.55.
CAS number. 130308-48-4.
7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Date of First Approval
22 June 2022
Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.

