Ilumya
Brand Information
| Brand name | Ilumya |
| Active ingredient | Tildrakizumab |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Ilumya.
Summary CMI
Ilumya™
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Ilumya?
Ilumya contains the active ingredient tildrakizumab. Ilumya is used to treat plaque psoriasis in adults with moderate to severe plaque psoriasis who are candidates for systemic therapy.
For more information, see Section 1. Why am I using Ilumya? in the full CMI.
2. What should I know before I use Ilumya?
Do not use if you have ever had an allergic reaction to Ilumya or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Ilumya? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Ilumya and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Ilumya?
- Ilumya is injected under the skin (subcutaneous).
- Your doctor will decide how much Ilumya you need.
More instructions can be found in Section 4. How do I use Ilumya? in the full CMI.
5. What should I know while using Ilumya?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Ilumya? in the full CMI.
6. Are there any side effects?
Common side effects include diarrhoea, nausea, upper respiratory tract infections, injection site pain, fatigue, headache, joint stiffness, back or limb pain. Serious side effects include signs of an allergic reaction, such as shortness of breath, wheezing or difficulty breathing, swelling of the face, lips, tongue or other parts of the body, rash, itching or hives on the skin; signs of an infection, such as fever, lack of energy, chills and sweats, confusion or delirium, pain when passing urine or blood in the urine.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Ilumya™ (e-loom-e-a)
Active ingredient: tildrakizumab
Consumer Medicine Information (CMI)
This leaflet provides important information about using Ilumya. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Ilumya.
Where to find information in this leaflet:
1. Why am I using Ilumya?
2. What should I know before I use Ilumya?
3. What if I am taking other medicines?
4. How do I use Ilumya?
5. What should I know while using Ilumya?
6. Are there any side effects?
7. Product details
1. Why am I using Ilumya?
Ilumya contains the active ingredient tildrakizumab.
Tildrakizumab is a humanised monoclonal antibody and belongs to a group of medicines called interleukin (IL) inhibitors. This medicine works by neutralising the activity of a protein called IL-23, a substance found in the body which is involved in normal inflammatory and immune responses. In patients with plaque psoriasis, the body produces increased amounts of this protein. Plaque psoriasis causes inflammation of the skin. This may lead to symptoms such as itching, pain and scaling.
Ilumya is used to treat a skin condition called plaque psoriasis in adults with moderate to severe disease who are candidates for systemic therapy.
Ilumya reduces the inflammation and other symptoms of the disease, and promotes skin clearance.
2. What should I know before I use Ilumya?
Warnings
Do not use Ilumya if:
- you are allergic to tildrakizumab, or any of the ingredients listed at the end of this leaflet.
Always check the ingredients to make sure you can use this medicine. - you have an active infection which your doctor thinks is important.
- the product appears cloudy, discoloured or contains particles.
- the expiry date on the pack has passed or if packaging is torn or shows signs of tampering. Return expired or damaged stock to your pharmacist.
Check with your doctor if you:
- currently have an infection or if you have long-term or repeated infection
- have tuberculosis or have been in contact recently with someone who has
- have recently had or plan to have a vaccination
- have or have had any type of cancer
- have any other medical conditions
- take any medicines for any other condition
- are not sure whether you should start taking this medicine
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant, think that you may be pregnant or intend to become pregnant.
The effects of Ilumya in pregnancy are unknown. It is preferable to avoid the use of Ilumya in pregnancy. If you are a woman of childbearing potential, you are advised to avoid becoming pregnant and must use an effective method of contraception whilst having treatment with Ilumya and for at least 17 weeks after treatment.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Tell your doctor if you have had or are due to have a vaccination. You should not receive certain types of vaccines (live vaccines) while taking Ilumya.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Ilumya.
4. How do I use Ilumya?
How Ilumya is given
- Ilumya is given as an injection under the skin (subcutaneous injection).
- Ilumya may be given to you by your doctor or nurse, or you may be taught how to inject yourself with the medicine.
- You and your doctor, nurse or pharmacist should decide if you should inject Ilumya yourself.
- Do not inject yourself if you have not been properly trained by your doctor, nurse or pharmacist. A caregiver may also give you your Ilumya injection after proper training.
How much to use
- Your doctor will decide how much Ilumya you need.
The usual dose of Ilumya is 100 mg given by a single subcutaneous injection or 200 mg given by two subcutaneous injections. - After the first dose you will need to take the same dose 4 weeks later and then all future doses once every 12 weeks.
- If your doctor gives you different dosing instructions follow your doctor's instructions.
- Inject the whole content of the syringe.
- Do not shake the syringe.
- Do not exceed the recommended dose.
How to inject Ilumya yourself
Follow all directions given to you by your doctor, nurse or pharmacist carefully, if they differ from these instructions follow your healthcare professional's instructions.
If you do not understand the instructions, ask your doctor, nurse or pharmacist for help.
How to use the pre-filled syringe
Read all the instructions on how to use the pre-filled syringe before beginning the injection.

Keep the instructions and refer to them as needed. These instructions will help you to inject correctly using the pre-filled syringe.
Preparation
- One syringe is needed for a 100 mg dose and two syringes are needed for a 200 mg dose as prescribed by your doctor.
- For a more comfortable injection, take the carton containing the Ilumya pre-filled syringe out of the refrigerator and let it sit at room temperature for 30 minutes before injecting.

- Check the expiration date on the carton and pre-filled syringe and discard if the date has passed.
- Assemble other necessary supplies, not included in the Ilumya pack:
- an alcohol swab
- a cotton ball or gauze
- and a sharps container or similar puncture proof container of hard plastic or glass, for syringe disposal. - Remove the pre-filled syringe from the carton when ready to inject.
- DO NOT pull off the needle cover until you are ready to inject.
- Inspect Ilumya visually and discard if it contains particles, is cloudy or discoloured, or if the syringe is damaged.
- Ilumya is a clear to slightly opalescent (milky) and colourless to slightly yellow, solution.
- Air bubbles may be present; there is no need to remove them.
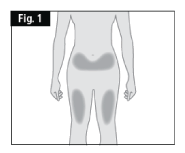
- Choose an injection site with clear skin and easy access such as front of thighs, or lower abdomen, but not the area five centimetres around the navel (belly button) (Fig. 1).
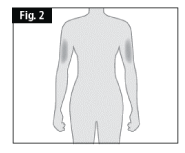
If a caregiver, or healthcare professional is giving you the injection, the outer upper arms may also be used (Fig. 2).
- DO NOT administer where the skin is tender, bruised, abnormally red, hard or affected by psoriasis.
- DO NOT inject into scars, stretch marks, or blood vessels.
Giving the Injection
Your Doctor, Pharmacist or Nurse will give you instructions on how to inject Ilumya subcutaneously and their instructions might differ from these instructions.
- When you are ready to inject, wash your hands thoroughly with soap and water.
- Find a clean, comfortable area to give the injection.
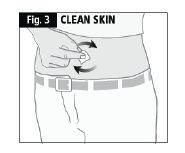
- Using a circular motion, clean the injection site with the alcohol swab. Leave to dry and do not touch the cleaned area again before injecting (Fig. 3).
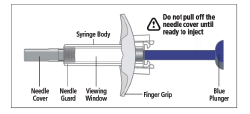
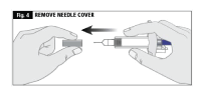
- While holding the body of the syringe, remove the needle cover as shown and discard (Fig. 4).
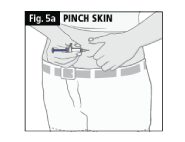
- To inject:
a. Gently press your skin between your thumb and index finger (Fig. 5a).
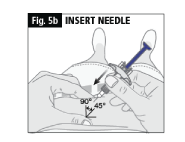
b. Insert the needle into the skin at a 45-90 degree angle (Fig. 5b).
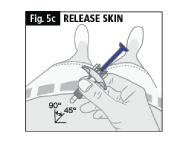
c. Release the skin before you push on the plunger (Fig. 5c).
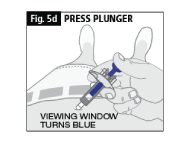
d. Slowly press down the blue plunger with your thumb as far as it will go (Fig. 5d). (This will activate the safety mechanism that ensures full retraction of the needle after the injection is given.)
The whole of the Dose Window will be blue when the injection is complete.
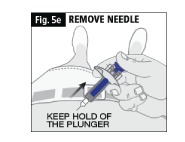
e. DO NOT LET GO OF THE PLUNGER until you have removed the needle from the skin (Fig. 5e).
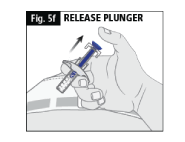
f. When you release the plunger the safety lock will draw the needle inside the needle guard (Fig. 5f).
g. There may be a small amount of blood at the injection site. You can press a cotton ball or gauze over the injection site and hold it for 10 seconds.
Do not rub the injection site.
You may cover the injection site with a small adhesive bandage, if needed.
- Dispose of the syringe in a sharps disposal container right away after use (Fig. 6).

Never try to reuse the pre-filled syringe.
- Repeat steps 1 to 6 with the second syringe in a different location of your skin if you are administering a dose of 200 mg.
How long to take Ilumya
- Ilumya is for long term treatment. Your doctor will regularly monitor your condition to check that the treatment is having the desired effect.
- Do not stop taking Ilumya just because you feel better.
It is important that you do not stop unless your doctor tells you to. - It is not dangerous to stop taking Ilumya but if you stop your psoriasis symptoms may come back.
If you forget to use Ilumya
Ilumya should be used regularly at the same time each day a dose is due. If you miss your dose at the usual time, inject the next dose as soon as you remember. Then talk to your doctor to discuss when you should inject the next dose.
Do not take a double dose to make up for the dose you missed.
If you use too much Ilumya
If you accidentally inject more Ilumya than you should or you take a dose sooner than according to the doctor's prescription, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using Ilumya?
Things you should do
- Keep all your doctor's appointments so that your progress can be checked.
- If you need to be vaccinated, tell your doctor you are taking Ilumya before you have the vaccination. You should not be given a live vaccination (e.g. oral polio vaccine) while being treated with Ilumya.
- Tell your doctor if you become pregnant while using Ilumya.
- Empty pre-filled syringes should be disposed of in a sharps container or similar puncture proof container composed of hard plastic or glass.
Call your doctor straight away or seek medical help immediately if you:
- notice any signs indicating a possible serious infection or an allergic reaction (see Serious Side effects).
Remind any doctor, dentist or pharmacist you visit that you are using Ilumya.
Things you should not do
- Do not stop using this medicine or change dosage without checking with your doctor first.
- Never leave the pre-filled syringe lying around where others might tamper with it.
- Do not use this medicine if the liquid contains easily visible particles, is cloudy or is distinctly brown.
- Do not use the pre-filled syringe if any of the outer packaging is damaged or the seals are broken. It may not be safe for you to use.
- Do not use the syringe if it is damaged or cap has been removed. Return damaged stock to your pharmacist.
Driving or using machines
Ilumya has no or little effect on the ability to drive and use machines.
Drinking alcohol
There is no information on the use of alcohol with Ilumya.
Looking after your medicine
Follow the instructions in the carton on how to take care of your medicine properly.
- Store Ilumya in a refrigerator (between 2°C and 8°C).
- Do not freeze.
- Do not shake.
- Keep the product in the original carton. Protect from light until the time of use.
- For purposes of travel or transporting between refrigerated storage, Ilumya can be stored for up to 30 days at or below 25°C.
- Once taken out of the refrigerator, do not refrigerate Ilumya again.
Keep it where young children cannot reach it.
When to discard your medicine
- Ilumya should be immediately used or discarded after 30 days out of the refrigerator or by the expiry date, whichever occurs first.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Gut-related:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Signs of an allergic reaction, such as:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Many medicines like Ilumya that may decrease the activity of your immune system, may increase the risk of cancer. Tell your doctor if you notice anything unusual or if you are concerned about any aspect of your health.
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Ilumya contains
| Active ingredient (main ingredient) | Each pre-filled syringe contains 100 mg tildrakizumab |
| Other ingredients (inactive ingredients) | Histidine, histidine hydrochloride monohydrate, polysorbate 80, sucrose and water for injections |
Do not take this medicine if you are allergic to any of these ingredients.
What Ilumya looks like
Ilumya is clear to slightly opalescent (milky). The colour may vary from colourless to slightly yellow solution. It is supplied in a carton pack containing 1 single use pre-filled syringe or 2 single use pre-filled syringes. (Aust R 290683).
Who distributes Ilumya
Sun Pharma ANZ Pty Ltd
ABN 17 110 871 826
Macquarie Park Sydney NSW 2113
Telephone 1 800 726 229
This leaflet was prepared in July 2025.
Brand Information
| Brand name | Ilumya |
| Active ingredient | Tildrakizumab |
| Schedule | S4 |
MIMS Revision Date: 01 September 2025
1 Name of Medicine
Tildrakizumab (rch).
2 Qualitative and Quantitative Composition
The single-use pre-filled syringe contains 100 mg/mL of tildrakizumab.
Tildrakizumab is a humanised IgG1/kappa monoclonal antibody produced by recombinant DNA technology in a Chinese hamster ovary (CHO) cells.
For a full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Solution for injection in a single-use pre-filled syringe.
The solution is clear to slightly opalescent and colourless to slightly yellow.
4 Clinical Particulars
4.1 Therapeutic Indications
Ilumya is indicated for the treatment of adults with moderate-to-severe plaque psoriasis who are candidates for systemic therapy.
4.2 Dose and Method of Administration
Ilumya is intended for use under the guidance and supervision of a physician experienced in the diagnosis and treatment of psoriasis. It is administered by subcutaneous injection.
Dosage. Adults. The recommended dose of Ilumya is 100 mg by subcutaneous injection at weeks 0, 4 and every 12 weeks thereafter.
At the physician's discretion, in patients with high disease burden or in patients above 90 kg of body weight who had unsatisfactory response at 100 mg, a dose of 200 mg may be considered.
If a dose is missed, administer the dose as soon as possible. Thereafter, resume dosing at the regularly scheduled interval.
Method of administration. Ilumya (1 mL) is administered by subcutaneous injection. Full instructions for use are provided in the Consumer Medicine Information.
After proper training in subcutaneous injection technique, patients may self-inject Ilumya if a healthcare professional determines that it is appropriate. However, the physician should ensure appropriate follow-up of patients.
Sites for injection include abdomen, thighs, or upper arm. Do not administer 5 cm around the navel or where the skin is tender, bruised, abnormally red, indurated or affected by psoriasis. Do not inject into scars, stretch marks, or blood vessels.
The pre-filled syringe should be visually inspected for particulate matter and discolouration prior to administration. Product exhibiting particulate matter or discolouration must not be used. Do not shake.
Ilumya does not contain any antimicrobial preservatives. Discard any unused product remaining in the pre-filled syringe. Ilumya pre-filled syringes are for single-use in one patient only.
When the 200 mg dose is prescribed by the healthcare professional, the patient will need to use 2 pre-filled syringes each time the product is administered.
Dosage adjustments. Renal or hepatic impairment. Ilumya has not been studied in these patient populations. No dose recommendations can be made.
Elderly (≥ 65 years). No dose adjustment is required (see Section 5.2 Pharmacokinetic Properties).
Paediatric population. The safety and efficacy of Ilumya in children and adolescents under 18 years of age has not yet been evaluated.
4.3 Contraindications
Hypersensitivity to the active substance or to any of the excipients (see Section 4.4 Special Warnings and Precautions for Use; Section 6.1 List of Excipients).
Clinically important active infection (e.g. active tuberculosis, see Section 4.4 Special Warnings and Precautions for Use).
4.4 Special Warnings and Precautions for Use
Infections. In clinical trials, there was no increased risk of infection in subjects treated with Ilumya, however patients with active infections or a history of recurrent infections were not included in clinical trials. Caution should be exercised when considering the use of Ilumya in patients with a chronic infection or a history of recurrent infection. If a patient develops a serious infection whilst on treatment with Ilumya, the patient should be closely monitored.
Ilumya must not be given to patients with active tuberculosis (TB).
Pre-treatment evaluation for tuberculosis. Prior to initiating treatment with Ilumya, patients should be evaluated for tuberculosis (TB) infection. Patients receiving Ilumya should be monitored for signs and symptoms of active TB during and after treatment. Anti-TB therapy should be considered prior to initiating Ilumya in patients with a past history of latent or active TB in whom an adequate course of treatment cannot be confirmed.
Hypersensitivity. Non-serious cases of urticaria occurred in Ilumya-treated subjects in clinical trials. If a serious hypersensitivity reaction occurs, administration of Ilumya should be discontinued immediately and appropriate therapy initiated.
Immunisations. Prior to initiating treatment with Ilumya, consider completion of all appropriate immunisations according to current immunisation guidelines. If a patient has received live viral or bacterial vaccination it is recommended to wait at least 4 weeks prior to starting treatment with tildrakizumab. Patients treated with Ilumya should not receive live vaccines during treatment and for at least 17 weeks after treatment (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Malignancy. In the initial placebo-controlled period of the Phase 2 and Phase 3 studies, a non-significant numerical imbalance between tildrakizumab-treated patients and those receiving placebo with malignancies was reported (see Section 4.8 Adverse Effects (Undesirable Effects)). As patients with prior or concurrent malignancy were excluded from the clinical trials, caution should be observed when treating patients with a history of malignancy or those who develop a malignancy during therapy.
Use in the elderly. See Section 5.2 Pharmacokinetic Properties.
Paediatric use. The safety and efficacy of Ilumya in children and adolescents under 18 years of age has not yet been evaluated.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Live vaccines should not be given concurrently with Ilumya (see Section 4.4 Special Warnings and Precautions for Use).
Results from a drug-drug interactions study conducted in psoriasis subjects suggest that Ilumya had no clinically relevant effect on cytochrome p450 (CYP) enzymes CYP1A2, CYP2C9, CYP2C19, CYP2D6 and CYP3A4 (see Section 5.2 Pharmacokinetic Properties, Drug interactions).
The safety of Ilumya in combination with other immunomodulatory agents or phototherapy has not been evaluated.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. The effect of Ilumya on human fertility has not been evaluated.
The effects of tildrakizumab were not directly assessed in dedicated animal fertility studies. However, no effects on fertility parameters such as reproductive organs, menstrual cycle length, and/or hormones were observed in male and female cynomolgus monkeys that were administered tildrakizumab at up to 100 mg/kg by subcutaneous injections once every 14 days (> 100 times the human exposure at the recommended dose based on AUC) for 9 months.
Use in pregnancy. (Category B1)
There is limited information regarding the use of Ilumya in pregnant women. As a precautionary measure, it is preferable to avoid the use of Ilumya during pregnancy. Women of childbearing potential should use an effective method of contraception during treatment and for at least 17 weeks after treatment.
Animal studies do not indicate direct or indirect harmful effect with respect to pregnancy, embryonic/fetal development, parturition or post-natal development. In a pre- and postnatal development toxicity study in monkeys given up to 100 mg/kg tildrakizumab by subcutaneous injections once every 14 days (> 100 times the human exposure at the recommended dose), no related increase in pregnancy loss was observed.
Tildrakizumab was shown to distribute across the placental barrier. After repeated dosing to pregnant cynomolgus monkeys, serum concentrations were quantifiable in the fetus however the reproduction toxicity studies did not reveal any untoward effects. No harmful effects were noted in neonates at maternal exposures up to 19 times the human exposure at the recommended dose. Two neonatal deaths from monkeys administered tildrakizumab at maternal exposure of > 100 times the human exposure at the recommended dose were attributed to possible viral infection and considered of uncertain relationship to the treatment. The clinical significance of these findings is unknown.
Use in lactation. It is not known whether tildrakizumab is excreted in human milk. In a pre-/postnatal development study tildrakizumab was detected at low levels in breast milk in monkeys dosed with tildrakizumab from gestation day 50 to parturition. The milk/serum ratio was ≤ 0.002. In the same study, serum levels of tildrakizumab in infants up to 91 days old were similar to, or exceed maternal serum levels.
A decision should be made whether to discontinue breast-feeding or to discontinue Ilumya taking into account the benefit of breast-feeding for the child and the benefit of therapy for the woman.
4.7 Effects on Ability to Drive and Use Machines
Ilumya has no or negligible influence on the ability to drive and use machines.
4.8 Adverse Effects (Undesirable Effects)
The most common adverse reactions observed with Ilumya in the pooled data from one Phase 2 and two Phase 3 studies in psoriasis patients for the placebo controlled period (16 weeks for the Phase 2 study and 12 weeks for the Phase 3 studies) were nasopharyngitis, headache, and site injection pain.
Most adverse reactions were considered mild and no adverse reaction led to discontinuation of treatment in > 1% of patients. For clinical management of specific adverse reactions, see Section 4.4 Special Warnings and Precautions for Use.
The frequency of adverse reactions is defined using the following convention: very common (≥ 1/10); common (≥ 1/100 to < 1/10); uncommon (≥ 1/1,000 to < 1/100); rare (≥ 1/10,000 to < 1/1,000); very rare (< 1/10,000); and not known (cannot be estimated from available data).
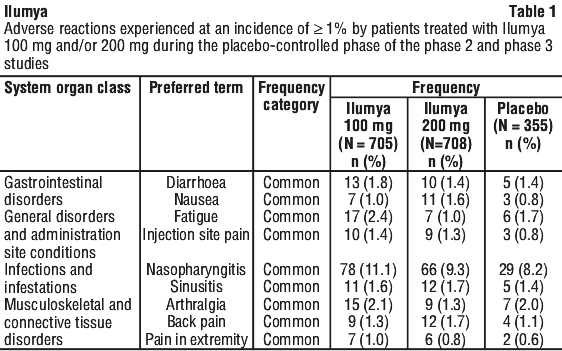
Table 1 provides a summary of the adverse reactions that were reported at a frequency of > 1% and at a higher rate in the Ilumya group than in the placebo group.
Immunogenicity. In the Phase 2 and Phase 3 psoriasis clinical studies 6.5% of patients treated continuously with Ilumya 100 mg developed antibodies to Ilumya. In the subset of these patients with treatment emergent neutralising antibodies, a small decrease in serum tildrakizumab concentrations and a corresponding increase in clearance was observed. Those patients who developed neutralising antibodies reported minor decreases in some efficacy parameters. No apparent association between the development of antibodies to Ilumya and the development of treatment emergent adverse events was seen.
Hypersensitivity reactions. Non-serious cases of urticaria occurred in Ilumya-treated subjects in clinical trials. If a serious hypersensitivity reaction occurs, administration of Ilumya should be discontinued immediately and appropriate therapy initiated. See Section 4.4 Special Warnings and Precautions for Use, Hypersensitivity.
Malignancy. In the initial placebo-controlled period of the Phase 2 and Phase 3 studies, a non-significant numerical imbalance in malignancy was reported between tildrakizumab-treated patients (0.2%) and those receiving placebo (0.0%). This imbalance was not observed in the controlled-data period (exposure-adjusted incidence rate of 1.2/100 patient years of exposure for tildrakizumab 100 mg, 1.7/100 patient years 200 mg, 2.6/100 patient years with etanercept, 0.9/100 patient years with placebo) or in the data collected in the open label extensions to the pivotal studies following up to 5 years of treatment (exposure adjusted rates of 0.7/100 patient with tildrakizumab 100 mg and 0.8/100 patient years with tildrakizumab 200 mg). The overall rate of malignancies reported with tildrakizumab across all studies was 0.422/100 patient years exposure (excluding non-melanoma skin cancer (NMSC)) and 0.348/100 patient years for non-melanoma skin cancer.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Intravenous doses up to 10 mg/kg have been safely administered in clinical trials.
In the event of overdose, it is recommended that the patient be monitored for any signs or symptoms of adverse reactions and appropriate symptomatic treatment be instituted immediately.
For information on the management of overdose, contact the Poison Information Centre on 131 126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
The expression of mRNA for IL-23p19 is elevated in psoriatic lesions compared to normal skin. In exploratory studies in patients with psoriasis, inflammatory infiltrates were decreased in lesional tissue biopsies after dosing with tildrakizumab. A decrease in expression of IL-23p19 was observed in lesional skin biopsies measured at baseline and up to two weeks post-treatment. Gene expression patterns of inflammation were normalised following treatment with tildrakizumab.
Mechanism of action. Tildrakizumab is a humanized IgG1/kappa monoclonal antibody that specifically binds to the p19 protein subunit of the interleukin 23 (IL-23) cytokine and inhibits its interaction with the IL-23 receptor.
IL-23 is a naturally occurring cytokine composed of 2 subunits (IL-23p19 and IL-12/23p40), that is involved in inflammatory and immune responses. Tildrakizumab inhibits the release of proinflammatory cytokines and chemokines. In in vitro models, tildrakizumab was shown to disrupt IL-23 mediated signalling and cytokine cascades by disrupting the interaction of IL-23 binding to its specific receptor, IL-23R without binding to IL-12 (composed of 2 subunits IL-12p35 and IL-12/23p40).
Clinical trials. The multicenter, randomized, double-blind, placebo-controlled trials reSURFACE 1 and reSURFACE 2 enrolled a total of 1862 patients 18 years of age and older with plaque psoriasis who had a minimum body surface area involvement of 10%, a Physician Global Assessment (PGA) score of ≥ 3 in the overall assessment (plaque thickness, erythema, and scaling) of psoriasis on a severity scale of 0 to 5, a Psoriasis Area and Severity Index (PASI) score ≥ 12, and who were candidates for phototherapy or systemic therapy.
In these studies, there was an initial 12 week placebo controlled period where patients were randomized to Ilumya (including 100 mg and 200 mg at 0, 4 and every twelve weeks thereafter [Q12W]) or placebo. In the active comparator study (reSURFACE 2), patients were also randomized to receive etanercept 50 mg twice weekly for 12 weeks, and weekly thereafter up to 28 weeks. After the initial 12 week treatment period, patients previously receiving placebo were randomized to either 100 mg or 200 mg Ilumya through week 28. After week 28, those patients receiving Ilumya from week 0 were eligible for dosage adjustment based on PASI response. In reSURFACE 2, non-responding or partial-responding patients receiving etanercept were switched to Ilumya 200 mg. The total treatment period was 52 or 64 weeks for the base studies, each study also incorporated an optional long-term extension phase.
All eligible subjects who completed Week 64 of reSURFACE 1 or Week 52 of reSURFACE 2, achieved at least 50% improvement in PASI from baseline and met all entry criteria were allowed to participate in a long term extension period to evaluate the long term safety and maintenance treatment of the effect of Ilumya in subjects with moderate-to-severe plaque psoriasis. Eligible patients received the same treatment they received at the completion of Week 64 or Week 52 of the respective studies, either Ilumya 100 mg or 200 mg, every 12 Weeks during the long-term extension for a minimum of 212 weeks.
Results obtained at Weeks 12, 28 and beyond (up to Week 64 in reSURFACE 1 and up to Week 52 in reSURFACE 2) are presented in Table 2.
Patients in all treatment groups (reSURFACE 1 and reSURFACE 2) had a median baseline PASI score ranging from 17.6 to 18.4. The baseline PGA score was marked or severe in 33.4% of patients. Of all patients enrolled, 35.8% had received prior phototherapy, 41.1% had received prior conventional systemic therapy, 16.7% had received prior biologic therapy for the treatment of psoriasis, and 7.7% had received at least one anti-TNF alpha agent. A total of 15.4% of study patients had a history of psoriatic arthritis. Baseline Dermatology Life Quality Index (DLQI) ranged from 13.2 to 14.8.
Studies reSURFACE 1 and reSURFACE 2 assessed the changes from baseline at Week 12 in the two co-primary endpoints: 1) PASI 75 and 2) PGA of "0" (cleared) or "1" (minimal), with at least a 2-point improvement from baseline. Other evaluated outcomes in reSURFACE 1 and reSURFACE 2 included the proportion of patients who achieved PASI 90 and PASI 100, the proportion of patients with DLQI 0 or 1, and maintenance of efficacy up to 64 weeks.
Examination of age, race, previous treatment with a biologic or traditional systemic therapy did not identify significant differences in PASI 75 response to Ilumya 100 mg among these subgroups at Week 12.
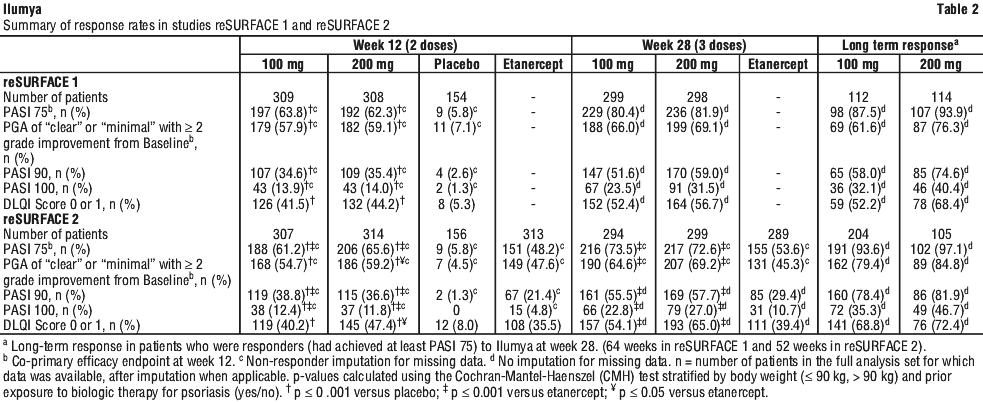
Of the patients continuing with the same dose of Ilumya, 87.5% of patients treated with Ilumya 100 mg, and 93.9% of patients treated with Ilumya 200 mg, maintained PASI 75 response at week 64. Of the patients re-randomized to receive placebo who did not relapse on placebo, 49% of patients originally treated with Ilumya 100 mg, and 56.7% of patients originally treated with Ilumya 200 mg, maintained PASI 75 response at Week 64.
Retreatment after relapse. In patients re-randomized to placebo in reSURFACE 1, 54.4% (who were originally treated with 100 mg) and 48.7% (who were originally treated with 200 mg) experienced relapse (defined as a reduction in maximum PASI response by 50%). These patients were restarted on their original dose of Ilumya upon relapse. After a minimum of 12 weeks from re-initiation of therapy, 85.7% (100 mg) and 83.3% (200 mg) had regained a PASI 75 response at Week 64.
Quality of life/patient-reported outcomes. At Week 12 and across studies, Ilumya was associated with statistically significant improvement in Health-related Quality of Life as assessed by the Dermatology Life Quality Index (DLQI). The proportion of patients treated with Ilumya who achieved DLQI 0 or 1 at W12 was 41% and 40%, in reSURFACE 1 and 2 respectively. Improvements were maintained over a year with DLQI 0/1 achieved by 63.7% (100 mg) and 73.3% (200 mg) in reSURFACE 1, and 68.8% (100 mg) and 72.4% (200 mg) in reSURFACE 2 respectively. At Week 52, 82% (100 mg) and 89% (200 mg) of patients in reSURFACE 1 and 87% (100 mg) and 91% (200 mg) of patients in reSURFACE 2, had a DLQI score < 5 points. Different subgroups of subjects contributed to the results at Week 12 and Week 52.
In reSURFACE 1, the physical and mental component summary scores of the Short Form Health Survey (SF-36) were significantly improved in patients treated with Ilumya compared with placebo.
Treatment maintenance in partial responders. 68.4% patients (reSURFACE 2) originally randomized to Ilumya 100 mg who achieved a PASI response of ≥ 50% but < 75% improvement from Baseline at Week 28 (3 doses) and remained on Ilumya 100 mg (n ~ 20), achieved a PASI 75 response at Week 52 (i.e. 3 additional doses).
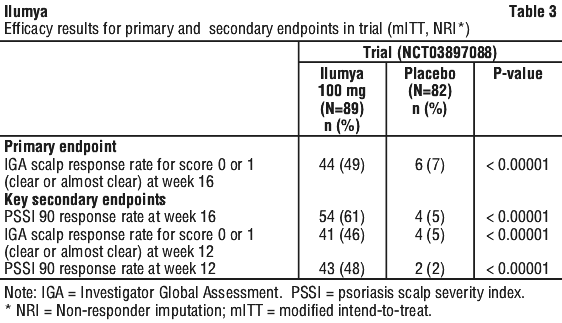
Psoriasis of the scalp. In a multicenter, randomized, double-blind, placebo-controlled trial (Trial [NCT03897088]) 231 subjects with moderate to severe psoriasis of the scalp were treated with Ilumya 100 mg (n=117) or placebo (n=114) at Week 0 and 4 and every 12 weeks thereafter. Of the 231 randomized subjects, 217 subjects completed Part 1 (Day 1 to Week 16). For Week 16 to Week 52, subjects randomized to placebo were switched to Ilumya receiving 100 mg at Weeks 16, 20, 32, and 44. Those in the Tildrakizumab 100 mg arm continued to receive Tildrakizumab 100 mg at Weeks 16, 28, 40, and 52. After Week 52, the study treatments were stopped, and all subjects entered the 20-week observational treatment-free safety follow-up period to monitor safety and tolerability following the last dose of treatment.
The primary endpoint was the proportion of subjects with IGA score for the scalp of "clear" and "almost clear" with at least 2-point reduction from Baseline at Week 16.
Other evaluated outcomes included the proportion of subjects achieving a) Psoriasis Scalp Severity Index (PSSI) 90 (≥ 90% improvement from Baseline in PSSI) at Week 16; b) subjects achieving PSSI 90 at Week 12; and c) IGA for the scalp score of "clear" or "almost clear" with at a least a 2-point reduction from Baseline at Week 12.
Subjects in the Ilumya and placebo treatment groups were predominantly men (60.2%) and White (78.9), with a mean age of 44.8 years. At baseline, these subjects had a median affected scalp surface area (SSA) of 50%, a median PASI score of 16.7, and PGA score of 3 ("moderate") or 4 ("severe") in 87.1% and 12.3%, respectively.
Clinical response in trial. The results from trial are presented in Table 3.
5.2 Pharmacokinetic Properties
Absorption. The subcutaneous formulation of tildrakizumab was characterized with an absolute bioavailability of 73-90% and slow absorption with time to reach maximum concentration at 6.2 days after injection. Steady-state was achieved by 16 weeks with the clinical regimen of 0, 4, and every 12 weeks thereafter, with 1.1-fold accumulation in exposure between Week-1 and Week-12 independent of dose.
At steady state, following administration of 100 mg of tildrakizumab in subjects with moderate to severe psoriasis geometric means (% CV) of AUC0-T and Cmax values were 305 microgram*day/mL (41%) and 8.1 microgram/mL (34%), respectively.
Tildrakizumab pharmacokinetics exhibited low to moderate variability (33-41%).
Distribution. Tildrakizumab has limited extravascular distribution with geometric mean (%CV) apparent volume of distribution (Vd/F) of 10.8 L (24%).
Metabolism. Tildrakizumab is catabolized into component amino acids by general protein degradation processes.
Small-molecule metabolic pathways (e.g. cytochrome P450 enzymes (CYPs), glucuronosyltransferases) do not contribute to its clearance.
Excretion. The geometric mean apparent clearance (CV %) was 0.32 L/day (38%) and the elimination half-life was 23.4 days (23%) in subjects with plaque psoriasis.
Dose linearity. Tildrakizumab exhibited dose-proportional pharmacokinetics in subjects with plaque psoriasis over a dose range from 50 mg to 200 mg, and in healthy subjects at doses from 50 mg to 400 mg following subcutaneous administration, with clearance being independent of dose.
Special populations. Elderly. Population pharmacokinetic analysis indicated that age did not have a clinically significant influence on the clearance of tildrakizumab in adult subjects with plaque psoriasis. Subjects who are 65 years or older had a similar tildrakizumab clearance as compared to subjects less than 65 years old.
Renal and hepatic impairment. No formal trial on the effect of hepatic or renal impairment on the pharmacokinetics of tildrakizumab has been conducted.
Body weight. Population pharmacokinetic modelling indicated that exposure decreased as body weight increased. The geometric mean exposure (AUCss - area under the concentration versus time curve at steady state) in adult patients weighing > 90 kg following a 100 mg or 200 mg subcutaneous dose was predicted to be about 30% lower than in an adult patient weighing ≤ 90 kg (see Section 4.2).
Drug interactions. A clinical pharmacology study, administering 200 mg of tildrakizumab subcutaneously on Day 1 and Day 29 demonstrated that tildrakizumab does not have a clinically relevant effect on the pharmacokinetics of caffeine (CYP1A2 probe substrate), warfarin (CYP2C9 probe substrate), omeprazole (2C19 probe substrate), dextromethorphan (CYP2D6 probe substrate) or midazolam (CYP3A4 probe substrate). (See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.)
5.3 Preclinical Safety Data
Genotoxicity. Ilumya has not been evaluated for genotoxic potential.
Carcinogenicity. Animal carcinogenicity studies have not been conducted with tildrakizumab. Studies in mouse tumour models showed that selective inhibition of IL-23p19 does not increase carcinogenic risk.
6 Pharmaceutical Particulars
6.1 List of Excipients
Histidine, histidine hydrochloride monohydrate, polysorbate 80, sucrose and water for injections.
6.2 Incompatibilities
Not known.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store at 2°C to 8°C. Refrigerate. Do not freeze.
Ilumya is stable for up to 30 days at 25°C.
Protect from light. Keep the pre-filled syringe in the outer carton until ready to use. Do not shake.
6.5 Nature and Contents of Container
Ilumya solution for injection is supplied as a single-use, pre-filled syringe comprising a clear glass barrel with a stainless-steel needle, latex-free rubber plunger stopper and needle shield. Each pre-filled syringe is assembled with a needle safety device for subcutaneous (sc) administration.
Pack size of 1 or 2 pre-filled syringes.
6.6 Special Precautions for Disposal
No special requirements for disposal.
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
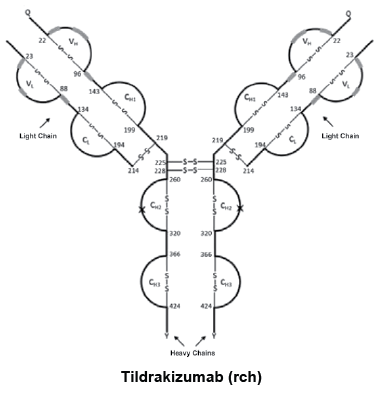
Ilumya (tildrakizumab) is a humanised IgG1/kappa monoclonal antibody produced by recombinant DNA technology in a Chinese hamster ovary (CHO) cells. It is composed of two identical heavy chains of 446 amino acids each and two identical light chains of 214 amino acids each linked by interchain disulfide bonds, with an approximate molecular weight of 147.0 kDa.
Chemical structure.
7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Date of First Approval
10 September 2018
Date of Revision
22 July 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.