IMODIUM
Brand Information
| Brand name | IMODIUM |
| Active ingredient | Loperamide hydrochloride |
| Schedule | Unscheduled | S2 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the IMODIUM.
What is in this leaflet
This leaflet answers some common questions about IMODIUM. It does not contain all the available information. It does not take the place of talking to your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of you taking IMODIUM against the benefits this medicine is expected to have for you.
If you have any concerns about using IMODIUM ask your doctor or pharmacist.
Keep this leaflet with your medicine. You may need to read it again.
What IMODIUM is used for
IMODIUM is used to treat sudden (acute) diarrhoea or long-lasting (chronic) diarrhoea.
IMODIUM is also used in patients who have had part of their intestines removed by surgery (intestinal resection).
IMODIUM makes the stools more solid and less frequent. It does this by normalising the muscle contractions of the intestine. This allows the bowel time to absorb water and produce a more solid stool.
Your doctor may have prescribed IMODIUM for another reason.
Ask your doctor if you have any questions about why IMODIUM has been prescribed for you.
Before you take IMODIUM
When you must not take it:
Do not take IMODIUM if:
- you have an allergy to IMODIUM or any of the ingredients listed at the end of this leaflet (see Product Description).
- you are constipated, or have to avoid becoming constipated
- you have severe diarrhoea with blood in the stools or fever
- you have inflammation of the lower bowels (for example, ulcerative colitis or pseudomembranous colitis as a result of using antibiotics)
- you are breast-feeding
- the packaging is torn or shows signs of tampering
Do not use IMODIUM after the expiry date (month and year) printed on the pack. If you take IMODIUM after the expiry date it may not work.
Do not give IMODIUM to children under the age of 12 years.
Before you start to take it:
You must tell your doctor or pharmacist if you:
- are pregnant or planning to become pregnant
- are breastfeeding
- have or have ever had liver disease
- have kidney disease
- have AIDS (Acquired Immune Deficiency Syndrome)
- have glaucoma (high pressure in the eye)
- have bladder problems
- regularly drink a lot of alcohol.
Taking other medicines:
Tell your doctor or pharmacist if you are taking any other medicines, including medicines you can buy without a prescription from a pharmacy, supermarket or health food shop.
In particular, tell your doctor or pharmacist if you are taking any of the following:
- sleeping tablets or medicines to treat anxiety and nervousness
- medicines to treat depression
- medicines that slow down the action of the stomach and intestines.
Taking IMODIUM
How to take:
IMODIUM capsule and caplet should be swallowed with the aid of liquid.
IMODIUM Melts should be placed on the tongue, allowed to dissolve and swallowed with saliva. No liquid intake in necessary.
How much to take:
Adults:
Sudden (acute) diarrhoea
The usual adult dose is two IMODIUM capsules or caplets or Melts initially, followed by one capsule or caplet or Melt after each loose unformed stool as needed.
Do not take more than eight capsules or caplets in one day.
Your doctor may prescribe a different dose for you.
IMODIUM will usually stop the symptoms of diarrhoea within 48 hours. If it does not, stop taking the medicine and see your doctor.
Chronic diarrhoea and intestinal resection
The usual adult dose is two IMODIUM capsules or caplets or Melts initially, followed by one capsule or caplet after each loose unformed stool until the diarrhoea is under control. Your doctor will then adjust the dose of IMODIUM to suit you.
Children:
IMODIUM should not be given to children under 12 years of age.
If you forget to take it:
If you forget to take IMODIUM after an unformed stool, take it when you remember or after your next unformed stool.
Do not take a double dose to make up for the dose you missed.
If you are not sure what to do, check with your doctor or pharmacist.
If you have taken too much (overdose):
Immediately telephone your doctor or the Poisons Information Centre for advice, or go to Accident and Emergency at your nearest hospital.
Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
Poisons Information Centre telephone numbers:
- Australia: 13 11 26
- New Zealand: 0800 POISON or 0800 764 766
Keep these telephone numbers handy in case of an emergency.
If you take too much IMODIUM you may have any of the following symptoms: sleepiness, nausea and vomiting, constipation, difficulty passing urine, weak breathing, muscle stiffness and uncoordinated movements.
Children are more sensitive than adults to medicines such as IMODIUM. An accidental overdose is especially dangerous. Make sure IMODIUM is kept out of reach of children.
While you are using IMODIUM
Things you must do:
- If you do not improve after taking IMODIUM for 48 hours tell your doctor or pharmacist
- Drink plenty of fluids, to avoid dehydration
- Tell your doctor if you become pregnant while taking IMODIUM
- Tell your doctor or pharmacist that you are taking IMODIUM if you are about to start taking a new medicine.
Things you must not do:
- Do not take more than 8 capsules or caplets in one day
- Do not use IMODIUM to treat any other complaint unless your doctor says to
- Do not give this medicine to anyone else, even if they have the same symptoms as you.
Things to be careful of:
Effects on driving and operating machinery
Tiredness, dizziness or drowsiness can be seen with diarrhoea. Therefore, it is advisable to use caution when driving or operating machinery.
Side Effects
All medicines can have side effects. Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some side effects.
Do not be alarmed by this list of possible side effects. You may not experience any of them.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor if you experience any of the following and they worry you:
- nausea or vomiting
- constipation
- dry mouth or bad taste
- headache
- stomach pain
- flatulence (wind)
- difficulty passing urine
- tiredness
- dizziness or drowsiness.
These side effects are usually mild. The above list does not represent all side effects that have been reported for IMODIUM. If you notice any side effects not mentioned in this leaflet, please inform your pharmacist or doctor.
Tell your doctor immediately and do not take your next dose of IMODIUM if you experience:
- asthma, wheezing or shortness of breath
- swelling of the face, lips or tongue which may cause difficulty in swallowing or breathing
- sudden or severe blisters, itching, skin rash, hives
- flaking or peeling of skin
- loss of or depressed level of consciousness.
These are serious side effects. You may need urgent medical attention. Serious side effects are rare.
Other side effects not listed above may also occur in some people.
Tell your doctor if you notice anything making you feel unwell when you are taking, or soon after you have finished taking IMODIUM.
Ask your pharmacist or doctor if you do not understand anything in this list.
After using IMODIUM
Storage
Keep your IMODIUM Capsules and Caplets in a cool dry place where the temperature stays below 30 degrees C.
Keep your IMODIUM Melts in a cool dry place where the temperature stays below 25 degrees C.
Do not store IMODIUM, or any other medicine, in the bathroom or near a sink. Do not leave medicines in the car or on window sills. Heat and dampness can destroy some medicines.
Keep your medicines where children cannot reach them. A locked cupboard at least one-and-a-half metres (1.5 m) above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop taking IMODIUM or they have passed their expiry date, ask your pharmacist what to do with any that are left over.
Product Description
What IMODIUM looks like:
IMODIUM capsules are small grey/green capsules.
Each pack contains 12 capsules.
IMODIUM caplets are light green, capsule-shaped tablet, marked with IMODIUM A-D on one side, scored on the other side and marked with 2 mg.
Each pack contains 8 or 20 caplets.
IMODIUM Melts are white to off-white circular tablets.
Each pack contains 10 tablets.
Ingredients
Each IMODIUM Capsule contains 2 mg of loperamide hydrochloride as the active ingredient.
The capsules also contain lactose, maize starch, purified talc and magnesium stearate, iron oxide yellow (E172), indigo carmine (E132), erythrosine (E127), titanium dioxide (E171) and gelatin.
The capsules do not contain gluten.
Each IMODIUM Caplet contains 2 mg of loperamide hydrochloride as the active ingredient.
The caplets also contain cellulose microcrystalline, dibasic calcium phosphate, magnesium stearate, silica, quinoline yellow (E104) and brilliant blue FCF (E133).
The caplets do not contain lactose or gluten.
Each IMODIUM Melts contains 2mg of loperamide hydrochloride as the active ingredient.
The Melts also contain gelatin, mannitol, aspartame, sodium bicarbonate and peppermint 51296 TP0551 flavour.
The Melts do not contain lactose or gluten.
Sponsor
Johnson & Johnson Pacific
45 Jones Street
Ultimo NSW 2007
Australia
For further enquiries call our Toll Free Info Line
Aus 1800 029 979
NZ 0800 446 147
Australian Register Number:
IMODIUM capsules - AUST R 13303
IMODIUM caplets - AUST R 56001
IMODIUM Melts - AUST R 83176
This leaflet was prepared in March 2009.
Brand Information
| Brand name | IMODIUM |
| Active ingredient | Loperamide hydrochloride |
| Schedule | Unscheduled | S2 |
MIMS Revision Date: 01 April 2022
1 Name of Medicine
Loperamide hydrochloride.
2 Qualitative and Quantitative Composition
Imodium Capsules contain Loperamide hydrochloride 2 mg.
Excipients with known effect. Lactose.
Imodium Capsules contain the inactive ingredients: maize starch, purified talc and magnesium stearate, iron oxide yellow CI77492, indigo carmine CI173015, titanium dioxide, erythrosine CI45430, iron oxide black CI177499, gelatin and water.
Imodium Zapid Tablets containing loperamide hydrochloride 2 mg and contain the inactive ingredients gelatin, mannitol, aspartame, sodium bicarbonate and mint flavour.
3 Pharmaceutical Form
Imodium Capsules are supplied in a gelatin capsule with a light grey body and dark green cap.
Imodium Zapid is a white to off-white circular freeze dried tablet.
4 Clinical Particulars
4.1 Therapeutic Indications
Imodium is indicated for:
The control and symptomatic treatment of acute nonspecific diarrhoea, and of chronic diarrhoea.
Reducing the volume of discharge in patients with ileostomies, colostomies, and other intestinal resections.
4.2 Dose and Method of Administration
Imodium capsule is administered orally with the aid of liquid.
Imodium Zapid tablet should be placed on the tongue. The tablet will dissolve and should be swallowed with saliva. No liquid intake is necessary.
Acute diarrhoea. The recommended initial dose of Imodium in adults is two capsules or tablets (4 mg) followed by one capsule or tablet (2 mg) after each unformed stool. Daily dose should not exceed eight capsules or tablets (16 mg). Clinical improvement is usually observed within 48 hours.
Chronic diarrhoea and reduction in volume of discharge of intestinal resections. The recommended initial dose of Imodium is two capsules or tablets (4 mg) followed by one capsule or tablet (2 mg) after each unformed stool until diarrhoea is controlled, after which dosage of Imodium should be reduced to meet individual requirements. When the optimal maintenance daily dosage has thus been established, this amount can then be administered as a single dose or in two or three divided doses.
The average daily maintenance dosage in clinical trials was two to four capsules (4-8 mg). A dosage of five capsules (10 mg) was rarely exceeded. A temporary exacerbation of diarrhoea was controlled by increasing the loperamide dosage to achieve further control followed by titration back to the established maintenance dose.
4.3 Contraindications
Hypersensitivity to any ingredients;
In patients with acute dysentery, which is characterised by blood in stools and high fever;
In patients with acute ulcerative colitis or Crohn's disease;
In patients with bacterial enterocolitis caused by invasive organisms including Salmonella, Shigella and Campylobacter;
In patients with pseudomembranous colitis associated with the use of broad-spectrum antibiotics;
In children under the age of 12 years.
In general, Imodium should not be used when inhibition of peristalsis is to be avoided due to the possible risk of significant sequelae including ileus, megacolon and toxic megacolon. Imodium must be discontinued promptly when constipation, abdominal distension or ileus develop.
Treatment of diarrhoea with Imodium is only symptomatic. Whenever an underlying aetiology can be determined, specific treatment should be given when appropriate.
4.4 Special Warnings and Precautions for Use
Fluid and electrolyte depletion may occur in patients who have diarrhoea. The use of Imodium does not preclude the administration of appropriate fluid and electrolyte therapy.
In acute diarrhoea, if clinical improvement is not observed in 48 hours, the administration of Imodium should be discontinued and patients should be advised to consult their physician.
Anticholinergic effects. In vitro studies have demonstrated anti-cholinergic properties. Hence, caution should be used in patients with glaucoma, urinary bladder neck obstruction, pyloric obstruction, significant gastric retention, or intestinal stasis.
Use in patients with AIDS. Use with caution in patients with AIDS. Patients with AIDS treated with Imodium for diarrhoea should have therapy stopped at the earliest signs of abdominal distension. There have been isolated reports of obstipation with an increased risk for toxic megacolon in AIDS patients with infectious colitis from both viral and bacterial pathogens treated with loperamide hydrochloride.
Renal impairment. Since the majority of the drug is metabolised, and the metabolites or the unchanged drug is excreted in the faeces, dose adjustments in patients with a kidney disorder are not required.
Use in hepatic impairment. Imodium must be used with caution in patients with hepatic insufficiency as it may result in a relative overdose leading to CNS toxicity. Abuse and misuse of loperamide, as an opioid substitute, have been described in individuals with opioid addiction (see Section 4.9 Overdose).
Use in the elderly. No data available.
Paediatric use. Imodium is contraindicated in children under the age of 12 years.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Although the pharmacological effect of Loperamide hydrochloride is not associated with a central action, patients with concomitant administration of tranquillisers or alcohol should be carefully observed.
Other drugs that affect loperamide hydrochloride theoretical interactions. Consideration should always be given with new drugs as to possible interaction with monoamine oxidase inhibitors. Theoretically, the combination of Imodium with monoamine oxidase inhibitors (which are also inhibitors of liver microsomal enzymes) may potentiate the action of loperamide by blocking its metabolic pathway.
Non-clinical data have shown that loperamide is a P-glycoprotein substrate. Concomitant administration of loperamide (16 mg single dose) with quinidine, or ritonavir, which are both P-glycoprotein inhibitors, resulted in a 2-3 fold increase in loperamide plasma levels. The clinical relevance of this pharmacokinetic interaction with P-glycoprotein inhibitors, when loperamide is given at recommended dosages (2 mg, up to 16 mg maximum daily dose), is unknown.
4.6 Fertility, Pregnancy and Lactation
Use in pregnancy. (Category B3)
Safe use of Imodium during pregnancy has not been established. Reproduction studies performed in rats and rabbits with high doses did not demonstrate evidence of impaired fertility or harm to the offspring due to loperamide hydrochloride. Higher doses impaired maternal and neonate survival, but even higher doses did not demonstrate teratogenicity. Such experience cannot exclude the possibility of damage to the foetus. Imodium should be used in only if the potential benefit justifies the risk to the foetus. Therefore, pregnant woman should be advised to consult their doctor for appropriate treatment.
Use in lactation. There is little information on the excretion of Imodium in human milk, but as small amounts of the drug are detectable in the milk of nursing mothers, the use of Imodium is not recommended in breast feeding subjects. Woman who are breastfeeding should be advised to consult their doctor for appropriate treatment. In a peri- and postnatal study, loperamide administered to female rats at dosage of 40 mg/kg indicated a possible adverse effect of lactation as evidenced in a decreased pup-survival rate.
Effects on fertility. No data available.
4.7 Effects on Ability to Drive and Use Machines
Tiredness, dizziness or drowsiness may occur in the setting of diarrhoeal syndromes treated with loperamide. Therefore it is advisable to use caution when driving a car or operating machinery.
4.8 Adverse Effects (Undesirable Effects)
The adverse effects reported during clinical investigations of Imodium are difficult to distinguish from symptoms associated with the diarrhoeal syndrome. Adverse experiences recorded during clinical studies with Imodium were generally of a minor and self-limiting nature. They were more commonly observed during treatment of chronic diarrhoea.
Acute diarrhea (12 years and over). The safety of loperamide HCl was evaluated in 2755 patients aged ≥ 12 years who participated in 26 controlled and uncontrolled clinical trials of loperamide HCl used for the treatment of acute diarrhea. Adverse reactions reported for ≥ 1% of loperamide HCl-treated patients are shown in Table 1.
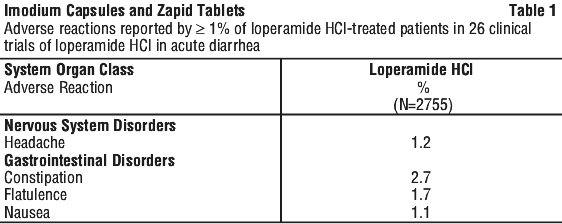
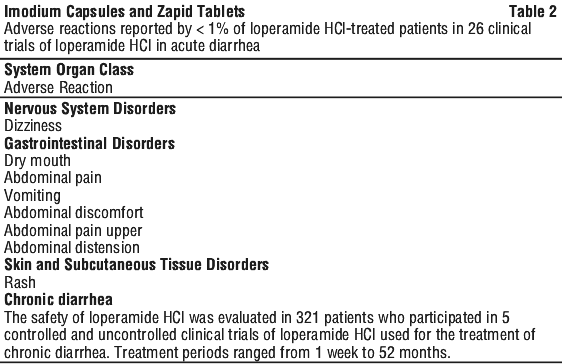


Very common ≥ 1/10; Common ≥ 1/100 and < 1/10; Uncommon ≥ 1/1,000 and < 1/100; Rare ≥ 1/10,000 and < 1/1,000; Very rare < 1/10,000; Not known (cannot be estimated from the available data).
Skin and subcutaneous tissue disorders. Very rare: rash, urticaria and pruritus.
Isolated occurrences of angioedema, and bullous eruptions including Stevens-Johnson syndrome, erythema multiforme, and toxic epidermal necrolysis have been reported with use of loperamide hydrochloride.
Immune system disorders. Very rare: Isolated occurrences of allergic reactions and in some cases severe hypersensitivity reactions including anaphylactic shock and anaphylactoid reactions have been reported with use of loperamide hydrochloride.
Gastrointestinal disorders. Very rare: abdominal pain, ileus, abdominal distension, nausea, constipation, vomiting, megacolon including toxic megacolon, flatulence, glossodynia1 and dyspepsia (see Section 4.3 Contraindications; Section 4.4 Special Warnings and Precautions for Use).
Renal and urinary disorders. Very rare: Isolated reports of urinary retention.
Psychiatric system disorders. Very rare: drowsiness.
Nervous system disorders. Very rare: Loss of consciousness, depressed level of consciousness, dizziness, coordination abnormality, hypertonia, somnolence.
Eye disorders. Very rare: Miosis.
General disorders and administration site conditions. Very rare: Fatigue.
Special senses. Very rare: taste disturbance.
A number of the adverse events reported during the clinical investigations and post-marketing experience with loperamide are frequent symptoms of the underlying diarrhoeal syndrome (abdominal pain/discomfort, nausea, vomiting, dry mouth, tiredness, drowsiness, dizziness, constipation and flatulence). These symptoms are often difficult to distinguish from undesirable drug effects.
With Imodium Zapid some subjects have complained about a burning or prickling sensation on the tongue immediately following its use.
1 Reported for orodispersible tablet only.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at: www.tga.gov.au/reporting-problems.
4.9 Overdose
Symptoms. In case of overdose (including relative overdose due to hepatic dysfunction), central nervous system depression (stupor, coordination abnormality, somnolence, miosis, muscular hypertonia, respiratory depression), urinary retention and paralytic ileus may occur. Children may be more sensitive to CNS effects than adults.
In clinical trials using loperamide hydrochloride, an adult took three 20 mg doses within a 24-hour period, was nauseated after the second dose, and vomited after the third dose.
In individuals who have intentionally ingested overdoses (reported in doses from 40 mg up to 792 mg per day) of loperamide HCl, QT interval and QRS complex prolongation and/or serious ventricular arrhythmias, including Torsade de Pointes, have been observed (see Section 4.4 Special Warnings and Precautions for Use). Fatal cases have also been reported. Abuse, misuse and/or overdose with excessively large doses of loperamide, may unmask Brugada syndrome. Upon cessation, cases of drug withdrawal syndrome, have been observed in individuals abusing, misusing or intentionally overdosing with excessively large doses of loperamide.
Treatment. If vomiting has occurred spontaneously, a slurry of 100 g of activated charcoal should be administered orally as soon as fluids can be maintained.
If vomiting has not occurred, gastric lavage should be performed, followed by administration of 100 g of activated charcoal slurry through gastric tube. In the case of overdosage, patient should be monitored for signs of CNS depression and/or respiratory depression for at least 24 hours. If CNS depression is observed, naloxone may be administered. If responsive to naloxone, vital signs must be monitored carefully for recurrence of symptoms of drug overdosage for at least 24 hours after the last dose of naloxone. In view of the prolonged action of loperamide and the short duration (one to three hours) of naloxone, the patient must be monitored closely and treated repeatedly with naloxone as indicated. Based on the fact that relatively little loperamide is excreted in urine, forced diuresis is not expected to be effective for Imodium overdosage.
Physical dependence to Imodium in humans has not been observed. However, studies in monkeys demonstrated that loperamide hydrochloride at high dose produced symptoms of physical dependence of the morphine type.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group- antipropulsive.
Mechanism of action. Loperamide binds to the opiate receptor in the gut wall. Consequently, it inhibits the release of acetylcholine and prostaglandins, thereby reducing propulsive peristalsis and increasing intestinal transit time. Studies suggest that loperamide may increase the tone of the anal sphincter, reducing incontinence and urgency.
Due to its high affinity for the gut wall and its high first-pass metabolism, loperamide hardly reaches the systemic circulation. In man, as a constipating agent, loperamide on a mg to mg basis is about 3 times more potent than diphenoxylate hydrochloride and 25 times more potent than codeine phosphate.
The onset of action, as determined in clinical studies with volunteers, indicated that clinical improvement occurs within 1-3 hours following drug administration (4 mg dose). The duration of action was determined from the interval between the time treatment was stopped due to constipation and the time bowel motion and stool consistency were again normal. In normal test subjects, a single 4 mg dose of loperamide significantly increased the median time of defaecation from 23 hours to 41 hours.
In those patients where biochemical and haematological parameters were monitored during clinical trials, no trends toward abnormality during loperamide therapy were noted. Similarly, urinalysis, ECG, and clinical ophthalmological examinations did not show trends towards abnormality.
CNS activity. Animal studies indicate that loperamide is devoid of analgesic properties (2-16 mg/kg). Studies in morphine-dependent monkeys demonstrated that loperamide hydrochloride in high S.C. doses prevented signs of morphine withdrawal. However, in humans the naloxone challenge pupil test which when positive indicated opiate-like effects, was negative when performed after a single high dose or after more than two years of therapeutic use (mean dose 4 mg/day) of loperamide hydrochloride.
Cardiovascular effects. In human volunteers, analysis of electrocardiograms obtained pre-therapy, and then two and six hours after administration of loperamide hydrochloride (16 mg), revealed no evidence of cardiovascular toxicity.
5.2 Pharmacokinetic Properties
Metabolism. The absorption, excretion and tissue distribution of a single oral dose of 3H-labelled loperamide was studied in rats (1.25 mg/kg) and man (2 mg). In man, peak plasma levels of about 2 nanogram/mL of intact drug occurred at 4 hours.
In the rat, approximately 15% of the administered dose was recovered after 96 hours. The highest residual concentration was found in the liver; the lowest in fatty tissue. About 60% of the administered dose was recovered from the faeces mainly as unchanged drug. Urinary excretion accounted for approximately 5% of which only 20% was unmetabolised loperamide. The existence of an enterohepatic shunt has been shown in rats and is assumed in man.
Excretion. The half-life of loperamide in man is about 11 hours with a range of 9-14 hours. Elimination mainly occurs by oxidative N-demethylation, which is the main metabolic pathway of loperamide. Excretion of the unchanged loperamide and the metabolites mainly occurs through the faeces. The combined cumulative urinary excretion of loperamide and its conjugates accounts for only about 2% of the administered dose.
Bioavailability. In a formal study Imodium Capsules and Caplets were not considered to be bioequivalent. As the antidiarrhoeal action of loperamide is considered to be due to a local effect, the clinical significance is unknown but is probably not significant.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
See Section 2 Qualitative and Quantitative Composition.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine. See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C.
6.5 Nature and Contents of Container
Imodium Capsules are available in cartons containing 8 and 20 capsules in a blister pack.
Imodium Zapid tablets are available in cartons containing 6 and 12 tablets in a blister pack.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Loperamide hydrochloride is 4-(4-chlorophenyl)-4-hydroxy-N, N-dimethyl-a, a-diphenyl-1-piperidinebutyramide monohydrochloride, a synthetic compound for oral use. It is a white to yellowish, amorphous or microcrystalline powder, insoluble in water.
Chemical structure.
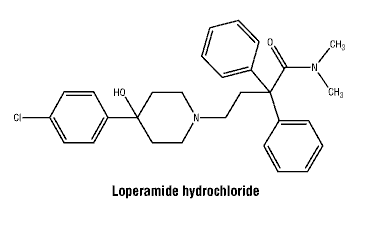
CAS number. 34552-83-5.
7 Medicine Schedule (Poisons Standard)
Pharmacy Medicine (S2).
Date of First Approval
01 August 1991
Date of Revision
30 November 2021
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.