Infanrix Hexa
Brand Information
| Brand name | Infanrix Hexa |
| Active ingredient | Diphtheria toxoid + Tetanus toxoid + Hepatitis B child vaccine + Pertussis vaccine + Poliomyelitis vaccine (inactivated), Haemophilus B conjugate vaccine |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Infanrix Hexa.
Summary CMI
INFANRIX HEXA
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about receiving this vaccine, speak to your doctor or pharmacist.
1. Why is my child being given INFANRIX HEXA?
INFANRIX HEXA is a vaccine used to prevent six diseases: diphtheria, tetanus, pertussis (whooping cough), hepatitis B, poliomyelitis (polio) and Haemophilus influenzae type b (Hib). INFANRIX HEXA contains the active ingredients of non-infectious substances from diphtheria bacteria, tetanus, purified proteins of pertussis bacteria, the surface protein of the hepatitis B virus (HBsAg, derived from genetically engineered yeast cells) and inactivated poliovirus.
For more information, see Section 1. Why is my child being given INFANRIX HEXA? in the full CMI.
2. What should I know before my child is given INFANRIX HEXA?
Do not use if your child has ever had an allergic reaction to INFANRIX HEXA or any of the ingredients listed at the end of the CMI.
Talk to your doctor if your child has any other medical conditions or takes any other medicines.
For more information, see Section 2. What should I know before my child is given INFANRIX HEXA? in the full CMI.
3. What if my child is taking other medicines?
Some medicines may interfere with INFANRIX HEXA and affect how it works.
A list of these medicines is in Section 3. What if my child is taking other medicines? in the full CMI.
4. How is INFANRIX HEXA given?
- INFANRIX HEXA is given as a 0.5 mL injection into the upper arm muscle, upper thigh or leg muscle.
- INFANRIX HEXA is usually given as a total of two or three doses with an interval of at least one month between each injection.
- If additional injections (boosters) are necessary, the doctor or nurse will tell you.
- Each dose is given on a separate visit. INFANRIX HEXA should not be given at birth.
More instructions can be found in Section 4. How is INFANRIX HEXA given? in the full CMI.
5. What should I know while my child is being given INFANRIX HEXA?
| Things you should do |
|
| Looking after the vaccine |
|
For more information, see Section 5. What should I know while my child is being given INFANRIX HEXA? in the full CMI.
6. Are there any side effects?
Most unwanted effects with INFANRIX HEXA are mild and usually clear up within a few days. These effects, as with other vaccines, generally occur around the injection site. Side effects are more likely to occur with booster dosing.
Side effects can include: pain, redness, swelling, a hard lump around the injection site, fever between 38°C and 39.5°C, generally feeling unwell, runny nose or loss of appetite, unusual crying (for more than 1 hour), nausea, vomiting, diarrhoea, headache, sleepiness, tiredness, nervousness, restlessness, fussiness or difficulty sleeping, skin rash, bruising, or purple or red-brown spots visible through the skin (purpura). For more information, including what to do if your child has any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
INFANRIX HEXA
Active ingredient(s): diphtheria toxoid, tetanus toxoid, pertussis toxoid, filamentous haemagglutinin and pertactin, Hepatitis B, Inactivated Poliovirus and Haemophilus influenzae type b
Consumer Medicine Information (CMI)
This leaflet provides important information about INFANRIX HEXA. You should also speak to your doctor, nurse or pharmacist if you would like further information or if you have any concerns or questions about your child receiving INFANRIX HEXA.
Where to find information in this leaflet:
1. Why is my child being given INFANRIX HEXA?
2. What should I know before my child is given INFANRIX HEXA?
3. What if my child is taking other medicines?
4. How is INFANRIX HEXA given?
5. What should I know while my child is being given INFANRIX HEXA?
6. Are there any side effects?
7. Product details
1. Why is my child being given INFANRIX HEXA?
INFANRIX HEXA contains the active ingredients of non-infectious substances from diphtheria bacteria, tetanus, purified proteins of pertussis bacteria, the surface protein of the hepatitis B virus (HBsAg, derived from genetically engineered yeast cells), inactivated poliovirus and Haemophilus influenzae type b.
INFANRIX HEXA is a vaccine used to prevent six diseases: diphtheria, tetanus, pertussis (whooping cough), hepatitis B, poliomyelitis (polio) and Haemophilus influenzae type b (Hib). The vaccine works by causing the body to produce its own protection (antibodies) against these diseases.
Diphtheria, tetanus, pertussis and Hib are all serious life-threatening diseases caused by bacterial infection. Hepatitis B and poliomyelitis are infectious diseases caused by viral infection.
Diphtheria
Diphtheria mainly affects the airways and sometimes the skin. Generally, the airways become inflamed (swollen) causing severe breathing difficulties and sometimes suffocation. The bacteria also release a toxin (poison), which can cause nerve damage, heart problems, and death. The risk of serious complications and death is greater in the very young and elderly.
Tetanus (Lockjaw)
Tetanus bacteria enter the body through wounded skin. Wounds that are especially prone to infection are burns, fractures, deep wounds or wounds contaminated with soil, dust, horse manure or wood splinters. The bacteria release a toxin (poison), which can cause muscle stiffness, painful muscle spasms, fits and death. The spasms can be strong enough to cause bone fractures of the spine. The death rate is 10% of cases.
Pertussis (Whooping cough)
Pertussis is a highly infectious illness. The disease affects the breathing tract causing severe spells of coughing that may interfere with normal breathing. The coughing is often accompanied by a ‘whooping’ sound. The cough may last for 1-2 months or longer. Pertussis can also cause inner ear infections, long-lasting bronchitis, pneumonia, fits, brain damage and death. The risk of severe complications and death is greatest in infants under 6 months of age. The death rate is 0.5% for infants under 6 months of age.
Hepatitis B
Hepatitis B is caused by the hepatitis B virus. It causes the liver to be become swollen (inflamed). The virus is found in body fluids such as blood, semen, vaginal secretions, or saliva of infected people. The virus can enter the bloodstream through:
- an infected mother passing the virus onto her baby during or shortly after birth
- sores, cuts or tiny wounds coming into contact with infected fluids (e.g. from a human bite, sharing razors or toothbrushes, or working with human blood or body fluids)
- injection (e.g. needlestick injury, or sharing needles for IV drug use)
- sexual intercourse.
Some people infected with hepatitis B may not look or feel sick. But others will get symptoms, which may not be seen for 6 weeks to 6 months after infection. Sometimes people will only have mild flu-like symptoms, but other people can become very ill. They may be extremely tired, and have dark urine, pale faeces, yellowish skin and/or eyes (jaundice), and other symptoms possibly requiring hospitalisation.
Most adults fully recover from the disease. However, some people, particularly children, who may not have had symptoms, can remain infected. They are called hepatitis B virus carriers. Hepatitis B carriers can infect others throughout their lives.
Babies infected with hepatitis B at birth almost always become carriers. Often they do not show symptoms, and seem healthy for many years. However, after 30, 40 or 50 years they can become sick and develop symptoms. For all chronic hepatitis B carriers there is a risk of serious liver disease, such as cirrhosis (liver scarring) and liver cancer.
There is no specific treatment for hepatitis B.
Poliomyelitis (Polio)
Polio is a viral infection that can have variable effects. Often it causes only a mild illness but in some people it causes permanent injury or death.
In its severest form, polio infection causes paralysis of the muscles (unable to move), including those needed for breathing and walking. Polio infection can leave a person unable to breathe without the help of an iron lung machine, unable to walk without leg braces, or confined to a wheelchair. The limbs affected by the disease may be painfully twisted (deformed).
Haemophilus influenzae type b (Hib)
Hib most frequently causes brain inflammation (swelling), which is generally seen in infants under 18 months of age. The death rate is 5-10% of infants in this age group. In 15-30% of surviving infants there will be some type of serious complication such as: mental retardation, cerebral palsy, deafness, epilepsy or partial blindness. Hib can also causes inflammation of the throat, which is mostly seen in children over 18 months of age. This occasionally causes death by suffocation. Less commonly, the bacteria can also infect the blood, heart, lungs, bones, joints, and tissues of the eyes and mouth.
Vaccination is the best way to protect against these diseases. INFANRIX HEXA cannot give your child diphtheria, tetanus, pertussis, hepatitis B, Hib or polio infection. The vaccine will not protect against diseases caused by other types of bacteria, viruses or organisms.
If a person is already infected with the hepatitis B virus at the time of vaccination, INFANRIX HEXA may not prevent the disease in these people.
2. What should I know before my child is given INFANRIX HEXA?
Warnings
Your child should not be given INFANRIX HEXA if:
- your child is allergic to INFANRIX HEXA or any of the ingredients listed at the end of this leaflet. Always check the ingredients to make sure your child can receive this vaccine
- your child is allergic to any other diphtheria, tetanus, hepatitis B, inactivated polio or Haemophilus influenzae type b vaccine.
Check with your doctor if your child:
- has a severe infection with a high temperature. A minor infection such as a cold should not be a problem, but talk to your doctor or nurse about this before being vaccinated
- has experienced any problems after receiving INFANRIX HEXA or another pertussis-containing vaccine, especially:
- a high temperature (over 40.5°C) within 48 hours of vaccination
- a collapse or shock-like state within 2 days of vaccination
- crying lasting 3 hours or more within 2 day of vaccination
- convulsions (seizures/fits) with or without a fever within 3 days of vaccination
- a disease of the brain within 7 days after previous vaccination with a pertussis containing vaccine. - has any medical conditions such as:
- brain disease or central nervous system (CNS) disease (for example epilepsy etc.)
- a bleeding problem or bruises easily
- lowered immunity due to medical treatment or a medical condition
- a tendency to febrile convulsions (seizures/fits due to a fever or high body temperature)
- a family history of seizures/fits
- a family history of Sudden Infant Death Syndrome (SIDS)
- allergy to the antibiotics neomycin and polymyxin.
Fainting can occur following, or even before, any needle injection, therefore, tell the doctor or nurse if your child fainted with a previous injection.
From vaccination, your child may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
3. What if my child is taking other medicines?
Tell your doctor, nurse or pharmacist if your child is taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with INFANRIX HEXA and affect how it works, such as:
- medicines which suppress the immune system, such as high-dose steroids.
Having other vaccines
Tell your doctor or nurse if your child has received another vaccine recently.
Some vaccines may be affected by other vaccines. Your doctor, nurse or pharmacist will be able to tell you what to do if INFANRIX HEXA is to be given with another vaccine.
4. How is INFANRIX HEXA given?
The doctor or nurse will give INFANRIX HEXA as an injection.
If you have any concerns about how this vaccine is to be given, talk to your doctor, nurse or pharmacist.
How much is given
- The dose of INFANRIX HEXA is 0.5 mL.
How is it given
- INFANRIX HEXA will be injected into the upper leg muscle or the upper arm muscle.
- The vaccine should never be injected into a vein, artery or the skin.
When it is given
INFANRIX HEXA is usually given as a total of two or three doses with an interval of at least one month between each injection.
- If additional injections (boosters) are necessary, the doctor or nurse will tell you.
- Each dose is given on a separate visit. INFANRIX HEXA should not be given at birth.
- It is important to return at the recommended times for follow-up doses.
- INFANRIX HEXA can be given as a booster dose when the child is 18 months old when boosting with hepatitis B and/or poliovirus and/or Haemophilus influenza type b, as well as diphtheria, tetanus and pertussis, is required.
- You should discuss with your doctor what is needed for your child.
If a dose is missed
If your child misses a scheduled dose, talk to your doctor or nurse and arrange another visit as soon as possible.
If too much INFANRIX HEXA is given
If you think that too much INFANRIX HEXA has been given, urgent medical attention may be required.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while my child is being given INFANRIX HEXA?
Things you should do
Keep your child's follow-up visits with the doctor or clinic. It is important the follow-up doses of INFANRIX HEXA are given at the correct times. This will ensure the best effect of the vaccine in protecting your child against diphtheria, tetanus, pertussis, hepatitis B, poliovirus infection and haemophilus influenzae type b.
Looking after the vaccine
INFANRIX HEXA is usually stored at the doctor's clinic or surgery, or at the pharmacy.
If you need to store INFANRIX HEXA always:
- keep INFANRIX HEXA in the refrigerator stored between 2°C and 8°C. THE PACK SHOULD NEVER BE FROZEN. FREEZING DESTROYS THE VACCINE
- keep INFANRIX HEXA in the original pack until it is time for it to be given
- INFANRIX HEXA should be used immediately after opening.
Keep it where young children cannot reach it.
Getting rid of any unwanted vaccine
If you no longer need to use this vaccine or it is out of date, take it to any pharmacy for safe disposal. Do not use this vaccine after the expiry date.
6. Are there any side effects?
All vaccines can have side effects. If your child experiences any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor, nurse or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
General disorders and administration site conditions:
| Speak to your doctor if your child has any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
General disorders and administration site conditions:
As with all vaccines given by injection there is a very small risk of such reactions. Allergy to INFANRIX HEXA vaccine is rare. Any such severe reactions will usually occur within the first few hours of vaccination. Respiratory disorders:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel worried.
Other side effects not listed here may occur in some children.
Reporting side effects
After you have received medical advice for any side affects your child experienced, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this vaccine.
Always make sure you speak to your doctor, nurse or pharmacist before you decide to stop your child taking any medicines.
7. Product details
This vaccine is only available with a doctor's prescription.
What INFANRIX HEXA contains
| Active ingredients (main ingredients) |
|
| Other ingredients (inactive ingredients) |
The following ingredients are present as residues:
|
Your child should not be given this vaccine if they are allergic to any of these ingredients.
The manufacture of this product includes exposure to bovine derived materials. No evidence exists that any case of vCJD (considered to be the human form of bovine spongiform encephalopathy) has resulted from the administration of any vaccine product.
What INFANRIX HEXA looks like
INFANRIX HEXA (AUST R 132881) comes in two parts. The first part is a white, milky liquid (0.5 mL) in a pre-filled syringe that consists of the combined diphtheria, tetanus, pertussis, hepatitis B and inactivated poliovirus vaccine.
The second part is the Hib vaccine and is a white pellet in a separate glass vial.
These parts are mixed together before use. When both parts are mixed the vaccine looks like a white, cloudy/milky liquid.
Who distributes INFANRIX HEXA
GlaxoSmithKline Australia Pty Ltd
Level 4, 436 Johnston Street,
Abbotsford, Victoria, 3067
Phone: 1800 033 109
www.gsk.com.au
Trade marks are owned by or licensed to the GSK group of companies.
©2025 GSK group of companies or its licensor.
This leaflet was prepared on 29 August 2025.
Version 11.0
Brand Information
| Brand name | Infanrix Hexa |
| Active ingredient | Diphtheria toxoid + Tetanus toxoid + Hepatitis B child vaccine + Pertussis vaccine + Poliomyelitis vaccine (inactivated), Haemophilus B conjugate vaccine |
| Schedule | S4 |
MIMS Revision Date: 01 September 2023
1 Name of Medicine
Combined diphtheria-tetanus-acellular pertussis (DTPa), hepatitis B, poliovirus and Haemophilus influenzae type b vaccine.
2 Qualitative and Quantitative Composition
1 dose (0.5 ml) contains:
diphtheria toxoid1 not less than 30 international units;
tetanus toxoid1 not less than 40 international units;
Bordetella pertussis antigens: pertussis toxoid (PT)1 25 microgram, filamentous haemagglutinin (FHA)1 25 microgram, pertactin (PRN)1 8 microgram;
hepatitis B surface antigen (HBs)2,3 10 microgram;
poliovirus (inactivated) (IPV) type 1 (Mahoney strain)4 40 D-antigen unit, type 2 (MEF-1 strain)4 8 D-antigen unit, type 3 (Saukett strain)4 32 D-antigen unit;
Haemophilus influenzae type b polysaccharide (polyribosylribitol phosphate, PRP)3 10 microgram, conjugated to tetanus toxoid as carrier protein 20-40 microgram.
1 Adsorbed on aluminium hydroxide hydrate (Al(OH)3) 0.5 milligram Al3+.
2 Produced in yeast cells (Saccharomyces cerevisiae) by recombinant DNA technology.
3 Adsorbed on aluminium phosphate (AlPO4) 0.32 milligram Al3+.
4 Propagated in VERO cells.
List of excipients with known effect. Infanrix Hexa also contains residual amounts of neomycin sulfate and polymyxin B sulfate, which are carried over from the manufacturing process.
For the full list of excipients, see Section 6.1 List of Excipients.
This medicine contains less than 1 mmol (39 mg) per dose of potassium and less than 1 mmol (23 mg) per dose of sodium, i.e. essentially 'potassium-free' and 'sodium-free'.
3 Pharmaceutical Form
Powder and suspension for injection.
The DTPa-HBV-IPV component is presented as a turbid white suspension. Upon storage, a white deposit and clear supernatant can be observed. This is a normal observation.
The Hib component is presented as a white pellet.
4 Clinical Particulars
4.1 Therapeutic Indications
Infanrix Hexa is indicated for primary immunisation of infants from the age of 6 weeks against diphtheria, tetanus, pertussis, hepatitis B, poliomyelitis, and Haemophilus influenzae type b.
Infanrix Hexa is also indicated for use as booster dose if boosting with hepatitis B, poliomyelitis, and Haemophilus influenzae type b, as well as diphtheria, tetanus and pertussis is required.
The use of Infanrix Hexa should be in accordance with official recommendations.
See Section 4.2 Dose and Method of Administration for further information.
4.2 Dose and Method of Administration
Infanrix Hexa is for deep intramuscular injection.
Before use of the vaccine, the Infanrix Hexa suspension should be well shaken in order to obtain a homogeneous turbid white suspension. The DTPa-HBV-IPV suspension and the Hib pellet should be inspected visually for any foreign particulate matter and/or any variation of physical aspect. In the event of either being observed, do not administer the vaccine.
Infanrix Hexa must be reconstituted by adding the entire contents of the prefilled syringe containing the liquid component to the vial containing the Hib pellet.
After the addition of the liquid component to the pellet, the mixture should be well shaken until the pellet is completely dissolved in the suspension.
It is good clinical practice to only inject a vaccine when it has reached room temperature. In addition, a vial at room temperature ensures sufficient elasticity of the rubber closure to minimise any coring of rubber particles. To achieve this, the vial should be kept at room temperature (25 ± 3°C) for at least five minutes before connecting the prefilled syringe and reconstituting the vaccine.
The reconstituted vaccine presents as a slightly more cloudy suspension than the liquid component alone. This is a normal observation.
The reconstituted vaccine should be inspected visually for any foreign particulate matter and/or abnormal physical appearance. In the event of either being observed, discard the vaccine.
After reconstitution, the vaccine should be injected immediately. However, the vaccine may be kept for up to 8 hours at room temperature.
Withdraw the entire contents of the vial.
Instructions for the pre-filled syringe. Hold the syringe by the barrel, not by the plunger.
Unscrew the syringe cap by twisting it anticlockwise.
To attach the needle, connect the hub to the Luer lock adaptor and rotate a quarter turn clockwise until you feel it lock.
Reconstitute the vaccine as described above.
Do not pull the syringe plunger out of the barrel. If it happens, do not administer the vaccine.
Dosage. After reconstitution, each dose consists of a 0.5 mL ready to use sterile suspension.
Administration. Infanrix Hexa is administered by deep intramuscular injection. The vaccine should never be administered intravenously.
Infanrix Hexa should be injected intramuscularly in the anterolateral aspect of the thigh or the deltoid region of the arm. The recommended dose (0.5 mL) of vaccine must be administered.
The primary immunisation course of Infanrix Hexa consists of two or three doses (of 0.5 mL) (see Section 5.1 Pharmacodynamic Properties, Clinical trials for schedules evaluated in clinical trials). See Table 1.
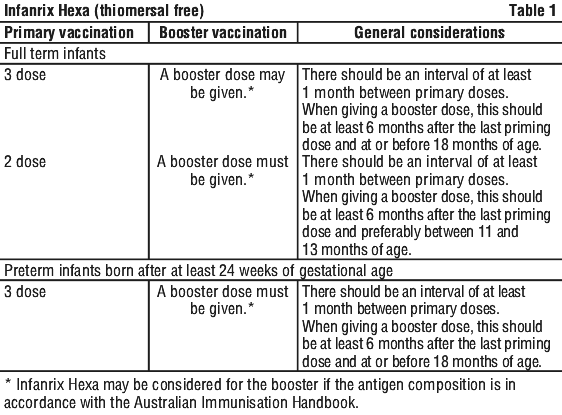
Locally established immunoprophylactic measures against hepatitis B should be maintained.
Further guidance regarding the use of vaccines can be found in the Australian Immunisation Handbook (see Section 5.1 Pharmacodynamic Properties, Clinical trials for schedules evaluated in clinical trials).
4.3 Contraindications
Infanrix Hexa should not be administered to subjects with known hypersensitivity to the active substances or to any of the excipients or residues (see Section 2 Qualitative and Quantitative Composition).
Infanrix Hexa should not be administered to subjects having shown signs of hypersensitivity after previous administration of diphtheria, tetanus, pertussis, hepatitis B, polio or Hib vaccines.
Infanrix Hexa is contraindicated if the child has experienced encephalopathy of unknown aetiology, occurring within 7 days following previous vaccination with pertussis containing vaccine. In these circumstances pertussis vaccination should be discontinued and the vaccination should be continued with diphtheria, tetanus, hepatitis B, inactivated polio and Hib vaccines.
4.4 Special Warnings and Precautions for Use
Do not administer Infanrix Hexa intravascularly or intradermally.
As with other vaccines, the administration of Infanrix Hexa should be postponed in subjects suffering from acute severe febrile illness (with fever > 38.5°C). The presence of a minor infection, however, is not a contraindication.
It is good clinical practice that immunisation should be preceded by a review of the medical history (especially with regard to previous immunisation and possible occurrence of undesirable events) and a clinical examination.
A protective immune response may not be elicited in all vaccinees (see Section 5.1 Pharmacodynamic Properties, Clinical trials).
Infanrix Hexa will not prevent disease caused by pathogens other than Corynebacterium diphtheriae, Clostridium tetani, Bordetella pertussis, hepatitis B virus, poliovirus or Haemophilus influenzae type b. However, it can be expected that hepatitis D will be prevented by immunisation as hepatitis D (caused by the delta agent) does not occur in the absence of hepatitis B infection. The vaccine will not prevent infection caused by other agents such as hepatitis A, hepatitis C and hepatitis E and other pathogens known to infect the liver.
If any of the following events are known to have occurred in temporal relation to receipt of whole cell or acellular pertussis containing vaccine, the decision to give further doses of vaccine containing the pertussis component should be carefully considered:
Temperature of ≥ 40.0°C within 48 hours, not due to another identifiable cause.
Collapse or shock-like state (hypotonic-hyporesponsive episode) within 48 hours of vaccination.
Persistent, inconsolable crying lasting ≥ 3 hours, occurring within 48 hours of vaccination.
Convulsions with or without fever, occurring within 3 days of vaccination.
No data currently exist on use of Infanrix Hexa in these children. There may be circumstances, such as a high incidence of pertussis, when the potential benefits outweigh possible risks, particularly since these events are not associated with permanent sequelae.
In children with progressive neurological disorders, including infantile spasms, uncontrolled epilepsy or progressive encephalopathy, it is better to defer pertussis (Pa or Pw) immunisation until the condition is corrected or stable. However, the decision to give pertussis vaccine must be made on an individual basis after careful consideration of the risks and benefits.
As with all injectable vaccines, appropriate medical treatment and supervision should always be readily available in case of a rare anaphylactic reaction following the administration of the vaccine.
Infanrix Hexa should be administered with caution to subjects with thrombocytopenia or a bleeding disorder since bleeding may occur following an intramuscular injection in these subjects.
A history of febrile convulsions, a family history of convulsions, or sudden infant death syndrome (SIDS) do not constitute contraindications for the use of Infanrix Hexa. Vaccinees with a history of febrile convulsions should be closely followed up as such adverse events may occur within 2 to 3 days postvaccination.
Data from clinical studies indicate that, when Infanrix Hexa is coadministered with pneumococcal conjugate vaccine, the rate of febrile reactions is higher compared to that occurring following the administration of Infanrix Hexa alone.
Increased reporting rates of convulsions (with or without fever) and hypotonic hyporesponsive episode (HHE) were observed with concomitant administration of Infanrix Hexa and Prevenar 13 (see Section 4.8 Adverse Effects (Undesirable Effects)).
Antipyretic treatment should be initiated according to local treatment guidelines.
Syncope (fainting) can occur following, or even before, any vaccination as a psychogenic response to the needle injection. It is important that procedures are in place to avoid injury from faints.
Infanrix Hexa should not be administered at birth. Infants born of HBsAg positive mothers should receive hepatitis B immune globulin and hepatitis B vaccine at birth.
The Hib component of the vaccine does not protect against diseases due to other strains of Haemophilus influenzae or against meningitis caused by other organisms.
Special populations. Human immunodeficiency virus (HIV) infection is not considered as a contraindication. However, in patients with immunodeficiency or in patients receiving immunosuppressive therapy, the expected immunologic response may not be achieved. No data currently exist on use of Infanrix Hexa in these patients.
Preterm infants. Clinical data indicate that Infanrix Hexa can be given to preterm infants, however, as expected in this population, a lower immune response has been observed for some antigens (see Section 4.8 Adverse Effects (Undesirable Effects); Section 5.1 Pharmacodynamic Properties, Clinical trials).
The potential risk of apnoea and the need for respiratory monitoring for 48-72h should be considered when administering the primary immunisation series to very preterm infants (born ≤ 28 weeks of gestation) and particularly for those with a previous history of respiratory immaturity. As the benefit of vaccination is high in these infants, vaccination should not be withheld or delayed.
Native populations (native Alaskans, native American Indians) with a high incidence of Haemophilus influenzae type b disease have shown a reduced antibody response to Haemophilus influenzae type b conjugate vaccines. The immunogenicity of Infanrix Hexa has not been studied in the Australian indigenous population and the possibility of a lower antibody response to the Hib component, than that seen in clinical studies, should be borne in mind.
Use in the elderly. No data available.
Paediatric use. See Section 4.4 Special Warnings and Precautions for Use.
Effects on laboratory tests. Since the Hib capsular polysaccharide antigen is excreted in the urine a positive urine test can be observed within 2 weeks following vaccination. Other tests should be performed in order to confirm Hib infection during this period.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Infanrix Hexa should not be mixed with other vaccines in the same syringe.
Infanrix Hexa can be given concomitantly with pneumococcal conjugate, MenC conjugate, MenACWY conjugate, MenB, rotavirus, measles, mumps, rubella and varicella vaccines. Data have shown no clinically relevant interference in the antibody response to each of the individual antigens.
When Infanrix Hexa was co-administered with MenB and pneumococcal conjugate vaccines, inconsistent results were seen across studies for responses to inactivated poliovirus type 2 and pneumococcal conjugate serotype 6B antigen and to the pertussis pertactin antigen but these data do not suggest clinically significant interference.
Data from clinical studies indicate that, when Infanrix Hexa is coadministered with pneumococcal conjugate vaccine, the rate of febrile reactions is higher compared to that occurring following the administration of Infanrix Hexa alone. (See Section 4.4 Special Warnings and Precautions for Use).
Data from clinical studies indicate a more frequent occurrence of fever, pain at the injection site, appetite lost and irritability when Infanrix Hexa is co-administered with MenB vaccine and 7-valent pneumococcal conjugate vaccine.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category B2)
As Infanrix Hexa is not intended for use in adults, adequate human data on use during pregnancy and adequate animal reproduction studies are not available.
Use in lactation. As Infanrix Hexa is not intended for use in adults, adequate human data on use during lactation are not available.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Clinical trial experience. The safety profile presented below is based on data from more than 16,000 subjects.
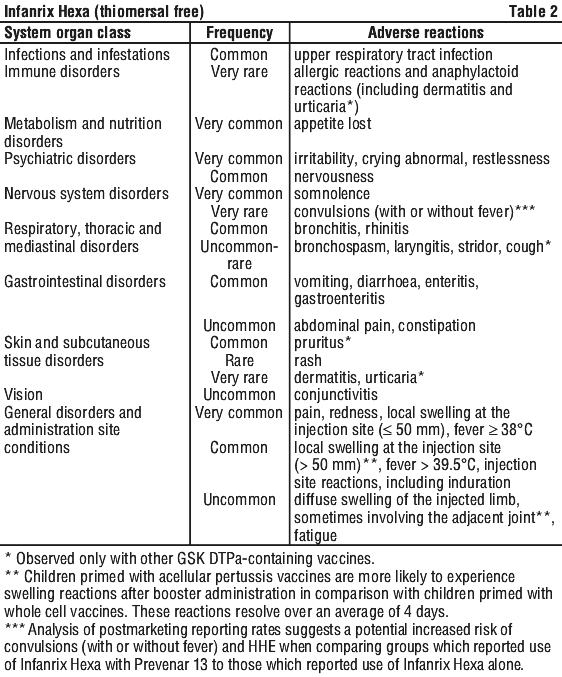
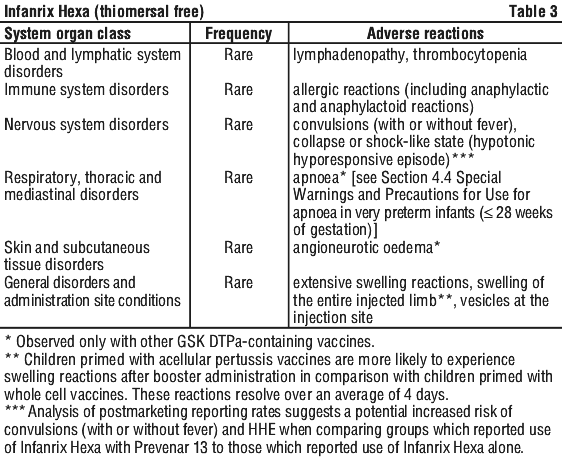
As has been observed for DTPa and DTPa containing combinations, an increase in local reactogenicity and fever was reported after booster vaccination with Infanrix Hexa with respect to the primary course. Table 2 contains data from subjects receiving booster vaccinations and those receiving primary doses.
Adverse reactions reported are listed according to the following frequency:
Very common: ≥ 1/10; Common: ≥ 1/100 to < 1/10; Uncommon: ≥ 1/1000 to < 1/100; Rare: ≥ 1/10,000 to < 1/1000; Very rare: < 1/10,000.
Safety in infants and toddlers born to mothers vaccinated with dTpa during pregnancy. In two clinical studies, Infanrix Hexa has been administered to more than 500 subjects born to mothers vaccinated with dTpa (n=341) or placebo (n=346) during the third trimester of pregnancy. The safety profile of Infanrix Hexa was similar regardless of exposure/non-exposure to dTpa during pregnancy.
Experience with hepatitis B vaccine which is the same antigen used in Infanrix Hexa. Meningitis, allergic reactions mimicking serum sickness, paralysis, encephalitis, encephalopathy, neuropathy, neuritis, hypotension, vasculitis, lichen planus, erythema multiforme, arthritis and muscular weakness, Guillain-Barré syndrome have been reported during postmarketing surveillance following GlaxoSmithKline Biologicals' hepatitis B vaccine in infants < 2 years old. The causal relationship to the vaccine has not been established.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Infanrix Hexa provides immunity against diphtheria, tetanus, pertussis, hepatitis B, poliovirus and Haemophilus influenzae type b by inducing the production of antibodies and the ability to mount an immunological memory.
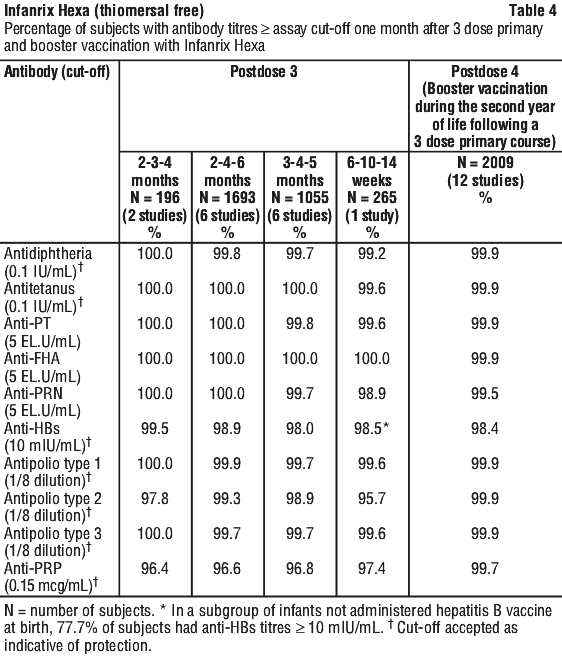
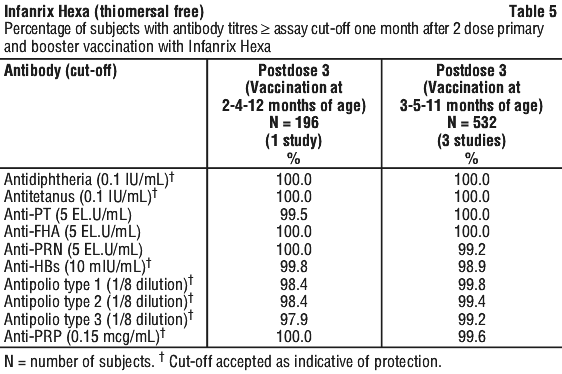
Clinical trials. Immunogenicity. The immunogenicity of Infanrix Hexa has been evaluated in clinical studies from 6 weeks of age. The vaccine was assessed in 2 dose and 3 dose priming schedules and as a booster dose. The results of these clinical studies are summarised in Tables 4 and 5.
After a 3 dose primary vaccination schedule, at least 95.7% of infants had developed seroprotective or seropositive antibody levels against each of the vaccine antigens. After booster vaccination (postdose 4), at least 98.4% of children had developed seroprotective or seropositive antibody levels against each of the vaccine antigens.
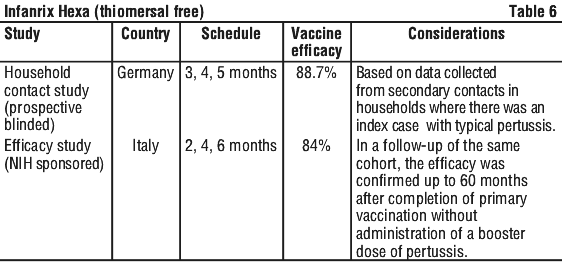
Protective efficacy against pertussis. The protective efficacy of the pertussis component of Infanrix (DTPa) against WHO defined typical pertussis (≥ 21 days of paroxysmal cough with laboratory confirmation) was demonstrated after 3 dose primary immunisation in the studies in Table 6.
Lower antibody concentrations against pertussis antigens post-primary (PT, FHA and PRN) and post-booster (PT, FHA) vaccination were observed in infants and toddlers born to mothers vaccinated with dTpa during pregnancy. The fold-increases of anti-pertussis antibody concentrations from the pre-booster to the 1-month post-booster time point were in the same range for infants and toddlers born to mothers vaccinated with dTpa or with placebo, demonstrating effective priming of the immune system.
In the absence of correlates of protection for pertussis, the clinical relevance of these observations remains to be fully understood. However, current epidemiological data on pertussis disease following the implementation of dTpa maternal immunisation do not suggest any clinical relevance of this immune interference.
Immunogenicity in preterm infants. The immunogenicity of Infanrix Hexa was evaluated across three studies including approximately 300 preterm infants (born after a gestation period of 24 to 36 weeks) following a 3 dose primary vaccination course at 2, 4 and 6 months of age. The immunogenicity of a booster dose at 18 to 24 months of age was evaluated in approximately 200 preterm infants.
One month after primary vaccination at least 98.7% of subjects were seroprotected against diphtheria, tetanus and poliovirus types 1 and 2; at least 90.9% had seroprotective antibody levels against the hepatitis B, PRP and poliovirus type 3 antigens; and all subjects were seropositive for antibodies against FHA and PRN while 94.9% were seropositive for anti-PT antibodies.
One month after the booster dose at least 98.4% of subjects had seroprotective or seropositive antibody levels against each of the antigens except against PT (at least 96.8%) and hepatitis B (at least 88.7%). The response to the booster dose in terms of fold increases in antibody concentrations (15 to 235-fold), indicate that preterm infants were adequately primed for all the antigens of Infanrix Hexa.
In a follow-up study, approximately 2.5 to 3 years after the booster dose, 85.3% of the children were still seroprotected against hepatitis B and at least 95.7% were seroprotected against the three poliovirus types and PRP.
Persistence of the immune response. The persistence of the immune response to a 3 dose primary and booster schedule with Infanrix Hexa was evaluated in children 4-8 years of age. Protective immunity against the three poliovirus types and PRP was observed in at least 91.0% of children and against diphtheria and tetanus in at least 64.7% of children. At least 25.4% (anti-PT), 97.5% (anti-FHA) and 87.0% (anti-PRN) of children were seropositive against the pertussis components.
With regards to hepatitis B, seropositive antibody concentration following a 3-dose primary and booster schedule with Infanrix Hexa have been shown to persist in ≥ 85% of subjects 4-5 years of age and in ≥ 72% of subjects 7-8 years of age, in ≥ 60% of subjects 12-13 years of age and in 53.7% of subjects 14-15 years of age. Additionally, following a 2-dose primary and booster schedule, seroprotective antibody concentrations against hepatitis B persisted in ≥ 48% of subjects 11-12 years of age.
Hepatitis B immunological memory was confirmed in children 4 to 15 years of age. These children had received Infanrix Hexa as primary and booster vaccination in infancy, and when an additional dose of monovalent HBV vaccine was administered, protective immunity was observed in at least 93% of subjects.
Postmarketing experience. Results of long term follow-up in Sweden demonstrate that acellular pertussis vaccines are efficacious in infants when administered according to the 3 and 5 months primary vaccination schedule, with a booster dose administered at approximately 12 months. However, data indicate that protection against pertussis may be waning at 7-8 years of age. This suggests that a second booster dose of pertussis vaccine is warranted in children aged 5-7 years who have previously been vaccinated following this schedule.
The effectiveness of the Hib component of Infanrix Hexa was investigated via an extensive postmarketing surveillance study conducted in Germany. The recommended schedule in Germany for all DTaP/Hib conjugate vaccines is a 3 dose primary series at the age of 2, 3 and 4 months with a booster at 11 to 14 months of age. Over a 7 year follow-up period, the effectiveness of the Hib components of two hexavalent vaccines, of which one was Infanrix Hexa, was 89.6% for a full primary series and 100% for a full primary series plus booster dose (irrespective of the Hib vaccine used for priming).
Infanrix Hexa has been the principal Hib containing vaccine available in Italy since 2006. The vaccine is administered at 3, 5 and 11 months of age and coverage has exceeded 95%. Hib disease has continued to be well controlled, with no more than three confirmed Hib cases reported annually between 2006 and 2011 in Italian children aged less than 5 years.
5.2 Pharmacokinetic Properties
Not relevant to vaccines.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
The final vaccine also contains the excipients lactose, medium 199 (as stabiliser containing amino acids, mineral salts, vitamins and other substances), sodium chloride, aluminium hydroxide hydrate, aluminium phosphate and water for injections. The vaccine also contains the following residues: neomycin sulfate and polymyxin B sulfate.
6.2 Incompatibilities
See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Infanrix Hexa should be stored at +2°C to +8°C.
The DTPa-HBV-IPV suspension and the reconstituted vaccine must not be frozen. Discard if it has been frozen.
Protect from light.
During transport, recommended conditions of storage must be respected.
Stability data indicate that the vaccine components are stable at temperatures up to 25°C for 72 hours. These data are intended to guide healthcare professionals in case of temporary temperature excursion only.
6.5 Nature and Contents of Container
The DTPa-HBV-IPV suspension is presented in a pre-filled syringe.
The lyophilised Hib component is presented in a glass vial.
Powder in a vial (type I glass) containing 1 dose with a stopper (butyl rubber) and 0.5 mL of suspension in a pre-filled syringe (type I glass) with a plunger stopper (butyl rubber) and with a rubber tip cap.
The tip cap and rubber plunger stopper of the pre-filled syringe and the stopper of the vial are not made with natural rubber latex.
This combination pack is supplied in packs of 1 and 10.
Not all pack sizes may be distributed in Australia.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Not relevant to vaccines.
7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Date of First Approval
15 June 2006
Date of Revision
18 July 2023
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.