Iomeron
Brand Information
| Brand name | Iomeron |
| Active ingredient | Iomeprol |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Iomeron.
Summary CMI
IOMERON®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using IOMERON®?
IOMERON® contains the active ingredient iomeprol. IOMERON® is used to make blood vessels and other parts of the body visible to X-rays.
For more information, see Section 1. Why am I using IOMERON®? in the full CMI.
2. What should I know before I use IOMERON®?
Do not use if you have ever had an allergic reaction to IOMERON® or any of the ingredients listed at the end of the CMI.
Talk to your radiologist or doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use IOMERON®? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with IOMERON® and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use IOMERON®?
- Your radiologist/doctor will inject the correct amount of IOMERON® into a blood vessel or body cavity, before taking the X-ray photographs.
More instructions can be found in Section 4. How do I use IOMERON®? in the full CMI.
5. What should I know while using IOMERON®?
| Things you should do |
|
| Driving or using machines |
|
For more information, see Section 5. What should I know while using IOMERON®? in the full CMI.
6. Are there any side effects?
Less serious side effects may include: flushing, injection-site pain/bleeding/swelling, itching, rash, sweating, weakness, fever, feeling unwell, tingling or numbness of skin, excessive hairiness, chills, swelling, fatigue, chest pain, back pain, low muscle strength, muscle stiffness, nervousness, skin discolouration, drowsiness, urinary incontinence, cough, limb numbness or weakness, palpitations, abnormal vision, watery or swollen eyes, eye pain, nausea, vomiting, diarrhoea, burping, abdominal pain, runny or blocked nose, dry mouth, increased salivation, altered taste, lip inflammation, sinus pressure/fullness, heat sensation, headache, dizziness, mood swings.
Serious side effects may include: life-threatening allergic reaction, reduced urine output, protein in urine, faecal incontinence, paralysis, swollen/red/sore tongue, sore throat or painful swallowing, heart attack, abnormal heart rate or rhythm, ECG changes, high or low blood pressure, collapse, fainting, shock, confusion, CNS disturbances (tremor, muscle spasms, loss of consciousness, language impairment, loss of smell or taste, seizures, coma), difficulty breathing, asthma. For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
IOMERON®
Active ingredient(s): Iomeprol
Consumer Medicine Information (CMI)
This leaflet provides important information about using IOMERON®. You should also speak to your radiologist, doctor or pharmacist if you would like further information or if you have any concerns or questions about using IOMERON®.
Where to find information in this leaflet:
1. Why am I using IOMERON®?
2. What should I know before I use IOMERON®?
3. What if I am taking other medicines?
4. How do I use IOMERON®?
5. What should I know while using IOMERON®?
6. Are there any side effects?
7. Product details
1. Why am I using IOMERON®?
IOMERON® contains the active ingredient iomeprol. IOMERON® is an iodine-containing dye, called a “contrast medium” and is injected into blood vessels or body cavities to make blood vessels and other parts of the body visible to X-rays.
IOMERON® is used for making blood vessels and other parts of the body visible to X-rays.
2. What should I know before I use IOMERON®?
Warnings
Do not use IOMERON® if:
- you are allergic to iomeprol, or any of the ingredients listed at the end of this leaflet.
- Always check the ingredients to make sure you can use this medicine.
- you suffer from multiple myeloma or Waldenstroem's paraproteinaemia (a disorder of the immuno-globulins-cells involved in the body's natural ability to fight disease).
- you suffer from kidney disease or a combination of severe liver and kidney disease.
- you underwent a myelography procedure during the last 48 hours.
Check with your doctor if you:
- have any other medical conditions such as:
- asthma
- hay fever or food allergy
- overactive thyroid gland or goitre
- diabetes and being treated with metformin (Diabex, Diaformin or Glucophage)
- phaeochromocytoma
- heart disease or problems with your heart, especially if you are elderly
- increased blood pressure
- any disorder or injuries affecting your brain or nervous system
- alcoholism
- addiction to, or dependence on drugs or medicines
- inflammation of the female sex organs
- myasthenia gravis
- sickle-cell disease - take any medicines for any other condition
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
If you received contrast media whilst pregnant your baby may be given a test for hypothyroidism in the period shortly after birth.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Because many substances do pass into human breast milk, stop breastfeeding your baby before you have an injection of IOMERON®, feed the baby with formula milk and do not start breastfeeding again until you have checked with your doctor that it is safe to do so.
Children
- In children, IOMERON® is injected only into the blood vessels.
3. What if I am taking other medicines?
Tell your radiologist, doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with IOMERON® and affect how it works:
- Neuroleptics (medicines used for the treatment of some mental disorders – sometimes also called major tranquillisers).
- Antidepressants (medicines used for the treatment of depression).
- Anticonvulsants (any medicine used for the treatment of epilepsy or fits).
- Certain medicines used to treat diabetes, such as metformin.
- Nephrotoxic drugs (medicines which can have a side effect of damaging the kidneys in certain doses and under certain conditions).
- Corticosteroids (a class of steroid hormones) may promote and affect the signs and symptoms of arachnoiditis (inflammation of arachnoid).
Check with your radiologist, doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect IOMERON®.
Your doctor may ask you not to take certain medicines for 48 hours before receiving an injection of IOMERON®.
4. How do I use IOMERON®?
How much to use
- The strength and amount of IOMERON® you will be given depends upon the parts of your body which are being examined and your body weight. Your radiologist/doctor will inject the correct amount of IOMERON® into a blood vessel or body cavity, before taking the X-ray photographs.
- You will probably be observed for at least 30 minutes after the X-ray.
When to use IOMERON®
- IOMERON® is used to make blood vessels and other parts of the body visible to x-rays, so that x-ray photographs can be taken.
- IOMERON® will be injected into a blood vessel or any other parts of the body by your radiologist/doctor.
If you use too much IOMERON®
You will be given IOMERON® by injection in a hospital environment so it is extremely unlikely you will be given too much.
However, if you experience any side effects after being given IOMERON® tell your radiologist or doctor immediately. Your radiologist or doctor will know how to treat you.
If you think that you have been given too much IOMERON®, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using IOMERON®?
Things you should do
Your radiologist or doctor will advise you what to do before, during and after your procedure.
You should have plenty to drink and can eat normally on the day of your x-ray up to two hours before receiving an injection of IOMERON®.
Driving or using machines
Do not drive a vehicle or operate machinery for at least 24 hours after receiving the injection of IOMERON® as delayed reactions to iodine-containing dyes may occur within this period.
Drinking alcohol
Tell your doctor if you drink alcohol.
Alcohol may increase the risk of seizure when contrast media such as IOMERON® is administered, so caution must be exercised.
Looking after your medicine
- You will not normally be required to store IOMERON®. It will usually be kept by your radiologist or in the radiology or pharmacy department of the hospital where you are having the x-ray.
- Your radiologist should store this product below 25°C and protect it from light. It is advisable to store the product out of reach of ionising radiation.
6. Are there any side effects?
Tell the radiologist/doctor immediately if you are not feeling well after receiving an injection of IOMERON®, during the x-ray procedure and afterwards.
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
General:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
General:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your radiologist, doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What IOMERON® contains
| Active ingredient (main ingredient) | Iomeprol |
| Other ingredients (inactive ingredients) | Trometamol Hydrochloric acid Water |
Do not take this medicine if you are allergic to any of these ingredients.
What IOMERON® looks like
IOMERON® is a sterile solution, which is supplied in vials with rubber stoppers. It is available in the following different strengths and sizes:
IOMERON® 300
30.62 g/50 mL injection vial (AUST R 63955)
61.24 g/100 mL injection vial (AUST R 63957)
IOMERON® 350
35.72 g/50 mL injection vial (AUST R 63960)
71.44 g/100 mL injection vial (AUST R 63961)
142.88 g/200 mL injection vial (AUST R 63963)
Who distributes IOMERON®
Sponsor:
Bracco Pty Ltd
14 Allambie Avenue, East Lindfield
NSW 2070
Australian distributor:
Regional Healthcare Group
Medi-Consumables Pty Ltd
(ACN 001 394 323)
3-11 Primrose Avenue
ROSEBERY NSW 2018
New Zealand distributor:
Regional Health Limited
PO Box 101-140
North Shore Mall Centre
Auckland
New Zealand
This leaflet was prepared in December 2025.
Brand Information
| Brand name | Iomeron |
| Active ingredient | Iomeprol |
| Schedule | S4 |
MIMS Revision Date: 01 July 2020
Notes
Distributed by Regional Health Care Products Group Medi-Consumables Pty. Ltd.
1 Name of Medicine
Iomeprol.
2 Qualitative and Quantitative Composition
Iomeron 300 - Each mL contains iomeprol 612.4 mg.
Iomeron 350 - Each mL contains iomeprol 714.4 mg.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Injection, solution. Clear solution, practically free from visible particles.
4 Clinical Particulars
4.1 Therapeutic Indications
Iomeron is indicated for use for the radiological visualisation of the vascular, renal and other physiological systems in adults and children. See Section 4.2 Dose and Method of Administration for a listing of specific indications for each Iomeron concentration.
4.2 Dose and Method of Administration
In relation to the procedure. Coagulation, flushing of catheters. Non-ionic contrast media have less anticoagulant activity in vitro than ionic media.
Medical personnel performing vascular catheterisation procedures should be aware of this and pay meticulous attention to the angiographic technique and catheter flushing so as to minimise the risk of procedure related thrombosis and embolism.
Observation of the patient. Intravascular administration of contrast media should, if possible, be done with the patient lying down. The patient should be kept under observation for at least 30 minutes after the administration.
Following intrathecal use, the patient should rest with the head and chest elevated for one hour and be kept well hydrated.
Thereafter, he/she may ambulate carefully but bending down must be avoided. If remaining in bed, the head and chest should be kept elevated for a few hours. Patients suspected of having a low seizure threshold should be observed during this period.
Pretesting. Sensitivity test doses are not recommended since severe or fatal reactions to contrast media are not predictable from a patient's history or a sensitivity test.
When examining the subarachnoidal space the total amount of administered contrast substance should be kept as low as possible within the recommended dose interval.
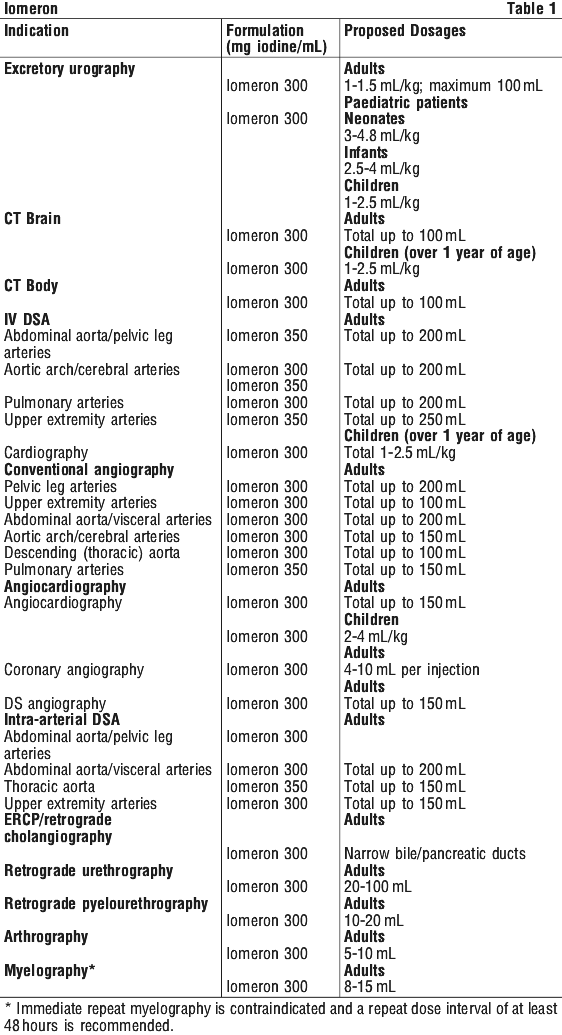
Instructions for use. See Table 1.
4.3 Contraindications
Hypersensitivity to iomeprol or other components in the formulations.
Spinal puncture should not be performed in the presence of significant local or systemic infection where bacteraemia is likely.
Immediate repeat myelography is contraindicated and a repeat dose interval of at least 48 hours is recommended.
Severe renal impairment. Iomeron should not be used in patients with severe impairment of renal function, as the kinetics and safety of Iomeron have not been established in this group of patients. Iomeron is also contraindicated in patients with a combination of severe hepatic and renal impairment which can delay contrast media excretion, therefore predisposing to untoward reactions.
Multiple myeloma, Waldenstroem's paraproteinaemia. The use of the product is generally contraindicated. It is necessary to consider that the presence of myelomatosis or paraproteinaemias is a factor predisposing to renal impairment following intravascular contrast media administration. Adequate hydration is recommended.
4.4 Special Warnings and Precautions for Use
Special warnings - common to all types of administration. In consideration of possible serious side effects, the use of iodine containing contrast media should be limited to cases for which there is a precise need for the examination. The need should be evaluated on the basis of the clinical status of the patient, in particular in relation to pathologies of the cardiovascular, urinary or hepatobiliary systems. The risks of the specific radiographic contrast procedure itself should be carefully evaluated in each patient. Contrast media designed for angiocardiographic procedures should be used in hospitals or clinics equipped and staffed for intensive care in emergencies.
For other more common diagnostic procedures calling for the use of iodinised contrast media, in the institutions, where such procedures are to take place, resuscitation equipment and therapeutic measures should be immediately available.
General. Adverse reactions to iodinated contrast media are more common in patients with a history of allergy, including hay fever, hives and food allergy.
Hypersensitivity or a previous history of a reaction to iodinated contrast media also increase the risk of recurrence of a severe reaction. Hence, before administering Iomeron, the patient should be questioned about his/her allergic history to determine his/her allergic disposition or history of hypersensitivity. A history of allergy, including asthma, while implying a greater than usual risk, is not, however, an absolute contraindication to the use of Iomeron.
Patients should be well hydrated prior to and following administration of any contrast media including Iomeron. The procedure of preparatory dehydration is dangerous and may lead to acute renal failure in patients with advanced renal disease, diabetics and other susceptible non-diabetic patients, particularly the elderly with pre-existing renal disease. The osmotic diuretic effect of contrast media appears to increase dehydration in these patients. High risk patients should therefore be identified and well hydrated before administration of Iomeron, preferably by maintaining i.v. infusion before and during the procedures and until the contrast medium has been cleared by the kidneys.
Special precaution. High concentrations of Iomeron by intra-arterial route in critical circulations (i.e. coronary and cerebral) may, due to their higher viscosity, induce or aggravate ischaemia in these circulations.
Management of severe allergic or cardiovascular reactions. Severe allergic or cardiovascular reactions, including fatalities, have occurred following administration of water soluble iodinated contrast media. Prior to administration of Iomeron, therefore, a planned course of action, including having appropriate and adequate facilities and personnel available, must be in place and be capable of immediate implementation, in case a serious reaction occurs.
Use with caution in the following circumstances. Paediatric use. Young infants (age < 1 year), especially neonates, are particularly susceptible to electrolyte imbalances and haemodynamic alterations. Care should be taken regarding the dosage to be used, the details of the procedure, and the patient's status. Safety and efficacy of Iomeron for use in myelography in children have not been adequately demonstrated.
Thyroid function in infants exposed to iodinated contrast media (ICM) should be evaluated and monitored. Decreased levels of thyroxine (T4) and triiodothyronine (T3) and increased level of thyroid stimulating hormone (TSH) were reported after exposure to ICM in infants, especially preterm infants, which remained for up to a few weeks or more than a month. Thyroid function in infants exposed to ICM should therefore be evaluated and monitored until thyroid function is normalised. Some patients were treated for hypothyroidism.
Use in the elderly. The elderly are at special risk of reactions due to high dosage of contrast media. Myocardial ischaemia, major arrhythmias and extrasystoles are more likely to occur in these patients. The frequently encountered combination of neurological disturbances and severe vascular pathologies constitutes a serious complication.
The probability of acute renal insufficiency is higher in these subjects.
History of hypersensitivity, allergic disposition. It is generally agreed that adverse reactions to iodinated contrast media are more common in patients having a history of allergy, including hay fever, hives and food allergy. Hypersensitivity or a previous history of a reaction to iodinated contrast media also increases the risk of recurrence of a severe reaction with non-ionic media.
Asthmatic patients. The risk of bronchospasm inducing reactions in asthmatic patients is higher after contrast media administration.
Hyperthyroidism, nodular goitre. The release of free inorganic iodine from iodinated molecules, especially from lipid soluble and water soluble ionic compounds, may affect thyroid function. These effects appear to be more evident in patients with hyperthyroidism or goitre. Iodine containing contrast media should not be administered to patients with known hyperthyroidism.
Use in renal impairment. Pre-existing renal impairment may predispose to acute renal dysfunction following intra-arterial or intravenous contrast media administration. Preventive measures include: identification of high risk patients; ensuring adequate hydration before contrast media administration, preferably by maintaining i.v. infusion before and during the procedures and until the contrast media has been cleared by the kidneys; avoiding whenever possible the administration of nephrotoxic drugs or major surgery or procedure such as renal angioplasty until the contrast media has been cleared; postponing a new contrast agent examination until renal function returns to pre-examination levels.
Diabetes mellitus. The presence of renal damage in diabetic patients is one of the factors predisposing to renal impairment following intravascular contrast media administration. This may precipitate lactic acidosis in patients who are taking biguanides. As a precaution, biguanides should be stopped 48 hours prior to the contrast media examination and reinstated only after control of renal function has been regained.
Phaeochromocytoma. These patients may develop severe (rarely uncontrollable) hypertensive crises following intravascular contrast media usage during radiological procedures. Premedication with alpha-receptor blockers is recommended because of the risk of blood pressure crises.
Sickle cell disease. Contrast media may promote sickling in individuals who are homozygous for sickle cell disease. Adequate hydration is recommended.
Myasthenia gravis. The administration of iodinated contrast media may aggravate myasthenia signs and symptoms.
Severe cardiovascular disease. There is an increased risk to severe reactions in individuals with severe cardiac disease and particularly in those with heart failure and coronary artery disease. The intravascular contrast media injection may precipitate pulmonary oedema in patients with manifest or incipient heart failure, whereas contrast media administration, in pulmonary hypertension and heart valvular diseases, may lead to pronounced haemodynamic changes. Ischaemic ECG changes and major arrhythmias are more common in elderly patients and in those with pre-existing cardiac disease - their frequency and severity appear to be related to the severity of cardiac impairment. Severe and chronic hypertension may increase the risk of renal damage following intravascular contrast media administration and the risks associated with the catheterisation procedure.
CNS disorders. Particular care should be paid to the intravascular and intrathecal administration of contrast media in patients with acute cerebral infarction, acute intracranial haemorrhage, and conditions involving blood-brain-barrier (BBB) damage, brain oedema and acute demyelination. Intrathecal administration of contrast media should be undertaken with caution in patients with a likelihood of reduced seizure threshold, since there is no barrier to contrast media in the CSF penetrating the brain. The presence of intracranial tumours or metastases and a history of epilepsy may increase the probability of the occurrence of convulsive seizures. Neurological symptoms due to degenerative, inflammatory or neoplastic cerebrovascular pathologies may be exacerbated by intravascular contrast media administration. Vasospasm and consequent cerebral ischaemic phenomena may be caused by intravascular injections of contrast media. Patients with symptomatic cerebrovascular diseases, recent stroke or frequent transient ischaemic attack have an increased risk of transient neurological complications.
Alcoholism. Acute and chronic alcoholism have been proven both experimentally and clinically to increase BBB permeability. This facilitates the passage of iodinated agents into the cerebral tissue, possibly leading to CNS disorders. Caution must be exercised in the intravascular and intrathecal administration of contrast media in alcoholics because of the possibility of a reduced seizure threshold.
Drug addiction. Caution must be exercised in drug addicts because of the possibility of a reduced seizure threshold.
Check the following before use. Hydration. Any severe disorders of water and electrolyte balance should be corrected. Adequate hydration must be ensured before examination, especially in patients with multiple myeloma, diabetes mellitus, polyuria, oliguria, hyperuricemia, as well as in babies, small children and elderly patients.
Dietary suggestion. Unless otherwise instructed by the doctor, a normal diet may be maintained on the day of the examination. Adequate fluid intake must be ensured. However, for two hours prior to the procedure the patient should refrain from eating.
History of hypersensitivity. In patients with an allergic disposition, known hypersensitivity to iodinated contrast media and a history of asthma, premedication with antihistamines and/or corticoids may be considered in order to prevent possible anaphylactoid reactions.
Anxiety. Pronounced states of excitement, anxiety and pain can be the cause of side effects or intensify contrast related reactions. These patients may be given a sedative.
Effects of laboratory tests. Thyroid function tests. Following administration of iodinated contrast media, the capacity of the thyroid tissue to take up radioisotopes for the diagnosis of thyroid disorders is reduced for up to two weeks, or even longer in individual cases. These findings have not been associated with clinical manifestations. Thyroid function tests not depending on iodine estimations, e.g. T3 resin uptake and total or free thyroxine (T4) assays are not affected.
Laboratory tests. High concentrations of contrast media in serum and urine can interfere with laboratory test results of bilirubin, proteins or inorganic substances (e.g. iron, copper, calcium, phosphate).
4.5 Interactions with Other Medicines and Other Forms of Interactions
No specific clinical studies on interactions with other drugs have been conducted with iomeprol. Nevertheless, in analogy to other iodinated contrast media, the following measures should be taken in respect of the drugs mentioned below.
Neuroleptics and phenothiazine derivative anti-depressants. Treatment should be discontinued 48 hours before the examination because they reduce the seizure threshold. Treatment should not be resumed until 24 hours post-procedure.
Anti-convulsants. Therapy must not be discontinued and should be administered in optimal dosage.
Oral cholecystographics. Interactions of renally excreted contrast media with oral cholecystographics contrast media are unlikely to occur.
Nephrotoxic drugs. Whenever possible, the administration of nephrotoxic drugs should be delayed until the contrast media has been cleared.
Biguanides. Intravascular contrast media administration may precipitate lactic acidosis in diabetic patients with renal damage who are taking biguanides.
Biguanides should be stopped 48 hours prior to contrast media examination and reinstated only after control of renal function has been regained.
Corticosteroids. Epidural and intrathecal corticosteroids should never be concurrently administered intrathecally when iodinated contrast media are used, because corticosteroids may promote and affect the signs and symptoms of arachnoiditis.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Fertility was not impaired in rats with daily exposures (based on the dosage) up to more than twice that recommended in patients.
Use in pregnancy. (Category B3)
There are no controlled studies in pregnant women to confirm the safety of iomeprol.
In animal studies, after pregnant rats were dosed with radio-labelled iomeprol i.v., maternal tissues, placentae and fetuses showed a comparable distribution of radioactivity.
Iomeprol was not teratogenic at doses of 2 or 4 g I/kg i.v., administered daily, in rabbits or rats, respectively, during pregnancy. However, there were slight increases in the frequency of skeletal anomalies in both rabbits and rats, although no statistically significant differences or dose related responses were observed.
Iomeprol should be used during pregnancy only if clearly needed. Infants born to women who received iodinated contrast media while pregnant should have testing for hypothyroidism in the neonatal period. Some patients were treated for hypothyroidism. Also see Paediatric use.
Use in lactation. There is no information on the excretion of iomeprol in the milk of animals. For testing perinatal and postnatal toxicity, rats were dosed with iomeprol at 0.6, 1.5 or 4 g I/kg i.v. daily during the final period of gestation and throughout lactation. Neonatal mortality was increased, and the survival index, viability index and mean bodyweight of the pups were lower at a dose of 4 g I/kg.
Since no data are available in respect of the adverse effects in nursing infants, use of iomeprol in lactation is not recommended. Also see "Paediatric use".
4.7 Effects on Ability to Drive and Use Machines
No data are available but, since delayed reactions can rarely occur after administration of iodinated contrast media, driving or operating machinery is not advisable for the first 24 hours following contrast medium examination.
4.8 Adverse Effects (Undesirable Effects)
The use of iodinated compounds may cause untoward effects which are generally of a mild to moderate nature, as well as more severe ones, with fatal anaphylactoid reactions. Symptoms are, however, usually mild, of short duration and self limiting. More severe neurological sequelae may be the result of complications of a pre-existing pathology. In myelography common adverse events are headache, nausea, vomiting and pain.
General or local sensation of heat is the most frequent complaint associated with administration of Iomeron.
Body as a whole. Common. Pain at the injection site, headache.
Uncommon. Abdominal, back and chest pain, chills, fatigue, fever, headache, malaise, mucous disorder, oedema.
Rare. Fatal anaphylactoid reactions.
Cardiovascular. Common. Flushing.
Uncommon. Angina pectoris, arrhythmia, bradycardia, ECG change, tachycardia, hypertension, hypotension, cardiac arrest, palpitation, peripheral vascular disorder, shock, syncope.
Rare. Cardiac failure, peripheral vasodilation with pronounced hypotension, hypertension, tachycardia or bradycardia, cyanosis, dyspnoea and circulatory collapse.
Central nervous system. Common. Disturbance of taste sensation.
Uncommon. Confusion, dizziness, dry mouth, emotional lability, hemiplegia, hypertonia, hypotonia, increased salivation, nervousness, neuropathy, paraesthesia, somnolence.
Rare. The intravenous or intra-arterial injection of contrast agents may cause symptoms related to CNS disturbances: tremor, muscular spasms, mental confusion, loss of consciousness, disturbances of visual field, muscular palsies, aphasia, convulsive seizures, and coma.
Dermatological. Uncommon. Angioedema, hirsutism, pruritus, rash, skin discolouration, subcutaneous nodule, sweating, urticaria, widespread erythema.
Skin reactions may be present in the form of diversified rashes or diffuse pomphus formation, and sometimes itching. Pain, haemorrhage and oedema may arise at the site of injection. In the case of extravasation a tissue reaction may ensue, but this is rare.
Endocrine. Uncommon. Thyroid function tests indicative of hypothyroidism or transient thyroid suppression have been uncommonly reported following iodinated contrast media administration to adult and paediatric patients, including infants. Some patients were treated for hypothyroidism.
Gastrointestinal. Common. Nausea.
Uncommon. Cheilitis, diarrhoea, dysphagia, eructation, glossitis, vomiting.
Ophthalmological. Uncommon. Abnormal vision, conjunctivitis, eye disorder, eye pain.
Muscular-skeletal. Uncommon. Asthenia.
Respiratory system. Uncommon. Apnoea, asthma, worsening of cough, dyspnoea, pharyngitis, rhinitis, sinusitis, upper respiratory infection, laryngeal oedema.
Urogenital. Uncommon. Incontinence, urinary urgency.
Rare. A transient renal failure with oliguria, proteinuria and an increase of serum creatinine level may arise, particularly in patients with pre-existing impairment of renal function.
Post marketing data. The following adverse effects have been reported in less than 3 out of 10,000 patients (rare < 0.03%).
Intravascular and intrathecal administration. Body as a whole. Shock, malaise, fatigue, hot flushes, facial flushing, cold sweat, local sensation of coldness, taste abnormality, thirst, injection site reaction.
Nervous. Hyperkinesia, encephalopathy, paralysis, oculomotor nerve paralysis, paraesthesia, dysarthria, giddiness, dysphonia, faecal incontinence, cerebral oedema.
Cardiovascular. Cardiac arrest, myocardial infarction, angina pectoris, extrasystoles, arrhythmia, ventricular or atrial fibrillation, tachycardia, palpitation, atrioventricular block, abnormal ECG, elevation of ST.
Respiratory. Respiratory arrest, pulmonary oedema, adult respiratory distress syndrome (ARDS), bronchospasm, asthma, pharynx oedema, laryngeal stridor, rhinitis, coughing, hyperventilation, hypoxia, pharyngolaryngeal abnormal sensation.
Skin and appendages. Angioedema, eczema, urticaria, wheals.
Vascular (extracardiac). Cerebrovascular disorder, transient ischaemic attack.
Gastrointestinal disorders. Acute pancreatitis, ileus, diarrhoea, abdominal pain, salivation, dysphagia.
Urogenital. Urinary incontinence, increase in blood urea nitrogen (BUN).
Special senses. Parosmia.
Vision. Transient cortical loss of vision, vision disturbance, conjunctivitis, lacrimation, photopsia, photophobic sensation.
Musculo-skeletal. Arthralgia, muscle stiffness.
Psychiatric. Amnesia, anorexia, anxiety, somnolence.
Liver and biliary tract. Liver function tests abnormalities.
Platelet, bleeding and clotting. Thrombocytopenia.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Overdose may lead to life threatening adverse effects mainly through effects on the pulmonary and cardiovascular system.
Treatment of overdosage is directed toward the support of all vital functions, and prompt institution of symptomatic therapy.
Iomeprol does not bind to plasma or serum proteins and is therefore dialysable.
Contact the Poisons Information Centre on 13 11 26 for advice on management.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Iomeron is a sterile aqueous contrast medium solution for intravascular and body cavities administration. The active ingredient of Iomeron formulations is iomeprol, a tri-iodinated, non-ionic contrast agent, and is indicated for use in X-ray examinations.
Animal studies. Results from studies in rats, mice and dogs demonstrate that iomeprol has an acute intravenous or intra-arterial toxicity similar to that of the other non-ionic contrast media, as well as a good systemic tolerability after repeated intravenous administrations in rats and dogs. After intravenous administration in rats iomeprol is distributed between plasma and the extracellular space. It does not bind to plasma proteins. It is not metabolised and is eliminated almost exclusively through the kidneys. In the rat, 94% of the administered dose is found unchanged in the urine within the first 8 hours.
Clinical trials. The efficacy and safety of Iomeron was studied in 31 controlled trials and over 60 uncontrolled trials for the indications listed below. In 22 controlled studies, safety was assessed for 24 hours after contrast administration, by physical examinations, monitoring of vital signs, patient monitoring of adverse events, ECG controls, and clinical laboratory investigations. Renal function was monitored to 72 hours after Iomeron administration in 3 studies which involved approximately 60 adults and 60 children, but was not assessed for periods longer than 72 hours in any study.
In myelography, the safety and efficacy of Iomeron following lumbar injection has been demonstrated. Other sites of injection have not been adequately assessed.
5.2 Pharmacokinetic Properties
The pharmacokinetic profile of intravascularly injected iomeprol was determined in 18 healthy volunteers who received the contrast medium intravenously at a concentration of 400 mg iodine/mL at three dose levels: 50 mL, 100 mL and 200 mL.
The plasma concentration curves, expressed as a function of time, showed the plasma kinetics are not dose dependent and are well described as a two compartment model with a rapid phase for drug distribution on and a slower phase for drug elimination. The mean half-lives of the distribution and elimination phases of iomeprol were 23 ± 14 minutes and 109 ± 20 minutes, respectively, with excretion in the urine of approximately 50% within the first 2 hours, 83% within 8 hours and 90% within 96 hours of administration.
Intrathecal administration. After intrathecal administration, iomeprol is completely absorbed from the cerebrospinal fluid in about 3 to 6 hours. The half-life of elimination is between 8 to 11 hours and is independent from the dose. Plasma concentrations were quantifiable up to 24 hours in 93% of the patients.
It is rapidly excreted from the body through the kidney as unchanged iomeprol. The majority of urinary excretion occurs in the first 24 hours post-dose with smaller percentage excreted during the first 24-48 hour period.
Renal impairment. Iomeprol is mainly eliminated by the kidney through glomerular filtration. The mean half-lives of elimination for subjects with mild renal impairment is of 3.67 hours; for those with moderate impairment 6.9 hours, and for those with severe impairment of 15.1 hours. In patients with mild and moderate renal impairment the cumulative urinary excretion of iomeprol between 4 and 8 hours, expressed as percentage of administered intravenous dose, is approximately 50% of the injected dose. In patients with severe renal impairment 50% of the injected dose was recovered in the urine between 16 and 84 hours after injection.
In renal impaired patients, the additional route of elimination is the biliary route. Mean cumulative 120 hour fecal excretion of the total iomeprol dose administered averages from 1.6% in normal subjects to 7.2% in severe renal impaired patients.
Haemodialysis. After a single dialysis session, approximately 58% of the dose is recovered in the dialysate.
5.3 Preclinical Safety Data
Genotoxicity. Iomeprol, with or without metabolic activation as appropriate, was not mutagenic in the Salmonella typhimurium reverse mutation assay and Schizosaccharomyces pombe assay. Iomeprol was not clastogenic in the rat micronucleus test in vivo or in the chromosomal aberration test with the Chinese hamster cells (with or without S-9) in vitro, and was not genotoxic in the Saccharomyces cerevisiae-gene conversion and DNA repair test.
Carcinogenicity. There are no data on the carcinogenicity of iomeprol.
6 Pharmaceutical Particulars
6.1 List of Excipients
Trometamol 1 mg, hydrochloric acid (d=1.18) 0.24 mg and water for injections q.s. to 1 mL.
6.2 Incompatibilities
In order to avoid possible incompatibilities, contrast media must not be mixed with other drugs.
See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Iomeron is available in vials containing contrast media solution and are not intended for the withdrawal of multiple doses.
The rubber stopper should never be pierced more than once. The use of proper withdrawal cannulas for piercing the stopper and drawing up the contrast medium is recommended. The contrast medium should not be drawn into the syringe until immediately before use. Solutions not used in one examination session must be discarded.
Store below 25°C. Protect from light. Although the sensitivity of iomeprol to X-rays is low, it is advisabIe to store the product out of reach of ionising radiation.
6.5 Nature and Contents of Container
Iomeron is available in the following strengths and pack sizes:
Iomeron 300: 50 mL, 100 mL vials.
Iomeron 350: 50 mL, 100 mL, 200 mL vials.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Iomeprol injection contains N,N'-bis-(2,3-dihydroxypropyl)- 5-[(hydroxyacetyl)-methylamino]- 2,4, 6-triiodo- 1,3-benzene-dicarboxamide and has a molecular weight of 777.09. Iomeprol is a white crystalline powder, which is highly water soluble. Organically bound iodine: 49% of its molecular weight.
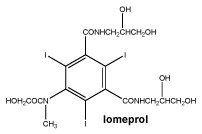
The iodine strength specific physicochemical characteristics of Iomeron for injection are as follows. See Table 2.
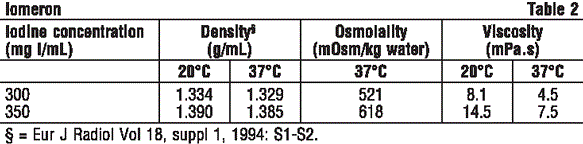
Chemical structure.
7 Medicine Schedule (Poisons Standard)
Not scheduled. Not considered by committee.
Date of First Approval
29 December 2000
Date of Revision
09 June 2020
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.