Isopto Carpine
Brand Information
| Brand name | Isopto Carpine |
| Active ingredient | Pilocarpine hydrochloride |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Isopto Carpine.
Summary CMI
Isopto Carpine®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Isopto Carpine?
Isopto Carpine contains the active ingredient Pilocarpine hydrochloride. Isopto Carpine is used to treat glaucoma, an eye condition that damages the optic nerve of your eye.
For more information, see Section 1. Why am I using Isopto Carpine? in the full CMI.
2. What should I know before I use Isopto Carpine?
Do not use if you have ever had an allergic reaction to Isopto Carpine or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Isopto Carpine? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Isopto Carpine and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Isopto Carpine?
- Follow the instructions provided by your doctor.
- Use 1-2 drops in the affected eye(s) 3-4 times daily.
More instructions can be found in Section 4. How do I use Isopto Carpine? in the full CMI.
5. What should I know while using Isopto Carpine?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Isopto Carpine? in the full CMI.
6. Are there any side effects?
Common side effects include eye pain, eye irritation, blurred vision, headache, vision problems, dizziness, feeling that something is in your eye(s).
Full CMI
Isopto Carpine® Eye Drops 1%, 2%, 4%
Active ingredient: Pilocarpine hydrochloride
Consumer Medicine Information (CMI)
This leaflet provides important information about using Isopto Carpine. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Isopto Carpine.
Where to find information in this leaflet:
1. Why am I using Isopto Carpine?
2. What should I know before I use Isopto Carpine?
3. What if I am taking other medicines?
4. How do I use Isopto Carpine?
5. What should I know while using Isopto Carpine?
6. Are there any side effects?
7. Product details
1. Why am I using Isopto Carpine?
Isopto Carpine contains the active ingredient Pilocarpine hydrochloride, which belongs to a class of drugs called Parasympathomimetics.
Isopto Carpine is used to treat glaucoma, an eye condition that damages the optic nerve of your eye. It is usually caused by a buildup of the fluid which flows through the eye. It is often associated with increased pressure of the eye which damages the optic nerve.
Isopto Carpine eye drops help to lower the increased eye pressure by increasing the amount of fluid that drains from the eye.
2. What should I know before I use Isopto Carpine?
Warnings
Do not use Isopto Carpine:
- If you are allergic to Pilocarpine hydrochloride, or any of the ingredients listed at the end of this leaflet. Always check the ingredients to make sure you can use this medicine. Symptoms of allergic reaction may include shortness of breath, difficulty breathing, swelling of the face, lips, tongue or other parts of the body, rash, hives.
- If you suffer from any symptoms such as eye pain, redness, sensitivity to light or blurred vision.
- If you have a certain type of condition that can cause "pupillary constriction" (pinpoint pupils).
- In children unless specifically advised to by your doctor.
Check with your doctor if you have:
- Asthma, symptoms such as shortness of breath, coughing, wheezing
- Ulcers in stomach or intestine with symptoms such as indigestion, feeling sick, vomiting, pain in upper part of stomach
- Thyroid problems
- Stomach cramps
- Trembling, rigid posture, slow movements and a shuffling, unbalanced walk
- Difficulty urinating, fever, feeling sick, vomiting (an obstruction of the urinary tract)
- Recent history of heart attack
- High or low blood pressure
- Problems with your retina with symptoms such as blurred vision, flashes of light.
- Diseases of cornea, symptoms include eye pain, feeling that something is in your eye(s), sensitivity to light.
- Symptoms such as pink or red discoloration of eye(s), itchy or burning eyes, excessive tearing.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Avoid use of Isopto Carpine during pregnancy.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Isopto Carpine may affect breast milk production. Your doctor will advise you regarding the possible risks and benefits of using Isopto Carpine during breastfeeding.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Isopto Carpine.
4. How do I use Isopto Carpine?
How much to use
- Follow the instructions provided and use Isopto Carpine until your doctor tells you to stop.
- Use 1-2 drops in the affected eye(s) 3-4 times daily.
When to use Isopto Carpine
- Isopto Carpine should be used every day, at about the same time each day unless your doctor tells you otherwise.
How to use Isopto Carpine
- Sitting or lying down position might make the process simpler.
- Remove contact lenses if you are wearing them before using the eye drops.
- Shake the bottle well prior to use.
Follow the steps below to use Isopto Carpine:
- Wash your hands thoroughly with soap and water.
- Remove the cap from the bottle.
- Mix the contents of the bottle by inverting 5 to 10 times.
- Hold the bottle upside down in one hand between your thumb and middle finger (refer diagram 1).
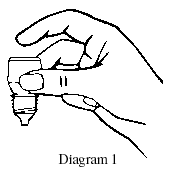
- Tilt your head back, gently pull down the lower eyelid of your eye to form a pouch/pocket.
- Place the tip of the bottle close to your eye. Do not touch the tip to your eye as this may cause injury to your eye.
- Do not touch the dropper tip to your eyelid or surrounding areas or any surface to avoid contamination of the dropper tip and solution.
- Release one drop into the pouch/pocket formed between your eye and eyelid by gently tapping or pressing the base of the bottle with your forefinger (refer diagrams 2 and 3).
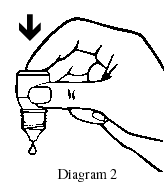

- Gently close your eye(s). Do not blink or rub your eye(s).
- When your eye is closed, place your index finger against the inside corner of your eye and press against your nose for about two minutes. This will help to prevent the medicine from draining through the tear duct to the nose and throat, from where it can be absorbed into other parts of your body and may result in less side effects.
- This will also help to prevent the unpleasant taste sensation that some people experience while using these eye drops.
- If necessary, repeat the above steps for the other eye.
- You may feel initial discomfort or a slight burning sensation in the eye shortly after using Isopto Carpine. If it continues, or is uncomfortable, contact your doctor or pharmacist.
- It is normal for a small amount of eye drops to spill onto your cheek since your eyelids can only hold less than one drop at a time. Wipe away any spillage with a tissue.
- If you want to use any other eye drops, wait at least 5 minutes after using Isopto Carpine.
- Replace the cap on the bottle and close it tightly.
- Always keep the bottle tightly closed when not in use.
- Wash your hands again with soap and water to remove any residue.
- Wait for 15 minutes before putting your contact lenses in.
- If you are being changed from one eye drop to another, follow your doctor's instructions carefully as to when to stop the old drops and when to start the new drops.
- Discard Isopto Carpine eye drops 4 weeks after opening.
If you forget to use Isopto Carpine
Isopto Carpine should be used regularly at the same time each day. If you miss your dose at the usual time, use it as soon as you remember, and then go back to using it as you would normally.
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Do not take a double dose to make up for the dose you missed.
If you use too much Isopto Carpine
If you think that you have used too much Isopto Carpine, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
If you accidentally use several Isopto Carpine eye drops in your eyes, wash your eyes with lukewarm water.
5. What should I know while using Isopto Carpine?
Things you should do
- Do not put Isopto Carpine into your eye(s) while wearing contact lenses.
- Remove soft contact lenses before using Isopto Carpine. Benzalkonium chloride, one of the ingredients may cause eye irritation and is known to discolour soft contact lenses.
- Put your lenses back in 15 minutes after using the eye drops.
Call your doctor straight away if you:
- develop any signs of an allergic reaction.
- become pregnant, plan to become pregnant or if you are breast feeding while using Isopto Carpine.
- take any medicines for any condition.
- have cornea problems, symptoms such as eye pain, feeling that something is in your eye(s), sensitivity to light.
Remind any doctor, dentist, pharmacist or specialist you visit that you are using Isopto Carpine.
Things you should not do
- Do not stop using this medicine suddenly.
- Do not give Isopto Carpine to anyone else, even if they seem to have the same condition as you.
- Do not touch the dropper tip to any surface.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Isopto Carpine affects you.
Isopto Carpine may cause blurred vision and difficulty in changing from very bright light to darkness.
Take caution while driving at night or while performing hazardous tasks in poor light.
Looking after your medicine
- Store below 25°C.
- Do not freeze
- Protect from light.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
When to discard your medicine
Discard Isopto Carpine eye drops 4 weeks after opening.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Eye Problems:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Eye problems:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Isopto Carpine contains
| Active ingredient (main ingredient) | Pilocarpine hydrochloride 1%, 2%, and 4% |
| Other ingredients (inactive ingredients) | Hypromellose 0.5%, benzalkonium chloride 0.01%, boric acid, sodium citrate and purified water. |
| Potential allergens | NA |
Do not take this medicine if you are allergic to any of these ingredients.
What Isopto Carpine looks like
Isopto Carpine is a clear eye drops solution which comes in 15 mL LDPE bottle dispenser.
Australian Registration Numbers
AUST R 25294 (1%)
AUST R 25295 (2%)
AUST R 25297 (4%).
Who distributes Isopto Carpine
This product is supplied in Australia by:
Novartis Pharmaceuticals Australia
Pty Limited
ABN 18 004 244 160
54 Waterloo Road
Macquarie Park NSW 2113
Telephone: 1800 671 203
Web Site: www.novartis.com.au
This product is supplied in New Zealand by:
Novartis New Zealand Limited
PO Box 99102
Newmarket
Auckland 1149
New Zealand
Free Phone: 0800 354 335.
® Registered Trademark
© Novartis Pharmaceuticals
Australia Pty Limited 2025
This leaflet was prepared in November 2025.
Internal document code:
iso091123c_V2 based on PI iso091123i
Brand Information
| Brand name | Isopto Carpine |
| Active ingredient | Pilocarpine hydrochloride |
| Schedule | S4 |
MIMS Revision Date: 01 December 2023
1 Name of Medicine
Pilocarpine hydrochloride.
2 Qualitative and Quantitative Composition
Pilocarpine hydrochloride 1%, 2%, and 4%, hypromellose 0.5%, benzalkonium chloride 0.01%, boric acid, sodium citrate and purified water.
3 Pharmaceutical Form
Isopto Carpine 1%/15 mL clear eye drops solution.
Isopto Carpine 2%/15 mL clear eye drops solution.
Isopto Carpine 4%/15 mL clear eye drops solution.
4 Clinical Particulars
4.1 Therapeutic Indications
In the treatment of glaucoma.
4.2 Dose and Method of Administration
Topically, 1-2 drops in the eye(s) 3-4 times daily.
4.3 Contraindications
Miotics are contraindicated in conditions where pupillary constriction is undesirable such as in acute iritis or anterior uveitis.
Hypersensitivity to the active substance or to any of the excipients.
4.4 Special Warnings and Precautions for Use
Identified precautions. Sensitivity is infrequently observed. If reaction occurs, discontinue use.
Retinal detachment has been reported when miotics are used in susceptible individuals, such as young patients with myopia or patients with history of retinal detachment.
Miotics should be avoided in acute inflammatory diseases of the anterior chamber.
A paradoxical rise in intraocular pressure may be observed in patients with severely compromised trabecular outflow.
Caution is advised in the presence of corneal or conjunctival damage to avoid excessive penetration which can produce systemic toxicity.
Isopto Carpine should be used with caution in patients with acute cardiac failure, bronchial asthma, peptic ulcer, hyperthyroidism, gastro-intestinal spasms, Parkinson's disease, urinary tract obstruction, recent myocardial infarction, hypertension and hypotension due to the risk of exacerbating these conditions.
Isopto Carpine contains benzalkonium chloride which may cause eye irritation and is known to discolour soft contact lenses. Avoid contact with soft contact lenses. Patients must be instructed to remove contact lenses prior to the application of Isopto Carpine and wait at least 15 minutes before reinsertion.
Nasolacrimal occlusion or gently closing the eyelid after administration is recommended. This may reduce the systemic absorption of medicinal products administered via the ocular route and result in a decrease in systemic adverse reactions.
Use in the elderly. No data available.
Paediatric use. No data available.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
No interaction studies have been performed.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Studies have not been performed to evaluate the effect of topical ocular administration of Isopto Carpine on fertility.
Use in pregnancy. (Category B3)
There are no or limited amount of data from the use of Isopto Carpine in pregnant women. Animal studies have, however, showed harmful effects of systemic pilocarpine exposure with respect to reproductive toxicity in rats. There are no adequate and well controlled studies in pregnant women. As a precautionary measure, it is preferable to avoid the use of Isopto Carpine during pregnancy.
Use in lactation. It is unknown whether pilocarpine is excreted in human milk. However, excretion in breast milk should be expected. There is also no information on the safety of pilocarpine ophthalmic formulations used during breastfeeding. However, a risk to the suckling child cannot be excluded. A decision must be made whether to discontinue breastfeeding or to abstain from Isopto Carpine, taking into account the benefit of breastfeeding for the child and the benefit of therapy for the woman.
4.7 Effects on Ability to Drive and Use Machines
Isopto Carpine has a major influence on the ability to drive and use machines. Miosis may cause blurred vision and difficulty in dark adaptation. Patients should be advised to exercise caution while driving at night or while performing hazardous tasks in poor light.
4.8 Adverse Effects (Undesirable Effects)
The following adverse reactions are classified according to the following convention: very common (≥ 1/10), common (≥ 1/100 to < 1/10), uncommon (≥ 1/1,000 to < 1/100), rare (≥ 1/10,000 to < 1/1,000), very rare (< 1/10,000), or not known (cannot be estimated from the available data) according to system organ classes. Within each frequency-grouping, adverse reactions are presented in order of decreasing seriousness. The adverse reactions have been observed during clinical trials and post-marketing experience with Isopto Carpine.
Nervous system disorders. Very common (≥ 10%): headache.
Common (≥ 1% to < 10%): dizziness.
Eye disorders. Very common (≥ 10%): vision blurred.
Common (≥ 1% to < 10%): visual impairment, visual acuity reduced, eye pain, photopsia, vitreous floaters, myodesopsia, eye irritation, ocular hyperaemia.
Uncommon (≥ 0.1% to < 1%): retinal tear, vitreous haemorrhage, eyelid oedema, miosis, vitreous detachment, glare, foreign body sensation in eyes.
Not known: intraocular pressure increased, corneal oedema.
Gastrointestinal disorders. Common (≥ 1% to < 10%): nausea.
Not known: vomiting.
Slight ciliary spasms may occur with resultant temporary reduction of visual acuity.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
An ocular overdose of Isopto Carpine can be flushed from the eye(s) with lukewarm water. In case of overdose, symptoms of toxicity may include: headache, salivation, sweating, syncope, bradycardia, hypotension, abdominal cramps, vomiting, asthma and diarrhoea. Treatment of overdose is supportive. In cases of severe systemic toxicity therapy with anticholinergics may be necessary.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Lowers intraocular pressure.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
No data available.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
See Section 2 Qualitative and Quantitative Composition.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Do not freeze. Protect from light.
Discard container 4 weeks after opening.
6.5 Nature and Contents of Container
15 mL LDPE bottle dispenser.
'How to Use' is supplied with this product.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure.
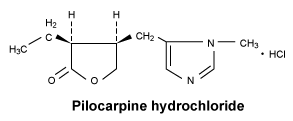
7 Medicine Schedule (Poisons Standard)
Prescription Only Medicine (Schedule 4).
Date of First Approval
15 October 1991
Date of Revision
09 November 2023
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.