Isovue
Brand Information
| Brand name | Isovue |
| Active ingredient | Iopamidol |
| Schedule | Unscheduled |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Isovue.
Summary CMI
ISOVUE®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using ISOVUE®?
ISOVUE® contains the active ingredient Iopamidol. ISOVUE® is used for making blood vessels and other parts of the body visible to X-rays.
For more information, see Section 1. Why am I using ISOVUE®? in the full CMI.
2. What should I know before I use ISOVUE®?
Do not use if you have ever had an allergic reaction to ISOVUE® or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use ISOVUE®? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with ISOVUE® and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use ISOVUE®?
- ISOVUE® is given as an injection before the X-ray procedure. Your radiologist will determine the correct dose for you.
More instructions can be found in Section 4. How do I use ISOVUE®? in the full CMI.
5. What should I know while using ISOVUE®?
| Things you should do |
|
| Driving or using machines |
|
For more information, see Section 5. What should I know while using ISOVUE®? in the full CMI.
6. Are there any side effects?
Less serious side effects of ISOVUE® may include: flushing, hives or rash, skin blisters, muscle pain or weakness, involuntary movements, leg weakness, sweating, fever, chills, very low body temperature, injection-site reactions, chest tightness, feeling unwell, tingling or numbness, wheezing or coughing, difficulty swallowing, difficulty urinating, vivid dreams, drowsiness, unusual bruising, reduced sensitivity, back or leg pain, shoulder pain, cold extremities, vision disturbances, eye irritation or swelling, conjunctivitis, nausea, vomiting, abdominal pain, loss of appetite, diarrhoea, heartburn, altered taste, nasal congestion or runny nose, sneezing, facial pressure, dry mouth, sore throat, palpitations or fast heartbeat, headache, dizziness, emotional stress.
Serious side effects may include: allergic reactions, bluish skin, kidney problems, neurological disturbances, blindness, dilated pupils, abnormal eye movements, rectal bleeding, swallowing difficulty with tongue swelling/redness, chest pain from angina, blood pressure changes, abnormal heart rhythms, circulatory collapse, heart attack, vein inflammation, coagulation problems, facial or throat swelling, faintness, restlessness, seizures, confusion, meningitis or meningitis-like reactions, agitation, memory loss, poor coordination, hallucinations, severe facial nerve pain, tinnitus, shortness of breath, asthma, fluid in the lungs, swelling of the voice box, hypothyroidism.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
ISOVUE®
Active ingredient(s): [iopamidol]
Consumer Medicine Information (CMI)
This leaflet provides important information about using ISOVUE®. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using ISOVUE®.
Where to find information in this leaflet:
1. Why am I using ISOVUE®?
2. What should I know before I use ISOVUE®?
3. What if I am taking other medicines?
4. How do I use ISOVUE®?
5. What should I know while using ISOVUE®?
6. Are there any side effects?
7. Product details
1. Why am I using ISOVUE®?
ISOVUE® contains the active ingredient iopamidol. ISOVUE® is an iodine-containing dye, called a “contrast medium”. ISOVUE® is given as an injection before the X-ray procedure.
ISOVUE® is used for making blood vessels and other parts of the body visible to X-rays.
2. What should I know before I use ISOVUE®?
Warnings
Do not use ISOVUE® if:
- you are allergic to iopamidol, or any of the ingredients listed at the end of this leaflet, or any other iodine-containing contrast media.
- Always check the ingredients to make sure you can use this medicine.
- You suffer from:
- A condition affecting the thyroid gland
- Decompensated cardiac failure or insufficiency
- A recent stroke or heart attack
- Any infection or open injury in or near the area to be examined
- An infection and ISOVUE® is to be injected into your spinal cord - If ISOVUE® is to be injected into your spinal cord within 48 hours of the first injection
Check with your radiologist or doctor if you:
- have any other medical conditions:
- heart disease or disease of the blood vessels
- respiratory conditions
- kidney disease or urinary problems
- any disorder or injuries affecting the brain or nervous system
- multiple myeloma or myelomatosis
- diabetes
- gout
- liver disease
- phaeochromocytoma
- sickle cell disease
- alcoholism
- abscesses
- homocystinuria - take any medicines for any other condition.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
It is not known if ISOVUE® harms the developing baby. ISOVUE® should therefore be used during pregnancy only if your doctor decides it is essential for your health. If you received contrast media whilst pregnant, your baby may be given a test for hypothyroidism in the period shortly after birth.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
It is not known if ISOVUE® passes into human milk. Stop breastfeeding your baby before you have an injection of ISOVUE® and do not start breastfeeding again until you have checked with your doctor that it is safe to do so.
Children
- In children, ISOVUE® is injected only into the blood vessels.
Elderly
- The elderly require special care when receiving an injection of ISOVUE®
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
In particular, you should tell your radiologist or doctor if you are taking any of the following:
- antidepressants
- antinauseants
- antihistamines
- drugs that act on the central nervous system
- anti-diabetic medicines metformin, buformin or phenformin
Some medicines may interfere with ISOVUE® and affect how it works.
The anti-diabetic medicines, metformin (Diabex®, Diaformin® or Glucophage®), buformin (not available in Australia) or phenformin (not available in Australia) may interact with ISOVUE® and cause harm to the kidneys. Check with your doctor or pharmacist which anti-diabetic medicine or medicines you are taking, and, if you are taking metformin, buformin or phenformin, stop taking these medicines at least 48 hours before your X-ray and do not start taking them again until at least 48 hours after your X-ray.
Check with your radiologist, doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect ISOVUE®.
4. How do I use ISOVUE®?
How much to use
- ISOVUE® is given as an injection before the X-ray procedure. Your radiologist will determine the correct dose for you.
If you use too much ISOVUE®
In the unlikely event of your receiving an overdose, your radiologist will know how to treat you.
5. What should I know while using ISOVUE®?
Call your doctor straight away if you:
- develop pain at the injection site, if you are not feeling well or experiencing side effects, after receiving an injection of ISOVUE®, during the X-ray procedure or afterwards.
- phone the Poisons Information Centre on 13 11 26, or
- contact your doctor, or
- go to the Emergency Department at your nearest hospital.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how ISOVUE® affects you.
As delayed reactions to iodine-containing dyes may occur, do not drive a vehicle or operate machinery for at least 24 hours after receiving the injection of ISOVUE®.
Drinking alcohol
Tell your doctor if you drink alcohol.
Isovue® should be administered with caution in patients with chronic alcoholism.
Looking after your medicine
- You will not normally be required to store ISOVUE®. It will usually be kept by your radiologist or in the radiology or pharmacy department of the hospital where you are having the X-ray.
- The expiry date of ISOVUE® can be found on the label of the ampoule or vial.
- ISOVUE® must be stored below 25°C and protected from light.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
General:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
General
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
7. Product details
This medicine is only available with a doctor's prescription.
What ISOVUE® contains
| Active ingredient (main ingredient) | Iopamidol |
| Other ingredients (inactive ingredients) | Trometamol Sodium calciumedetate Hydrochloric acid or sodium hydroxide (to adjust pH) |
Do not take this medicine if you are allergic to any of these ingredients.
What ISOVUE® looks like
ISOVUE® is a clear, colourless to pale yellow liquid contained in a glass ampoule or vial.
ISOVUE® 300 INJECTION (equivalent to 300 mg iodine/mL)
Each mL of ISOVUE® 300 contains 612 mg of iopamidol. It is available in the following pack sizes:
- 50 mL vial (AUST R 47068)
- 100 mL vial (AUST R 47069)
ISOVUE® 370 INJECTION (equivalent to 370 mg iodine/mL)
Each mL of ISOVUE® 370 contains 755 mg of iopamidol. It is available in the following pack sizes:
- 50 mL vial (AUST R 47070)
- 100 mL vial (AUST R 47071)
- 200 mL vial (AUST R 47072)
Who distributes ISOVUE®
Sponsor:
Bracco Pty. Ltd.
14 Allambie Avenue, East Lindfield
NSW 2070
ealang57@gmail.com
Distributed by:
Regional Health Care Products Group
Medi-Consumables Pty. Ltd.
3-11 Primrose Avenue
ROSEBERY NSW 2018, AUSTRALIA
Regional Health Limited
P O Box 101-140
North Shore Mail Centre
Auckland, New Zealand
This leaflet was prepared in December 2025.
Brand Information
| Brand name | Isovue |
| Active ingredient | Iopamidol |
| Schedule | Unscheduled |
MIMS Revision Date: 01 August 2021
Notes
Distributed by Regional Health Care Products Group Medi-Consumables Pty. Ltd.
1 Name of Medicine
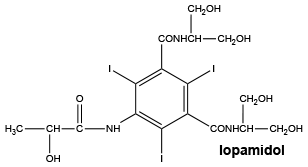
Iopamidol.
2 Qualitative and Quantitative Composition
Isovue 300 (iopamidol injection 61%) provides 612 mg iopamidol.
Isovue 370 (iopamidol injection 76%) provides 755 mg iopamidol.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Injection, solution. Clear colourless liquid.
4 Clinical Particulars
4.1 Therapeutic Indications
In adults, Isovue is indicated for angiography throughout the cardiovascular system, including cerebral and peripheral arteriography, peripheral venography (phlebography), coronary arteriography and ventriculography, selective visceral arteriography and aortography, intravenous excretory urography and contrast enhancement of computed tomographic head and body imaging.
Isovue is also indicated for myelography (lumbar, total columnar) and for contrast enhancement in computed tomography of the central nervous system.
In children, Isovue is indicated for intravascular use only, including intravenous excretory urography.
4.2 Dose and Method of Administration
Depending on age, weight, cardiac output, general state of health, the clinical problem, examination technique, kind and volume of the region to be examined, an appropriate dose should be used. Examinations of infants and children always require individualisation of dosages, to be chosen in the first place in relation to the region to be demonstrated, the weight and the age.
It is desirable that solutions of radiopaque diagnostic agents for intrathecal or intravascular use be at body temperature when injected. Isovue solutions should be used only if clear and within the normal colourless to pale yellow range. In the event that crystallisation of the medium has occurred, the solution should not be used. Many radiopaque contrast agents are incompatible in vitro with some antihistamines and many other drugs; therefore, no other pharmaceuticals should be admixed with contrast agents. Isovue contains no antimicrobial agent. Use only once and discard any residue.
Intravascular use in adults. Cerebral arteriography. Isovue 300 should be used. The usual individual injection by carotid puncture or transfemoral catheterisation is 5 to 10 mL, with multiple doses totalling in the range of 20 mL.
Peripheral arteriography. Isovue 300 usually provides adequate visualisation. For injection into the femoral artery or subclavian artery, 5 to 40 mL may be used; for injection into the aorta for a distal runoff, 25 to 50 mL may be used.
Peripheral venography (phlebography). Isovue 300 should be used. The usual dose is 30 to 50 mL per lower extremity.
Selective visceral arteriography and aortography. Isovue 370 should be used. Doses up to 50 mL may be required for injection into the larger vessels such as the aorta or celiac artery; doses up to 10 mL may be required for injection into the renal arteries.
Coronary arteriography and ventriculography. Isovue 370 should be used. The usual dose for selective coronary artery injections is 2 to 10 mL. The usual dose for ventriculography, or for nonselective opacification of multiple coronary arteries following injection at the aortic root, is 25 to 50 mL. ECG monitoring is essential.
Excretory urography. Isovue 370 should be used. The usual adult dose is 30 to 50 mL administered by rapid intravenous injection.
Computed tomography. Isovue 300 may be used. Head imaging: the suggested dose range is 50 to 100 mL by intravenous administration; imaging may be performed immediately after completion of administration. Body imaging: the usual adult dose is 100 mL administered by rapid intravenous (within approximately one minute) bolus injection. Imaging is performed immediately after injection. Equivalent doses of Isovue 370, based on organically bound iodine content, may also be used.
Intravascular use in children. Isovue 300 should be used in children. For children up to 8 kg bodyweight, the usual dose is 3 to 5 mL per kg bodyweight administered by rapid infusion. For children over 8 kg bodyweight, the usual dose is 1 to 2 mL per kg bodyweight administered by rapid infusion.
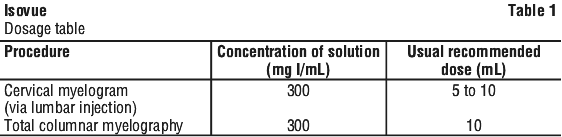
Intrathecal use in adults. Isovue 300 should be used for lumbar and total columnar myelography. A total dose in excess of 3000 mg iodine, and Isovue at concentrations greater than 300 mg I/mL should not be used intrathecally.
In patients with a history of seizure activity who are not on anti-convulsant therapy, premedication barbiturate or phenytoin should be considered. Lumbar puncture is usually made between L3 and L4; if pathology is suspected at this level, the interspace immediately above or below may be selected. A lateral cervical puncture may also be used.
Rate of injection. To avoid excessive mixing with cerebrospinal fluid and consequent loss of contrast as well as premature cephalad dispersion, injection must be made slowly over one to two minutes; the needle may then be removed. An interval of at least 48 hours should be allowed before repeat examination; however, whenever possible five to seven days is recommended. The recommended adult doses of Isovue should not be exceeded. See Table 1.
Aspiration of Isovue is unnecessary following intrathecal administration (see Section 5 Pharmacological Properties).
Suggestions for usual patient management. Preprocedure. See Section 4.4 Special Warnings and Precautions for Use regarding discontinuation of neuroleptic agents.
Maintain normal diet up to 2 hours before procedure.
Ensure hydration - fluids up to time of procedure.
During procedure. Use minimum dose and concentration required for satisfactory contrast.
Inject slowly over 1 to 2 minutes to avoid excessive mixing.
Avoid abrupt or active patient movement as it causes excessive mixing with CSF.
Instruct patient to remain passive. Move patient slowly and only as necessary.
To maintain as a bolus, move medium to distal area very slowly under fluoroscopic control.
In all positioning techniques keep the patient's head elevated above highest level of spine.
Do not lower head of table more than 15° during thoraco-cervical procedures.
In patients with excessive lordosis, consider lateral position for injection and movement of the medium cephalad.
Avoid intracranial entry of a bolus.
Avoid early and high cephalad dispersion of the medium.
At completion of direct cervical or lumbo-cervical procedures, raise head of table steeply (45°) for about 2 minutes to restore medium to lower levels.
Postprocedure. Raise head of stretcher to at least 15° before moving patient onto it.
Movement onto stretcher, and off the stretcher to bed, should be done slowly with patient completely passive, maintaining head up position.
Before moving patient onto bed, raise head of bed 15° to 30° and maintain the patient in this position under close observation for 12 to 24 hours.
Advise patient to remain still in bed, in head up position for the first 24 hours.
Obtain visitors' co-operation in keeping the patient quiet and in head up position, especially in first few hours.
Encourage oral fluids and diet as tolerated.
Intrathecal use in children. Myelography. Use in paediatric myelography is not recommended as, at present, there is insufficient data to support such use.
4.3 Contraindications
General. Isovue should not be administered to patients with known hypersensitivity to iopamidol. Iodine containing contrast media, whether ionic or non-ionic, should not be administered to patients with thyrotoxicosis or decompensated cardiac insufficiency.
Intravascular use. Iodine containing contrast agents for intravascular use are contraindicated in certain specific procedures and situations such as carotid angiography during the progressive period of stroke; coronary arteriography in the first 4 weeks after myocardial infarction; and the presence of infection or open injury in or near the region to be examined.
Intrathecal use. Intrathecal administration of corticosteroids with Isovue is contraindicated. Because of overdosage considerations, immediate repeat myelography in the event of technical failure is contraindicated and an interval of at least 48 hours should ordinarily be allowed before repeat examination (see Section 4.2 Dose and Method of Administration). Spinal puncture should not be performed in the presence of significant local or systemic infection where bacteraemia is likely.
4.4 Special Warnings and Precautions for Use
Diagnostic procedures which involve the use of any radiopaque diagnostic agent should be carried out under the direction of knowledgeable personnel.
Isovue does not contain any preservatives. Any contrast medium solution left over from the examination must be discarded. In rare cases, crystals may form in the solution of Isovue even before the container is opened. Solutions in which this change has taken place should not be used.
General. Fatal reactions have been associated with the administration of water soluble contrast media, most of which have been attributed to chemical, allergic or idiosyncratic effects. Therefore, it is of utmost importance that a course of action be carefully planned in advance for the immediate treatment of serious reactions, and that adequate and appropriate facilities and personnel be readily available in case of a severe reaction. Most adverse reactions to injectable contrast agents appear within one to three minutes after the start of injection but delayed reactions may occur. Before a contrast medium is injected, the patient should be questioned on previous tolerance to iodine containing contrast media and for a history of allergy. Although a history of allergy, including asthma, may imply greater than usual risk, it does not necessarily contraindicate the use of the medium. With Isovue, as with any other iodinated contrast medium, the possibility cannot be ruled out that, irrespective of the amount and type of contrast medium administered, an individual patient may be particularly sensitive to such compounds. The use of a small i.v. test dose as a predictive measure is not advised as it may precipitate a severe, or even fatal, reaction in allergic patients. In cases with a history of allergy or iodine hypersensitivity an antihistamine or corticoid may be administered prophylactically. Fluid intake should not be restricted before the use of Isovue in any patient and, in particular, in patients with multiple myeloma, juvenile type diabetes or diabetes of long standing, polyuria, oliguria, gout or in debilitated patients. Before the procedure, compensation must be made for disturbances of water and electrolyte balance and patients should be kept well hydrated after the procedure.
Intravascular use. In patients with severe impairment of hepatic or renal function, decompensated cardiac failure, uncontrolled cardiac arrhythmias, advanced vascular disease, pulmonary emphysema, poor general health, cerebral arteriosclerosis, juvenile type diabetes or diabetes of long standing, cerebral spasmodic conditions, or epilepsy, latent thyroid hyperfunction and multiple myeloma, the need for examination merits particularly careful consideration. Diabetics with serum creatinine above 500 micromol/L should not be examined unless the possibility of benefit clearly outweighs the additional risk. Caution is also necessary in patients suffering from phaeochromocytoma, sickle cell disease, chronic alcoholism and allergic manifestations. Premedication with alpha-receptor blockers is recommended in phaeochromocytoma patients because of the risk of blood pressure crises. Renal toxicity has been reported in a few patients with liver dysfunction who were given oral cholecystographic agents followed by parenterally administered contrast agents.
Studies in vitro utilizing animal blood show that radiopaque contrast agents including Isovue can produce a slight depression of plasma coagulation factors including prothrombin time, partial thromboplastin time and fibrinogen and a slight tendency to cause platelet or red blood cell aggregation, or both. Muscle cramps and involuntary movements which follow the use of conventional ionic contrast media in arteriography may also occur with Isovue but are less marked. Convulsions may be precipitated, particularly in patients with a history of epilepsy.
Intrathecal use. Isovue should be administered with caution in patients with increased intracranial pressure or suspicion of intracranial tumor, abscess or haematoma, history of convulsive disorders, chronic alcoholism and multiple sclerosis. Particular attention must be given to the concentration of the contrast agent, the dose and the technique as well as the state of hydration of the patient. If frankly bloody cerebrospinal fluid is observed, the possible benefits of a myelographic examination should be considered in terms of risk to the patient. Patients on anticonvulsant medication should be maintained on this therapy. However, this does not totally eliminate the risk of seizures occurring and such patients should be kept under observation for at least 48 hours. Should a seizure occur, appropriate treatment should be instituted immediately. In patients with a history of seizures but who are not on anticonvulsant therapy, appropriate preventive medication should be considered. Inadvertent intracranial entry of a large or concentrated bolus of the contrast medium, which increases the risk of neurotoxicity, can be prevented by careful patient management. Also, effort should be directed to avoid rapid dispersion of the medium causing inadvertent rise to intracranial levels (e.g. by active patient movement). If such intracranial entry of the medium occurs, prophylactic anticonvulsant treatment with diazepam or barbiturates orally for 24 to 48 hours should be considered. Focal and generalized motor seizures have been reported after intrathecal use of water soluble contrast agents, including Isovue. Therefore, avoid:
deviations from recommended neuroradiologic procedure or patient management;
use in patients with a history of epilepsy;
overdosage;
intracranial entry of a bolus or premature diffusion of a high concentration of the medium;
medication with neuroleptic drugs or phenothiazine antinauseants or other medication that may lower the seizure threshold;
lowering of the head from its elevated position during the procedure, on the stretcher, and in bed;
excessive and particularly active patient movement or straining.
In patients with renal impairment. Caution must be exercised in patients with impaired renal function, particularly when large doses are given.
In diabetic patients. Biguanide oral anti-diabetic agents, e.g. metformin, buformin and phenformin are excreted unchanged through the kidneys.
They therefore compete for excretion in the kidneys with contrast media and this could lead to a reduction in kidney function. In Australia, the only biguanide oral anti-diabetic agent available is metformin (Diabex, Diaformin or Glucophage). Patients with non-insulin-dependent diabetes mellitus who are taking metformin should therefore be instructed not to take any doses of this agent (metformin) for 48 hours before, and 48 hours after the X-ray examination. The same restriction should be applied to patients, particularly overseas visitors, who are taking other biguanide oral anti-diabetic agents which may be sourced from outside Australia (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Other. Other precautions which apply to the various radiographic contrast procedures are the same for Isovue as they are for conventional ionic media.
The risk of the procedure itself should be evaluated carefully in each patient. Such precautions include:
Angiography. The possibility of dislodging plaques or damaging or perforating the vessel wall should be borne in mind during catheter manipulation and contrast medium injection. Angiography should be avoided whenever possible in patients with homocystinuria because of the risk of inducing thrombosis and embolism.
Cerebral angiography. Use with caution in patients with extreme senility, advanced atherosclerosis, or severe hypotension. The procedure may be hazardous in subarachnoid haemorrhage and in migraine (because of ischaemic complications).
Peripheral angiography. Pulsation should be present in the artery to be injected. In thromboangiitis obliterans (Buerger's Disease) or ischaemia associated with ascending infection, angiography should be performed with extreme caution, if at all.
Cardioangiography. Caution is advised in the administration of large volumes to patients with incipient heart failure because of the possibility of aggravating the pre-existing condition. Hypotension should be corrected promptly since it may induce serious arrhythmias. Patients should be observed for several hours following the procedure to detect delayed haemodynamic effects. The risk of cardioangiography in patients with chronic pulmonary emphysema should be weighed carefully against the necessity for performing this procedure.
Caution is advised with dosage in patients with right ventricular failure, pulmonary hypertension, or stenotic pulmonary vascular beds because of the haemodynamic changes which may occur after injection into the right heart outflow tract.
Excretory urography. It is advisable to allow an interval of at least 48 hours before repeating excretory urography. Dehydration should be avoided in the elderly, particularly those with polyuria, oliguria, advanced vascular disease or pre-existing dehydration. Myelomatosis.
Use in the elderly. No data available.
Paediatric use. Young infants (age < 1 year), especially neonates, are particularly susceptible to electrolyte imbalances and haemodynamic alterations. Care should be taken regarding the dosage to be used, the details of the procedure, and the patient's status. Safety and efficacy of Isovue for use in myelography in children have not been adequately demonstrated.
Thyroid function in infants exposed to iodinated contrast media (ICM) should be evaluated and monitored. Decreased levels of thyroxine (T4) and triiodothyronine (T3) and increased level of thyroid stimulating hormone (TSH) were reported after exposure to ICM in infants, especially preterm infants, which remained for up to a few weeks or more than a month. Thyroid function in infants exposed to ICM should therefore be evaluated and monitored until thyroid function is normalised. Some patients were treated for hypothyroidism.
Effects on laboratory tests. Intravascular iodinated contrast media can interfere with simultaneous or subsequent laboratory tests or diagnostic procedures as a result of their chemical properties or effects on vascular and organ physiology.
Intravascular contrast media have been reported to affect the following laboratory diagnostic procedures. See Table 2.
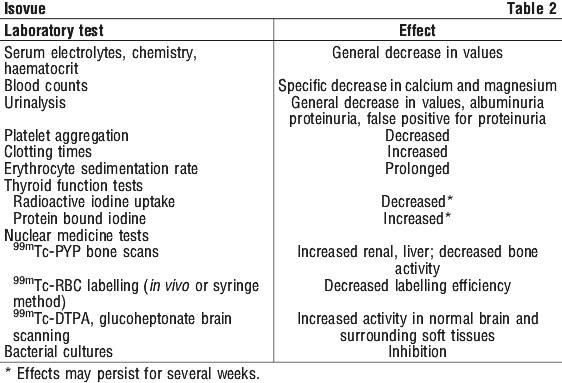
Any test which might be affected by the contrast medium should be performed prior to its administration.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Metformin and other biguanide oral anti-diabetic agents. Biguanide oral anti-diabetic agents, e.g. metformin, buformin and phenformin are excreted unchanged through the kidneys.
They therefore compete for excretion in the kidneys with contrast media and this could lead to a reduction in kidney function. In Australia, the only biguanide oral anti-diabetic agent available is metformin (Diabex, Diaformin or Glucophage). Patients with non-insulin-dependent diabetes mellitus who are taking metformin should therefore be instructed not to take any doses of this agent (metformin) for 48 hours before, and 48 hours after the X-ray examination. The same restriction should be applied to patients, particularly overseas visitors, who are taking other biguanide oral anti-diabetic agents which may be sourced from outside Australia (see Section 4.4 Special Warnings and Precautions for Use).
Intravascular use. Intravascular contrast media have been reported to interact with other drugs to generally produce an effect which is additive to the pharmacological action of the drug. The duration of the interaction may bear no relationship to the severity and clinical outcome. Interactions have been reported to have occurred between intravascular contrast media and the following classes of drugs; antiarrhythmics, anticoagulants, antiplatelet drugs, beta-adrenergic receptor blocking agents, cardiac glycosides, diuretics, nephrotoxic drugs and rauwolfia alkaloids.
Intrathecal use. Use of medications that may lower the seizure threshold (phenothiazine derivatives, including those used for their antihistaminic properties; tricyclic antidepressants; monoamine oxidase (MAO) inhibitors, central nervous system (CNS) stimulants; analeptics; antipsychotic agents) should be avoided. Although the contributory role of such medications has not been fully established, it is advisable to discontinue these agents for at least 48 hours before and for at least 24 hours following intrathecal use. These agents should not be used for controlling the nausea and vomiting which may accompany the procedure.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Intravenously administered iopamidol does not induce adverse effects on fertility and general reproductive performance in rats.
Use in pregnancy. It is not known whether iopamidol crosses the placental barrier or reaches foetal tissues.
However, many injectable contrast agents cross the placental barrier in humans and appear to enter foetal tissues.
Intravenously administered iopamidol does not induce adverse effects on fertility and general reproductive performance in rats. No teratogenic effects attributable to iopamidol have been observed in teratology studies performed in animals. There are, however, no adequate and well controlled studies in pregnant women. Because animal teratology studies are not always predictive of human response, Isovue should be used during pregnancy only if its use is deemed essential for the patient's welfare.
Infants born to women who received iodinated contrast media while pregnant should have testing for hypothyroidism in the neonatal period. Some patients were treated for hypothyroidism. Also, see Section 4.4 Special Warnings and Precautions for Use, Paediatric use.
Use in lactation. It is not known whether iopamidol is excreted in human milk. However, many injectable contrast agents are excreted unchanged in human milk.
Use in nursing mothers is not recommended unless alternative arrangements can be made for feeding the infant. Also, see Section 4.4 Special Warnings and Precautions for Use, Paediatric use.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Severe and life threatening reactions, including fatal reactions, have been reported with both ionic and non-ionic contrast media.
Transitory changes may occur in red cell and leucocyte counts, serum calcium, serum creatinine, serum glutamic oxaloacetic transaminase (SGOT) and uric acid in urine; transient albuminuria may occur. These laboratory findings have not been associated with clinical manifestations. Studies in vitro utilising animal blood show that radiopaque contrast agents including Isovue can produce a slight depression of plasma coagulation factors including prothrombin time, partial thromboplastin time, and fibrinogen, and a slight tendency to cause platelet or red blood cell aggregation or both.
Intravascular use. Isovue causes less pain/ heat sensation following intra-arterial or intravenous administration than the conventional ionic contrast media.
Otherwise, the spectrum of adverse reactions is similar to that seen with the conventional ionic contrast media, although generally less pronounced.
Gastrointestinal. Nausea, vomiting, abdominal discomfort/ pain, anorexia, diarrhoea, rectal haemorrhage.
Cardiovascular. Ventricular and supraventricular ectopic beats, angina, hypotension, hypertension, bradycardia, tachycardia, ECG changes, changes in cardiac output and other haemodynamic parameters, circulatory collapse, ventricular fibrillation, arrhythmia, palpitations, myocardial infarction, cardiac arrest, thrombophlebitis, disseminated intravascular coagulation, angioedema.
Nervous system. Sensation of heat/ pain, headache, dizziness, faintness, restlessness, EEG changes, convulsions, confusion, coma, meningismus, meningitis-like reaction, meningitis, agitation, amnesia, abnormal coordination.
Respiratory. Respiratory distress, bronchospasm, dyspnoea, asthma, pulmonary oedema.
Endocrine. Uncommon: Thyroid function tests indicative of hypothyroidism or transient thyroid suppression have been uncommonly reported following iodinated contrast media administration to adult and paediatric patients, including infants. Some patients were treated for hypothyroidism.
Ocular. Vision disturbances, periorbital oedema, blindness, itchy eyes, mydriasis, photophobia, conjunctivitis, abnormal lacrimation, oculogyric crisis.
Nasal, oral, pharyngeal. Altered taste sensation, nasal congestion, rhinitis, sneezing, oedema (laryngeal, tongue, facial), glossitis, dry mouth, pharyngitis.
General. Flushing, urticaria, itching, exanthema, rash, bullous eruption, muscle cramps, myalgia, myopathy, involuntary movements, paraplegia, sweating, fever, chills, shivering, rigors, hypothermia, skin cold/ clammy, injection site reaction, tightness/ pain in chest, anaphylactoid reaction, cyanosis, malaise, paraesthesia, coughing, dysphagia, urinary retention, acute renal failure, abnormal renal function, encephalopathy, paroniria, somnolence, purpura, hypoaesthesia.
Myelography. Experience with Isovue at present is not as extensive as with conventional ionic contrast media and it is likely that the full spectrum of adverse effects associated with its use has not yet been identified. The clinician should therefore remain alert for adverse effects other than those listed below, particularly those already reported in the literature for other non-ionic water soluble contrast agents. The most frequently reported adverse reactions with intrathecal use of Isovue are headache, nausea and vomiting. These reactions usually occur 1 to 10 hours after injection, almost all occurring within 24 hours. They are usually mild to moderate in degree lasting for a few hours and usually disappearing within 24 hours. Rarely, headaches may be severe or persist for days. Headache is often accompanied by nausea and vomiting, and tends to be more frequent and persistent in patients not optimally hydrated and during examination of the thoracic and cervical regions. Other reported adverse effects include backache, neck stiffness, numbness and paraesthesias, leg or sciatic-type pain, pyrexia, muscle weakness, hot flushes, diarrhoea, heartburn, radicular irritation (lumbosacral), hypotension, hypertension, shoulder pain, leg cramps, meningeal irritation, tachycardia, emotional stress, dizziness, confusion, hallucinations, lightheadedness, syncope, cold extremities, neurogenic bladder, respiratory distress, rash and injection site pain. Other uncommon effects include major motor seizures, facial neuralgia, tinnitus, sweating and drowsiness. Early onset of seizures (less than two hours) is indicative of early substantial intracranial entry. Transitory EEG changes occur and usually take the form of slow wave activity.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Treatment of an overdose of an injectable radiopaque contrast medium is directed towards the support of all vital functions, and prompt institution of symptomatic therapy. In myelography, even use of a recommended dose can produce mental aberrations tantamount to overdosage if incorrect management of the patient during or immediately following the procedure permits inadvertent early intracranial entry of a large portion of the medium.
Contact the Poisons Information Centre on 13 11 26 for advice on management.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Isovue (iopamidol injection) is a non-ionic, radiopaque contrast medium for intrathecal or intravascular administration.
Intravascular injection of Isovue opacifies blood vessels in the path of its flow, permitting radiographic visualization of the vasculature until significant haemodilution occurs. Being a contrast medium with lower osmolality than the conventional ionic contrast media, Isovue has less potential for deleterious effects on the myocardium and the peripheral vasculature.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Following intravenous administration, Isovue is distributed into the extracellular compartment.
Plasma half life is approximately 2 hours.
Following intrathecal administration, Isovue is rapidly absorbed into the bloodstream from the cerebrospinal fluid (CSF). Peak serum levels are reached in 1-4 hours. Isovue undergoes no significant metabolism in the body. Less than 1% is bound to the plasma proteins. Isovue is excreted primarily by the kidneys, approximately 50% of an intravenously administered dose being recovered during the first 2 hours and 90-98% being recovered in 96 hours.
Approximately 1% is excreted via the faeces. Approximately 85% of an intrathecal dose is excreted in the urine in 48 hours.
Experience with Isovue suggests that in peripheral arteriography it causes less discomfort (e.g. pain and/or warmth) than seen with conventional ionic contrast media. Animal studies in rabbits indicate that, following intracarotid administration, Isovue does not cross the blood brain barrier to any significant extent.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Isovue 300. 1 mg trometamol and 0.302 (0.271-0.331) mg sodium calcium edetate per mL. The solution contains approximately 0.037 mg (0.002 mEq) sodium and 300 mg organically bound iodine per mL.
Isovue 370. 1 mg trometamol and 0.373 (0.337-0.411) mg sodium calcium edetate per mL. The solution contains approximately 0.046 mg (0.002 mEq) sodium and 370 mg organically bound iodine per mL.
6.2 Incompatibilities
Many radiopaque contrast agents are incompatible in vitro with some antihistamines and many other drugs; therefore, no other pharmaceuticals should be admixed with contrast agents.
See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Protect from light.
6.5 Nature and Contents of Container
Isovue is available in the following strengths and pack sizes:
Isovue 300. 50 mL, 100 mL vials.
Isovue 370. 50 mL, 100 mL, 200 mL vials.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
The pH of Isovue contrast media has been adjusted to 6.5-7.5 with hydrochloric acid. The osmolality of Isovue is substantially lower than that of conventional ionic contrast agents of equivalent iodine concentration. See Table 3.

Chemical structure.
7 Medicine Schedule (Poisons Standard)
Not scheduled. Not considered by committee.
Date of First Approval
09 December 1994
Date of Revision
17 June 2021
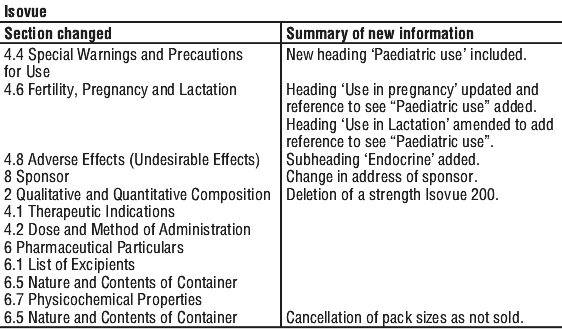
Summary Table of Changes
Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.