Jespect
Brand Information
| Brand name | Jespect |
| Active ingredient | Japanese encephalitis virus vaccine (inactivated) |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Jespect.
Summary CMI
JESPECT®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using JESPECT?
JESPECT contains the active ingredient Inactivated Japanese Encephalitis Vaccine (adsorbed). JESPECT is used to help prevent you against Japanese Encephalitis.
For more information, see Section 1. Why am I using JESPECT? in the full CMI.
2. What should I know before I use JESPECT?
Do not use if you have ever had an allergic reaction to JESPECT or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use JESPECT? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with JESPECT and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use JESPECT?
- JESPECT is an injection given by a health professional.
More instructions can be found in Section 4. How do I use JESPECT? in the full CMI.
5. What should I know while using JESPECT?
| Things you should do | Tell your doctor, nurse or pharmacist before you receive the vaccine:
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using JESPECT? in the full CMI.
6. Are there any side effects?
The most common side effects include headache, muscle pain, tiredness and nausea. Severe allergic reactions are possible, but rare.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
1. Why am I using JESPECT®?
JESPECT® contains the active ingredient Inactivated Japanese Encephalitis Vaccine (adsorbed).
JESPECT® is a vaccine for persons 18 years of age or older used to help prevent Japanese encephalitis (en-kef-al-i-tis), a severe and often fatal infection of the brain. Some of those who recover from the disease are often left with brain damage.
Japanese encephalitis is caused by the Japanese encephalitis virus that mainly occurs in Asia. The virus is transmitted to humans by infected mosquitoes that feed on human blood. JESPECT® is used to vaccinate persons 18 years and older who:
- Plan to live in or travel to areas where Japanese Encephalitis is common or seasonal. Your doctor will explain your risk of catching the disease.
- Work with Japanese encephalitis virus.
JESPECT® works by getting your body to produce its own protection against the Japanese encephalitis virus.
The vaccine does not contain live virus and cannot give you the illness.
After you have been given JESPECT®, your body makes substances called antibodies. These antibodies fight the Japanese encephalitis virus. When you come into contact with the virus, your body is usually ready to destroy it.
Most people who receive two doses of the vaccine will produce enough antibodies to protect against the disease. However, as with all vaccines, 100% protection cannot be guaranteed.
JESPECT® does not protect against encephalitis caused by other viruses or bacteria.
2. What should I know before I use JESPECT®?
Warnings
Do not use JESPECT® if:
- you are allergic to Japanese Encephalitis vaccine or any of the ingredients listed at the end of this leaflet. Always check the ingredients to make sure you can use this medicine.
Check with your doctor if you:
- take any medicines for any other condition
- have a high temperature (fever)
- have an immune system that does not work properly
- tend to bleed or bruise easily
- have received or are going to receive other vaccines
- you are under 18 years of age.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Your doctor will discuss the possible risks and benefits of having JESPECT® during pregnancy and breastfeeding.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with JESPECT® and affect how it works.
Medicines that may reduce the effect of JESPECT® include:
- medicines that suppress your immune system, for example, corticosteroids.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect JESPECT®.
4. How do I use JESPECT®?
How much to use
JESPECT® is given to you by a trained health professional as an injection into the upper arm.
- JESPECT® is given as two injections, 28 days apart.
- Each dose of the vaccine is 0.5 mL
When to use JESPECT®
- The vaccination course of JESPECT® should be completed at least one week before potential exposure to the Japanese encephalitis virus.
If you miss the second dose of vaccine, talk to your doctor. It is important that you are given the second dose so that your body can produce enough antibodies to protect against the Japanese encephalitis virus.
Your doctor may decide that you need a booster dose 1–2 years after receiving the vaccine. Your doctor will tell you if a booster dose is required.
If you use too much JESPECT®
As each JESPECT® pre-filled syringe contains only one dose, overdosage is unlikely.
If you think that you have used too much JESPECT®, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26 in Australia or 0800 POISON (0800 764 766) in New Zealand), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using JESPECT®?
Things you should do
Call your doctor straight away if you:
- Notice sudden signs of allergy such as:
- rash, itching or hives on the skin, swelling of the face, lips, tongue or other parts of the body
- shortness of breath, wheezing or trouble breathing
- collapse
Remind any doctor, dentist or pharmacist you visit that you are using JESPECT®.
Driving or using machines
Do not drive or use machines if you are feeling unwell after vaccination. Wait until any effects of the vaccine have worn off before you drive or use machines.
Looking after your medicine
JESPECT® is usually stored in the doctor's surgery or clinic, or at the pharmacy. However, if you need to store JESPECT®:
- Keep it in the refrigerator, between 2°C and 8°C. DO NOT FREEZE. Freezing destroys the vaccine.
- Keep it in the original pack until it is time for it to be injected
- Keep it where children cannot reach it.
- Do not use JESPECT® after the expiry date printed on the pack.
- Do not use JESPECT® if the packaging is torn, shows signs of tampering or does not look quite right.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away. |
Signs of a severe allergic reaction
These side effects are very rare. | Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What JESPECT® contains
| Active ingredient (main ingredient) | Inactivated Japanese encephalitis virus strain SA14–14–2 produced in Vero cells: 6 AgU (antigen units). |
| Other ingredients (inactive ingredients) |
|
| Potential allergens | Sulfites: JESPECT may contain trace amounts of sodium metabsulfite. |
Do not take this medicine if you are allergic to any of these ingredients.
What JESPECT® looks like
JESPECT® is supplied as a single dose (0.5 mL) in a needle-less pre-filled glass syringe. The vaccine should appear as a clear liquid with a white precipitate and when shaken, it should appear as a white, cloudy suspension.
Registration number
Australia
AUST R 150602
New Zealand
TT50-9020
Who distributes JESPECT®
Sponsor
JESPECT® is distributed in Australia by:
Seqirus Pty Ltd
ABN: 26 160 735 035
63 Poplar Road
Parkville, VIC 3052 Australia
Telephone: 1800 642 865
www.cslseqirus.com.au
JESPECT® is distributed in New Zealand by:
Seqirus (NZ) Ltd
PO Box 62 590
Greenlane, Auckland 1546
New Zealand
This leaflet was prepared in September 2025
JESPECT® is a trademark of Valneva Scotland Ltd
Brand Information
| Brand name | Jespect |
| Active ingredient | Japanese encephalitis virus vaccine (inactivated) |
| Schedule | S4 |
MIMS Revision Date: 01 December 2020
1 Name of Medicine
Inactivated Japanese encephalitis vaccine (adsorbed).
2 Qualitative and Quantitative Composition
Jespect Japanese encephalitis (JE) virus, purified inactivated vaccine is a sterile, ready to use suspension for intramuscular (IM) injection. The vaccine is prepared by propagating Japanese encephalitis virus strain (SA14-14-2) in Vero cells. No preservatives or antibiotics are added to the formulation.
Jespect is a clear liquid with white precipitate and when shaken before use, a white/cloudy suspension forms. Jespect is supplied in pre-filled syringes without needles. Each 0.5 mL dose of vaccine contains 6 antigen units (AgU) of purified, inactivated JE virus. Each dose of vaccine also contains the following excipients: 0.1% aluminium hydroxide hydrate corresponding to 0.25 mg aluminium, and Phosphate Buffered Saline.
Phosphate Buffered Saline has the following saline composition:
sodium chloride - 9 mg/mL;
monobasic potassium phosphate - 0.144 mg/mL;
dibasic sodium phosphate - 0.795 mg/mL.
3 Pharmaceutical Form
Jespect is supplied as a 0.5 mL suspension in a pre-filled syringe (Type I glass) with a plunger stopper (chlorobutyl elastomer) in a pack size of 1 needle-less syringe.
To attach needle, remove the syringe cap by gently twisting it. Do not attempt to snap or pull the tip off as this may damage the syringe.
4 Clinical Particulars
4.1 Therapeutic Indications
Jespect is indicated for active immunisation against Japanese encephalitis (JE) virus for persons 18 years of age and older.
Jespect should be considered for use in persons who plan to reside in or travel to areas where JE is endemic (common) or epidemic (seasonal), especially during the transmission season.
Jespect is indicated for persons who work with JE virus in laboratories and in industry.
4.2 Dose and Method of Administration
Primary vaccination. The primary vaccination series consists of a total of two doses of 0.5 mL each according to the following schedule.
First dose at Day 0 (elected date).
Second dose: 28 days after first dose.
It is recommended that persons who receive the first dose of Jespect complete the 2-dose primary vaccination course.
If the primary immunisation schedule of 2 doses is not completed, sufficient protection against Japanese encephalitis virus might not be achieved. Clinical trial data indicates that administration of the second dose up to 11 months after the first dose results in high seroconversion rates (see Section 5.1 Pharmacodynamic Properties, Clinical trials, Incomplete primary immunisation).
Booster dose. See Section 5.1 Pharmacodynamic Properties, Clinical trials for clinical trial data for potential booster doses.
The vaccine should be inspected visually for presence of particulate matter and discolouration prior to administration. Discard the product if particulates are present, if it appears discoloured or if the syringe appears to be physically damaged. Any unusual product or waste material should be disposed of in accordance with local requirements.
Jespect should be well shaken before administration to obtain a homogenous suspension. Once shaken, the vaccine should appear as a white cloudy suspension.
Jespect should be administered by IM injection into the deltoid muscle. It should never be injected IV.
In exceptional circumstances, for those patients with thrombocytopenia or bleeding disorders where bleeding may occur following IM administration, Jespect may be administered subcutaneously. Subcutaneous administration could lead to a suboptimal response to the vaccine.
Jespect pre-filled syringe is for single use only in one individual only. Inject the entire contents of the syringe.
4.3 Contraindications
Jespect should not be administered to individuals who have previously experienced a serious reaction (e.g. anaphylaxis) to this vaccine or who are known to be hypersensitive to any of the vaccine components or to the residues protamine sulphate, formaldehyde, bovine serum albumin, host cell DNA, sodium metabisulphite or host cell protein. Individuals who show hypersensitivity reactions after receiving the first dose of the vaccine should not be given the second dose.
4.4 Special Warnings and Precautions for Use
As with other injectable vaccines, appropriate medical treatment and supervision should always be available in the event of anaphylactic reaction. Adrenaline should always be readily available whenever the injection is given.
Jespect will not protect against encephalitis caused by other organisms. As with any other vaccine, vaccination with Jespect may not result in protection in all cases. The primary vaccination series (2 doses) should be completed at least one week prior to potential exposure to Japanese encephalitis virus. Seroconversion rates after one dose are limited (see Section 5.1 Pharmacodynamic Properties, Clinical trials, Table 3).
Jespect should be administered by IM injection into the deltoid muscle. It should never be injected IV. In exceptional circumstances, for those patients with thrombocytopenia or bleeding disorders where bleeding may occur following IM administration, Jespect may be administered subcutaneously. Subcutaneous administration could lead to a suboptimal response to the vaccine (see Section 4.2 Dose and Method of Administration).
As with other vaccines, Jespect should not be administered in persons with acute severe febrile illness.
Safety and efficacy of Jespect have not been established in persons with a history of flavivirus (including JE virus, yellow fever virus, Dengue virus and Murray Valley encephalitis virus) infection and/or vaccination against JE or yellow fever (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Paediatric use. The safety and efficacy of Jespect in persons under 18 years of age have not been established.
Use in the elderly. Special studies in the geriatric population have not been performed; however, Jespect has been administered to 118 subjects ≥ 65 years of age. No overall differences in safety and effectiveness were observed between these subjects and younger subjects.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Concomitant administration of Jespect with inactivated hepatitis A vaccine (Havrix 1440) has been explored in one clinical trial (study IC51-308). There was no interference with the immune response to the JE virus and HAV, respectively (see Section 5.1 Pharmacodynamic Properties, Clinical trials).
Vaccination with Jespect in persons with vaccine induced tick born encephalitis (TBE) antibodies resulted in comparable SCR and GMT at Day 56 compared to TBE naive persons.
Vaccination with Jespect concurrently with or after administration of other flavivirus vaccines (including yellow fever vaccine) has not been assessed.
If co-administration with other vaccines is indicated, injections should be given into separate limbs. It should be noted that adverse reactions might be intensified.
In patients receiving immunosuppressive therapy or patients with immunodeficiency an adequate immune response may be diminished.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No significant effects on mating performance or fertility were observed in vaccine treated rats administered with IM Jespect prior to mating (3 weeks (regimen 1 only) and 1 week) during gestation (day 6). The effect of Jespect administration on male fertility has not been evaluated (see Use in pregnancy).
Use in pregnancy. (Category B1)
In a three phase reproductive study of female rats administered approximately the clinical IM dose of Jespect (5 microgram) prior to mating (3 weeks (regimen 1 only) and 1 week) and during gestation (day 6), there were no significant toxicological effects in the dams. High JEV-specific antibody titres were detected in maternal blood during gestation (day 5 and 20) in fetal cord blood and at the end of gestation (day 20), and in pups at weaning (lactation day 21).
In a reproductive and pre-/post-natal toxicity study, no vaccine-related effects were detected on reproduction, foetal weight, survival and development of the off-springs. However, incomplete ossification of parts of the skeleton was observed in the group receiving 2 doses, but not in the group receiving 3 doses. There is currently no evidence that this phenomenon is treatment related.
There are limited data from the use of Jespect in pregnant women. The vaccine should be used during pregnancy only when clearly needed and the possible advantages outweigh the possible risks for the fetus.
Use in lactation. It is not known whether vaccine antigens or antibodies induced by the vaccine are excreted in human milk. Therefore, caution should be exercised when Jespect is administered to a nursing woman.
In a three phase reproductive study of maternal treatment of rats with IM Jespect prior to mating and during gestation, assessed to lactation day 21, high JEV-specific antibody titres were detected in maternal blood during gestation, in fetal cord blood at the end of gestation and in pups at weaning (see Use in pregnancy).
Adequate human data on Jespect use during lactation are not available. Jespect should be administered to women who are breastfeeding only when clearly needed.
4.7 Effects on Ability to Drive and Use Machines
No studies on the effects on the ability to drive and use machines have been performed.
4.8 Adverse Effects (Undesirable Effects)
The safety of Jespect has been assessed in different controlled clinical trials in which more than 5,405 healthy adults were included of which 4,248 healthy adults received the vaccine.
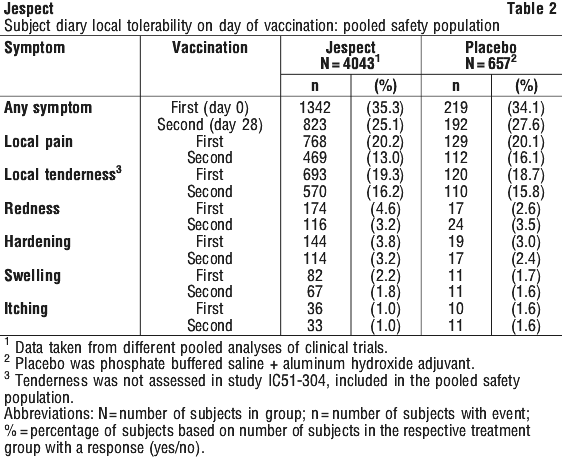
Approximately 40% of treated subjects can be expected to experience adverse reactions. They usually occur within the first three days after vaccination, are usually mild and disappear within a few days. No increase in the number of undesirable reactions was noted between first and second doses. The incidence of systemic and local symptoms was less prominent after the second vaccination.
Most commonly reported adverse reactions included headache and myalgia occurring in approximately 20% and 13% of subjects, respectively. The treatment-related, treatment-emergent adverse events seen in clinical trials are described in Table 1.
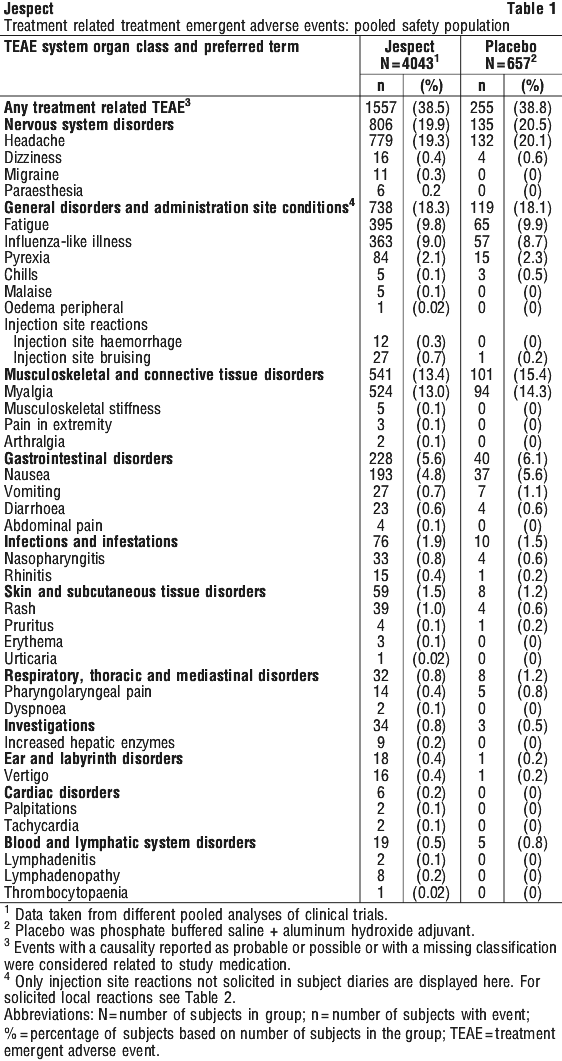
Nervous system disorders. Paraesthesia, neuritis, syncope.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
There have been no cases of overdosage reported.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. No data available.
Clinical trials. Efficacy (immunogenicity) of Jespect has been studied in approximately 2,228 healthy, adult subjects in 10 multi-centre clinical trials conducted in the US and Europe.
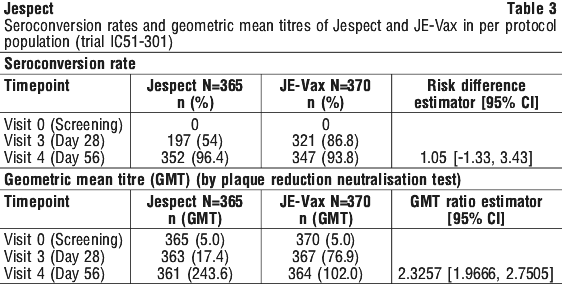
Immunogenicity of the vaccine was evaluated in a randomized, active-controlled, observer-blinded, multicenter Phase 3 clinical trial (trial IC51-301) conducted in the US, Germany and Austria in healthy male and female subjects ≥ 18 years of age (mean age: 41.3 years; 60.8% female; race: Caucasian 80.8%, Asian 0.8%, Black/African American 13.1%, and Other 5.3%). Subjects were randomised to receive either two separate doses of 6 microgram/0.5 mL of Jespect (on a 0, 28-day schedule by IM injection) (n = 430) or three separate doses of 1.0 mL of the JE vaccine JE-Vax (on a 0, 7 and 28 day schedule by subcutaneous injection) (n = 437). The co-primary endpoint was seroconversion rate (anti-JE virus antibody titer ≥ 1:10) and geometric mean titer (GMT) at Day 56 as assessed by a Plaque Reduction Neutralisation Test (PRNT) for the entire study population.
By Day 56, the proportion of subjects who had seroconverted was similar for both treatment groups (96.4% vs. 93.8% for Jespect and JE-Vax, respectively) (see Table 3). Geometric mean titers increased by Day 56 to 243.6 for Jespect and to 102.0 for JE-Vax, respectively. The immune responses elicited by Jespect were non-inferior to those induced by JE-Vax (see Table 3).
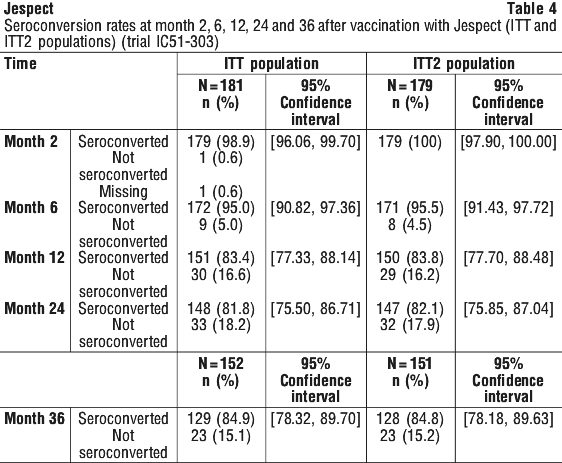
Antibody persistence. Antibody persistence was assessed in an uncontrolled Phase 3 follow-up clinical trial (trial IC51-303) of subjects who completed the pivotal immunogenicity study (trial IC51-301) or the pivotal safety study (trial IC51-302), and who received at least one dose of Jespect. The primary objective of the study was the evaluation of the immune response (seroconversion rate) to Jespect 24 months after the first vaccination. Secondary objectives included the evaluation of the immune response to Jespect 6, 12, 24 and 36 months after the first vaccination and to evaluate the safety of Jespect during the respective study period (see Section 4.8 Adverse Effects (Undesirable Effects)). A total of 3,258 healthy male and female subjects were enrolled (mean age: 35.6 years, 56.5% female, race: Caucasian 90.2%, Asian 1.7%, Black/African American 4.9%, Other 3.2%) of which 2,283 subjects received Jespect, 338 subjects received JE-Vax, and 637 subjects received placebo in the respective previous study. Long-term immunogenicity to Jespect was assessed in a subset of 181 subjects up to 24 months, and a subset of 152 subjects at 36 months (ITT population). Immunogenicity data covering a period up to 36 months after the first vaccination are described below.
Seroconversion rates for anti-JE virus antibodies at Months 2, 6, 12, 24 and 36 are summarized in Table 4 for the ITT population. At Month 2, 98.9% of subjects had seroconverted (95% CI: 96.06, 99.70), at Month 6, 95.0% of subjects had seroconverted (95% CI: 90.82, 97.36). By Month 12, the percentage of subjects who had seroconverted was 83.4% (95% CI: 77.33, 88.14). Seroconversion at 24 months and 36 months was 81.8% (95% CI: 75.50, 86.71) and 84.9% (95% CI: 78.32, 89.70) respectively.
In the ITT2 population, which is a subset of the ITT population with positive PRNT results at Month 2, per definition, 100% of the subjects (N = 179) had seroconverted at Month 2 (95% CI: 97.90%, 100.00%). By Month 6 and Month 12, the number of subjects of the ITT2 population who had seroconverted was 95.5% (95% CI: 91.43, 97.72) and 83.8% (95% CI: 77.70, 88.48) respectively (see Table 4). The number of subjects of the ITT2 population who had seroconverted at 24 months was 82.1% (95% CI: 75.85, 87.04) and at 36 months was 84.8% (95% CI: 78.18, 89.63).
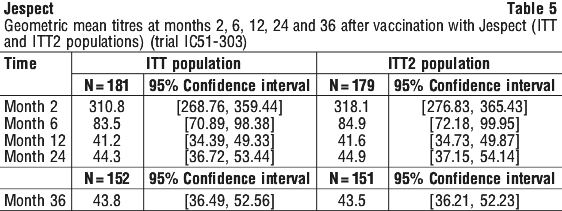
Vaccination with inactivated JE vaccine is known to produce antibodies that decline with time in a proportion of the vaccinated population. The observed decline in GMT is therefore as expected and compares well with data from other inactivated JE vaccines.
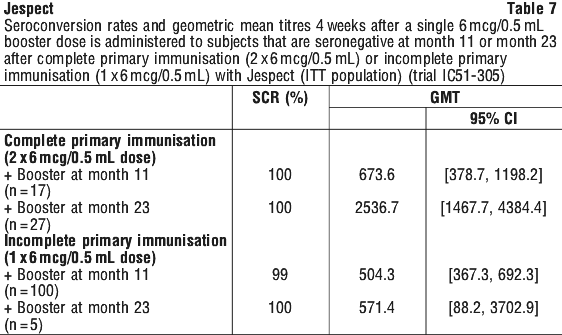
Results of another Phase 3 follow-up clinical trial (trial IC51-305) support these findings. In this study, 58.3% and 48.3% of subjects who received the recommended primary vaccination series (see Section 4.2 Dose and Method of Administration) showed persisting protective antibodies 12 and 24 months (respectively) after completion of primary vaccination. See Incomplete primary immunisation.
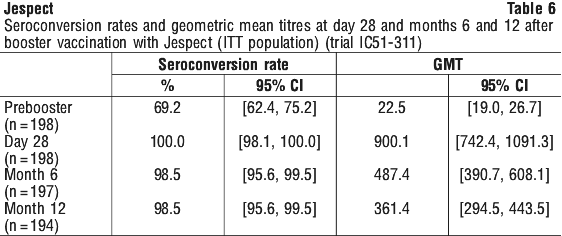
Booster immunisation. The effect of booster vaccination of Jespect on long-term immunogenicity was investigated in an uncontrolled, open label, Phase 3 clinical trial in healthy male and female adults (trial IC51-311). Subjects (n = 198) were administered one 6 microgram/0.5 mL dose of Jespect 15 months after their first dose (Day 0) of the recommended primary vaccination series (see Section 4.2 Dose and Method of Administration). The primary endpoint was assessment of the effect of booster vaccination on immunogenicity in terms of seroconversion rate 12 months after administration of the booster dose (equivalent to 27 months after first dose (Day 0) of the recommended primary vaccination series). Secondary endpoints included assessment of seroconversion 28 days and 6 months after administration of the booster dose, and GMT 28 days, 6 months and 12 months after booster vaccination (equivalent to 16, 21 and 27 months after first dose (Day 0) of the recommended primary vaccination series).
Pre-booster vaccination, 69.2% of subjects had seroconverted and GMT was 22.5 (95% CI: 19.0, 26.7).
Seroconversion was 100% at 28 days after administration of the booster vaccination and remained high (98.5%) to 12 months after administration of the booster vaccination (equivalent to 27 months after first dose (Day 0) of the recommended primary vaccination series) (see Table 6).
Geometric mean titers were highest 28 days after receiving the booster vaccination and declined to 361.4 after 12 months (equivalent to 27 months after first dose (Day 0) of the recommended primary vaccination series) (see Table 6).
Administration of the second injection of the primary immunisation series up to 11 months after the first dose results in 99% seroconversion and GMT of 504.3 (95% CI 367.3, 692.3) (see Table 7).
5.2 Pharmacokinetic Properties
No data available.
5.3 Preclinical Safety Data
Animal studies in mice, rats and rabbits have shown that the Jespect vaccine induces the immune system to produce neutralising antibodies against Japanese encephalitis virus which correlate with protection and survival. Mice treated with Jespect vaccine or human Jespect antisera demonstrated protection against lethal Japanese encephalitis challenge from several different strains (SA14, Beijing and KE-093) in a generally dose- or titre-dependent manner, respectively.
Genotoxicity. Jespect has not been evaluated for genotoxic potential.
Carcinogenicity. Jespect has not been evaluated for carcinogenic potential.
6 Pharmaceutical Particulars
6.1 List of Excipients
See Section 2 Qualitative and Quantitative Composition.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store in a refrigerator at 2° to 8°C. Do not freeze. Store in the original package in order to protect from light.
Do not use if the package is torn or damaged.
Do not use the vaccine after the expiration date shown on the label.
Upon storage, a fine white deposit with a clear colourless supernatant can be observed. Shake well before use.
6.5 Nature and Contents of Container
Packed in a pre-filled syringe (Type I glass) with a plunger stopper (chlorobutyl elastomer).
Jespect is available in a pack size of 1 needle-less syringe containing 0.5 mL dose.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure. No data available.
CAS number. Not applicable.
7 Medicine Schedule (Poisons Standard)
Prescription Only Medicine (S4).
Date of First Approval
30 January 2009
Date of Revision
12 October 2020
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.