Kenalog in Orabase
Brand Information
| Brand name | Kenalog in Orabase |
| Active ingredient | Triamcinolone acetonide |
| Schedule | S3 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Kenalog in Orabase.
Summary CMI
Kenalog in Orabase
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Kenalog in Orabase?
Kenalog in Orabase contains the active ingredient triamcinolone acetonide. It is used for relieving tenderness, pain, inflammation and ulceration of the inside of the mouth or the gums.
For more information, see Section 1. Why am I using Kenalog in Orabase? in the full CMI.
2. What should I know before I use Kenalog in Orabase?
Do not use if you have ever had an allergic reaction to Kenalog in Orabase or any of the ingredients listed at the end of the CMI.
Talk to your doctor, dentist or pharmacist if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Kenalog in Orabase? in the full CMI.
3. What if I am taking other medicines?
It is unlikely that other medicines will affect the way Kenalog in Orabase works. Nevertheless, you should tell your doctor, dentist, or pharmacist about other medicines you are taking, even if those medicines have been bought without a prescription.
For more information, see Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Kenalog in Orabase?
- Dab a small quantity, about 6-10 mm onto the sore area in your mouth. DO NOT RUB IT IN.
- Kenalog in Orabase should be applied at night before bed, or after meals if using it multiple times a day.
- Use Kenalog in Orabase until all symptoms have resolved, or for as long as your doctor, dentist, pharmacist, or other healthcare professional tells you to.
More instructions can be found in Section 4. How do I use Kenalog in Orabase? in the full CMI.
5. What should I know while using Kenalog in Orabase?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Kenalog in Orabase? in the full CMI.
6. Are there any side effects?
Side effects for Kenalog in Orabase are extremely rare. Side effects that may occur include worsening of the sores inside the mouth, new patches of irritation or sensitivity in the mouth, and weakness or dizziness. If corticosteroids are used for long periods during pregnancy, the mother may develop fluid retention, or the baby may be affected. However these effects are very rare, and usually only occur when using much higher doses of corticosteroids than the amount present in Kenalog in Orabase.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Kenalog in Orabase
Active ingredient: triamcinolone acetonide
Consumer Medicine Information (CMI)
This leaflet provides important information about using Kenalog in Orabase. You should also speak to your doctor, dentist or pharmacist if you would like further information or if you have any concerns or questions about using Kenalog in Orabase.
Where to find information in this leaflet:
1. Why am I using Kenalog in Orabase?
2. What should I know before I use Kenalog in Orabase?
3. What if I am taking other medicines?
4. How do I use Kenalog in Orabase?
5. What should I know while using Kenalog in Orabase?
6. Are there any side effects?
7. Product details
1. Why am I using Kenalog in Orabase?
Kenalog in Orabase is an oral sticky paste that contains the active ingredient triamcinolone acetonide, which belongs to a class of medicines known as corticosteroids.
Corticosteroids, including triamcinolone acetonide, are used to relieve itching, burning, inflammation and allergies.
Kenalog in Orabase is used for relieving tenderness, pain, inflammation and ulceration of the inside of the mouth or the gums. This can include sores caused by injury or by dentures (false teeth) rubbing on the gums or by conditions such as lichen planus.
The sticky paste keeps the medicine on the sore area and protects it while the medicine relieves the soreness, reduces the swelling and accelerates the healing process.
It may be prescribed by your doctor or bought at a pharmacy when recommended by your dentist, pharmacist or other healthcare professional.
2. What should I know before I use Kenalog in Orabase?
Warnings
Do not use Kenalog in Orabase if:
- You are allergic to triamcinolone acetonide or any of the ingredients listed at the end of this leaflet.
- Some symptoms of an allergic reaction may include shortness of breath, wheezing or difficulty in breathing; swelling of the face, lips, tongue, or any other parts of the body; rash, itching or hives on the skin.
- Always check the ingredients to make sure you can use this medicine. Check with your doctor or pharmacist if you are unsure. - You have an infection in your mouth or throat
- The packaging is faulty or has been tampered with.
In this case, show it to your pharmacist. - The expiry date printed on the pack has passed.
Check with your doctor, dentist, or pharmacist if you:
- Have any other medical conditions, especially the following:
- Infections in the mouth or throat, especially herpes (cold sores)
- Tuberculosis (TB)
- Stomach ulcers
- Diabetes. - Take any medicines for any other condition
- Have had any allergy to any other medicines, or any other substances, such as foods, preservatives, or dyes.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy
Tell your doctor or dentist if you are pregnant or become pregnant while you are using this medicine.
3. What if I am taking other medicines?
It is unlikely that other medicines will affect the way Kenalog in Orabase works. Nevertheless, you should tell your doctor, dentist or pharmacist about other medicines you are taking, even if those medicines have been bought without a prescription.
Check with your doctor, dentist or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Kenalog in Orabase.
4. How do I use Kenalog in Orabase?
Where to use it
- Kenalog in Orabase is only for use in your mouth.
- You should not swallow this medicine.
- You should not put it in your eyes.
How to use it
- To open, pierce the sealed tube end with the cap point.
- Dab a small quantity, about 6-10 mm (about half the size of a five cent coin) onto the sore area in your mouth. You may need to use a little more if the sore is large. For the best results, use only enough to coat the sore with a thin film.
- DO NOT RUB IT IN. Do not attempt to spread the paste as it might cause a gritty sensation in the mouth and cause the paste to crumble. After applying it correctly, a smooth, slippery film should develop.
- Wipe the neck of the tube and replace the cap tightly after use.
When to use it
Apply the preparation at bedtime, to allow the medication to work overnight.
Sometimes it may be necessary to apply the preparation two or three times a day, preferably after meals.
How long to use it for
Use Kenalog in Orabase until all symptoms have resolved, or for as long as your doctor, dentist, pharmacist, or other healthcare professional tells you to.
If you forget to use Kenalog in Orabase
If it is almost time for your next dose, skip the dose you missed and use your next dose when you are meant to.
Do not use a double dose to make up for the dose you missed.
If you use too much Kenalog in Orabase
If you think that you have used too much Kenalog in Orabase, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using Kenalog in Orabase?
Things you should do
- Tell your doctor if you become pregnant while you are using this medicine.
- Stop using it if your condition is getting worse, or if you feel any weakness or dizziness while using it. Speak to your doctor or pharmacist if you notice any of these things.
- Remind any doctor, dentist or pharmacist you visit that you are using Kenalog in Orabase.
Things you should not do
- Do not swallow Kenalog in Orabase
- Do not put it in your eyes.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Kenalog in Orabase affects you.
Looking after your medicine
Keep Kenalog in Orabase in a cool dry place, away from moisture or heat, where the temperature stays below 25°C.
For example, do not store it:
- In the bathroom or near a sink, or
- In the car or on window sills
Heat and dampness can destroy some medicines.
Keep it where young children cannot reach it.
A locked cupboard at least one and a half metres above the ground is a good place to store medicines.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor, dentist or pharmacist if you have any further questions about side effects.
Side effects for Kenalog in Orabase are extremely rare.
Side effects
| Side effects | What to do |
| Stop using Kenalog in Orabase and speak to your doctor if you notice any of these side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Kenalog in Orabase contains
| Active ingredient (main ingredient) |
|
| Other ingredients (inactive ingredients) |
|
Do not take this medicine if you are allergic to any of these ingredients.
What Kenalog in Orabase looks like
Kenalog in Orabase is an opaque light brown dental paste in a 5 g sealed aluminium tube.
(AUST R 19205)
Who distributes Kenalog in Orabase
Aspen Pharmacare Australia Pty Ltd
34-36 Chandos St
St Leonards NSW 2065
Australia
This leaflet was prepared in September 2024.
Brand Information
| Brand name | Kenalog in Orabase |
| Active ingredient | Triamcinolone acetonide |
| Schedule | S3 |
MIMS Revision Date: 01 November 2022
1 Name of Medicine
Triamcinolone acetonide.
2 Qualitative and Quantitative Composition
Each gram of Kenalog in Orabase provides 1 mg (0.1%) triamcinolone acetonide.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
An opaque light brown ointment.
4 Clinical Particulars
4.1 Therapeutic Indications
Acute and chronic lesions of the oral mucosa including recurrent ulcerative aphthous stomatitis; erosive lichen planus; denture stomatitis; traumatic lesions including denture sore spots; desquamative gingivitis and stomatitis. Lesions of traumatic origin and most nonrecurring conditions heal rapidly and permanently. Chronic and recurrent lesions are promptly relieved, but symptoms usually recur after termination of therapy. However, Kenalog in Orabase is effective in suppressing subsequent recurrences.
4.2 Dose and Method of Administration
Press a small dab (about 1 cm) to the lesion until a thin film develops. A larger quantity may be required for coverage of some lesions. For optimal results use only enough to coat the lesion with a thin film. Do not rub in. Attempting to spread this preparation may result in a granular, gritty sensation and cause it to crumble. After application, however, a smooth, slippery film develops.
The preparation should be applied at bedtime to permit steroid contact with the lesion throughout the night. Depending on the severity of symptoms, it may be necessary to apply the preparation 2 or 3 times a day, preferably after meals.
4.3 Contraindications
Patients with a history of hypersensitivity to any of its components. Because it contains a corticosteroid, it is contraindicated in the presence of fungal or bacterial infections of the mouth or throat. It should not be used in herpetic lesions of known viral origin such as herpes labialis, or intraoral lesions such as primary herpetic gingival stomatitis and herpanginas.
4.4 Special Warnings and Precautions for Use
The small amount of steroid released when the preparation is used as recommended makes systemic effects very unlikely; however, they are a possibility when topical corticosteroid preparations are used over a long period of time, and any unusual symptoms such as weakness or dizziness should be called to the physician's attention by the patient.
If local irritation or sensitisation should develop, the preparation should be discontinued and appropriate therapy instituted.
Patients with tuberculosis, peptic ulcer or diabetes mellitus should not be treated with any corticosteroid preparation without the advice of the patient's physician.
It should be borne in mind that the normal defensive responses of the oral tissues are depressed in patients receiving topical corticosteroid therapy. Virulent strains of oral microorganisms may multiply without producing the usual warning symptoms of oral infections.
If significant regeneration or repair of oral tissues has not occurred in seven days, additional investigation into the oral lesion is advised.
Use in the elderly. No data available.
Paediatric use. No data available.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
No data available.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category C)
Safe use of this preparation during pregnancy has not been established with respect to possible adverse reactions upon fetal development. Use during pregnancy is not recommended unless the physician or dentist feels that the benefits outweigh the risks.
In animal experiments, corticosteroids have been found to cause malformations of various kinds (cleft palate, skeletal malformations) and abortion. These findings do not seem to be relevant to humans. Reduced placental and birth weight have been recorded in humans after long-term treatment. Since the possibility of suppression of the adrenal cortex in the newborn baby after long-term treatment must be considered, the needs of the mother must be carefully weighed against the risk to the fetus when prescribing these drugs. The short-term use of corticosteroids antepartum for the prevention of respiratory distress syndrome does not seem to pose a risk for the newborn infant. Maternal pulmonary oedema has been reported with tocolysis and fluid overload.
Use in lactation. No data available.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
Intolerance to the preparation is extremely rare. No topical reactions to the corticosteroid have been observed with intraoral application.
Prolonged administration may elicit the adverse reactions known to occur with systemic steroid preparations, for example adrenal suppression, alteration of glucose metabolism, protein catabolism, peptic ulcer activations and others.
These are usually reversible and disappear when the hormone is discontinued.
4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Kenalog is a corticosteroid with anti-inflammatory, antipruritic and antiallergic action, which may provide prompt relief of oral tenderness, pain, inflammation and ulceration. The emollient dental paste is an adhesive vehicle for applying medication to the oral surfaces. This adhesive maintains the medication in close contact with the lesion and provides a protective covering which augments the effects of the steroid.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
No data available.
5.3 Preclinical Safety Data
No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Gelatin, pectin, carmellose sodium and plastibase (a plasticised hydrocarbon gel - a polyethylene and mineral oil gel base).
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25⁰C.
6.5 Nature and Contents of Container
5 g dental paste in a sealed aluminium tube.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure.
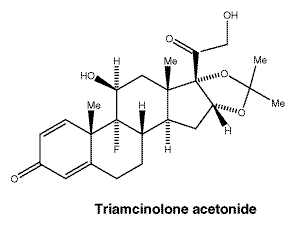
7 Medicine Schedule (Poisons Standard)
Schedule 3 - Pharmacist Only Medicine.
Date of First Approval
30 September 1991
Date of Revision
15 September 2022
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.