Lanoxin Tablets & Liquid
Brand Information
| Brand name | Lanoxin Tablets & Liquid |
| Active ingredient | Digoxin |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Lanoxin Tablets & Liquid.
Summary CMI
LANOXIN tablets & oral liquid
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I being given LANOXIN?
LANOXIN contains the active ingredient digoxin. It is used to treat certain heart problems like chronic heart failure, and certain types of irregular heartbeats.
For more information, see Section 1. Why am I being given LANOXIN? in the full CMI.
2. What should I know before I use LANOXIN?
Do not use if you have ever had an allergic reaction to digoxin, other cardiac glycosides, or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use LANOXIN? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with LANOXIN and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use LANOXIN?
- Tablets: Swallow LANOXIN tablets with a glass of water.
- Oral liquid: Accurately measure the dose with the calibrated dropper provided. Do not dilute.
- Your doctor will tell you how much LANOXIN to take and how long you use it for.
- It does not matter if you take LANOXIN before or after food.
More instructions can be found in Section 4. How do I use LANOXIN? in the full CMI.
5. What should I know while using LANOXIN?
| Things you should do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using LANOXIN? in the full CMI.
6. Are there any side effects?
Side effects of LANOXIN generally occur when using higher doses than needed. Tell your doctor immediately if you notice any side effects while using LANOXIN.
Side effects include nausea, vomiting or loss of appetite; diarrhoea; lower stomach pain; slow or irregular heart beats (may be fast in children); unusual tiredness or extreme weakness.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
LANOXIN tablets & oral liquid
Active ingredient: digoxin
Consumer Medicine Information (CMI)
This leaflet provides important information about using LANOXIN tablets & oral liquid.
You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using LANOXIN.
Where to find information in this leaflet:
1. Why am I being given LANOXIN?
2. What should I know before I use LANOXIN?
3. What if I am taking other medicines?
4. How do I use LANOXIN?
5. What should I know while using LANOXIN?
6. Are there any side effects?
7. Product details
1. Why am I being given LANOXIN?
LANOXIN contains the active ingredient digoxin. It belongs to a group of medicines known as cardiac glycosides. They work by slowing down the rate while increasing the force of your heart when it beats.
It is used to treat certain heart problems, such as:
Chronic heart failure
Heart failure is when your heart can't pump strongly enough to supply blood needed throughout the whole body. It is not the same as a heart attack and does not mean that your heart stops.
Certain types of irregular heartbeats
Irregular heart rhythms caused by an electrical problem in the upper chamber of your heart. They cause your heart to beat too fast or in an uneven way.
Ask your doctor if you have any questions about why LANOXIN has been prescribed for you.
Your doctor may have prescribed it for another reason.
There is no evidence that LANOXIN is addictive.
2. What should I know before I use LANOXIN?
Warnings
You must not be given LANOXIN if:
- you are allergic to digoxin, other cardiac glycosides, or any of the ingredients listed at the end of this leaflet.
Symptoms of an allergic reaction may include shortness of breath, wheezing or difficulty in breathing; swelling of the face, lips, tongue, or any other parts of the body; rash, itching or hives on the skin. - you have or have had any of the following medical conditions:
- intermittent or complete heart block or second degree atrioventricular block, (conditions where the heart misses beats) especially if there is a history of Stokes-Adams attacks
- ventricular tachycardia and ventricular fibrillation (a type of irregular heart rhythm)
- irregular heart rhythm caused by cardiac glycoside overdose.
- certain other irregularities of heart rhythm. - the expiry date printed on the pack has passed.
- the packaging is torn or shows signs of tampering.
Check with your doctor if:
- you have allergies to any other medicines, foods, preservatives or dyes.
- you have or have had any other medical conditions, especially the following:
- heart disease
- heart disease caused by lack of vitamin B, known as ‘Beri-Beri disease’
- lung problems
- liver or kidney problems – effect of LANOXIN may be increased because of slower removal from the body
- thyroid disease – patients with low or high thyroid gland activity may be more or less sensitive to the effects of LANOXIN
- high or low levels of calcium, potassium in your blood
- low level of magnesium in your blood
- digestion problems.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Tell your doctor if you are pregnant or intend to become pregnant.
Pregnant women may require an adjustment of the dose of this medicine during pregnancy. It may have harmful effects on the developing baby if the dose is too high.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with LANOXIN and affect how it works. These include:
- amphetamine, appetite suppressants, medicines for colds, sinus problems, hay fever or other allergies – may increase the risk of heart rhythm problems
- medicines for heart problems, including high blood pressure (hypertension) and irregular heartbeat (arrhythmia)
- diuretics (water tablets)
- potassium supplements
- medicines to treat all types of cancer
- medicines for depression
- medicines for bacterial infection (antibiotics)
- medicines for fungal infections (antifungals)
- medicines for stomach problems, including for nausea, vomiting, diarrhoea or indigestion
- some laxatives
- medicines for relieving asthma attacks or other breathing problems
- corticosteroids (e.g. hydrocortisone, dexamethasone, prednisolone, prednisone)
- medicines for diabetes (e.g. insulin, glucagon)
- cholestyramine, a medicine used for high cholesterol
- phenytoin, a medicine used to control epilepsy
- indomethacin – medication used for inflammation, gout or arthritis
- St John's Wort.
These medicines may be affected by LANOXIN or may affect how well it works. You may need different amounts of your medicine, or you may need to take different medicines. You doctor will advise you.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect LANOXIN.
4. How do I use LANOXIN?
How much to take
Your doctor will tell you how much LANOXIN you will receive. This depends on your condition and other factors such as your weight, age and other medicines you may be taking.
How to take it
Tablets:
Swallow LANOXIN tablets with a glass of water.
Oral liquid:
Accurately measure the dose with the calibrated dropper provided. Do not dilute.
Using the dropper will make sure that you get the correct dose.
Do not change your dose of LANOXIN unless your doctor tells you to do so.
After you begin taking LANOXIN, your doctor may sometimes check its level in your blood. This will help your doctor find out if your dose needs to be changed.
When to take it
Take LANOXIN at the same time each day.
This will have the best effect. It will also help you remember when to take it.
It does not matter if you take LANOXIN before or after food.
How long to take it
Continue taking LANOXIN for as long as your doctor tells you.
LANOXIN helps control your condition but does not cure it. Therefore, you must take LANOXIN every day.
If you forget to take it
Take it as soon as you remember. However, if it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Do not take a double dose to make up for the dose that you missed.
This may increase the chance of you getting an unwanted side effect.
If you are not sure what to do, ask your doctor or pharmacist.
If you take too much
If you think that you or someone you know has taken too much LANOXIN, medical attention may be needed.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
Symptoms of an overdose to LANOXIN include loss of appetite, nausea, vomiting, diarrhoea, or extremely slow heartbeat. In infants and small children, the earliest signs of overdose are changes in the rate and rhythm of the heartbeat. Carers should be aware that children may not show symptoms as soon as adults.
5. What should I know while using LANOXIN?
Things you should do
Remind any other doctors, dentists and pharmacists you visit that you are being given LANOXIN.
If you plan to have surgery that needs a general anaesthetic, tell your doctor or dentist that you are being given LANOXIN.
If you need to have any medical tests while you are being given it, tell your doctor.
This medicine may affect the results of some tests.
Be sure to keep all your doctor's appointments so that your progress can be checked.
Your doctor will check your progress and may want to take some blood tests from time to time. This helps to prevent unwanted side effects.
Call your doctor straight away if you:
- become pregnant while using LANOXIN.
- notice any side effects from this medicine.
Things you should not do
Do not give LANOXIN to anyone else, even if their symptoms seem similar or they have the same condition as you.
Do not take LANOXIN to treat any other complaints unless your doctor tells you to.
Do not stop taking LANOXIN or change the dose even if you are feeling better, without checking with your doctor.
Driving or using machines
Be careful driving or operating machinery until you know how LANOXIN affects you.
LANOXIN may cause dizziness, blurred vision, and faintness in some patients. Make sure you know how you react to it before you drive a car, operate machinery, or do anything else that could be dangerous if this happens.
Looking after your medicine
Store LANOXIN-PG 62.5 microgram tablets in the bottle in a cool, dry place away from light where the temperature stays below 30°C.
Store LANOXIN 250 microgram tablets in the blister pack and LANOXIN oral liquid in the glass bottle in a cool, dry place away from light where the temperature stay below 25°C.
For example, do not store LANOXIN:
- in the bathroom or near a sink, or
- in the car or on window sills.
Heat and dampness can destroy some medicines.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Serious side effects | What to do |
| Tell your doctor if you have any of these less serious side effects. They may be caused by a dose of LANOXIN that is too high for you. |
Serious side effects
| Serious side effects | What to do |
| Tell your doctor immediately or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. Serious side effects are rare. |
| Tell your doctor immediately or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Your doctor may do regular checks on your blood levels of digoxin and body salts, as well as your kidney function to make sure this medicine is working safely for you
Tell your doctor if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What LANOXIN-PG 62.5 microgram tablets contain
| Active ingredient (main ingredient) | digoxin 62.5 microgram/tablet |
| Other ingredients (inactive ingredients) |
|
| Potential allergens |
|
What LANOXIN 250 microgram tablets contain
| Active ingredient (main ingredient) | digoxin 250 microgram/tablet |
| Other ingredients (inactive ingredients) |
|
| Potential allergens |
|
What LANOXIN PAEDIATRIC ELIXIR oral liquid contains
| Active ingredient (main ingredient) | digoxin 250 microgram/1 mL |
| Other ingredients (inactive ingredients) |
|
| Potential allergens |
|
Do not take this medicine if you are allergic to any of these ingredients.
What LANOXIN tablets & oral liquid look like
LANOXIN-PG 62.5 microgram tablets
Blue, biconvex, round tablet coded “U3A” on the upper face, bottom face plain. Available in glass bottles of 200 tablets.
LANOXIN 250 microgram tablets
White, biconvex, round tablet scored and marked “X3A” on one side. Available in blister packs of 100 tablets.
LANOXIN PAEDIATRIC ELIXIR 50 microgram/mL oral liquid
Clear, bright yellow liquid with a characteristic odour. Available in a 60 mL bottle with a calibrated dropper.
Do not dilute.
Australian Registration numbers:
- LANOXIN-PG 62.5 microgram tablets: AUST R 11108
- LANOXIN 250 microgram tablets: AUST R 15333
- LANOXIN PAEDIATRIC ELIXIR 50 microgram/mL oral liquid: AUST R 42757.
Who distributes LANOXIN
Aspen Pharmacare Australia Pty Ltd
34-36 Chandos Street
St Leonards NSW 2065
Australia
This leaflet was revised in October 2025.
Brand Information
| Brand name | Lanoxin Tablets & Liquid |
| Active ingredient | Digoxin |
| Schedule | S4 |
MIMS Revision Date: 01 November 2025
1 Name of Medicine
Digoxin.
2 Qualitative and Quantitative Composition
Lanoxin tablets. Contain digoxin 250 microgram. Excipients with known effect: sugars (as lactose monohydrate).
Lanoxin PG tablets. Contain digoxin 62.5 microgram. Excipients with known effect: sugars (as lactose monohydrate).
Lanoxin paediatric elixir. Each mL contains digoxin 50 microgram. Excipients with known effect: sugars (as sucrose), alcohol (as ethanol 10.6% v/v), hydroxybenzoates.
Lanoxin adult injection. Each 2 mL contains digoxin 500 microgram. Excipients with known effect: alcohol (as ethanol 10.6% v/v).
Lanoxin infants injection. Each 2 mL contains digoxin 50 microgram. Excipients with known effect: alcohol (as ethanol 10.6% v/v).
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Lanoxin tablets. White, biconvex, round tablet scored and coded "X3A" on the upper face. Bottom face plain.
Lanoxin PG tablets bottle. Blue, biconvex, round tablet, coded "U3A" on the upper face. Bottom face plain.
Lanoxin PG tablets blister pack. Blue, biconvex, round tablet, coded "Wellcome U3A" on the upper face.
Lanoxin paediatric elixir. Clear, bright yellow liquid with a characteristic odour. Free of obvious visible contamination.
Lanoxin adult injection. Clear colourless liquid, free of obvious visible contamination.
Lanoxin infants injection. Clear colourless liquid, free of obvious visible contamination.
4 Clinical Particulars
4.1 Therapeutic Indications
Congestive heart failure. Lanoxin is useful regardless of whether the failure is predominantly of the left or right ventricle or involves both sides of the heart. It is particularly useful in heart failure resulting from chronic overload (hypertension, valvular lesions, atherosclerotic heart disease) in which the supply of energy is not impaired. Lanoxin does not cause major benefit in situations in which the metabolic energy supply is compromised as in thyrotoxicosis, hypoxia and severe thiamine deficiency.
Atrial fibrillation. Because Lanoxin depresses conduction in the atrioventricular bundle, producing a slower ventricular beat, it is valuable in atrial fibrillation. It will frequently convert atrial flutter into fibrillation and, upon withdrawal of the drug, normal sinus rhythm may be restored.
Paroxysmal atrial tachycardia. Lanoxin may relieve or prevent an attack, but its use in paroxysmal ventricular tachycardia is dangerous.
4.2 Dose and Method of Administration
Dosage of Lanoxin must be adjusted individually for each patient and the suggested doses are intended only as a guide. Use of the drug in heart failure should be based on the principles that optimum doses are not necessarily maximum tolerated doses and that a small dose does improve cardiac performance.
When the decision to treat a patient with Lanoxin has been made, two important factors must be considered. First, how urgent is it to achieve a therapeutic response, and second, are there any factors present likely to influence patient drug sensitivity? A stable therapeutic blood (and tissue) level of Lanoxin can be achieved if the drug is started on an appropriate maintenance dosage regimen, but 5 to 7 days will elapse before an optimal effect is achieved. If there is no clinical urgency, this approach is preferred for outpatient and domiciliary digitalisation. In emergencies, a loading or digitalising dose of Lanoxin may be given for a more rapid onset of the clinical effect. The distribution of Lanoxin into body fat is poor. Dosage in obesity should therefore be based on lean bodyweight and not total bodyweight.
Monitoring. The suitability of dosage regimens can be assessed by determination of the serum digoxin concentration using radioimmunoassay. Blood for digoxin level determination must be taken at least 6 hours after the last dose.
There are no rigid guidelines as to the range of serum concentrations that are most efficacious. Several post hoc analyses of heart failure patients in the digitalis investigation group trial suggest that the optimal trough digoxin serum level may be 0.5 to 1.0 nanogram/mL. Digoxin toxicity is more commonly associated with serum digoxin concentration greater than 2 nanogram/mL. However, in deciding whether a patient's symptoms are due to digoxin, the clinical state together with serum electrolyte levels and thyroid function are important factors. (See Section 4.9 Overdose.)
Other glycosides, including digoxin metabolites, spironolactone and its metabolites, certain herbs and endogenous digoxin-like immunoreactive substances may interfere with some assays. The clinician should always be wary of values which do not seem commensurate with the clinical state of the patient.
Ambulatory patients. In general, these patients are seen in domiciliary or outpatient practice, are relatively well, but require Lanoxin. Continued maintenance treatment is generally thought necessary for patients who have been treated with Lanoxin for heart failure.
Adults and children over 10 years. For patients with normal renal function 250 microgram as a single daily dose or, when required, up to 500 microgram daily in divided doses. The patient should be reviewed within 7 to 10 days and the dosage adjusted if necessary.
Elderly. The elderly require smaller doses of Lanoxin because impairment of renal function in the elderly is common and also because the sensitivity of the myocardium to the drug is thought to be increased. Therefore, it is important to assess renal function in elderly patients on Lanoxin therapy. Consideration may be given to determining serum digoxin concentration after 5 to 7 days' therapy. A usual dose is 125 microgram daily. This may be given in divided doses as 1 Lanoxin PG tablet twice daily.
Emergency administration. This is necessary when the condition of the patient warrants the rapid achievement of a therapeutic response to Lanoxin, for example in pulmonary oedema or atrial fibrillation. These circumstances would usually require the patient to be treated in a hospital with ancillary services available.
A therapeutic effect may be achieved more rapidly by giving a loading or digitalising dose.
Adults and children over 10 years. 1. Rapid oral digitalisation. 0.75 to 1.5 mg (10 to 20 microgram/kg) as a single dose, or as 3 or 4 divided doses at intervals of four to six hours, followed by appropriate maintenance treatment.
2. Emergency parenteral digitalisation. (In patients who have not been given cardiac glycosides within the preceding two weeks.) The total digitalising dose with parenteral Lanoxin is 0.5 to 1 mg. This should be given slowly by intravenous route over at least 5 minutes in divided doses each of 0.25 to 0.5 mg, at intervals of four to six hours.
Elderly. 1. Rapid oral digitalisation. In elderly patients a smaller initial dose of 500 to 750 microgram may be given as a single dose, or as 3 divided doses at intervals of four to six hours, followed by appropriate maintenance treatment.
2. Emergency parenteral digitalisation. The total digitalising dose with parenteral Lanoxin in the elderly is 250 to 500 microgram. This may be given slowly over at least 5 minutes by the intravenous route, either as a single dose or in divided doses each of 125 to 250 microgram at intervals of four to six hours.
Infants and children under 10 years. Maintenance. 10 to 20 microgram/kg bodyweight daily in single or divided doses. The lower dose range applies in neonates and when Lanoxin is administered parenterally.
Digitalisation (all routes). 10 to 20 microgram/kg, repeated every six hours until a therapeutic result is obtained, usually after 2 to 4 doses.
Slow intravenous infusion is the preferred route when oral administration is not indicated. Intramuscular injection is not generally advised because it may cause unpredictable absorption, intense local irritation and muscle necrosis. Digoxin should not be given subcutaneously. When given intravenously, Lanoxin should be infused over a period of at least 5 minutes. Lanoxin injection may be added to a 5% glucose, a 0.9% saline, or a 4% glucose plus 0.18% saline infusion fluid. Intravenous administration produces an appreciable effect within 5 to 10 minutes, a maximum effect being achieved in about 2 hours. After oral administration, the onset of effect occurs in 30 to 60 minutes and maximum effect is reached in 4 to 6 hours.
4.3 Contraindications
Lanoxin is contraindicated in the following conditions:
Intermittent complete heart block or second degree atrioventricular block, especially if there is a history of Stokes-Adams attacks.
Ventricular tachycardia and ventricular fibrillation.
Arrhythmias caused by cardiac glycoside intoxication.
Supraventricular arrhythmias associated with an accessory atrioventricular pathway, as in the Wolff-Parkinson-White syndrome, unless the electrophysiological characteristics of the accessory pathway and any possible deleterious effect of digoxin on these characteristics have been evaluated. If an accessory pathway is known or suspected to be present and there is no history of previous supraventricular arrhythmias, Lanoxin is similarly contraindicated.
Hypertrophic obstructive cardiomyopathy, unless there is concomitant atrial fibrillation and heart failure, but even then caution should be exercised if Lanoxin is to be used.
Hypersensitivity to digoxin, other digitalis glycosides, or any other constituent.
4.4 Special Warnings and Precautions for Use
Use with caution in the following circumstances:
Carotid sinus hypersensitivity. Digitalis glycosides may cause an increase in vagal tone.
Diuretics. Concurrent use with Lanoxin can predispose the patient to toxicity reactions, due to the electrolyte disturbances caused by diuretics (primarily hypokalaemia but also hypomagnesaemia and, with thiazides, hypercalcaemia). Periodic electrolyte determinations must be performed in patients receiving Lanoxin and a diuretic and corrective measures undertaken if required.
Acute glomerulonephritis, accompanied by heart failure. Use of a low total daily dose is recommended, administered in divided doses, with constant ECG monitoring; use of antihypertensives and diuretics is also recommended and Lanoxin should be withdrawn as soon as possible.
Hypercalcaemia, hyperkalaemia. Increased risk of digitalis induced arrhythmias; primarily heart block.
Hypocalcaemia. Lanoxin may be ineffective; administration of calcium may be necessary.
Hypokalaemia, hypomagnesaemia. Increased risk of digitalis toxicity. (Hypokalaemia can result from drugs, dialysis, mechanical suction of gastrointestinal secretions, malnutrition, diarrhoea, prolonged vomiting, old age, and long standing heart failure.)
Hypothyroidism, hyperthyroidism. Administering Lanoxin to a patient with thyroid disease requires care. Initial and maintenance doses of Lanoxin should be reduced when thyroid function is subnormal. Hyperthyroid patients may be less sensitive and require larger doses, while hypothyroid patients may be more sensitive and require smaller doses; dosage adjustment may become necessary as they become euthyroid.
Idiopathic hypertrophic subaortic stenosis. Use of Lanoxin may aggravate left ventricular outflow restrictions.
Ischaemic heart disease, acute myocardial infarction, acute myocarditis, myxoedema, or severe pulmonary disease. Increased sensitivity of the myocardium to the effects of Lanoxin and increased risk of digitalis induced arrhythmias.
Renal function impairment. Reduced volume of distribution and excretion of Lanoxin, hence possibility of toxicity; dosage reduction may be required; in addition, time to achieve a new or steady state concentration is increased.
Atrioventricular block. Many benefits of Lanoxin on arrhythmias result from a degree of atrioventricular conduction blockade. However, when incomplete atrioventricular block already exists the effects of a rapid progression in the block should be anticipated. In complete heart block the idioventricular escape rhythm may be suppressed.
Sick sinus syndrome. Possible worsening of sinus bradycardia or sinoatrial (SA) block.
Myocardial infarction. The administration of Lanoxin in the period immediately following myocardial infarction is not contraindicated. However, the use of inotropic drugs in some patients in this setting may result in undesirable increases in myocardial oxygen demand and ischaemia, and some retrospective follow-up studies have suggested Lanoxin to be associated with an increased risk of death. The possibility of arrhythmias arising in patients who are likely to be haemodynamically unstable must be borne in mind. The limitations imposed thereafter on direct current cardioversion must also be remembered.
Cardiac amyloidosis. Treatment with Lanoxin should generally be avoided in patients with heart failure associated with cardiac amyloidosis. Extreme caution should be exercised in situation where digoxin is needed to control the ventricular rate in patients with atrial fibrillation and cardiac amyloidosis.
Myocarditis. Lanoxin can rarely precipitate vasoconstriction and therefore should be avoided in patients with myocarditis.
Beri beri. Patients with beri beri heart disease should be treated first with thiamine, or they may fail to respond adequately to Lanoxin.
Constrictive pericarditis. Lanoxin should not be used in constrictive pericarditis.
Arrhythmias. May be precipitated by digoxin toxicity, some of which can resemble arrhythmias for which the drug could be advised. For example, atrial tachycardia with varying atrioventricular block requires particular care, as clinically the rhythm resembles atrial fibrillation. Serum digoxin concentration may be very helpful in making a decision whether to treat with further digoxin, but toxic doses of other glycosides may cross react in the assay and wrongly suggest apparently satisfactory measurements. Observations during the temporary withholding of digoxin might be more appropriate.
Rapid intravenous injection. Can cause vasoconstriction producing hypertension and/or reduced coronary flow. A slow injection rate is therefore important in hypertensive heart failure and acute myocardial infarction.
In malabsorption syndrome, or gastrointestinal reconstruction. Patients may require larger doses of Lanoxin.
Respiratory disease. Patients with severe respiratory disease may have an increased myocardial sensitivity to digitalis glycosides.
Digoxin improves exercise tolerance in patients with impaired left ventricular systolic function and normal sinus rhythm. This may or may not be associated with an improved haemodynamic profile. Digoxin slows the resting ventricular rate in patients with chronic atrial fibrillation yet it is less effective in controlling exercise induced or stress induced tachycardia in patients with atrial fibrillation.
In patients receiving diuretics and an ACE inhibitor, or diuretics alone, the withdrawal of digoxin has been shown to result in clinical deterioration.
The use of therapeutic doses of digoxin may cause prolongation of the PR interval and depression of the ST segment on the electrocardiogram. Digoxin may produce false positive ST-T changes on the electrocardiogram during exercise testing. These electrophysiological effects reflect an expected effect of the drug and are not indicative of toxicity.
In cases where cardiac glycosides have been taken in the preceding two weeks the recommendations for initial dosing of a patient should be reconsidered and a reduced dose is advised. A reduction in both initial and maintenance doses should be considered if the patients are elderly or there are other reasons for the renal clearance of digoxin being reduced.
Patients receiving digoxin should have their serum electrolytes and renal function (serum creatinine concentration) assessed periodically; the frequency of assessments will depend on the clinical setting.
Hypoxia increases risk of toxicity to digoxin.
Although many patients with chronic congestive cardiac failure benefit from acute administration of digoxin, it does not lead to constant, marked or lasting haemodynamic improvement in some. It is therefore important to evaluate the response of each patient individually when Lanoxin is continued long-term.
The intramuscular route is painful and is associated with muscle necrosis. This route cannot be recommended.
Direct current cardioversion. The risk of provoking dangerous arrhythmias with direct current cardioversion is greatly increased in the presence of digitalis toxicity and is in proportion to the cardioversion energy used.
For elective direct current cardioversion of a patient who is taking digoxin, the drug should be withheld for 1-2 days before cardioversion is performed. In emergencies, such as cardiac arrest, when attempting cardioversion the lowest effective energy should be applied.
Direct current cardioversion is inappropriate in the treatment of arrhythmias thought to be caused by cardiac glycosides.
Many beneficial effects of digoxin on arrhythmias result from a degree of atrioventricular conduction blockade. However, when incomplete atrioventricular block already exists the effects of a rapid progression in the block should be anticipated. In complete heart block, the idioventricular escape rhythm may be suppressed.
The administration of digoxin in the period immediately following myocardial infarction is not contraindicated. However, the use of inotropic drugs in some patients in this setting may result in undesirable increases in myocardial oxygen demand and ischaemia, and some retrospective follow-up studies have suggested digoxin to be associated with an increased risk of death. The possibility of arrhythmias arising in patients who may be hypokalaemic after myocardial infarction and are likely to be cardiologically unstable must be borne in mind. The limitations imposed thereafter on direct current cardioversion must also be remembered.
Use in the elderly. Appropriate studies on the relationship of age to the effects of digitalis glycosides have not been performed in the elderly. However, elderly patients are more likely to have age related hepatic and renal function impairment, which may require lower doses of Lanoxin. In addition, elderly patients may have a decreased volume of distribution for Lanoxin and electrolyte imbalances (e.g. hypokalaemia), which may require lower doses to prevent digoxin toxicity. Digoxin induced loss of appetite is a significant risk in frail elderly patients.
Paediatric use. Digitalis glycosides are a major cause of accidental poisoning in children. The tolerance of newborn infants to Lanoxin is variable, since their renal clearance of the medication is reduced. Premature and immature infants are especially sensitive. Dosage should be reduced and individualised according to the infant's degree of maturity, since renal clearance increases as the infant matures. Children over one month of age generally require proportionally larger doses than adults on the basis of bodyweight or body surface area.
Effects on laboratory tests. Laboratory test interference. Falsely elevated serum levels of digoxin may occur when samples from patients receiving enzalutamide are analysed using the chemiluminescent microparticle immunoassay (CMIA), independently of being treated with digoxin. In case of doubtful results, it is recommended to confirm digoxin serum levels with an alternative assay without known interference, in order to avoid any unnecessary discontinuation or decrease in the dose of digoxin (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
4.5 Interactions with Other Medicines and Other Forms of Interactions
These may arise from effects on the renal excretion, tissue binding, plasma protein binding, distribution within the body, gut absorptive capacity and sensitivity to Lanoxin. Consideration of the possibility of an interaction whenever concomitant therapy is contemplated is the best precaution and a check on serum digoxin concentration is recommended when any doubt exists.
Serum levels of digoxin may be increased by concomitant administration of the following:
Amiodarone, captopril, flecainide, prazosin, quinidine, spironolactone, propafenone, tetracycline, macrolide antibiotics e.g. erythromycin and clarithromycin (and possibly other antibiotics), gentamicin, itraconazole, isavuconazole, posaconazole, epoprostenol (transient), quinine, trimethoprim, alprazolam, diphenoxylate with atropine, indomethacin, propantheline, nefazodone, atorvastatin, cyclosporin, vasopressin receptor antagonists (tolvaptan and conivaptan), carvedilol, ritonavir/ ritonavir containing regimens, telaprevir, dronedarone, ranolazine, telmisartan, lapatinib, ticagrelor, daclatasvir, flibanserin, mirabegron, simeprevir, velpatasvir, canagliflozin, ivacaftor and vandetanib, venetoclax, vemurafenib and osimertinib.
Serum levels of digoxin may be reduced by concomitant administration of the following:
Antacids, kaolin-pectin, some bulk laxatives, acarbose, cholestyramine, sulphasalazine, adrenaline, salbutamol, neomycin, rifampicin, some cytostatics, phenytoin, metoclopramide, penicillamine and St John's wort (Hypericum perforatum).
Proton pump inhibitors (PPI) are able to increase plasma levels of digoxin by inhibiting its efflux. Metabolism of digoxin in the gastrointestinal tract is inhibited by omeprazole, resulting in increased plasma levels of digoxin. Similar effects have been reported with pantoprazole and rabeprazole to a lesser extent.
Angiotensin converting enzyme inhibitors may also increase or cause no change in serum digoxin levels.
Antiarrhythmic agents - although quinidine, procainamide, disopyramide, phenytoin, propranolol and lignocaine have been used effectively with Lanoxin to treat arrhythmias, these agents may have negative inotropic effects with larger than usual doses, especially in patients with Lanoxin toxicity (propranolol has negative inotropic effects with usual doses).
Adrenergic blockers can have additive negative effects on AV conduction, which can result in complete heart block. Although such combined therapy may be useful in controlling atrial fibrillation, digoxin dosage in patients receiving such therapy should be carefully individualised given the considerable variability of these interactions. Lanoxin, in association with beta-adrenoreceptor blocking drugs, may increase atrioventricular conduction time through an additive effect. This can result in complete heart block.
Calcium channel blocking agents may either increase or cause no change in serum digoxin levels. Verapamil, felodipine and tiapamil increase serum digoxin levels. Nifedipine and diltiazem may increase or have no effect on serum digoxin levels. Isradipine causes no change in serum digoxin levels. Additionally, concurrent use of verapamil and diltiazem with Lanoxin may result in excessive bradycardia due to additive depression of AV nodal conduction.
Magnesium sulfate, parenteral - must be administered with extreme caution, especially if intravenous calcium salts are also employed; cardiac conduction changes and heart block may occur.
Drugs that deplete body potassium (e.g. alcohol, diuretics, lithium salts, carbenoxolone, amphotericin B, β2-adrenergic bronchodilators, corticosteroids, corticotrophin, edetate disodium, insulin, sodium bicarbonate, laxatives, sodium polystyrene sulfonate) or that reduce extracellular potassium (e.g. glucagon, large doses of dextrose, dextrose-insulin infusions) can predispose digitalised patients to toxicity, frequent potassium determination recommended.
Drugs that increase body potassium (e.g. ACE inhibitors, spironolactone, amiloride, indomethacin, high doses of potassium containing penicillins, cyclosporin, potassium supplements, succinylcholine chloride, tacrolimus) can predispose the patient to heart block in the presence of Lanoxin. Patients receiving Lanoxin are also more susceptible to the effects of suxamethonium exacerbated hyperkalaemia. Careful monitoring of serum potassium is required.
Sympathomimetics (e.g. ephedrine, adrenaline, pseudoephedrine), parenteral calcium salts, rauwolfia alkaloids and succinylcholine should be used with caution since the risk of arrhythmias may be increased. If concurrent use with Lanoxin is required then extreme caution and electrocardiogram monitoring (ECG) is important.
St John's wort (Hypericum perforatum) - concurrent administration of digoxin and St John's wort may result in a decrease in the serum digoxin level resulting in diminished efficacy. In patients who are taking digoxin and St John's wort, the serum digoxin level should be closely monitored and administration of St John's wort should be stopped. The serum digoxin level may increase when the administration of St John's wort is stopped, therefore, resulting in the need to adjust the dose of digoxin.
Phosphodiesterase inhibitors (e.g. milrinone) does not alter steady-state digoxin levels.
Enzalutamide - determination of serum digoxin concentrations with the chemiluminescent microparticle immunoassay (CMIA) while using enzalutamide may cause falsely elevated serum digoxin levels. Results should be confirmed by another type of assay (see Section 4.4 Special Warnings and Precautions for Use).
Digoxin is a substrate of P-glycoprotein. Thus, inhibitors of P-glycoprotein may increase blood concentrations of digoxin by enhancing its absorption and/or by reducing its renal clearance.
4.6 Fertility, Pregnancy and Lactation
Use in pregnancy. (Category A)
The use of digoxin in pregnancy is not contraindicated, although the dosage may be less predictable in pregnant than in nonpregnant women with some requiring an increased dosage of digoxin during pregnancy. As with all drugs, use should be considered only when the expected clinical benefit of treatment to the mother outweighs any possible risk to the developing foetus.
Digoxin readily crosses the placenta. No significant adverse effects have been observed in the foetus or neonate when maternal serum digoxin concentrations are maintained within the normal range. Although it has been speculated that a direct effect of digoxin on the myometrium may result in relative prematurity and low birthweight, a contributing role of the underlying cardiac disease cannot be excluded (see Section 5.3 Preclinical Safety Data, Carcinogenicity).
Adverse foetal effects have been reported in mothers with digitalis toxicity, including foetal deaths.
Use in lactation. Although digoxin is excreted in breast milk, the quantities are minute and breastfeeding is not contraindicated.
Effects on fertility. There is no information available on the effect of digoxin on human fertility.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Digoxin has a narrow therapeutic index, the adverse reactions of digoxin are in general dose dependent and occur at doses higher than those needed to achieve a therapeutic effect. Hence, adverse reactions are less common when Lanoxin is used within the recommended dosage range or therapeutic serum concentration range and when there is careful attention to concurrent medications and conditions.
Adverse reactions are listed below by system organ class and frequency. Frequencies are defined as: very common (≥ 1/10), common (≥ 1/100 and < 1/10), uncommon (≥ 1/1000 and < 1/100), rare (≥ 10,000 and < 1/1000), very rare (≥ 1/10,000), including isolated reports. Very common, common and uncommon events were generally determined from clinical trial data. The incidence in placebo was taken into account. Adverse drug reactions identified through postmarketing surveillance were considered to be rare or very rare (including isolated reports).
Blood and lymphatic system disorders. Very rare: thrombocytopaenia.
Metabolism and nutrition disorders. Common: possible electrolyte imbalance.
Psychiatric disorders. Uncommon: depression.
Very rare: psychosis, apathy, confusion.
Nervous system disorders. Common: CNS disturbances, dizziness, drowsiness, headache.
Eye disorders. Common: blurred vision or visual disturbances (e.g. coloured haloes around objects - yellow, green or white vision).
Cardiac disorders. Common: arrhythmia, conduction disturbances, bigeminy, trigeminy, PR prolongation, sinus bradycardia.
Very rare: supraventricular tachyarrhythmia, atrial tachycardia (with or without block), atrioventricular block, junctional (nodal) tachycardia, ventricular arrhythmia, ventricular fibrillation, ventricular premature contraction, ST segment depression.
Gastrointestinal disorders. Common: nausea, vomiting, diarrhoea, lower stomach pain, anorexia or loss of appetite.
Very rare: intestinal ischaemia, intestinal necrosis.
Skin disorders. Common: skin rashes of urticarial or scarlatiniform character, may be accompanied by pronounced eosinophilia.
Reproductive system and breast disorders. Very rare: gynaecomastia can occur with long-term administration.
General disorders and administration site conditions. Very rare: fatigue, malaise, weakness.
Some side effects, including nausea and vomiting and some arrhythmias may also be some of the symptoms of toxicity. These are principally associated with overdose but may occur from a temporarily high serum concentration due to rapid Lanoxin absorption. It is inadvisable to rely on nausea as an early warning of excessive digoxin dosage. If there is any doubt about the cause of these symptoms, Lanoxin should be withdrawn until the cause is determined.
Cardiac. Digoxin toxicity can cause various arrhythmias and conduction disturbances. Usually an early sign of cardiac arrhythmia is the occurrence of ventricular premature contractions; they can proceed to bigeminy or even trigeminy. Atrial tachycardias, frequently an indication for digoxin, may occur with excessive dosage of the drug. Atrial tachycardia with some degree of atrioventricular block is particularly characteristic of toxicity or intolerance, and the pulse rate may not necessarily be fast.
In adults. In adults, the most common arrhythmia is premature ventricular beats (extrasystoles); paroxysmal and nonparoxysmal nodal rhythms, atrioventricular (AV) interference, and paroxysmal atrial tachycardia with block are also common; increasing AV block may occur; death may occur from ventricular fibrillation.
In children. The side effects of digoxin in infants and children differ from those seen in adults in several respects. Although digoxin may produce anorexia, nausea, vomiting, diarrhoea and CNS disturbances in young patients, they are rarely the initial symptoms of overdosage. Rather, the earliest and most frequent manifestation of excessive dosing with digoxin in infants and children is the appearance of cardiac arrhythmias, including sinus bradycardia. In children, the use of digoxin may produce any type of arrhythmia. Premature ventricular systoles are rare, while nodal and atrial systoles are more frequent; atrial arrhythmias, atrial ectopic rhythms and paroxysmal atrial tachycardia (particularly with AV block) are common; ventricular arrhythmias are rare. An increase in PR interval may occur in newborns. Sinus bradycardia may be a sign of impending digoxin intoxication, especially in infants, even in the absence of first degree heart block. Any arrhythmia or alteration in cardiac conduction that develops in a child taking digoxin should be assumed to be caused by digoxin, until further evaluation proves otherwise.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Symptoms and signs. See Section 4.8 Adverse Effects (Undesirable Effects).
The symptoms and signs of toxicity are generally similar to those described in Section 4.8 Adverse Effects (Undesirable Effects) but may be more frequent and can be more severe.
Signs and symptoms of digoxin toxicity become more frequent with levels above 2.0 nanogram/mL although there is considerable interindividual variation. However, in deciding whether a patient's symptoms are due to digoxin, the clinical state, together with serum electrolyte levels and thyroid function are important factors (see Section 4.2 Dose and Method of Administration).
Adults. In adults without heart disease, clinical observation suggests that an overdose of digoxin of 10 to 15 mg was the dose resulting in death of half of the patients. If more than 25 mg of digoxin was ingested by an adult without heart disease, death or progressive toxicity responsive only to digoxin immune Fab (Digibind) resulted.
Cardiac manifestations. Cardiac manifestations are the most frequent and serious sign of both acute and chronic toxicity. Peak cardiac effects generally occur 3 to 6 hours following overdosage and may persist for the ensuing 24 hours or longer. Lanoxin toxicity may result in almost any type of arrhythmia. Multiple rhythm disturbances in the same patient are common. These include paroxysmal atrial tachycardia with variable atrioventricular (AV) block, accelerated junctional rhythm, slow atrial fibrillation (with very little variation in the ventricular rate) and bidirectional ventricular tachycardia.
Premature ventricular contractions (PVCs) are often the earliest and most common arrhythmia. Bigeminy or trigeminy also occur frequently.
Sinus bradycardia and other bradyarrhythmias are very common.
First, second, third degree heart blocks and AV dissociation are also common.
Early toxicity may only be manifested by prolongation of the PR interval.
Ventricular tachycardia may also be a manifestation of toxicity.
Cardiac arrest from asystole or ventricular fibrillation due to Lanoxin toxicity is usually fatal.
Acute massive Lanoxin overdosage can result in mild to pronounced hyperkalaemia due to inhibition of the sodium potassium (Na+-K+) pump. Hypokalaemia may contribute to toxicity (see Section 4.4 Special Warnings and Precautions for Use).
Noncardiac manifestations. Gastrointestinal symptoms are very common in both acute and chronic toxicity. The symptoms precede cardiac manifestations in approximately half of the patients in most literature reports. Anorexia, nausea and vomiting have been reported with an incidence up to 80%. These symptoms usually present early in the course of an overdose.
Neurologic and visual manifestations occur in both acute and chronic toxicity. Dizziness, various CNS disturbances, including headache, disorientation, confusion, fatigue and malaise are very common, more rarely are hallucinations and psychosis. The most frequent visual disturbance is an aberration of colour vision (predominance of yellow green). These neurological and visual symptoms may persist even after other signs of toxicity have resolved.
In chronic toxicity, nonspecific extracardiac symptoms, such as malaise and weakness, may predominate.
Children. In children aged 1 to 3 years without heart disease, clinical observation suggests that an overdose of Lanoxin of 6 to 10 mg was the dose resulting in death in half of the patients. If more than 10 mg of Lanoxin was ingested by a child aged 1 to 3 years without heart disease, the outcome was uniformly fatal when Digibind Fab fragment treatment was not given.
Most manifestations of toxicity in children occur during or shortly after the loading phase with Lanoxin.
Cardiac manifestations. The same arrhythmias or combination of arrhythmias that occur in adults can occur in paediatrics. Sinus tachycardia, supraventricular tachycardia, and rapid atrial fibrillation are seen less frequently in the paediatric population.
Paediatric patients are more likely to present with an AV conduction disturbance or a sinus bradycardia.
Ventricular ectopy is less common, however, in massive overdose, ventricular ectopy, ventricular tachycardia and ventricular fibrillation have been reported.
In neonates, sinus bradycardia or sinus arrest and/or prolonged PR intervals are frequent signs of toxicity. Sinus bradycardia is common in young infants and children. In older children, AV blocks are the most common conduction disorders.
Any arrhythmia or alteration in cardiac conduction that develops in a child taking Lanoxin should be assumed to be caused by digoxin, until further evaluation probes otherwise.
Extracardiac manifestations. The frequent extracardiac manifestations similar to those seen in adults are gastrointestinal, CNS and visual. However, nausea and vomiting are not frequent in infants and small children.
In addition to the undesirable effects seen with recommended doses, weight loss in older age groups and failure to thrive in infants, abdominal pain due to mesenteric artery ischaemia, drowsiness and behavioural disturbances including psychotic manifestations have been reported in overdose.
Treatment. After recent ingestion, such as accidental or deliberate self poisoning, the load available for absorption may be reduced in conscious patients, who are not exhibiting signs of heart block or sinus bradycardia, by gastric lavage. In patients with signs of heart block or sinus bradycardia, gastric lavage can cause an increase in vagal tone and result in worsening of cardiac toxicity.
Patients with massive digitalis ingestion should receive large doses of activated charcoal to prevent absorption and bind digoxin in the gut during enteroenteric recirculation.
Supportive and symptomatic treatment should be initiated depending on the type of cardiotoxicity and continuous ECG monitoring is advisable, along with monitoring of serum electrolytes (especially potassium, calcium and perhaps magnesium).
If hypokalaemia is present, it should be corrected with potassium supplements either orally or intravenously depending on the urgency of the situation. In cases where a large amount of Lanoxin has been ingested hyperkalaemia may be present due to release of potassium from skeletal muscle. Before administering potassium in digoxin overdose, the serum potassium level must be known.
Bradyarrhythmias may respond to atropine but temporary cardiac pacing may be required. Ventricular arrhythmias may respond to lignocaine or phenytoin.
Dialysis is not particularly effective in removing digoxin from the body in potentially life threatening toxicity.
Rapid reversal of the complications that are associated with serious poisoning by digoxin, digitoxin and related glycosides has followed intravenous administration of digoxin immune Fab (Digibind) when other therapies have failed. Digibind is the only specific treatment for digoxin toxicity and is very effective. For details consult the literature supplied with Digibind.
For information on the management of overdose, contact the Poison Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Digoxin is a cardiac glycoside with molecular haemodynamic, neurohumoral and electrophysiological effects contributing to its efficacy in heart failure.
Primary action of digoxin is to reversibly inhibit the alpha subunit of sodium potassium adenosine triphosphatase (ATPase). This results in inhibition of sodium potassium exchange allowing calcium to accumulate in cardiac myocytes by augmenting calcium ion influx. This is responsible for the positive inotropic effect of digoxin or increase force of contraction. This action is more pronounced in decompensated heart failure with systolic dysfunction. Extracellular potassium promotes dephosphorylation of the ATPase and decreases the affinity of the enzyme for digoxin.
Digoxin increases contractility without increasing ventricular rate.
Digoxin increases the sensitivity of baroreceptors thereby leading to reduced sympathetic drive.
Digoxin increases parasympathetic tone. Increase in efferent vagal impulses results in reduced sympathetic tone and diminished impulse conduction rate through the atria and atrioventricular node. Thus a major beneficial effect of digoxin is reduction in ventricular rate. Digoxin prolongs the effective refractory period.
Digoxin decreases plasma renin activity and serum aldosterone levels. Reduced activation of the sympathetic nervous system and renin angiotensin system appears to be independent of its inotropic action in heart failure. The modulatory neurohumoral action appears to contribute significantly to the efficacy of digoxin.
In patients with heart failure, digoxin decreases vascular resistance and venous tone as a consequence of altered autonomic activity.
Digoxin indirectly improves renal function secondary to increase in renal perfusion resulting in diuresis.
One large randomised placebo controlled trial showed a trend towards decrease risk of death associated with worsening heart failure and fewer hospitalisations in the digoxin treated patients (DGI study). There was no adverse impact on overall mortality.
At higher concentrations cardiac glycosides can increase sympathetic activity and directly affect automaticity.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. The absorption of digoxin from the gastrointestinal tract is variable depending on the formulation used. Approximately 70% of a dose is absorbed from digoxin tablets and 80% from digoxin paediatric elixir. Absorption of digoxin is from the stomach and upper part of the small intestine, presumably by a passive, nonsaturable process. Delayed gastric emptying or the presence of food may slow the absorption rate but not the extent of absorption of orally administered digoxin. When taken with meals high in fibre, digoxin may bind to fibre, therefore, amount absorbed from an oral dose may be reduced. Gastric pH does not affect the degree of digoxin absorption. Intestinal absorption of the drug may be impaired in patients with certain malabsorption states, but absorption is not substantially changed by partial gastrectomy or jejunoileal bypass.
In undigitalised patients after oral administration onset of peak effect occurs in 0.5-2 hours and peak effect occurs in 2-6 hours. After IV administration of a single dose of digoxin in previously undigitalised patients, the onset of action occurs in 5-30 minutes and peak effects occur in 1-5 hours. Pharmacological effect may persist 3-4 days after withdrawal of digoxin in digitalised patients.
If plasma concentrations of digoxin are to be determined, blood samples should be obtained at least 6-8 hours after the daily dose and preferably just prior to the next scheduled daily dose. Therapeutic plasma concentrations of digoxin in adults are generally 0.5-2 nanogram/mL. In some patients with atrial fibrillation, slowing of ventricular rate may require steady-state plasma concentrations of 2-4 nanogram/mL. In adults toxicity is usually but not always associated with plasma concentrations greater than 2 nanogram/mL. Although neonates and infants appear to tolerate higher plasma concentrations of digoxin than do adults, evidence suggests that plasma concentrations greater than 2 nanogram/mL are associated with little, if any, additional therapeutic benefit in these patients.
Distribution. With therapeutic plasma concentrations, about 20-30% of digoxin in blood is bound to plasma proteins. Digoxin protein binding is not appreciably changed in uraemic patients. Patients with severe renal impairment have smaller apparent volumes of distribution of digoxin than do normal subjects.
The initial distribution of digoxin from the central to the peripheral compartment generally lasts from 6 to 8 hours. This is followed by a more gradual decline in serum digoxin concentration, which is dependent upon digoxin elimination from the body. The volume of distribution is large (Vdss = 510 litres in healthy volunteers), indicating digoxin to be extensively bound to body tissues. The volume of distribution is reduced in renal failure patient to around 3-4 L/kg.
The highest digoxin concentrations are seen in the heart, liver and kidney, that in the heart averaging 30-fold that in the systemic circulation. Although the concentration in skeletal muscle is far lower, this store cannot be overlooked since skeletal muscle represents 40% of total bodyweight. Of the small proportion of digoxin circulating in plasma, approximately 25% is bound to protein.
Metabolism. The main metabolites of digoxin are dihydrodigoxin and digoxygenin. About 10% of the patients harbour the enteric organism Eubacterium lentum which can convert digoxin into cardio-inactive metabolites.
In most patients, only small amounts of digoxin are metabolised, but the extent of metabolism is variable and may be substantial in some patients. Some metabolism presumably occurs in the liver, but digoxin is also apparently metabolised by bacteria within the lumen of the large intestine following oral administration and possibly after biliary elimination following parenteral administration.
Elimination. The major route of elimination is renal excretion of the unchanged drug.
Digoxin has an elimination half-life of 36-48 hours. In patients with renal failure the elimination half-life is increased, for example in anephric patients the half-life is about 4.5 days or longer. Elimination half-life is prolonged in hypothyroid patients and decreased in hyperthyroid patients. In undigitalised patients, institution of a fixed daily dose of digoxin maintenance therapy without an initial loading dose results in steady-state plasma concentrations after 4-5 elimination half-lives (about 7 days in patients with normal renal function).
Digoxin is excreted mainly in the urine, principally as unchanged drug, by glomerular filtration and active tubular secretion; tubular resorption may also occur. In most patients, small amounts of reduced metabolites are also excreted in urine. However, in some patients, about 40% or more of orally administered digoxin excreted in urine will consist of reduced metabolites. In healthy individuals, about 50-70% of an IV dose is excreted unchanged in urine. Small amounts of cardioactive metabolites and unchanged digoxin are also excreted in the bile and faeces.
Digoxin is a substrate for P-glycoprotein. As an efflux protein on the apical membrane of enterocytes, P-glycoprotein may limit the absorption of digoxin. P-glycoprotein in renal proximal tubules appears to be an important factor in the renal elimination of digoxin (see Section 4.8 Adverse Effects (Undesirable Effects)).
Since most of the drug is bound to the tissues rather than circulating in the blood, digoxin is not effectively removed from the body during cardiopulmonary bypass. Furthermore, only small amounts of drug will be removed from the body during peritoneal dialysis and haemodialysis.
5.3 Preclinical Safety Data
Genotoxicity. No data are available on whether or not digoxin has mutagenic, carcinogenic or teratogenic effects; however, maternally administered digoxin has been used to treat foetal tachycardia and congestive heart failure.
Carcinogenicity. See above.
6 Pharmaceutical Particulars
6.1 List of Excipients
Lanoxin tablets. Lactose monohydrate, maize starch, oxidised maize starch, rice starch and magnesium stearate.
Lanoxin PG tablets. Lactose monohydrate, maize starch, oxidised maize starch, rice starch, magnesium stearate, povidone and indigo carmine.
Lanoxin paediatric elixir. Sucrose, dibasic sodium phosphate heptahydrate, citric acid, ethanol, propylene glycol, quinoline yellow, lime flavour oil D20673, methyl hydroxybenzoate and purified water.
Lanoxin adult injection. Propylene glycol 41.5% w/v, ethanol, citric acid, sodium phosphate, water for injections.
Lanoxin infants injection. Propylene glycol 41.5% w/v, ethanol, citric acid, sodium phosphate, water for injections.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
See Table 1.

6.5 Nature and Contents of Container
Lanoxin 250 microgram tablet blister pack. 50, 100 tablets in PVC/PVDC/Al blister pack.
Lanoxin digoxin 250 microgram tablet bottle. 30, 250 and 1000 tablets.
Lanoxin PG digoxin 62.5 microgram tablet bottle. 60, 200 tablets in a glass bottle. HDPE CRC cap.
Lanoxin PG digoxin 62.5 microgram tablet blister pack. 200 tablets.
Lanoxin paediatric elixir 50 microgram/mL. 60 mL and 100 mL glass bottles with calibrated dropper. PP child resistant cap. Do not dilute.
Lanoxin injection. Glass ampoule.
Adult. 500 microgram/2 mL; 5s.
Infant. 50 microgram/2 mL; 5s.
(Note: Not all products or pack sizes may be marketed.)
6.6 Special Precautions for Disposal
In Australia, an unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure.
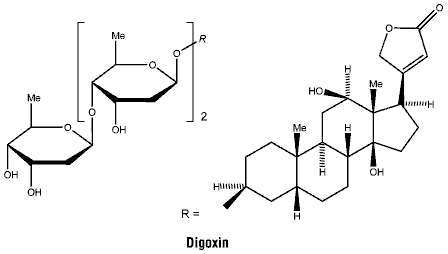
CAS number. The CAS number is 20830-75-5.
7 Medicine Schedule (Poisons Standard)
S4.
Date of First Approval
29 October 2008
Date of Revision
01 October 2025
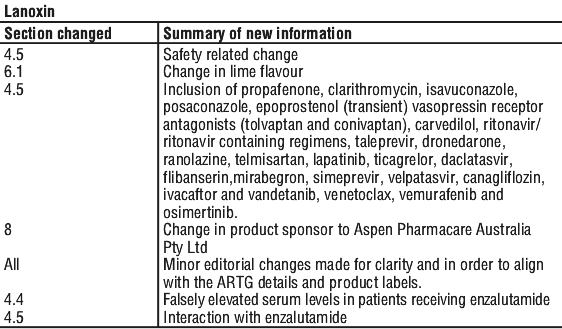
Summary Table of Changes
Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.