Leukeran Tablets
Brand Information
| Brand name | Leukeran Tablets |
| Active ingredient | Chlorambucil |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Leukeran Tablets.
Summary CMI
LEUKERAN tablets
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using LEUKERAN?
LEUKERAN contains the active ingredient chlorambucil. It is used to treat some types of cancers and certain blood disorders.
For more information, see Section 1. Why am I using LEUKERAN? in the full CMI.
2. What should I know before I use LEUKERAN?
Do not use if you have ever had an allergic reaction to chlorambucil or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant, plan to become pregnant or father a child, or are breastfeeding.
For more information, see Section 2. What should I know before I use LEUKERAN? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with LEUKERAN and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use LEUKERAN?
- The dose may vary from person to person. Your doctor will tell you how much to take, and when to take it.
- Swallow the tablet whole with a glass of water. Do not crush or chew.
- It should be taken on an empty stomach, at least 1 hour before or 3 hours after meals.
More instructions can be found in Section 4. How do I use LEUKERAN? in the full CMI.
5. What should I know while using LEUKERAN?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using LEUKERAN? in the full CMI.
6. Are there any side effects?
Tell your doctor as soon as possible if you do not feel well while you are taking LEUKERAN, as any side effects may potentially be serious.
Common side effects include unusual tiredness, being short of breath and looking pale; bruising or bleeding more easily than normal (e.g. more frequent nose bleeds); frequent infections such as fever, sore throat or mouth ulcers; nausea and vomiting; and convulsions in children with nephrotic syndrome.
More serious side effects include signs of an allergic reaction, such as wheezing; swelling of the lips and mouth; difficulty in breathing; severe rash; and fainting.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
LEUKERAN tablets
Active ingredient: chlorambucil
Consumer Medicine Information (CMI)
This leaflet provides important information about using LEUKERAN.
You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using LEUKERAN.
Where to find information in this leaflet:
1. Why am I using LEUKERAN?
2. What should I know before I use LEUKERAN?
3. What if I am taking other medicines?
4. How do I use LEUKERAN?
5. What should I know while using LEUKERAN?
6. Are there any side effects?
7. Product details
1. Why am I using LEUKERAN?
LEUKERAN contains the active ingredient chlorambucil. It belongs to a group of medicines called cytotoxics and is used to treat some types of cancer and certain blood disorders.
Ask your doctor if you have any questions about why LEUKERAN has been prescribed for you.
Your doctor may have prescribed it for another reason.
There is no evidence that this medicine is addictive.
2. What should I know before I use LEUKERAN?
Warnings
Do not take LEUKERAN if:
- you have a prior resistance to chlorambucil
- you or allergic to chlorambucil or any of the ingredients listed at the end of this leaflet.
- Symptoms of an allergic reaction may include shortness of breath, wheezing or difficulty in breathing; swelling of the face, lips, tongue, or any other parts of the body; rash, itching or hives on the skin. - the expiry date printed on the pack has passed.
- the packaging is torn or shows signs of tampering.
Check with your doctor if you:
- have or have had any other conditions, especially the following:
- recently been vaccinated with a ‘live’ organism vaccine or are planning to be vaccinated
- recently received radiotherapy or chemotherapy treatment
- are taking or likely to be taking other cytotoxic drugs
- epilepsy, fits, convulsions, or head trauma
- liver or kidney disease. - take any medicines for any other condition
- you are planning to become pregnant or intending to father a child
- have had any allergy to any other medicines, or any other substances, such as foods, preservatives, or dyes.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Do not take LEUKERAN if you are pregnant, planning to become pregnant or likely to father a child unless you and your doctor have discussed the risks and benefits involved.
LEUKERAN may harm the eggs and sperm so reliable contraceptive methods must be taken to avoid pregnancy whilst you or your partner is taking this medicine.
Do not take LEUKERAN if you are breast feeding unless you and your doctor have discussed the risks and benefits involved.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with LEUKERAN and affect how it works. You may need to take different amounts of your medicine, or you may need to take different medicines. These include:
- phenylbutazone
- vaccinations with ‘live’ organism vaccines.
Your doctor or pharmacist has more information on medicines to be careful with or avoid while taking medicine.
4. How do I use LEUKERAN?
How much to take
Your doctor will decide what dose and for how long you will be taking LEUKERAN. This depends on many factors such as your weight and your response to the treatment.
Your doctor may change the dose and frequency of your medicine as your condition changes.
Your doctor may order regular blood tests while you are taking LEUKERAN to monitor your blood cell count and to change your dosage if necessary.
How to take it
- Swallow the tablet whole with a glass of water.
- Do not break, crush or chew the tablet.
Provided the outer coating of the tablet is intact, there is no risk in handling LEUKERAN tablets.
When to take it
LEUKERAN should be taken daily on an empty stomach, at least one hour before or three hours after meals.
How long to take it
Continue taking LEUKERAN for as long as your doctor tells you.
If you forget to use LEUKERAN
LEUKERAN should be used regularly and at the same time each day.
Do not take a double dose to make up for the dose you missed.
If you use too much LEUKERAN
If you think that you have taken too many LEUKERAN tablets, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using LEUKERAN?
Things you should do
- Take LEUKERAN exactly as your doctor has prescribed.
Otherwise, your doctor may think that it was not effective and change your treatment unnecessarily. - Tell any other doctors, dentists and pharmacists who are treating you that you are taking LEUKERAN, especially if you are being started on any new medicines.
- If you are about to undergo surgery or an operation, tell your doctor or surgeon that you are taking LEUKERAN.
- Tell your doctor immediately if you become pregnant, are trying to become pregnant or trying to father a child while taking LEUKERAN.
Things you should not do
- Do not break, crush or chew the tablet.
Provided the outer coating of the tablet is intact, there is no risk in handling LEUKERAN tablets. - Do not stop taking LEUKERAN or change the dose without first checking with your doctor.
- Do not give this medicine to anyone else, even if their symptoms seem like yours.
- Do not use LEUKERAN to treat any other complaints unless your doctor tells you to.
Driving or using machines
Be careful driving or operating machinery until you know how LEUKERAN affects you.
Looking after your medicine
Store LEUKERAN tablets in the refrigerator where the temperature is between 2-8°C. Do not freeze.
Keep your tablets in the bottle, protected from light until it is time to take them.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
6. Are there any side effects?
Tell your doctor as soon as possible if you do not feel well while you are taking LEUKERAN.
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Serious effects
| Serious side effects | What to do |
Associated with the drop in production of bone marrow cells:
Other:
| Tell your doctor as soon as possible if you notice any of these common side effects. They may potentially be serious, and you may require medical attention. |
More serious side effects
| Serious side effects | What to do |
Allergic type symptoms:
| Call your doctor or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
LEUKERAN tablets are only available with a doctor's prescription.
What LEUKERAN contains
| Active ingredient (main ingredient) |
|
| Other ingredients (inactive ingredients) |
|
| Potential allergens |
|
Do not take this medicine if you are allergic to any of these ingredients.
What LEUKERAN looks like
LEUKERAN tablets are brown, film-coated, round, biconvex tablets, engraved "GX EG3"on one side and "L" on the other. Available in bottles of 25 tablets.
Australian Registration number: AUST R 73026
Who distributes LEUKERAN
Aspen Pharmacare Australia Pty Ltd
34-36 Chandos St
St Leonards NSW 2065
Australia
This leaflet was revised in August 2025.
Brand Information
| Brand name | Leukeran Tablets |
| Active ingredient | Chlorambucil |
| Schedule | S4 |
MIMS Revision Date: 01 June 2018
1 Name of Medicine
Leukeran chlorambucil 2 mg tablet.
2 Qualitative and Quantitative Composition
Leukeran tablets contain 2 mg chlorambucil.
Chlorambucil is a white or off-white crystalline or granular powder. It is practically insoluble in water, freely soluble in acetone and in alcohol.
3 Pharmaceutical Form
Tablets, film-coated.
4 Clinical Particulars
4.1 Therapeutic Indications
Treatment of Hodgkin's disease, certain forms of non-Hodgkin's lymphoma, chronic lymphocytic leukaemia, Waldenstrom's macroglobulinaemia, advanced ovarian adenocarcinoma.
Leukeran has a significant effect in a proportion of patients with breast cancer.
4.2 Dose and Method of Administration
The literature should be consulted for full details of the treatment schedules used.
Leukeran is an active cytotoxic agent for use only under the direction of physicians experienced in the administration of such agents.
Leukeran is administered orally, and should be taken daily on an empty stomach (at least one hour before meal or three hours after meal).
Hodgkin's disease. Used as a single agent a typical dosage is 0.2 mg/kg/day for four to eight weeks. Leukeran is usually included in combination therapy and a number of regimens have been used. Leukeran may also be used as an alternative to nitrogen mustard with a reduction in toxicity but similar therapeutic results.
Non-Hodgkin's lymphoma. Used as a single agent the usual dosage is 0.1 to 0.2 mg/kg/day for four to eight weeks initially; maintenance therapy is then given either by a reduced daily dosage or intermittent courses of treatment.
Leukeran is useful in the management of patients with advanced diffuse lymphocytic lymphoma and those who have relapsed after radiotherapy.
There is no significant difference in the overall response rate obtained with chlorambucil as a single agent and combination chemotherapy in patients with advanced non-Hodgkin's lymphoma.
Chronic lymphocytic leukaemia. Treatment with Leukeran is usually started after the patient has developed symptoms or when there is evidence of impaired bone marrow function (but not marrow failure) as indicated by the peripheral blood count.
Initially Leukeran is given at a dosage of 0.15 mg/kg/day until the total leucocyte count has fallen to 10,000/microlitre. Treatment may be resumed 4 weeks after the end of the first course and continued at a dosage of 0.1 mg/kg/day.
In a proportion of patients, usually after about 2 years of treatment, the blood leucocyte count is reduced to the normal range, enlarged spleen and lymph nodes become impalpable and the proportion of lymphocytes in the bone marrow is reduced to less than 20%.
Patients with evidence of bone marrow failure should first be treated with prednisolone and evidence of marrow regeneration should be obtained before commencing treatment with Leukeran.
Waldenstrom's macroglobulinaemia. Leukeran is the treatment of choice in this indication. Starting doses of 6 to 12 mg daily until leucopenia occurs are recommended followed by 2 to 8 mg daily indefinitely.
Ovarian carcinoma. Used as a single agent a typical dosage is 0.2 mg/kg/day for four to six weeks. A dosage of 0.3 mg/kg/day has been given until leucopenia had been induced.
Maintenance dosage of 0.2 mg/kg/day has been given aiming to keep the total leucocyte count below 4000/mm3. In practice, maintenance courses tend to last 2 to 4 weeks with intervals of 2 to 6 weeks between each course.
Advanced breast cancer. Used as a single agent a typical dosage is 0.2 mg/kg/day for six weeks. Leukeran may be given in combination with prednisolone at a dose range of 14 to 20 mg daily, regardless of bodyweight, over four to six weeks provided there is no serious haemopoietic depression. Leukeran may also be given in combination with methotrexate, 5-fluorouracil, and prednisolone at a dosage of 5 to 7.5 mg/m2/day.
Children. Leukeran may be used in the management of Hodgkin's disease and non-Hodgkin's lymphomas in children. The dosage regimens are similar to those used in adults.
Special populations. Hepatic impairment. Patients with hepatic impairment should be closely monitored for signs and symptoms of toxicity. Since chlorambucil is primarily metabolized in the liver, dose reduction should be considered in patients with severe hepatic impairment. However, there are insufficient data in patients with hepatic impairment to provide a specific dosing recommendation.
Older people. No specific studies have been carried out in older people. However, monitoring of renal or hepatic function is advised. In the event of impairment, caution should be exercised. While clinical experience has not revealed age-related differences in response, drug dosage should be titrated carefully in older patients, usually initiating therapy at the low end of the dosage range.
4.3 Contraindications
Chlorambucil should not be used in patients whose disease has demonstrated a prior resistance to the agent. Leukeran is contraindicated in patients with known hypersensitivity to chlorambucil or to any ingredient of the preparation.
4.4 Special Warnings and Precautions for Use
Leukeran is an active cytotoxic agent for use only under the direction of physicians experienced in the administration of such agents.
Immunisation using a live organism vaccine has the potential to cause infection in immunocompromised hosts. Therefore, immunisations with live organism vaccines are not recommended.
Patients who will potentially have autologous stem cell transplantation should not be treated with chlorambucil long term.
Monitoring. Since Leukeran is capable of producing irreversible bone marrow suppression, blood counts should be closely monitored in patients under treatment.
At therapeutic dosage Leukeran depresses lymphocytes and has less effect on neutrophil and platelet counts and on haemoglobin levels. Discontinuation of Leukeran is not necessary at the first sign of a fall in neutrophils but it must be remembered that the fall may continue for 10 days or more after the last dose.
Leukeran should not be given to patients who have recently undergone radiotherapy or received other cytotoxic agents. When lymphocytic infiltration of the bone marrow is present or the bone marrow is hypoplastic, the daily dose should not exceed 0.1 mg/kg bodyweight.
Children with nephrotic syndrome, patients prescribed high pulse dosing regimens and patients with a history of seizure disorder should be closely monitored following administration of Leukeran as they may have an increased risk of seizures. As with any potentially epileptogenic drug, caution should be exercised when administering chlorambucil to patients with a history of seizure disorder or head trauma, or who are receiving other potentially epileptogenic drugs.
Renal impairment. Patients with evidence of impaired renal function should be carefully monitored as they are prone to additional myelosuppression associated with azotaemia.
Hepatic impairment. The metabolism of Leukeran is still under investigation and consideration should be given to dose reduction in patients with gross hepatic dysfunction.
Effects on fertility. Chlorambucil may cause suppression of ovarian function and amenorrhoea has been reported following chlorambucil therapy.
Azoospermia has been observed as a result of therapy with chlorambucil although it is estimated that a total dose of at least 400 mg is necessary.
Varying degrees of recovery of spermatogenesis have been reported in patients with lymphoma following treatment with chlorambucil in total doses of 410 to 2600 mg.
As with all cytotoxic chemotherapy, adequate contraceptive precautions should be advised when either partner is receiving Leukeran.
In rats, chlorambucil has been shown to damage spermatogenesis and cause testicular atrophy.
Use in pregnancy. (Category D)
As with other cytotoxic agents, Leukeran can produce spontaneous abortion, foetal loss and birth defects. The use of chlorambucil should be avoided whenever possible during pregnancy, particularly during the first trimester. In any individual case, the potential hazard to the foetus must be balanced against the expected benefit to the mother.
Use in lactation. Mothers receiving Leukeran should not breastfeed.
Mutagenicity and carcinogenicity. As with other cytotoxic agents chlorambucil is mutagenic in in vitro and in vivo genotoxicity tests and carcinogenic in animals and humans.
Chlorambucil has been shown to cause chromatid or chromosome damage in man. Acute secondary haematologic malignancies (especially leukaemia and myelodysplastic syndrome) have been reported, particularly after long term treatment (see Section 4.8 Adverse Effects (Undesirable Effects)).
A comparison of patients with ovarian cancer who received alkylating agents with those who did not, showed that the use of alkylating agents, including chlorambucil, significantly increased the incidence of acute leukaemia.
Acute myelogenous leukaemia has been reported in a small proportion of patients receiving chlorambucil as long-term adjuvant therapy for breast cancer.
The leukaemogenic risk must be balanced against the potential therapeutic benefit when considering the use of chlorambucil.
Teratogenicity. Chlorambucil has been shown to induce developmental abnormalities, such as short or kinky tail, microcephaly and exencephaly, digital abnormalities including ectro-, brachy-, syn- and polydactyly and long-bone abnormalities such as reduction in length, absence of one or more components, total absence of ossification sites in the embryo of mice and rats following a single oral administration of 4-20 mg/kg. Chlorambucil has also been shown to induce renal abnormalities in the offspring of rats following a single intraperitoneal injection of 3-6 mg/kg.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Vaccinations with live organism vaccines are not recommended in immunocompromised individuals (see Section 4.4 Special Warnings and Precautions for Use).
Purine nucleoside analogues (such as fludarabine, pentostatin and cladribine) increased the cytotoxicity of chlorambucil in vitro; however, the clinical significance of this finding is unknown.
Animal studies indicate that patients who receive phenylbutazone may require a reduction of the standard chlorambucil doses because of the possibility of enhanced chlorambucil toxicity.
4.6 Fertility, Pregnancy and Lactation
Please see Section 4.4 (Special Warnings and Precautions for Use).
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
For this product there is no modern clinical documentation which can be used as support for determining the frequency of undesirable effects. Undesirable effects may vary in their incidence depending on the dose received and also when given in combination with other therapeutic agents.
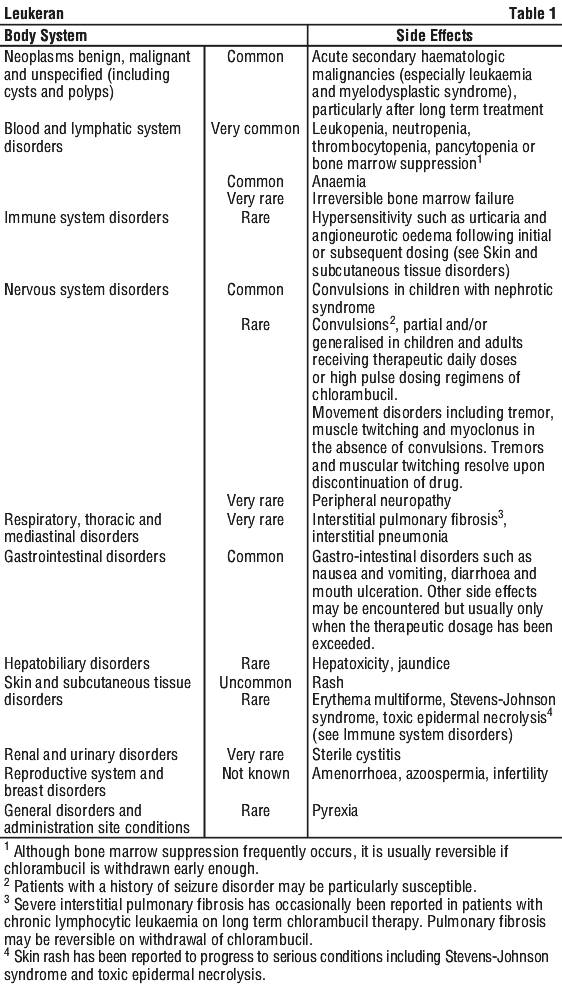
The following convention has been utilised for the classification of frequency: very common (≥ 1/10), common (≥ 1/100 and < 1/10), uncommon (≥ 1/1000 and < 1/100), rare (≥ 1/10,000 and < 1/1000), very rare (< 1/10,000) and not known (cannot be estimated from the available data). See Table 1.
4.9 Overdose
Treatment. Reversible pancytopenia was the main finding of inadvertent overdoses of chlorambucil. Neurological toxicity ranging from agitated behaviour and ataxia to multiple grand mal seizures has also occurred. As there is no known antidote, the blood picture should be closely monitored and general supportive measures should be instituted, together with appropriate blood transfusion if necessary. Chlorambucil is not dialysable.
Further management should be as clinically indicated. For information on the management of overdose, contact the Poison Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Chlorambucil is an aromatic nitrogen mustard derivative which acts as a bifunctional alkylating agent. Alkylation takes place through the formation of a highly reactive ethylenimonium radical. A probable mode of action involves cross linkage of the ethylenimonium derivative between two strands of helical DNA and subsequent interference with replication.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
After oral administration of 14C-chlorambucil, maximum plasma radioactivity occurs between 40 and 70 minutes later. Studies have shown that chlorambucil disappears from the plasma with a mean terminal phase life of 1.5 hours and that its urinary excretion is low. A high level of urinary radioactivity after oral or intravenous administration of 14C-labelled chlorambucil indicates that the drug is rapidly and completely absorbed from the gastrointestinal tract after oral dosage.
In a study of 12 patients administered chlorambucil 0.2 mg/kg bodyweight orally, the mean dose adjusted maximum plasma concentration of chlorambucil (492 ± 160 nanogram/mL) occurred between 0.25 and 2 hours after administration. The mean (± SD) terminal plasma elimination half-life was 1.3 ± 0.5 hours.
The metabolism of chlorambucil in man appears to be similar to that in laboratory animals and involves β oxidation of the butyric acid side chain. Bis-2-chlorethyl-2(4-aminophenyl) acetic acid (phenylacetic acid mustard (PAAM)) is a major metabolite of chlorambucil. Chlorambucil and its metabolites are extensively bound to plasma and tissue proteins. In vitro, chlorambucil is 99% bound to plasma proteins, specifically albumin. In a study of 12 patients administered chlorambucil 0.2 mg/kg body weight orally, the mean dose adjusted peak plasma concentration of PAAM (306 ± 73 nanogram/mL) was reached within 1-3 hours. The mean terminal elimination plasma half-life was 1.8 ± 0.4 hours. The significant contribution of PAAM to the alkylating activity of the drug was evident as the mean area under the plasma concentration time curve (AUC) of PAAM was approximately 1.33 times greater than the AUC of chlorambucil.
5.3 Preclinical Safety Data
No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Each Leukeran tablet contains the following excipients: lactose, microcrystalline cellulose, colloidal anhydrous silica, stearic acid, hypromellose, titanium dioxide, macrogol 400, iron oxide yellow (CI77492) and iron oxide red (CI77491).
6.2 Incompatibilities
No data available.
6.3 Shelf Life
36 months.
6.4 Special Precautions for Storage
Storage. Store at 2°C to 8°C. (Refrigerate. Do not freeze). Protect from light.
6.5 Nature and Contents of Container
Presentation. Leukeran tablets are brown film-coated, round, biconvex tablets engraved "GX EG3" on one side and "L" on the other. They each contain 2 mg chlorambucil and are supplied in bottles of 25 and 50* tablets.
(*Not currently distributed in Australia).
6.6 Special Precautions for Disposal
Safe handling of Leukeran tablets. The handling of Leukeran tablets should follow standard guidelines for the handling of cytotoxic drugs.
Provided the outer coating of the tablet is intact, there is no risk in handling Leukeran tablets. Leukeran tablets should not be divided.
6.7 Physicochemical Properties
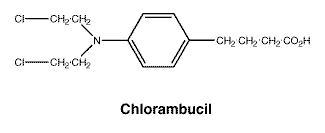
The chemical name for chlorambucil is 4-[bis(2-chloroethyl)amino] benzenebutanoic acid, it has a molecular weight of 304.2, its molecular formula is C14H19Cl2NO2.
Chemical structure. The chemical structure is:
7 Medicine Schedule (Poisons Standard)
S4.
Date of First Approval
23 November 2000
Date of Revision
03 May 2018
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.