Lucinda
Brand Information
| Brand name | Lucinda |
| Active ingredient | Drospirenone |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Lucinda.
Summary CMI
Lucinda
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Lucinda?
Lucinda contains the active ingredient drospirenone. Lucinda is a contraceptive pill and is used to prevent pregnancy. For more information, see Section 1. Why am I using Lucinda? in the full CMI.
2. What should I know before I use Lucinda?
Do not use if you have ever had an allergic reaction to drospirenone or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Lucinda? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Lucinda and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Lucinda?
- Always take this medicine exactly as your doctor has told you. Check with your doctor or pharmacist if you are not sure.
- Each blister of Lucinda contains 24 white active tablets and 4 green placebo tablets. The two differently coloured tablets are arranged in order.
- The first tablet of the treatment should be taken on the first day of menstrual bleeding. Thereafter, tablet taking is continuous.
More instructions can be found in Section 4. How do I use Lucinda? in the full CMI.
5. What should I know while using Lucinda?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Lucinda? in the full CMI.
6. Are there any side effects?
Call your doctor straight away if you: have severe pain or swelling in either of your legs, unexplained chest pain, breathlessness, an unusual cough, if you cough up blood (possibly indicating a thrombosis); have a sudden, severe stomach ache or look jaundiced (possibly indicating liver problems); feel a lump in your breast (possibly indicating breast cancer); have a sudden or severe pain in the lower abdomen or stomach area (possibly indicating an ectopic pregnancy, this is a pregnancy outside the womb); are to be immobilised or are to have surgery (consult your doctor at least four weeks in advance); have unusual, heavy vaginal bleeding; suspect that you are pregnant.
Tell your doctor if you notice any of the following and they worry you: headache, tiredness, dizziness, mood changes, nausea, changes in weight, acne, sweating, breast tenderness or pain and changes in your period.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Lucinda
Active ingredient(s): drospirenone
Consumer Medicine Information (CMI)
This leaflet provides important information about using Lucinda. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Lucinda.
Where to find information in this leaflet:
1. Why am I using Lucinda?
2. What should I know before I use Lucinda?
3. What if I am taking other medicines?
4. How do I use Lucinda?
5. What should I know while using Lucinda?
6. Are there any side effects?
7. Product details
1. Why am I using Lucinda?
Lucinda contains the active ingredient drospirenone.
Each blister of Lucinda contains 24 white tablets (also called active tablets) and 4 green tablets (also called placebo tablets that do not contain active substance). The two differently coloured tablets are arranged in order.
Each of the 24 white active tablets contains a small amount of one type of female sex hormone, the progestogen drospirenone. For this reason, Lucinda is called a progestogen-only-pill (POP). POPs don't contain any estrogen hormone, so Lucinda can be used by patients who do not tolerate estrogens.
Lucinda provides high contraceptive efficacy. The contraceptive effect of Lucinda is based on the inhibition of ovulation and changes in the cervical mucus, and effects on the endometrium, which becomes thinner.
Vaginal bleeding may occur at irregular intervals during the use of Lucinda. You also may not have any bleeding at all.
Lucinda is a contraceptive pill and is used to prevent pregnancy.
2. What should I know before I use Lucinda?
Warnings
Do not use Lucinda if:
- you are allergic to drospirenone, or any of the ingredients listed at the end of this leaflet.
Always check the ingredients to make sure you can use this medicine. - you have a blood clot in a blood vessel, eg. of the legs (deep venous thrombosis), or of the lungs (pulmonary embolism), or other organs.
- you have (or have ever had) liver disease and your liver function is still not normal.
- your kidneys are not working well (renal failure).
- you have or are suspected to have a cancer that is sensitive to sex-steroids, such as certain types of breast cancer.
- you have any unexplained vaginal bleeding.
If any of these conditions appear when using Lucinda, stop taking this medicine and immediately contact your doctor.
Check with your doctor if you:
- have ever had a thrombosis (formation of a blood clot in a blood vessel).
- have liver cancer, if you have jaundice (yellowing of the skin) or a liver disease and your liver is not working normally.
- have ever had a breast cancer.
- have or have had a chloasma (yellowish-brown pigmentation patches on the skin, particularly on the face). If this is the case, you will need to avoid exposure to the sun or ultraviolet radiation during treatment with Lucinda.
- have diabetes.
- have high blood pressure.
- have kidneys that are not working well. Your doctor will do a blood test to check your potassium levels during the first cycle.
- have any other medical conditions.
- take any medicines for any other condition.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
If you experience a worsening or first appearance of any of these conditions, you should contact your doctor.
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Do not use Lucinda if you are pregnant or think you might be pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Lucinda may be used while you are breastfeeding.
No effects on breastfed newborns/infants are anticipated. However, very small amounts of drospirenone are excreted in breast milk.
Breast cancer
Regularly check your breasts and contact your doctor as soon as possible if you feel any lump in your breasts.
- Breast cancer has been found slightly more often in patients who take other types of contraceptives that include a hormone called estrogen (these are called combined oral contraceptives (COCs) or combined pills), than in patients of the same age who do not take the combined pill.
- If patients stop taking the combined pill, the risk gradually decreases, so that 10 years after stopping the risk is the same as for patients who have never taken the combined pill.
- Breast cancer is rare under 40 years of age.
- Breast cancers found in patients who take the combined pill, seem less likely to spread than breast cancers found in patients who do not take the combined pill.
- It is not known whether the difference in breast cancer risk is caused by the combined pill. It may be that patients were examined more often, so that the breast cancer is noticed earlier.
- The risk of breast cancer in users of progestogen-only pills like Lucinda is believed to be similar to that in patients who use the combined pill, but the evidence is less conclusive.
Thrombosis (formation of a blood clot in a blood vessel)
See your doctor immediately, if you notice possible signs of a thrombosis.
Thrombosis is the formation of a blood clot, which may block a blood vessel. A thrombosis sometimes occurs in the deep veins of the legs (deep venous thrombosis). If this clot breaks away from the veins where it is formed, it may reach and block the arteries of the lungs, causing a so-called “pulmonary embolism”.
There may be a slightly increased risk of thrombosis with progestogen-only pills. The risk of thrombosis is higher if a member of your family (a sibling or a parent) has had thrombosis at a relatively early age, with increasing age, obesity, prolonged immobilisation, major surgery or major trauma.
There is no apparent risk of having a heart attack or stroke (a blood clot in the brain) with a progestogen-only pill, unless you also have high blood pressure.
Bone metabolism
- Treatment with Lucinda leads to decreased hormone (estradiol) levels, to a level corresponding with the early phase of the menstrual cycle.
- It is currently unknown whether the decrease in this hormone level may have a clinically relevant effect on bone mineral density.
- Loss of bone mineral density is of particular concern during adolescence and early adulthood, a critical period of bone accretion.
- It is unknown if bone mineral density decrease in this population will reduce peak bone mass and increase the risk for fracture in later life.
- Your doctor will decide if Lucinda is an appropriate treatment for you, taking into account if you have significant risk factors for osteoporosis.
Psychiatric disorders
- Some patients using hormonal contraceptives including Lucinda have reported depression or depressed mood.
- Depression can be serious and may sometimes lead to suicidal thoughts.
- If you experience mood changes and depressive symptoms contact your doctor for further medical advice as soon as possible.
Medical examination
- Before you start taking Lucinda for the first time or if you re-start the treatment after some time of not taking it, your doctor will ask you some questions about your health and will do a complete physical examination, including blood pressure measurements.
- Your doctor will tell you how often you should go for control visits.
Children and adolescents
Lucinda is used after menarche (the first menstrual bleeding).
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Lucinda and affect how it works.
These includes medicines used for the treatment of:
- epilepsy (eg. primidone, phenytoin, barbiturates, carbamazepine, oxcarbazepine, felbamate, topiramate);
- tuberculosis (eg. Rifampicin, rifabutin);
- HIV infections (eg. ritonavir, nevirapine, nelfinavir) or other infectious diseases (eg. griseofulvin);
- hepatitis C virus infections (eg. boceprevir, telaprevir);
- high blood pressure in the blood vessels in the lungs (eg. bosentan);
- depressive mood (eg. herbal remedy St John's wort);
- certain bacterial infections (eg. clarithromycin, erythromycin);
- fungal infections (eg. fluconazole, itraconazole, ketoconazole, voriconazole);
- high blood pressure (hypertension), angina or certain heart rhythm disorders (eg. diltiazem).
Your doctor can tell you if you need to take additional contraceptive precautions (for example condoms) and if so, how long.
Lucinda may also influence the effect of other medicines, causing either an increase in effect (eg. cyclosporine) or a decrease in effect (eg. lamotrigine).
If Lucinda is taken at the same time as potassium-sparing diuretics or aldosterone antagonists, which can increase serum potassium levels, your doctor could recommend testing your serum potassium levels during the first treatment cycle with Lucinda.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Lucinda.
Avoid grapefruit or grapefruit juice while you are taking Lucinda.
4. How do I use Lucinda?
How much to take
Always take this medicine exactly as your doctor has told you. Check with your doctor or pharmacist if you are not sure.
Each blister of Lucinda contains 24 white active tablets and 4 green placebo tablets.
Take one tablet of Lucinda every day with a little water if necessary. You may take the tablets with or without food. You must take the tablets every day around the same time of the day so the interval between two tablets is always 24 hours.
Do not confuse the tablets. Note:
- Because of the different composition of the tablets, it is necessary to begin with the first white active tablet on the upper left and to take the tablets every day.
- For the correct order, follow the direction of the arrows and the sequence of numbers on the blister.
- The first tablet of the treatment should be taken on the first day of menstrual bleeding. Thereafter tablet-taking is continuous.
- Take a white active tablet for the first 24 days and then a green placebo tablet for the last 4 days.
- You must then start a new pack straightaway (24 white active tablets followed by 4 green placebo tablets) without a break in daily tablet intake. There is therefore no gap between two packs.
You may have some bleeding during the use of Lucinda, or you may also have no bleeding at all, but you must continue to take your tablets as normal without interruption.
If you use Lucinda in this manner, you are still protected against pregnancy during the 4 days when you are taking a placebo tablet.
If you start taking the first pack of tablets on days 2 to 5 of the menstrual cycle, a barrier method should be used until you have completed 7 days of uninterrupted white active tablets.
Preparation of blister
To help you keep track, stickers, each with the 7 day of the weeks are provided in the pack.
Choose the sticker that starts with the day you begin taking the tablets (for example, if you start menstruating on a Thursday, use the sticker that starts with “THU”) and place it on the blister card over the words “Place day label here” so that the first day is above the tablet marked “START”.
There will be a day indicated above every tablet, so you can see whether you have taken a certain pill. The arrows and the consecutive numbers show the order you are to take the pills.
Starting your first pack of Lucinda
If you have not used a hormonal contraceptive in the previous month
Begin with Lucinda on the first day of your period. When doing so, you are immediately protected against pregnancy and you do not need to use extra protective measures such as a condom.
If you start Lucinda on days 2 to 5 of your period you must use extra protective measures such as a condom until you have taken 7 white active tablets.
When changing from a combined pill, vaginal ring or transdermal patch
Option 1
You should start Lucinda on the day:
- after the last active tablet (the last tablet containing the active substances) of your previous pill; or
- on the day of removal of your vaginal ring; or
- on the day of removal of your transdermal patch.
This means no tablet, ring or patch-free break.
If you follow these instructions, no additional contraceptive precautions are necessary.
Option 2
You can also start Lucinda, at the latest, on the day following either:
- the placebo interval of your present contraceptive tablet; or
- the hormone-free interval following removal of your vaginal ring; or
- a patch-free break.
In these cases, make sure you use an additional barrier method of contraception for the first 7 days of taking Lucinda.
When changing from another progestogen-only pill (POP)
You may switch any day from another POP and start taking Lucinda the next day. Additional contraceptive precautions are not necessary.
When changing from a progestogen-only injection or implant or from a progestogen-releasing intrauterine system (IUS)
You should start Lucinda the day when the next injection is due or on the day that your implant or your IUS is removed. Additional contraceptive precautions are not necessary.
After having a baby
You can start Lucinda any day between day 21 to 28 after having your baby. If you start later than day 28, but before the menstruation has returned, you must be sure you are not pregnant and you must use a barrier method such as a condom until you have completed the first 7 days of tablet-taking.
After miscarriage
You should follow the advice of your doctor.
When to take Lucinda
- Take one tablet of Lucinda every day at about the same time of the day so that the interval between two tablets is always 24 hours.
If you forget to use Lucinda
Lucinda should be used regularly at the same time each day. If you are less than 24 hours late in taking any active white tablet, contractive protection is not reduced. You should take the tablet as soon as you remember and take further tablets at the usual time without further contraceptive protection being required.
If you are more than 24 hours late in taking any white active tablet, contraceptive protection may be reduced. Refer to the table below for further instructions. The more consecutive tablets you have missed the higher the risk that the contraceptive efficacy is decreased.
Instructions for missed Lucinda tablets
| Days during cycle | Instructions for missed tablet if you are more than 24 hours late |
| Days 1-7 | Take the missed tablets as soon as you remember, even if this means taking two tablets at the same time, and use an additional method of contraception for the next 7 days. |
| Days 8-17 | Take the missed tablets as soon as you remember, even if that means taking two tablets at the same time. Continue taking the white tablets at the usual time. If you have taken the active white tablets correctly in in the 7 days before the first missed tablet, no additional contraceptive protection is required. If you have missed more than one tablet, then extra contraceptive precautions should be used until you have taken 7 days of uninterrupted white active tablets. |
| Days 18-24 | Two options are possible in this time frame as follows:
|
The last four green tablets in the 4th row of the strip are the placebo tablets. If you forget one of these tablets, this has no effect on the reliability of Lucinda. Throw away the forgotten placebo tablet.
If you have missed tablets and you do not experience a withdrawal bleed in the placebo tablet interval, the possibility of pregnancy should be considered.
What to do in the case of vomiting or severe diarrhoea
- If you vomit or have severe diarrhoea, there is a risk that the active substance in the pill will not be fully absorbed by your body. This situation is almost the same as forgetting a tablet. In these cases, an additional method of contraception may be needed, ask your doctor for advice.
- If you vomit or have severe diarrhoea within 3-4 hours after taking your white active tablet of Lucinda, you must take another white tablet from another blister pack as soon as possible. If possible, take it within 12 hours of when you normally take your pill. Additional contraceptive precautions are not necessary. If this is not possible, or 12 hours have passed, you should follow the advice given in the section “If you forget to take Lucinda” above.
If you use too much Lucinda
There have been no reports of serious harmful effects from taking too many Lucinda tablets at one time.
Symptoms that may occur are nausea, vomiting and slight vaginal bleeding.
If you think that you have used too much Lucinda, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26); or - contact your doctor; or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
However, in case of overdose, ask your doctor for advice because blood tests should be done.
5. What should I know while using Lucinda?
Lucinda (placebo tablet) contains lactose (milk sugar). If you have an intolerance to some sugars, contact your doctor before you start to take Lucinda.
Things you should do
Regular check-ups
When you are using Lucinda, your doctor will tell you to return for regular check-ups. In general, the frequency and nature of these check-ups will depend on your personal situation.
Call your doctor straight away if you:
- have severe pain or swelling in either of your legs, or unexplained pains in the chest;
- have breathlessness, an unusual cough, especially when you cough up blood (possibly indicating a thrombosis);
- have sudden, severe stomach ache or look jaundiced (possible indicating liver problems);
- feel a lump in your breast (possibly indicating breast cancer);
- have sudden or severe pain in the lower abdomen or stomach area (possibly indicating an ectopic pregnancy, which is a pregnancy outside the womb);
- are to be immobilised or are to have surgery (consult your doctor at least four weeks in advance);
- have unusual, heavy vaginal bleeding;
- suspect that you are pregnant.
Remind any doctor, dentist or pharmacist you visit that you are using Lucinda.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Lucinda affects you.
No effects on ability to drive and use machines have been observed in users of oral hormone contraceptives, although no studies have been performed with Lucinda.
Looking after your medicine
Follow the instructions on the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink; or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date which is stated on the packaging after EXP. The expiry date refers to the last day of that month.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Changes in your reproductive system:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Other side effects such as*:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Lucinda contains
| Active ingredient (main ingredient) | Drospirenone |
| Other ingredients (inactive ingredients) | White active tablets: Microcrystalline cellulose Methacrylic acid polymer Triethyl citrate Magnesium stearate Green placebo tablets: Lactose monohydrate Maize starch Quinoline yellow aluminium lake Indigo carmine aluminium lake Sunset yellow FCF aluminium lake Magnesium stearate |
| Potential allergens | Lactose |
Do not take this medicine if you are allergic to any of these ingredients.
What Lucinda looks like
Each blister of Lucinda contains 24 white active tablets and 4 green placebo tablets (AUST R 427710).
The active tablet is a white to off white, round, film-coated tablet debossed with a “J” on side and a “1” on the other side.
The placebo tablet is a green mottled, round, biconvex, film-coated tablet, debossed with "J" on one side and "2" on the other side.
Who distributes Lucinda
Generic Health Pty Ltd
Suite 2, Level 2
19-23 Prospect Street
Box Hill, VIC, 3128
Australia
 +61 3 9809 7900
+61 3 9809 7900
This leaflet was prepared in October 2024.
Brand Information
| Brand name | Lucinda |
| Active ingredient | Drospirenone |
| Schedule | S4 |
MIMS Revision Date: 01 January 2026
Notes
Distributed by Generic Health Pty Ltd
1 Name of Medicine
Drospirenone.
2 Qualitative and Quantitative Composition
Each white active tablet contains 4 mg of drospirenone.
The green placebo tablet does not contain any active substances.
List of excipient(s) with known effect. Green placebo tablets contain lactose.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Active tablet. White to off-white, round tablets debossed with a "J" on side and a "1" on the other side.
Placebo tablet. Green mottled, round, biconvex tablets, debossed with "J" on one side and "2" on the other side.
4 Clinical Particulars
4.1 Therapeutic Indications
Lucinda is indicated for use as an oral contraceptive.
4.2 Dose and Method of Administration
How to take Lucinda. One tablet is to be taken daily for 28 consecutive days: one white active tablet daily during the first 24 days; and one green inactive tablet daily during the 4 following days. Tablets must be taken every day, at about the same time of the day, so that the interval between two tablets is always 24 hours. Tablets should be taken in the order shown on the blister. Stickers marked with the 7 days of the week are provided. The patient should choose the sticker that starts with the day they begin taking the tablets and stick it on the blister.
The first tablet of the treatment should be taken on the first day of menstrual bleeding. Thereafter, tablet taking is continuous. A subsequent pack is started immediately after finishing the previous pack, without a break in daily tablet intake.
How to start Lucinda. No preceding hormonal contraceptive use (in the past month). Tablet-taking has to start on day 1 of the patient's natural cycle (first day of her menstrual bleeding). When doing so, no additional contraceptive measures are necessary.
Starting on days 2-5 is allowed, but during the first pill pack a barrier method should be used until the patient has completed 7 days of uninterrupted white tablet-taking.
Following first-trimester abortion. After first-trimester abortion, it is recommended to start Lucinda immediately after abortion took place. In that case there is no need to use an additional contraceptive method.
Following delivery or second-trimester abortion. Contraceptive treatment with Lucinda is recommended to start between 21 and 28 days after delivery or second trimester abortion. If contraceptive treatment with Lucinda is initiated later but before the menstruations have returned, pregnancy must be ruled out and an additional method of contraception should be used for the first week.
For breastfeeding patients, see Section 4.6. Fertility, Pregnancy and Lactation.
Changing from a combined hormonal contraceptive (combined oral contraceptive (COC), vaginal ring or transdermal patch). The patient should start Lucinda preferably on the day after the last active tablet (the last tablet containing the active substances) of their previous COC or on the day of removal of their vaginal ring or transdermal patch. In these cases, the use of an additional contraceptive is not necessary.
The patient may also start Lucinda at the latest on the day following the usual tablet-free, ring-free, patch-free or placebo tablet interval of their previous combined hormonal contraceptive, but during the first 7 days of tablet taking an additional barrier method is recommended.
Changing from a progestogen-only method (progestogen-only pill (POP), injection, implant) or from a progestogen-releasing intrauterine system (IUS). The patient may switch any day from another POP and should start Lucinda the day after, within 24 hours of discontinuing the previous POP. A patient may switch from an implant or following IUS removal on the same day that the implant or IUS is removed. A patient may switch from using an injectable contraceptive and should start Lucinda on the day the next injection was due to occur. In all of these cases, the use of an additional contraceptive is not necessary.
Management of missed tablets. The management of missed tablets can be guided by the following two basic rules:
1. Seven days of uninterrupted taking of active tablets is required to attain adequate suppression of the hypothalamic-pituitary-ovarian-axis. Active tablet-taking must never be discontinued for longer than 7 days.
2. The greater the number of white active tablets that are missed, and the closer they are to the green placebo tablets, the higher the risk of a pregnancy.
If the patient is less than 24 hours late in taking any white active tablet, contraceptive protection is not reduced. The patient should take the tablet as soon as they remember and should take further tablets at the usual time.
If the patient is more than 24 hours late in taking any white active tablet, contraceptive protection may be reduced.
The following advice can be given if a white active tablet is missed during:
Day 1-7. The patient should take the last missed white active tablet as soon as they remember, even if this means taking two tablets at the same time. The patient should then continue to take tablets at the usual time.
In addition, a barrier method such as a condom should be used until they have completed 7 days of uninterrupted white active table-taking.
If intercourse took place in the preceding 7 days, the possibility of a pregnancy should be considered.
Days 8-17. The patient should take the last missed tablet as soon as they remember, even if this means taking two tablets at the same time. The patient should then continue to take tablets at the usual time.
Provided that the patient has taken the active tablets correctly in the 7 days preceding the first missed tablet, there is no need to use extra contraceptive precautions. However, if they have missed more than 1 tablet, they should be advised to use extra precautions until they have completed 7 days of uninterrupted white active tablet-taking.
Days 18-24. Contraceptive reliability is reduced.
Contraceptive protection can still be provided, by adhering to either of the following two options:
a) The patient should take the last missed tablet as soon as they remember, even if this means taking two tablets at the same time. They then continue to take tablets at the usual time until the white active tablets are used up. The 4 green placebo tablets from the last row should be discarded, and the next blister pack started straight away. The patient is unlikely to have a withdrawal bleed until the end of the active tablets section of the second pack, but they may experience spotting or breakthrough bleeding on active tablet-taking days.
b) Alternatively, the patient may be advised to discontinue active tablet-taking from the current blister pack. They should immediately commence taking the green placebo tablets for a maximum of 3 days, such that the total number of green placebo tablets plus missed active white tablets is not more than 4. The patient should subsequently commence taking active white tablets from a new blister pack.
Provided that in the 7 days preceding the first missed tablet the patient has taken all tablets correctly, there is no need to use extra contraceptive precautions.
If the patient has missed a tablet during the preceding 7 days, then the patient should use extra contraceptive precautions for the next 7 days and follow option (a) above.
If the patient missed tablets and subsequently has no withdrawal bleed in the placebo tablet interval, the possibility of a pregnancy should be considered.
Please note: If the patient is not sure about the number or colour of tablets missed and what advice to follow, a barrier method should be used until they have completed 7 days of uninterrupted white active tablet-taking.
Green placebo tablets missed. Contraceptive protection is not reduced. Green tablets from the last (4th) row of the blister can be disregarded. However, the missed tablets should be discarded to avoid unintentionally prolonging the placebo tablet phase.
Advice in case of gastrointestinal disturbances. In case of severe gastrointestinal disturbances (e.g. vomiting or diarrhoea), absorption may not be complete and additional contraceptive measures should be taken.
If vomiting or diarrhoea occurs within 3-4 hours after tablet-taking, a new (replacement) tablet should be taken as soon as possible. The new tablet should be taken within 12 hours of the usual time of tablet-taking if possible. If more than 12 hours elapse, the advice concerning missed tablets, as given in Section 4.2 Dose and Method of Administration, Management of missed tablets, is applicable. If the patient does not want to change their normal tablet-taking schedule, they have to take the extra tablet(s) from another blister pack.
Paediatric population. Safety and efficacy of Lucinda have been established in patients of reproductive age. Safety and efficacy are expected to be the same for post pubertal adolescents under the age of 18 and patients 18 years and older. Use of this product before menarche is not indicated.
Method of administration. For oral use.
4.3 Contraindications
Progestogen-only contraceptives (POCs) like Lucinda should not be used in the presence of any of the conditions listed below. Should any of the conditions appear for the first time during Lucinda use, the medicinal product should be discontinued immediately:
Active venous thromboembolic disorder.
Presence or history of severe hepatic disease as long as liver function values have not returned to normal.
Severe renal insufficiency or acute renal failure.
Known or suspected sex-steroid sensitive malignancies.
Undiagnosed vaginal bleeding.
Known or suspected pregnancy.
Hypersensitivity to the active substance or to any of the excipients listed in Section 6.1 List of Excipients.
4.4 Special Warnings and Precautions for Use
If any of the conditions/risk factors mentioned below are present, the benefits of Lucinda should be weighed against the possible risks for each individual patient and discussed with the patient before they decide to start using Lucinda. In the event of aggravation, exacerbation or first appearance of any of these conditions, the patient should contact their physician. The physician should then decide whether Lucinda use should be discontinued.
Hyperkalaemia. Drospirenone is an aldosterone antagonist with potassium sparing properties. In most cases, no increase of potassium levels is to be expected. However, it's recommended to check serum potassium levels during the first treatment cycle in patients presenting with renal insufficiency and pre-treatment serum potassium in the upper reference range, and during concomitant use of potassium sparing medicinal products (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Circulatory disorders. From epidemiological studies there is little evidence for an association between progestogen-only preparations and an increased risk of myocardial infarction or cerebral thromboembolism. Rather, the risk of cardiovascular and cerebral events is related to increasing age, hypertension, and smoking. In patients with hypertension the risk of stroke may be slightly enhanced by progestogen-only preparations.
Although not statistically significant, some studies indicate that there may be a slightly increased risk of venous thromboembolism (deep venous thrombosis, pulmonary embolism) associated with the use of progestogen-only preparations. Generally recognised risk factors for venous thromboembolism (VTE) include a positive personal or family history (VTE in a sibling or a parent at a relatively early age), age, obesity, prolonged immobilisation, major surgery or major trauma.
Treatment should be stopped at once if there are symptoms of an arterial or venous thrombotic event or suspicion thereof and discontinuation of Lucinda should be considered in case of prolonged immobilisation due to surgery or illness.
Bone metabolism. Treatment with Lucinda leads to decreased estradiol serum levels, to a level corresponding with the early follicular phase. It is currently unknown whether the decrease in estradiol serum levels may have a clinically relevant effect on bone mineral density. Loss of bone mineral density is of particular concern during adolescence and early adulthood, a critical period of bone accretion. It is unknown if bone mineral density decrease in this population will reduce peak bone mass and increase the risk for fracture in later life.
Breast cancer. A meta-analysis from 54 epidemiological studies reported that there is a slightly increased relative risk (RR = 1.24) of having breast cancer diagnosed in patients who are currently using oral contraceptives (OCs), mainly using estrogen-progestogen preparations. The excess risk gradually disappears during the course of the 10 years after cessation of COC use. Because breast cancer is rare in patients under 40 years of age, the excess number of breast cancer diagnoses in current and recent COC patients is small in relation to the overall risk of breast cancer. These studies do not provide evidence for causation. The observed pattern of increased risk may be due to an earlier diagnosis of breast cancer in OC patients, the biological effects of OCs or a combination of both. The breast cancers diagnosed in patients of OCs tend to be less advanced clinically than the cancers diagnosed in those who have never used OCs.
The risk of having breast cancer diagnosed in patients of progestogen-only preparations is possibly of similar magnitude to that associated with COC. However, for progestogen-only preparations, the evidence is based on much smaller populations of patients and so is less conclusive than that for COCs.
Other tumours. In rare cases, benign liver tumours, and even more rarely, malignant liver tumours have been reported in patients of combined hormonal contraceptives. In isolated cases, these tumours have led to life-threatening intra-abdominal haemorrhages. A hepatic tumour should be considered in the differential diagnosis when severe upper abdominal pain, liver enlargement or signs of intra-abdominal haemorrhage occur.
Ectopic pregnancy. The protection with traditional POPs against ectopic pregnancies is not as good as with COCs, which has been associated with the frequent occurrence of ovulations during the use of POPs. Despite the fact that Lucinda consistently inhibits ovulation, ectopic pregnancy should be taken into account in the differential diagnosis if the patient presents amenorrhoea or abdominal pain.
Liver function. Discontinue Lucinda if jaundice develops. Steroid hormones may be poorly metabolised in patients with impaired liver function. Acute or chronic disturbances of liver function may require the discontinuation of Lucinda use until markers of liver function return to normal and Lucinda causation has been excluded.
Diabetes. Although progestogens may have an effect on peripheral insulin resistance and glucose tolerance, there is no evidence for a need to alter the therapeutic regimen in diabetics using POPs such as Lucinda. However, diabetic patients should be carefully observed during the first months of use. Special attention should be paid to diabetic patients with vascular involvement.
Other conditions. If a sustained hypertension develops during the use of Lucinda, or if a significant increase in blood pressure does not adequately respond to antihypertensive therapy, the discontinuation of Lucinda should be considered.
Like with any other hormonal contraceptive, chloasma may occasionally occur, especially in patients with a history of chloasma gravidarum. Patients with a tendency to chloasma should avoid exposure to the sun or ultraviolet radiation whilst taking Lucinda.
Depressed mood and depression are well-known undesirable effects of hormonal contraceptive use (see Section 4.8 Adverse Effects (Undesirable Effects)). Depression can be serious and is well-known risk factor for suicidal behaviour and suicide. Patients should be advised to contact their physician in case of mood changes and depressive symptoms, including shortly after initiating the treatment.
The following conditions have been reported both during pregnancy and during sex steroid use, but an association with the use of progestogens has not been established: jaundice and/or pruritus related to cholestasis; gallstone formation; porphyria; systemic lupus erythematosus; haemolytic uraemic syndrome; Sydenham's chorea; herpes gestationis; otosclerosis-related hearing loss; (hereditary) angioedema.
Excipients with known effect. Each green placebo tablet contains 62.668 mg of lactose monohydrate. Patients with rare hereditary problems of galactose intolerance, lactase deficiency or glucose-galactose malabsorption should not take this medicine.
Medical examination/consultation. Prior to the initiation or reinstitution of Lucinda a complete medical history (including family history) should be taken and pregnancy must be ruled out. Blood pressure should be measured, and a physical examination should be performed, guided by the contraindications (see Section 4.3 Contraindications) and warnings (see Section 4.4 Special Warnings and Precautions for Use). The patient should also be instructed to carefully read the patient leaflet and to adhere to the advice given. The frequency and nature of examinations should be based on established practice guidelines and be adapted to the individual patient.
Patients should be advised that OCs do not protect against HIV infections (AIDS) and other sexually transmitted diseases.
Changes in the menstrual bleeding pattern. Disruption of the menstrual bleeding pattern may occur during use of hormonal contraceptives that inhibit ovulation, including Lucinda (see Section 5.1 Pharmacodynamic Properties).
If the bleeding is very frequent and irregular, another contraceptive method should be considered. If the symptoms persist, an organic cause should be ruled out. Management of amenorrhoea during treatment depends on whether or not the tablets have been taken in accordance with the instructions and may include a pregnancy test.
The treatment should be stopped if a pregnancy occurs.
Reduced efficacy. The efficacy of POPs may be reduced in the event of missed tablets (see Section 4.2 Dose and Method of Administration), gastro-intestinal disturbances (see Section 4.2 Dose and Method of Administration) or concomitant medication (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Use in hepatic impairment. Discontinue Lucinda if jaundice develops. Steroid hormones may be poorly metabolised in patients with impaired liver function. Acute or chronic disturbances of liver function may require the discontinuation of Lucinda use until markers of liver function return to normal and Lucinda causation has been excluded.
Use in renal impairment. Lucinda is contraindicated for use in patients with severe renal insufficiency or acute renal failure.
Use in the elderly. No data available.
Paediatric use. See Section 4.2 Dose and Method of Administration, Paediatric population.
Effects on laboratory tests. The use of contraceptive steroids may influence the results of certain laboratory tests, including biochemical parameters of the liver, thyroid, adrenal and renal function, serum levels of (carrier) proteins, e.g. corticosteroid binding globulin and lipid/lipoprotein fractions, parameters of carbohydrate metabolism and parameters of coagulation and fibrinolysis.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Influence of other medicinal products on Lucinda. Interactions can occur between Lucinda and other medicinal products that induce microsomal enzymes. This can result in increased clearance of sex hormones and may lead to breakthrough bleeding and/or contraceptive failure.
Management. Enzyme induction can already be observed after a few days of treatment. Maximum enzyme induction is generally seen within a few weeks. After drug therapy is discontinued, enzyme induction may be sustained for about 4 weeks.
Short-term treatment. Patients on treatment with enzyme inducing drugs should temporarily use a barrier method or another method of contraception in addition to the POP. The barrier method must be used during the whole time of the concomitant drug therapy and for 28 days after its discontinuation.
If the drug therapy runs beyond the end of the active tablets in the POP pack, the placebo tablets must be discarded, and the next POP pack should be started right away.
Long-term treatment. In patients on long-term treatment with enzyme-inducing active substances, another reliable, nonhormonal, method of contraception is recommended.
The following interactions have been reported in the literature (mainly with combined contraceptives but occasionally also with POPs).
Substances increasing the clearance of contraceptive hormones (diminished contraceptive efficacy by enzyme induction) e.g. barbiturates, bosentan, carbamazepine, phenytoin, primidone, rifampicin and HIV medication ritonavir, nevirapine and efavirenz and possibly also felbamate, griseofulvin, oxcarbazepine, topiramate and products containing the herbal remedy St. John's wort (Hypericum perforatum).
Substances with variable effects on the clearance of contraceptive hormones. When co-administered with sex hormones, many combinations of HIV protease inhibitors (e.g. ritonavir, nelfinavir) and non-nucleoside reverse transcriptase inhibitors (e.g. nevirapine, efavirenz) and/or combinations with hepatitis C virus (HCV) medicinal products (e.g. boceprevir, telaprevir), can increase or decrease plasma concentrations of progestins. The net effect of these changes may be clinically relevant in some cases.
Therefore, the prescribing information of concomitant HIV/HCV medications should be consulted to identify potential interactions and any related recommendations. In case of any doubt, an additional barrier contraceptive method should be used by patients on protease inhibitor or non-nucleoside reverse transcriptase inhibitor therapy.
Substances decreasing the clearance of contraceptive hormones (enzyme inhibitors). The clinical relevance of potential interactions with enzyme inhibitors remain unknown.
Concomitant administration of strong or moderate CYP3A4 inhibitors such as azole antifungals (e.g. fluconazole, itraconazole, ketoconazole, voriconazole), verapamil, macrolides (e.g. clarithromycin, erythromycin), diltiazem and grapefruit juice can increase plasma concentrations of the progestogen.
In a multiple dose study evaluating the daily (10 days) co-administration of the strong CYP3A4 inhibitor ketoconazole with two drospirenone-containing hormone presentations (drospirenone 3 mg + estradiol 1.5 mg and drospirenone 3 mg + ethinylestradiol 0.02 mg) the AUC(0-24h) of drospirenone was 2.3-fold and 2.7-fold respectively.
Influence of Lucinda on other medicinal products. Hormonal contraceptives may affect the metabolism of certain other active substances. Accordingly, plasma and tissue concentrations may either increase (e.g. cyclosporine) or decrease (e.g. lamotrigine).
Based on in vitro studies and in vivo interaction studies in female volunteers using omeprazole, simvastatin and midazolam as marker substrate, a clinically relevant interaction of drospirenone with the cytochrome P450 mediated metabolism of other active substances is unlikely.
Pharmacodynamic interactions. Published data did not show a significant effect on serum potassium following the concomitant use of drospirenone and ACE-inhibitors or NSAIDs in patients without renal insufficiency. The concomitant use of Lucinda with aldosterone antagonists or potassium-sparing diuretics has not been studied. In this case, serum potassium should be tested during the first treatment cycle (see Section 4.4 Special Warnings and Precautions for Use).
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Lucinda is indicated for the prevention of pregnancy.
Use in pregnancy. (Category B3)
Lucinda is contraindicated in pregnancy. If pregnancy occurs during treatment with Lucinda, further intake should be stopped.
Epidemiological studies have revealed neither an increased risk of birth defects in children born to patients who used drospirenone prior to pregnancy, nor a teratogenic effect when drospirenone was taken inadvertently during pregnancy.
Drospirenone and/or its metabolites crossed the placenta and entered the foetus when administered orally to pregnant rats and rabbits.
Animal studies revealed adverse effects on embryofoetal development. With dosing during the period of major organogenesis, drospirenone impaired foetal growth and development in rats at doses ≥ 15 mg/kg/day and in rabbits at ≥ 30 mg/kg/day (yielding systemic exposure 14 and 4 times higher in the respective species than that in patients at the maximum recommended human dose of Lucinda, based on plasma AUC). Treatment at 100 mg/kg/day in rabbits caused abortions (relative exposure, 15). Feminisation of male foetuses was observed in rats with subcutaneous administration at ≥ 3 mg/kg/day during the period of sexual differentiation, consistent with drospirenone's known anti-androgenic activity.
Based on these animal data, undesirable effects due to hormonal action of the active compound cannot be excluded.
Use in lactation. Negligible amounts of drospirenone are excreted in the breast milk. The daily dose of drospirenone in the baby is < 1% of the maternal dose. Thus, at therapeutic doses of Lucinda, no effects on the breastfed newborns/infants are anticipated. Based on the available data drospirenone may be used during lactation.
4.7 Effects on Ability to Drive and Use Machines
No studies on the influence on the ability to drive and use machines have been performed with Lucinda.
No effects on ability to drive and use machines have been observed in patients of oral hormonal contraceptives.
4.8 Adverse Effects (Undesirable Effects)
Changes in the bleeding pattern as an adverse reaction frequently reported in the clinical trials (see Section 5.1 Pharmacodynamic Properties).
The most commonly reported adverse reactions in long-term clinical trials of more than 9 cycles of treatment with drospirenone (2,700 patients) were acne (3.8%), metrorrhagia (2.9%), headache (2.7%) and breast pain (2.2%).
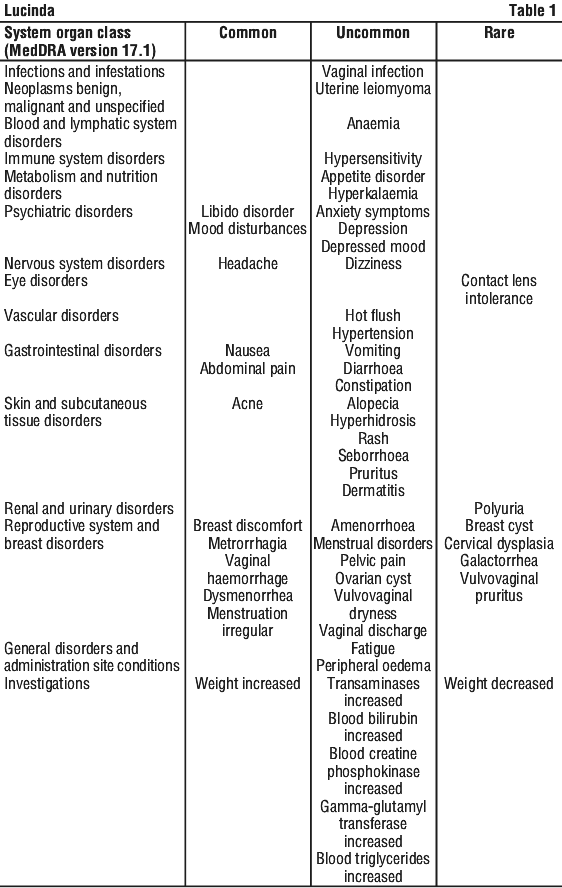
Tabulated list of adverse reactions. Adverse reactions that have been reported in short- and long-term clinical trials with drospirenone are listed in Table 1.
All adverse reactions are listed by system organ class and frequency: very common (> 1/10), common (≥ 1/100 to < 1/10), uncommon (≥ 1/1,000 to < 1/100), rare (≥ 1/10,000 to < 1/1,000).
4.9 Overdose
There have been no reports of serious deleterious effects from overdose. Symptoms that may occur in this case are nausea, vomiting and slight vaginal bleeding. There are no antidotes and further treatment should be symptomatic.
Drospirenone is a spironolactone analogue which has antimineralocorticoid properties. Serum potassium and sodium, and evidence of metabolic acidosis, should be monitored in cases of overdose.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: hormonal contraceptives for systemic use, progestogens.
ATC code: G03AC10.
Mechanism of action. Lucinda is a progestogen-only pill (POP) which contains the progestogen drospirenone, derived from spironolactone.
In a therapeutic dosage, drospirenone also possesses antiandrogenic and mild antimineralocorticoid properties. It has no estrogenic, glucocorticoid and antiglucocorticoid activity. This gives drospirenone a pharmacological profile closely resembling the natural hormone progesterone.
There are indications from clinical studies that for combined hormonal contraceptives containing 3 mg drospirenone and 0.02 mg ethinylestradiol, the mild antimineralocorticoid properties result in a mild antimineralocorticoid effect.
Pharmacodynamic effects. The contraceptive effect of Lucinda is achieved primarily by inhibition of ovulation. Drospirenone exhibits a strong anti-gonadotropic activity inhibiting follicular stimulation and ovulation by suppression of the luteinising hormone (LH). In addition, drospirenone has an effect on the cervix increasing the viscosity of the cervical mucus. Drospirenone also exerts progestational effects on the endometrium, which becomes thinner.
Clinical trials. Clinical efficacy and safety. The ovulation inhibition potential of drospirenone (drospirenone 4 mg non-micronised administered daily for 24 days) as reflected by the ovarian activity (follicular growth, endogenous estradiol and progesterone serum concentrations [Hoogland score]) in comparison to 0.075 mg of desogestrel administered daily for 28 days over two treatment cycles was assessed in a randomised, open-label Phase II study conducted in 60 healthy young patients. In cycle 1, no ovulation was observed in either treatment. Whereas one ovulation was observed for drospirenone and 0.075 mg of desogestrel group in cycle 2.
In a Phase II study performed in 130 patients, drospirenone maintained the inhibition of ovulation in spite of four fixed scheduled delayed intakes of 24 hours each on day 3, 6, 11 and 22.
In two multicentre Phase III European clinical trials, one single-arm study and one controlled study vs desogestrel 0.075 mg, 1,596 patients have been treated for 9 up to 13 consecutive cycles with drospirenone and 341 with desogestrel for 9 months. In the pooled analysis of these two studies the following Pearl Indexes (PI) were calculated:
PI (18-45 years of age), user + method failure: 0.73 (upper limit 95% confidence interval (CI) 1.43);
PI (18-35 years of age), user + method failure: 0.93 (upper limit 95% CI 1.84).
In a single arm multicentre Phase III clinical trial performed in 39 US sites, the efficacy population consisted of 915 non-breastfeeding subjects aged ≤ 35 years with 5,337 evaluable cycles. The PI (95% CI) for evaluable cycles based on 915 subjects, 12 confirmed on-drug pregnancies and 5337 evaluable cycles was 2.9 (1.5; 5.1).
Bleeding pattern. The bleeding pattern during use of drospirenone was assessed in a 9-month comparative, double blind trial vs desogestrel 0.075 mg, used continuously.
The occurrence of a withdrawal bleeding (defined as a bleeding starting during the 4 hormone-free days of drospirenone lasting for up to 8 consecutive days), was highest - occurring in less than 40% - during the first cycles and decreased with time. After 9 months of use, a withdrawal bleeding was recorded in less than 20% of patients.
The mean number of bleeding/spotting days in the drospirenone group vs the desogestrel group during the cycles 2-4 was 13.1 ± 13.0 vs 16.9 ± 16.9, respectively. The mean number of bleeding/spotting days during cycles 7-9, was 9.7 ± 10.4 vs 10.8 ± 13.3, respectively.
In the same study, the proportion of subjects without any bleeding/spotting (amenorrhea) during cycles 2-4 was 20.1% for drospirenone and 13.5% for desogestrel. The proportion of subjects with amenorrhea increased in cycles 7-9 to 26.7% for drospirenone and to 32.1% in the desogestrel group.
The number of subjects with prolonged bleeding (> 10 consecutive days) for drospirenone vs desogestrel was 18.1% and 26.1%, respectively, during cycles 2-4 and 9.1% and 16.7%, respectively, during cycles 7-9.
The rate of subjects who withdrew from the study due to bleeding related adverse events was 3.3% in the drospirenone group and 6.6% in the desogestrel group.
Paediatric population. A Phase III study was conducted in Europe to evaluate tolerability, safety and acceptability of drospirenone. 103 adolescents were included in a 6-cycle core part and 7 additional cycles (extension phase) for a total of 13 cycles, drospirenone was well tolerated and accepted by the subjects.
Bleeding pattern with drospirenone was assessed and data were generally consistent with those from the Phase III studies in adults. Drospirenone was associated with a decrease in the percentage of subjects experiencing bleeding or spotting over time.
5.2 Pharmacokinetic Properties
Absorption. Orally administered drospirenone is rapidly and almost completely absorbed. Maximum concentrations of drospirenone in plasma of about 28 nanogram/mL are reached at about 3-4 h after single ingestion. Concomitant ingestion of food has no influence on the extent of absorption of drospirenone.
The pharmacokinetics of drospirenone after single and repeated dose has been studied in comparison with the marketed product containing 3 mg of micronised drospirenone in combination with ethinylestradiol. After multiple dose administration, the relative bioavailability of drospirenone was 76.51% for AUCt,ss. The accumulation ratio expressed by Rac (AUC) was 1.9256 while it was 2.7684 for the combined product. These findings indicate that the total exposure to drospirenone is lower for drospirenone than for the combined product on the market in a cycle of 28 days.
Distribution. Drospirenone is 95-97% protein bound in serum. Drospirenone binds to serum albumin and does not bind to sex hormone binding globulin (SHBG) or corticosteroid binding globulin (CBG). The mean apparent volume of distribution of drospirenone is 4 L/kg.
Metabolism. Drospirenone is extensively metabolised after oral administration. Two major non-pharmacologically active metabolites in the plasma are the acid form of drospirenone, generated by opening of the lactone ring, and the 4,5-dihydro-drospirenone-3-sulfate, both of which are formed without involvement of the cytochrome P450 system. Drospirenone is also subject to oxidative metabolism catalysed by CYP3A4 .
In vitro, drospirenone is capable of inhibiting, from a weak to moderate level, the cytochrome P450 enzymes CYP1A1, CYP2C9, CYP2C19 and CYP3A4.
Elimination. After oral administration, plasma drospirenone levels decrease with a terminal half-life of 32 h.
The metabolic clearance rate of drospirenone in serum is 1.5 ± 0.2 mL/min/kg. Drospirenone is excreted only in trace amounts in unchanged form. The metabolites of drospirenone are excreted with the faeces and urine at an excretion ratio of about 1.2 to 1.4.
Linearity/non-linearity. The pharmacokinetics of oral drospirenone is dose proportional following single doses ranging from 1-10 mg.
Steady-state conditions. During a treatment cycle, maximum steady-state concentrations of drospirenone in serum of about 40 nanogram/mL are reached after about 7 days of treatment. Plasma drospirenone levels accumulate by a factor of about 2 as a consequence of the ratio of terminal half-life and dosing interval.
Special populations. Effect of renal impairment. No studies have been conducted to evaluate the effect of renal impairment on the pharmacokinetics of drospirenone. However, steady-state serum drospirenone levels in patients under treatment with a COC containing drospirenone with mild renal impairment (creatinine clearance CLcr, 50-80 mL/min) were comparable to those of patients with normal renal function. The serum drospirenone levels were on average 37% higher in patients with moderate renal impairment (CLcr, 30-50 mL/min) compared to those in patients with normal renal function. Drospirenone treatment was also well tolerated by patients with mild and moderate renal impairment. Drospirenone treatment did not show any clinically significant effect on serum potassium concentration.
Effect of hepatic impairment. No studies have been conducted to evaluate the effect of hepatic disease on the pharmacokinetics of drospirenone. However, steroid hormones may be poorly metabolised in patients with impaired liver function.
In a single dose study in patients taking a COC containing drospirenone, oral clearance (CL/F) was decreased approximately 50% in volunteers with moderate hepatic impairment as compared to those with normal liver function. The observed decline in drospirenone clearance in volunteers with moderate hepatic impairment did not translate into any apparent difference in terms of serum potassium concentrations. Even in the presence of diabetes and concomitant treatment with spironolactone (two factors that can predispose a patient to hyperkalaemia) an increase in serum potassium concentrations above the upper limit of the normal range was not observed. It can be concluded that drospirenone is well tolerated in patients with mild or moderate hepatic impairment (Child-Pugh B).
Ethnic groups. No studies were performed to assess pharmacokinetics in ethnic groups.
5.3 Preclinical Safety Data
Genotoxicity. Drospirenone was found to induce chromosome aberrations in human peripheral lymphocytes. However, drospirenone was not mutagenic in bacterial and mammalian cell gene mutation assays in vitro, and was not clastogenic in mouse micronucleus assays in vivo. Interactions between drospirenone and the DNA of liver cells, which indicate a genotoxic potential, were found in in vitro and in vivo studies in rats. No such finding was observed in human liver cells in vitro.
Carcinogenicity. No treatment-related increase in tumour incidence was observed with drospirenone in 2-year studies in mice and rats, involving oral administration at doses up to 10 mg/kg/day (yielding systemic exposure 4.5 and 12 times higher in the respective species than that in patients at the maximum recommended human dose of drospirenone, based on plasma AUC).
Although these long-term animal studies did not indicate carcinogenic activity for drospirenone, it should be borne in mind that sex steroids can promote the growth of certain hormone-dependent tissues and tumours.
6 Pharmaceutical Particulars
6.1 List of Excipients
Each active tablet contains the following excipients: microcrystalline cellulose, methacrylic acid copolymer, triethyl citrate, and magnesium stearate.
Each placebo tablet contains the following excipients: lactose monohydrate, maize starch, quinoline yellow aluminium lake, indigo carmine aluminium lake, sunset yellow FCF aluminium lake and magnesium stearate.
6.2 Incompatibilities
Not applicable.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Protect from light. Store in original container.
6.5 Nature and Contents of Container
Lucinda tablets are available in PVC/Aclar blister containing 28 tablets (24 active tablets and 4 placebo tablets).
Pack sizes: calendar packs containing 1 x 28, 3 x 28 and 4 x 28 tablets.*
* Not all pack sizes may be marketed.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Drospirenone is soluble in methylene chloride, soluble in acetone and in methanol, sparingly soluble in ethyl acetate and in alcohol, and practically insoluble in hexane.
Chemical structure.
Molecular weight: 366.5 g/mol.
CAS number. 67392-87-4.
7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Date of First Approval
31 October 2024
Date of Revision
30 June 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.

