Melpha Injection
Brand Information
| Brand name | Melpha Injection |
| Active ingredient | Melphalan |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Melpha Injection.
Summary CMI
MELPHA® injection
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using MELPHA?
MELPHA contains the active ingredient Melphalan Hydrochloride. MELPHA is used to treat multiple myeloma, a cancer of the blood.
For more information, see Section 1. Why am I using MELPHA? in the full CMI.
2. What should I know before I use MELPHA?
Do not use if you have ever had an allergic reaction to MELPHA or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use MELPHA? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with MELPHA and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use MELPHA?
- Your doctor will decide what dose and for how long you will be given MELPHA injection.
More instructions can be found in Section 4. How do I use MELPHA? in the full CMI.
5. What should I know while using MELPHA?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using MELPHA? in the full CMI.
6. Are there any side effects?
Tell your doctor as soon as possible if you do not feel well while you are being given MELPHA injection.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
MELPHA® injection
Active ingredient(s): [Melphalan]
Consumer Medicine Information (CMI)
This leaflet provides important information about using MELPHA. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using MELPHA.
Where to find information in this leaflet:
1. Why am I using MELPHA?
2. What should I know before I use MELPHA?
3. What if I am taking other medicines?
4. How do I use MELPHA?
5. What should I know while using MELPHA?
6. Are there any side effects?
7. Product details
1. Why am I using MELPHA?
MELPHA contains the active ingredient Melphalan Hydrochloride. MELPHA injection contains melphalan Hydrochloride as the active ingredient. It belongs to a group of medicines called cytotoxics.
MELPHA is used to used to treat multiple myeloma, a cancer of the blood.
2. What should I know before I use MELPHA?
Warnings
Do not use MELPHA if:
- you are allergic to Melphalan Hydrochloride, or any of the ingredients listed at the end of this leaflet.
- Always check the ingredients to make sure you can use this medicine.
Check with your doctor if you:
- have any other medical conditions
- take any medicines for any other condition
Tell your doctor as soon as possible if you do not feel well while you are being given MELPHA injection.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
You must not be given this medicine if you are trying to become pregnant or to father a child.
You must not be given MELPHA injection if you are pregnant or breast feeding unless you and your doctor have discussed the risks and benefits involved.
Use in Children
- The safety and effectiveness in children has not been established.
- If you are not sure if you should be given this medicine, talk to your doctor.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may be affected by MELPHA injection or may affect how well it works.
Some medicines may interfere with MELPHA and affect how it works.
You may need to take different amounts of your medicine or you may need to take different medicines. These include:
- nalidixic acid
- cyclosporin
- cisplatin
- carmustine
- other cytotoxic drugs
- vaccinations with 'live' organism vaccines.
Medicines that may increase the effect of MELPHA include:
Your doctor has more information on medicines to be careful with or avoid while being given this medicine.
Medicines that may reduce the effect of MELPHA include:
Your doctor has more information on medicines to be careful with or avoid while being given this medicine.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect MELPHA.
4. How do I use MELPHA?
How much to take / use
- MELPHA for Injection is administered intravenously.
The usual IV dose is 16 mg/m2. The drug is administered as a single infusion over 15 to 20 minutes. MELPHA is administered at 2-week intervals for four doses, then, after adequate recovery from toxicity, at 4-week intervals. - Follow the instructions provided and use MELPHA until your doctor tells you to stop.
When to take / use MELPHA
- MELPHA should be used to treat multiple myeloma, a cancer of the blood.
- Your doctor will decide what dose and for how long you will be given MELPHA injection.
- Your doctor may order regular blood tests while you are being given MELPHA injection in order to monitor your blood cell count and to change your dosage if necessary.
- MELPHA injection will be given to you under the supervision of a doctor.
- It is a sterile powder which is dissolved and then further diluted before intravenous infusion or 'drip' into a vein.
How to administer MELPHA (relevant for devices)
MELPHA for Injection must be reconstituted AT ROOM TEMPERATURE by rapidly injecting 10 mL of the supplied diluent directly into the vial of lyophilised powder using a sterile needle (21 Gauge or higher Gauge size) and syringe. Immediately shake vial vigorously (for approximately 5 minutes) until a clear solution is obtained. This provides a 5-mg/mL solution of melphalan and has a pH of approximately 6.5. Rapid addition of the diluent, as a single quantity, followed by immediate vigorous shaking is important for proper dissolution.
If you forget to use MELPHA
Your doctor will decide what dose and for how long you will be given MELPHA injection.
MELPHA until your doctor tells you to stop.
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Do not take a double dose to make up for the dose you missed.
- Your doctor will decide what dose and for how long you will be given MELPHA injection.
- Your doctor may order regular blood tests while you are being given MELPHA injection in order to monitor your blood cell count and to change your dosage if necessary.
- MELPHA injection will be given to you under the supervision of a doctor.
If you use too much MELPHA
As MELPHA injection is given to you under the supervision of your doctor, it is very unlikely that you will receive too much.
However, if you experience any side effects after being given MELPHA injection, tell your doctor immediately.
Symptoms of overdose may include severe nausea and vomiting, convulsions and stomach pain.
If you think that you have used too much MELPHA, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using MELPHA?
Things you should do
It is important that you visit your doctor regularly, so your doctor can check your progress and make sure your medicine is working.
Call your doctor straight away if you:
- especially if you are about to be started on any new medicines or radiotherapy.
- Do not have any vaccinations without your doctor's approval.
- Tell your doctor if you become pregnant, are trying to become pregnant or trying to father a child.
- If you are about to undergo surgery or an operation, tell your doctor or surgeon that you are being given MELPHA injection.
- This medicine can lower the number of white blood cells and platelets in your blood. This means that you have an increased chance of getting an infection or bleeding.
- The following precautions should be taken to reduce your risk of infection or bleeding:
- avoid people who have infections
- Check with your doctor immediately if you think you may be getting an infection, or if you get a fever, chills, cough, hoarse throat, lower back or side pain or find it painful or difficult to urinate.
- be careful when using a toothbrush, tooth pick or dental floss
- Check with your doctor before having dental work.
- Your doctor, dentist, nurse or pharmacist may recommend other ways to clean your teeth and gums.
- be careful not to cut yourself when using sharp objects such as a razor or nail cutters
- avoid contact sports or other situations where you may bruise or get injured.
Remind any doctor, dentist or pharmacist you visit that you are using MELPHA.
Things you should not do
- Do not stop using this medicine without consultation with your doctor.
Bleeding and Infection
- This medicine can lower the number of white blood cells and platelets in your blood. This means that you have an increased chance of getting an infection or bleeding.
- The following precautions should be taken to reduce your risk of infection or bleeding:
- avoid people who have infections
- Check with your doctor immediately if you think you may be getting an infection, or if you get a fever, chills, cough, hoarse throat, lower back or side pain or find it painful or difficult to urinate.
- be careful when using a toothbrush, tooth pick or dental floss
- Check with your doctor before having dental work.
- Your doctor, dentist, nurse or pharmacist may recommend other ways to clean your teeth and gums.
- be careful not to cut yourself when using sharp objects such as a razor or nail cutters
- avoid contact sports or other situations where you may bruise or get injured.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how MELPHA affects you.
MELPHA may cause dizziness in some people. Be careful driving or operating machinery until you know how MELPHA injection affects you.
Drinking alcohol
Tell your doctor if you drink alcohol.
Alcohol may have different effect on different people. Consult your doctor if you are drinking alcohol while taking MELPHA.
Looking after your medicine
- Keep MELPHA injection in a cool, dry place where it stays below 25°C. protect from light
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
When to discard your medicine (as relevant)
Return any unused or expired medicine to your pharmacist.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
General side effect on body:
| Speak to your doctor if you have any of these less serious side effects and they worry you. Your doctor will frequently test your blood count, but you should tell him at once if you notice any signs of fever, infection, unexpected bruising, bleeding or signs of blood in your urine. |
Serious side effects
| Serious side effects | What to do |
Bleeding-related:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What MELPHA contains
| Active ingredient (main ingredient) | Melphalan Hydrochloride |
| Other ingredients (inactive ingredients) | Each vial of sterile powder for injection contains 50 mg melphalan Hydrochloride and povidone. Each vial of sterile diluent contains sodium citrate Dihydrate, propylene glycol, ethanol and water for injections. |
| Potential allergens | NA |
Do not take this medicine if you are allergic to any of these ingredients.
What MELPHA looks like
MELPHA injection is supplied in a carton containing:
- one clear glass vial containing sterile, white or off-white powder for injection
- one 10 mL clear glass vial containing a sterile, clear, colourless solution of diluent.
The diluent is used to dissolve the powder. (Aust R 287038).
Who distributes MELPHA
Australia
Sponsor: Emcure Pharmaceuticals Pty Ltd.
Distributed by: Arrotex Pharmaceuticals Pty Ltd
15-17 Chapel Street
Cremorne VIC 3121
patient.safety@arrotex.com.au
New Zealand
Sponsor & Distributor:
Arrotex Pharmaceuticals (NZ) Limited
C/o Quigg Partners
Level 7, The Bayleys Building
36 Brandon Street
Wellington 6011 New Zealand
patient.safety@arrotex.com.au
This leaflet was prepared in July 2025.
Brand Information
| Brand name | Melpha Injection |
| Active ingredient | Melphalan |
| Schedule | S4 |
MIMS Revision Date: 01 November 2025
Notes
Distributed by Arrotex Pharmaceuticals Pty Ltd.
1 Name of Medicine
Melphalan hydrochloride 50 mg powder for injection.
2 Qualitative and Quantitative Composition
Each single-use vial contains melphalan hydrochloride equivalent to 50 mg melphalan and povidone.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Melpha for Injection is supplied as a sterile, no pyrogenic, freeze-dried powder.
4 Clinical Particulars
4.1 Therapeutic Indications
Melpha Injection is indicated for the treatment of patients with multiple myeloma for whom oral therapy is not appropriate.
4.2 Dose and Method of Administration
Dosage. Melpha for Injection is reconstituted using the sterile diluent provided.
Melpha for Injection is administered intravenously. The usual IV dose is 16 mg/m2. The drug is administered as a single infusion over 15 to 20 minutes. Melpha is administered at 2-week intervals for four doses, then, after adequate recovery from toxicity, at 4-week intervals. Available evidence suggests about one third to one half of the patients with multiple myeloma show a favourable response to the drug. Experience with oral melphalan suggests that repeated courses should be given since improvement may continue slowly over many months, and the maximum benefit may be missed if treatment is abandoned prematurely. Dose adjustment on the basis of blood cell counts at the nadir and day of treatment should be considered.
Patients with renal impairment. Melphalan clearance, though variable, is decreased in patients with renal impairment. Dosage reduction of up to 50% should be considered in patients with moderate to severe renal impairment and subsequent dosage determined according to the degree of haematological suppression.
Administration precautions. As with other toxic compounds, caution should be exercised in handling and preparing the solution of Melpha. Appropriate guidelines for the handling of cytotoxic compounds should be consulted. Skin reactions associated with accidental exposure may occur. The use of disposable latex or PVC gloves, facemask, protective goggles and a disposable apron is recommended. If the solution of Melpha contacts the skin or mucosa, immediately wash the skin or mucosa thoroughly with soap and cold water. In such instances it may be prudent to seek medical advice.
In case of contact with eyes, immediate irrigation with sodium chloride eyewash should be carried out and medical attention sought without delay. If sodium chloride solution is not available, large volumes of water may be used.
Any spillage should be dealt with immediately (by personnel wearing suitable protective clothing), by mopping with damp, disposable paper towels which are placed in a high-risk waste disposal bag. Contaminated surfaces should be washed with copious quantities of water.
Preparation for administration/stability. Melpha for Injection must be reconstituted at room temperature by rapidly injecting 10 mL of the supplied diluent directly into the vial of lyophilised powder using a sterile needle (21 gauge or higher gauge size) and syringe. Immediately shake vial vigorously (for approximately 5 minutes) until a clear solution is obtained. This provides a 5-mg/mL solution of melphalan and has a pH of approximately 6.5. Rapid addition of the diluent, as a single quantity, followed by immediate vigorous shaking is important for proper dissolution.
Immediately dilute the dose to be administered in 0.9% Sodium Chloride Injection to a concentration not greater than 0.45 mg/mL.
Administer the diluted product over a minimum of 15 minutes.
Complete administration within 60 minutes of reconstitution. Parenteral drug products should be visually inspected for particulate matter and discolouration prior to administration whenever solution and container permit. If either occurs, do not use this product.
The time between reconstitution/dilution and administration of Melpha should be kept to a minimum because reconstituted and diluted solutions of Melpha are unstable. Over as short a time as 30 minutes, a citrate derivative of melphalan has been detected in reconstituted material from the reaction of Melpha with Sterile Diluent for Melpha. Upon further dilution with saline, nearly 1% label strength of melphalan hydrolyses every 10 minutes.
A precipitate forms if the reconstituted solution is stored at 5°C. Do not refrigerate the reconstituted product. Melpha Injection contains no antimicrobial agent. It is for single use in one patient only. Any unused solution should be discarded (see Section 6.6 Special Precautions for Disposal).
4.3 Contraindications
Melpha should not be used in patients whose disease has demonstrated prior resistance to this agent. Patients who have demonstrated hypersensitivity to melphalan should not be given the drug.
4.4 Special Warnings and Precautions for Use
Identified precautions. Melphalan hydrochloride should be administered in carefully adjusted dosage under the direction of physicians experienced in the use of cytotoxic agents.
Immunisation using a live organism vaccine has the potential to cause infection in immunocompromised hosts. Therefore, immunisations with live organism vaccines are not recommended.
Severe bone marrow suppression with resulting infection or bleeding may occur. Since melphalan hydrochloride is a potent myelosuppressive agent, it is essential that careful attention should be paid to the monitoring of blood counts to avoid the possibility of excessive myelosuppression and the risk of irreversible bone marrow aplasia. Therefore, the following tests should be performed at the start of therapy and prior to each subsequent dose of melphalan hydrochloride: platelet count, haemoglobin, white blood cell count, and differential. Thrombocytopenia and/or leucopoenia are indications to withhold further therapy until the blood counts have sufficiently recovered. Frequent blood counts are essential to determine optimal dosage and to avoid toxicity. Dose adjustment on the basis of blood counts at the nadir and day of treatment should be considered. See Section 4.2 Dose and Method of Administration.
Blood counts may continue to fall after treatment is stopped, so at the first sign of an abnormally large fall in leucocyte or platelet counts, treatment should be temporarily interrupted. Controlled trials comparing intravenous (IV) to oral melphalan have shown more myelosuppression with the IV formulation. Melphalan hydrochloride should be used with extreme caution in patients whose bone marrow reserve may have been compromised by prior irradiation or chemotherapy or whose marrow function is recovering from previous cytotoxic therapy.
Hypersensitivity reactions including anaphylaxis have occurred in approximately 2% of patients who received the IV formulation. See Section 4.8 Adverse Effects (Undesirable Effects). These reactions usually occur after multiple courses of treatment. Treatment is symptomatic. The infusion should be terminated immediately, followed by the administration of volume expanders, pressor agents, corticosteroids, or antihistamines at the discretion of the physician. If a hypersensitivity reaction occurs, IV or oral melphalan should not be readministered since hypersensitivity reactions have also been reported with oral melphalan.
Melphalan hydrochloride may cause local tissue damage should extravasation occur, and consequently it should not be administered by direct injection into a peripheral vein. In all instances where the use of melphalan hydrochloride Injection is considered for chemotherapy, the physician must evaluate the need and usefulness of the drug against the risk of adverse events.
Use in renal impairment. Melphalan clearance may be reduced in patients with renal impairment, who may also have uraemic bone marrow suppression. Dosage reduction may therefore be necessary (see Section 4.2 Dose and Method of Administration), and these patients should be closely observed.
In one trial, increased bone marrow suppression was observed in patients with BUN levels ≥ 30 mg/Dl (≥ 10.71 mmol/L). A 50% reduction in the IV melphalan dose decreased the incidence of severe bone marrow suppression in the latter portion of this study.
Use in the elderly. Clinical experience with melphalan has not identified differences in responses between the elderly and younger patients. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Paediatric use. Safety and efficacy in children have not been established.
Effects on laboratory tests. Periodic complete blood counts with differentials should be performed during the course of treatment with Melpha. At least one determination should be obtained prior to each dose. Patients should be observed closely for consequences of bone marrow suppression, which include severe infections, bleeding, and symptomatic anaemia.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Cyclosporin. Impaired renal function has been described in bone marrow transplant patients who were conditioned with high-dose intravenous melphalan and who subsequently received cyclosporin to prevent graft-versus-host disease.
Cisplatin/carmustine. Cisplatin may affect melphalan kinetics by inducing renal dysfunction and subsequently altering melphalan clearance. IV melphalan may also reduce the threshold for Carmustine lung toxicity.
Nalidixic acid. When nalidixic acid and IV melphalan are given simultaneously, the incidence of severe haemorrhagic necrotic enterocolitis has been reported to increase in paediatric patients.
Vaccines. Vaccinations with live organism vaccines are not recommended in immunocompromised individuals. See Section 4.4 Special Warnings and Precautions for Use.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Melphalan causes suppression of ovarian function in premenopausal women, resulting in amenorrhoea in a significant number of patients. Reversible and irreversible testicular suppression have also been reported.
No fertility studies have been conducted in animals. However, there is evidence from some animal studies that melphalan can have an adverse effect on spermatogenesis. Therefore, it is possible that melphalan may cause temporary or permanent sterility in male patients.
Use in pregnancy. In view of its mutagenic properties and structural similarity to known teratogenic compounds, it is possible that melphalan could cause congenital defects in the offspring of patients treated with the drug.
Melphalan may cause foetal harm when administered to a pregnant woman.
As with all cytotoxic chemotherapy, adequate contraceptive precautions should be practised when either partner is receiving melphalan hydrochloride.
The use of melphalan hydrochloride should be avoided whenever possible during pregnancy, particularly during the first trimester. In any individual case the potential hazard to the foetus must be balanced against the expected benefit to the mother.
Although adequate animal studies have not been conducted with IV melphalan, oral and IV melphalan has been shown to be teratogenic and embryogenic in animal studies. A single dose of 5 mg/kg IP (30 mg/m2) given on day 6 or day 9 of gestation in the rat was embryolethal and teratogenic, and a single dose of 3 mg/kg IP (18 mg/m2) was teratogenic when administered on day 6. Malformations resulting from melphalan administration included alterations of the brain (underdevelopment, deformation, meningocele and encephalocele) and eye (anophthalmia and microphthalmos), reduction of the mandible and tail, as well as exomphaly (umbilical hernia).
In a repeat-dose embryotoxicity study in rats, (0.33, 1 and 3 mg/kg/day PO on gestation days 7-17; total doses: 22, 66 and 198 mg/m2 PO, respectively; cf. clinical dose of 16 mg/m2 IV), all doses were maternotoxic (reduced weight gain, and mortality occurred at the high dose). Intrauterine deaths, reduced foetal and pup weights and pup weight gain over the lactation period were seen in the mid and high dose groups but pup survival over the lactation period was reduced at all doses. Melphalan showed a reduction in ossification at ≥ 1 mg/kg/day and an increased incidence of rib anomalies and impairment of pup development (delayed eruption of incisors, significantly different open-field behaviour) at the high dose.
No animal studies have been conducted to investigate the peri- and post-natal effects of melphalan.
Use in lactation. It is not known whether this drug is excreted in human milk. Mothers receiving melphalan hydrochloride should not breast-feed.
4.7 Effects on Ability to Drive and Use Machines
No studies have been conducted on the effects on the viability and the ability to operate machines. However, the possibility will have to be taken into consideration, that the alcohol quantity in these pharmaceutical medicinal product can impair the competency to drive and the ability to operate machines.
4.8 Adverse Effects (Undesirable Effects)
The following information on adverse reactions is based on data from both oral and IV administration of melphalan as a single agent, using several different dose schedules for treatment of a wide variety of malignancies.
Haematologic. The most common side effect is bone marrow suppression, leading to leucopenia, thrombocytopenia and anaemia. White blood cell count and platelet count nadirs usually occur 2 to 3 weeks after treatment, with recovery in 4 to 5 weeks after treatment. Irreversible bone marrow failure has been reported. Acute leukaemia has also been reported (see Section 5.3 Preclinical Safety Data, Carcinogenicity).
Gastrointestinal. Gastrointestinal disturbances such as nausea, vomiting and diarrhoea are very common. At high doses of melphalan, stomatitis is very common and rare at conventional doses. The incidence of diarrhoea, vomiting and stomatitis becomes the dose limiting toxicity in patients given high i.v. doses of melphalan in association with heamopoietic stem cell rescue. Cyclophosphamide pre-treatment appears to reduce the severity of gastrointestinal damage induced by high-dose melphalan and the literature should be consulted for details. Oral ulceration occurs infrequently. Hepatic toxicity, including veno-occlusive disease, has been reported.
Hypersensitivity. Acute hypersensitivity reactions including anaphylaxis were reported in 2.4% of 425 patients receiving melphalan for Injection for myeloma (see Section 4.4 Special Warnings and Precautions for Use). These reactions were characterised by urticaria, pruritus, skin rashes, oedema, and in some patients, tachycardia, bronchospasm, dyspnoea, and hypotension. These patients appeared to respond to antihistamine and corticosteroid therapy. If a hypersensitivity reaction occurs, IV or oral melphalan should not be readministered since hypersensitivity reactions have also been reported with oral melphalan. Cardiac arrest has also been reported rarely in association with such events.
Miscellaneous. Other reported adverse reactions include skin hypersensitivity, skin ulceration at injection site, skin necrosis rarely requiring skin grafting, maculopapular rashes, pruritus, vasculitis, allergic reaction, and interstitial pneumonitis. A subjective and transient sensation of warmth and/or tingling is very common.
Hepatic disorders ranging from abnormal liver function tests to clinical manifestations such as hepatitis and jaundice have been reported. Veno-occlusive disease has been reported in association with these cases.
There have been case reports of interstitial pneumonitis and pulmonary fibrosis. There have also been case reports of fatal pulmonary fibrosis and haemolytic anaemia occurring after melphalan treatment.
Alopecia is very common at high doses and common at conventional doses.
Temporary significant elevation of the blood urea has been seen in the early stages of melphalan therapy in myeloma patients with renal damage.
Muscular atrophy, muscle fibrosis, myalgia and increases in blood creatine phosphokinase are very commonly observed following isolated limb perfusion, while compartment syndrome is commonly observed. The incidence of muscle necrosis and rhabdomyolysis are not known in this setting.
Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Symptoms and signs. Overdoses resulting in death have been reported. Overdoses, including doses up to 290 mg/m2, have produced the following symptoms: severe nausea and vomiting, decreased consciousness, convulsions, muscular paralysis, and cholinomimetic effects. Damage to the gastrointestinal lining may also ensue. Severe mucositis, stomatitis, colitis, diarrhoea, and haemorrhage of the gastrointestinal tract occur at high doses (> 100 mg/m2). Elevations in liver enzymes and veno-occlusive disease occur infrequently. Significant hyponatremia caused by an associated inappropriate secretion of antidiuretic hormone (ADH) syndrome has been observed. Nephrotoxicity and adult respiratory distress syndrome have been reported rarely.
Treatment. The principal toxic effect is bone marrow suppression, leading to leucopenia, thrombocytopenia and anaemia. Haematologic parameters should be closely followed for at least 4 weeks following overdosage until there is evidence of recovery. An uncontrolled study suggests that administration of autologous bone marrow or haematopoietic growth factors (i.e. filgrastim) may shorten the period of pancytopenia. General supportive measures together with appropriate blood and platelet transfusions and antibiotics should be instituted as deemed necessary by the physician. This drug is not removed from plasma to any significant degree by haemodialysis or haemoperfusion. A paediatric patient survived a 254 mg/m2 overdose treated with standard supportive care.
For information on the management of overdose, contact the Poison Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Cytotoxic and alkylating agent.
Melphalan is a bifunctional alkylating agent. Formation of carbonium intermediates from each of the two bis-2-chloroethyl groups enables alkylation through covalent binding with the 7-nitrogen of guanine on DNA, cross-linking two DNA strands and thereby preventing cell replication. Like other bifunctional alkylating agents, it is active against both resting and rapidly dividing tumour cells.
Clinical trials. A randomised trial compared prednisone plus IV melphalan to prednisone plus oral melphalan in the treatment of myeloma. As discussed below, overall response rates at week 22 were comparable; however, because of changes in trial design, conclusions as to the relative activity of the two formulations after week 22 are impossible to make. Both arms received oral prednisone starting at 0.8 mg/kg per day with doses tapered over 6 weeks. Melphalan doses in each arm were:
Arm 1 Oral melphalan 0.15 mg/kg per day x 7 followed by 0.05 mg/kg per day when WBC began to rise.
Arm 2 IV melphalan 16 mg/m2 once every 2 weeks x 4 (over 6 weeks) followed by the same dose every 4 weeks.
Doses of melphalan were adjusted according to the following criteria (see Table 1).
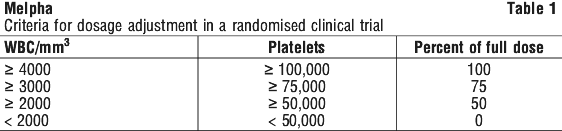

Severe myelotoxicity (WBC ≤ 1000 and/or platelets ≤ 25,000) was more common in the IV melphalan arm (28%) than in the oral melphalan arm (11%).
An association was noted between poor renal function and myelosuppression; consequently, an amendment to the protocol required a 50% reduction in IV melphalan dose if the BUN was ≥ 30 mg/Dl (≥ 10.71 mmol/L). The rate of severe leucopenia in the IV arm in the patients with BUN over 30 mg/Dl (≥ 10.71 mmol/L) decreased from 50% (8/16) before protocol amendment to 11% (3/28) (P = 0.01) after the amendment.
Before the dosing amendment, there was a 10% (8/77) incidence of drug-related death in the IV arm. After the dosing amendment, this incidence was 3% (3/108). This compares to an overall 1% (1/100) incidence of drug-related death in the oral arm.
5.2 Pharmacokinetic Properties
Absorption. The pharmacokinetics of melphalan after IV administration has been extensively studied in adult patients. Following injection, drug plasma concentrations declined rapidly in a biexponential manner with distribution phase and terminal elimination phase half-lives of approximately 10 and 75 minutes, respectively. Estimates of average total body clearance varied among studies, but typical values of approximately 7 to 9 mL/min per kg (250 to 325 mL/min per m2) were observed. One study has reported that on repeat dosing of 0.5 mg/kg every 6 weeks, the clearance of melphalan decreased from 8.1 mL/min per kg after the first course, to 5.5 mL/min per kg after the third course, but did not decrease appreciably after the third course. Mean (±SD) peak melphalan plasma concentrations in myeloma patients given IV melphalan at doses of 10 or 20 mg/m2 were 1.2 ± 0.4 and 2.8 ± 1.9 microgram/mL, respectively.
Distribution. The steady-state volume of distribution of melphalan is 0.5 L/kg. Penetration into cerebrospinal fluid (CSF) is low. The extent of melphalan binding to plasma proteins ranges from 60% to 90%. Serum albumin is the major binding protein, while α1-acid glycoprotein appears to account for about 20% of the plasma protein binding.
Approximately 30% of the drug is (covalently) irreversibly bound to plasma proteins. Interactions with immunoglobulins have been found to be negligible.
Metabolism. Melphalan is metabolised primarily by chemical hydrolysis to monohydroxymelphalan and dihydroxymelphalan. Aside from these hydrolysis products, no other melphalan metabolites have been observed in humans.
Excretion. Melphalan is eliminated from plasma. Although the contribution of renal elimination to melphalan clearance appears to be low, one study noted an increase in the occurrence of severe leucopoenia in patients with elevated blood urea nitrogen (BUN) after 10 weeks of therapy.
5.3 Preclinical Safety Data
Genotoxicity. Chromosome aberrations have been observed in patients being treated with Melphalan. Melphalan has been shown to cause chromatid and chromosome damage in human lymphocytes at a single dose of 20 mg IV (~10.6 mg/m2, comparable to a therapeutic dose of 16 mg/m2) and in rat bone marrow cells at a single intramuscular dose of 6 mg/m2. Melphalan also showed mutagenic effects on germ cells in male mice at 17.1-21.9 mg/m2.
Carcinogenicity. "Secondary malignancies, including acute nonlymphocytic leukaemia, myeloproliferative syndrome, and carcinoma, have been reported in patients with cancer treated with alkylating agents (including melphalan). Some patients also received other chemotherapeutic agents or radiation therapy. Precise quantitation of the risk of myeloproliferative syndrome, or carcinoma is not possible. Melphalan, in common with other alkylating agents, has been reported to be leukaemogenic. There have been reports of acute leukaemia occurring after melphalan treatment for diseases such as amyloid, malignant melanoma, multiple myeloma, macroglobulinaemia, cold agglutinin syndrome and ovarian cancer. Published reports of leukaemia in patients who have received melphalan (and other alkylating agents) suggest that the risk of leukaemogenesis increases with chronicity of treatment and with cumulative dose.
A comparison of patients with ovarian cancer who received alkylating agents with those who did not showed that the use of alkylating agents, including melphalan, significantly increased the incidence of acute leukaemia.
The potential benefits from melphalan hydrochloride therapy must be weighed on an individual basis against the possible risk of the induction of a second malignancy.
Although adequate and well-controlled carcinogenicity studies have not been conducted in animals, there is clear evidence from animal studies that melphalan is carcinogenic. Intraperitoneal (IP) administration of melphalan in rats (5.4 or 10.8 mg/m2) and mice (2.25 or 4.5 mg/m2) three times per week for 6 months followed by a 12 months postdose observation produced peritoneal sarcoma in rats, and lung tumors and lymphosarcomas (males) in mice. Lung tumours were also increased in two other studies in mice (total dose: 144 mg/m2 dermal given as 10 injections over a period of 10 weeks; 3.2-51 mg/m2 IP given as 12 injections over a period of 4 weeks) while in one of these studies (dermal), skin papillomas were increased although non-significantly."
6 Pharmaceutical Particulars
6.1 List of Excipients
Sodium citrate dihydrate, propylene glycol, ethanol, water for injection to a total of 10 mL.
6.2 Incompatibilities
Melpha Injection is not compatible with infusion solutions containing dextrose and it is recommended that only Sodium Chloride Intravenous Infusion 0.9% w/w be used.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C.
Protect from light.
6.5 Nature and Contents of Container
Melpha for Injection is supplied in a carton containing one single-use clear glass vial of freeze-dried melphalan hydrochloride equivalent to 50 mg melphalan and one 10-mL clear glass vial of sterile diluent.
6.6 Special Precautions for Disposal
Melpha Injection solution should be disposed in a manner appropriate for toxic chemicals, for example, high-temperature incineration.
6.7 Physicochemical Properties
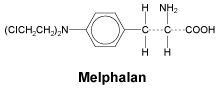
Melphalan hydrochloride, also known as L-phenylalanine mustard, phenylalanine mustard, L-PAM, or L-sarcolysin, is a phenylalanine derivative of nitrogen mustard. Melphalan hydrochloride is a bifunctional alkylating agent that is active against selected human neoplastic diseases. It is known chemically as 4-[bis (2-chloroethyl) amino]-L-phenylalanine. The molecular formula is C13H18Cl2N2O2.HCl and the molecular weight is 341.67.
Melphalan is the active L-isomer of the compound and was first synthesised in 1953. Melphalan is practically insoluble in water and freely soluble in methanol, soluble in conc. HCl.
Chemical structure. The structural formula is:
7 Medicine Schedule (Poisons Standard)
S4 - Prescription only medicine.
Date of First Approval
15 March 2018
Date of Revision
23 September 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.