Metomax
Brand Information
| Brand name | Metomax |
| Active ingredient | Metoclopramide hydrochloride + Paracetamol |
| Schedule | S3 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Metomax.
Summary CMI
METOMAX®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I taking METOMAX?
METOMAX contains the active ingredients metoclopramide hydrochloride (as monohydrate) and paracetamol. METOMAX is used for the relief of headache, nausea, and vomiting associated with migraine.
For more information, see Section 1. Why am I taking METOMAX? in the full CMI.
2. What should I know before I take METOMAX?
Do not take if you have ever had an allergic reaction to paracetamol and/or metoclopramide, or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I take METOMAX? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with METOMAX and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I take METOMAX?
- The initial dosage for adults is 1 to 2 capsules and then 1-2 capsules every 4 hours, as needed. Do not take more than 6 capsules in 24 hours, and do not take it for longer than 48 hours at a time unless advised to by a doctor.
- METOMAX should be taken at the first sign of a migraine attack.
- Swallow the capsule with a glass of water.
More instructions can be found in Section 4. How do I take METOMAX? in the full CMI.
5. What should I know while taking METOMAX?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while taking METOMAX? in the full CMI.
6. Are there any side effects?
Very common side effects of METOMAX include restlessness, drowsiness, fatigue, and lethargy. Common side effects include insomnia, headache, dizziness, nausea, or bowel upsets. Serious side effects of METOMAX include unusual changes in mood, different types of muscle spasms or rigidity, an allergic reaction, difficulty breathing, severe drowsiness/fatigue, or a bluish discolouration of the skin.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
METOMAX®
Active ingredient(s): paracetamol & metoclopramide hydrochloride (as monohydrate)
Consumer Medicine Information (CMI)
This leaflet provides important information about using METOMAX. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using METOMAX.
Where to find information in this leaflet:
1. Why am I taking METOMAX?
2. What should I know before I take METOMAX?
3. What if I am taking other medicines?
4. How do I take METOMAX?
5. What should I know while taking METOMAX?
6. Are there any side effects?
7. Product details
1. Why am I taking METOMAX?
METOMAX contains the active ingredients paracetamol and metoclopramide hydrochloride (as monohydrate). METOMAX is used for the relief of headache, nausea and vomiting associated with migraine.
Metoclopramide helps control nausea and vomiting caused by migraine and other illnesses. It works by blocking the action of a chemical in the brain which causes nausea and vomiting. It also acts in the stomach and upper intestine to increase muscle contractions.
Paracetamol is an analgesic. It provides effective temporary relief from pain (such as headache).
2. What should I know before I take METOMAX?
Warnings
Do not take METOMAX if:
- you have any of the following health conditions:
- porphyria (causing pain, nausea and vomiting)
- pain in the stomach, chest, legs or back
- bleeding from the stomach and/or digestive tract
- intestinal blockage
- recent surgery on the stomach and/or digestive tract
- phaeochromocytoma (a rare tumour of the adrenal gland)
- epilepsy (fits). - you are allergic to metoclopramide, paracetamol or any of the ingredients listed at the end of this leaflet.
Some symptoms of an allergic reaction may include;
- shortness of breath
- wheezing or difficulty in breathing
- swelling of the face, lips, tongue, or any other parts of the body
- rash, itching or hives on the skin. - the expiry date printed on the pack has passed.
- the packaging is torn or shows signs of tampering, or if the capsules do not look quite right.
Check with your doctor if you:
- have any other medical conditions, especially the following:
- epilepsy - metoclopramide may increase the risk of you having a fit
- breast cancer
- liver or kidney disease
- Parkinson's disease, a condition affecting muscle control and movement
- you have had movements that you cannot control, mainly of the tongue, mouth, jaw, arms and legs after taking metoclopramide or medicines used to calm emotional and mental problems.
Tell your doctor or pharmacist if you plan to have surgery.
METOMAX should not be taken immediately after certain types of operations.
Tell your doctor or pharmacist if you have had any allergy to any other medicines, or any other substances, such as foods, preservatives, or dyes.
Talk to your doctor or pharmacist if you are not sure whether you should start taking METOMAX.
During treatment with METOMAX, tell your doctor straight away if you have severe illnesses, including severe renal impairment or sepsis (when bacteria and their toxins circulate in the blood leading to organ damage), or you suffer from malnutrition, chronic alcoholism or if you are also taking flucloxacillin (an antibiotic). A serious condition called metabolic acidosis (a blood and fluid abnormality) has been reported in patients in these situations when paracetamol is taken at regular doses for a prolonged period or when paracetamol is taken together with flucloxacillin. Symptoms of metabolic acidosis may include: serious breathing difficulties with deep rapid breathing, drowsiness, feeling sick (nausea) and being sick (vomiting).
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Do not take METOMAX if you are pregnant or intend to become pregnant.
It may affect the developing baby if you take it during pregnancy. Do not take METOMAX if you are breastfeeding or intend to breastfeed.
METOMAX passes into breast milk and, although the effect on your baby is not known, there is a possibility that your breastfed baby may be affected.
Use in Children
Do not give METOMAX to children and adolescents under 18 years of age.
The safety of this medicine in children under 18 years of age has not been established.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and METOMAX may interfere with each other. These include:
- medicines used to prevent blood clots
- medicines used to treat epilepsy
- pain relievers such as codeine and morphine
- some medicines found in travel sickness, hay fever and allergy, stomach cramps, and cough and cold preparations
- medicines used to treat anxiety or help you to sleep
- medicines used to treat certain mental and emotional conditions, such as schizophrenia
- tetracycline antibiotics
- levodopa, a medicine used in the treatment of Parkinson's disease
- digoxin, a medicine used to treat heart failure
- flucloxacillin, an antibiotic, due to a serious risk of blood and fluid abnormality (called metabolic acidosis) that must have urgent treatment. See Section 2. What should I know before I take METOMAX? for further information.
- other paracetamol-containing products.
These medicines may be affected by METOMAX or may affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines.
Your doctor or pharmacist has a more complete list of medicines to avoid while taking METOMAX.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect METOMAX.
4. How do I take METOMAX?
How much to take
- The initial dose for adults is 1 to 2 capsules and then 1-2 capsules every 4 hours, as needed.
- Do not take more than 6 capsules in 24 hours.
Do not take more than the recommended dose.
How to take METOMAX
Swallow the capsules whole with a full glass of water.
When to take METOMAX
METOMAX should be taken at the first sign of a migraine attack.
How long to take it for
METOMAX is not intended for long-term use.
Do not take it for longer than 48 hours at a time unless advised to by a doctor.
If you forget to take METOMAX
If symptoms persist, take your next dose when you are meant to.
Do not take a double dose to make up for the dose you missed.
This may increase the chance of you getting an unwanted side effect.
If you are not sure what to do, ask your doctor or pharmacist.
If you have trouble remembering when to take your medicine, ask your pharmacist for some hints.
If you take too much METOMAX
If you think that you or anyone else has taken too much METOMAX, urgent medical attention may be required.
You should immediately:
- phone the Poisons Information Centre
(Australia telephone 13 11 26) for advice, or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
There is a risk of delayed, serious liver damage.
5. What should I know while taking METOMAX?
Things you should do
- Tell your doctor or pharmacist if nausea, vomiting or headache persists or worsens.
- Tell all doctors, dentists and pharmacists who are treating you that you are taking METOMAX.
- If you are about to be started on any new medicine, tell your doctor or pharmacist that you are taking METOMAX.
Tell your doctor straight away if:
- you become pregnant while you are taking METOMAX
Things you should not do
- Do not take METOMAX for longer than 48 hours at a time unless advised to by a doctor.
- Do not give this medicine to anyone else, even if they have similar symptoms.
- Do not take METOMAX to treat any other complaints unless your doctor or pharmacist tells you to.
- Do not take METOMAX with other products containing paracetamol, unless advised to do so by a doctor or pharmacist.
No more than 4 g of paracetamol should be taken in any 24 hour period.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how METOMAX affects you.
METOMAX may cause drowsiness, tiredness or dizziness in some people. If any of these occur, do not drive, operate machinery or do anything else that could be dangerous.
Drinking alcohol
Avoid drinking alcohol while taking METOMAX.
Combining METOMAX with alcohol can make you more sleepy or drowsy.
Looking after your medicine
- Store below 25°C.
- Keep your capsules in the original container until it is time to take them.
Store METOMAX in a cool dry place away from moisture, heat or sunlight. For example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
Do not take this medicine after the expiry date.
If you no longer need to take this medicine or if the expiry date has passed, you can take it to any pharmacy for safe disposal.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away if you notice any of these serious side effects. This includes serious side effects which may require medical attention or hospitalisation |
| Call your doctor straight away or go to the Emergency Department at your nearest hospital if you notice any of these very serious side effects. These side effects are rare, but serious and require urgent medical attention or hospitalisation. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available from your pharmacist without a doctor's prescription.
What METOMAX contains
| Active ingredient (main ingredient) | paracetamol 500 mg metoclopramide hydrochloride (as monohydrate) 5 mg |
| Other ingredients (inactive ingredients) |
|
| Potential allergens | sulfites |
Do not take this medicine if you are allergic to any of these ingredients.
What METOMAX looks like
METOMAX is a capsule marked with a Greek alpha symbol on the green section and "P500|M5" on the yellow section (AUST R 121343).
Each pack contains 10 capsules.
Who distributes METOMAX
Alphapharm Pty Ltd trading as Viatris
Level 1, 30 The Bond
30-34 Hickson Road
Millers Point NSW 2000
www.viatris.com.au
Phone: 1800 274 276
This leaflet was prepared in November 2025.
METOMAX® is a Viatris company trade mark
METOMAX_cmi\Nov25/00
Brand Information
| Brand name | Metomax |
| Active ingredient | Metoclopramide hydrochloride + Paracetamol |
| Schedule | S3 |
MIMS Revision Date: 01 January 2026
1 Name of Medicine
Paracetamol.
Metoclopramide hydrochloride (as monohydrate).
2 Qualitative and Quantitative Composition
Each Metomax capsule contains 500 mg paracetamol and 5.25 mg metoclopramide hydrochloride monohydrate equivalent to 5 mg metoclopramide hydrochloride.
Excipients with known effect. Sulfites.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Metomax capsules. The capsules are size 0, hard gelatin capsules with a green cap and yellow body. Printed in black ink is "α" on the cap and "P500/M5" on the body.
4 Clinical Particulars
4.1 Therapeutic Indications
Symptomatic relief of headache, nausea and vomiting associated with migraine.
4.2 Dose and Method of Administration
Metomax should be taken at the first warning of a migraine headache. If symptoms persist, further doses may be taken at four hourly intervals. Total dosage in any 24 hour period should not exceed the quantity stated.
The dosage recommendations given below should be strictly adhered to if side effects of the dystonic type are to be avoided. It should be noted that a total daily dosage of metoclopramide should not normally exceed 0.5 mg/kg bodyweight with a maximum of 30 mg daily. Metomax should only be used after careful examination to avoid masking an underlying disorder e.g. cerebral irritation.
Maximum treatment duration is 5 days. Keep to the recommended dose. Do not use for longer than a few days at a time unless advised to by a doctor.
Adults 18 years and over. The recommended dose is 1 or 2 capsules initially and then 1-2 capsules every 4 hours (maximum dose of 6 capsules in 24 hours).
Children and adolescents under 18 years. Metomax is not recommended for use in children under 18 years.
Renal and hepatic impairment. Therapy should be initiated at half the normal dose since adverse effects are likely to be exacerbated. The dose should then be adjusted depending on the clinical response.
4.3 Contraindications
Wherever stimulation of gastrointestinal motility might be dangerous, e.g. in the presence of gastrointestinal haemorrhage, mechanical obstruction or perforation.
Stimulation of gastrointestinal motility may aggravate these conditions.
Phaeochromocytoma. Hypertensive crisis have been reported with the use of metoclopramide in these patients. This is probably due to the release of catecholamines from the tumour. Such hypertensive crises may be controlled by phentolamine.
Epilepsy. The frequency and severity of seizures may be increased in epileptic patients given metoclopramide. Metoclopramide should not be used in patients with epilepsy since it may increase the frequency and severity of seizures.
Patients with porphyria. Metoclopramide should not be administered to patients receiving other drugs which are likely to cause extrapyramidal reactions, since the frequency and severity of extrapyramidal reactions may be increased.
Known hypersensitivity or intolerance to paracetamol, metoclopramide or any of the excipients in Metomax capsules.
Children and adolescence under 18 years.
Insufficient safety data exist to support the use of Metomax (metoclopramide/ paracetamol combination) in pregnancy or during lactation (see Section 4.4 Special Warnings and Precautions for Use).
4.4 Special Warnings and Precautions for Use
High anion gap metabolic acidosis. Cases of high anion gap metabolic acidosis (HAGMA) due to pyroglutamic acidosis have been reported in patients with severe illness such as severe renal impairment and sepsis, or patients with malnutrition and other sources of glutathione deficiency (e.g. chronic alcoholism) who were treated with paracetamol at therapeutic dose for a prolonged period or a combination of paracetamol and flucloxacillin. If HAGMA due to pyroglutamic acidosis is suspected, prompt discontinuation of paracetamol and close monitoring is recommended. The measurement of urinary 5-oxoproline may be useful to identify pyroglutamic acidosis as underlying cause of HAGMA in patients with multiple risk factors.
Persistent tardive dyskinesia. Tardive dyskinesia may appear in some patients on long-term therapy or may appear after drug therapy has been discontinued. The risk appears to be greater in elderly patients on high dose therapy, especially females. The symptoms are persistent and can oftentimes appear to be irreversible. The syndrome is characterised by rhythmical involuntary movement of the tongue, face, mouth or jaw (e.g. protrusion of tongue, puffing of cheeks, puckering of mouth, chewing movements). Sometimes these may be accompanied by involuntary movements of extremities. There is no known effective treatment for tardive dyskinesia; however, in some patients symptoms may lessen or resolve after metoclopramide treatment is stopped. Antiparkinson agents usually do not alleviate the symptoms of this syndrome.
Although the risk of tardive dyskinesia with metoclopramide has not been extensively studied, one published study reported a tardive dyskinesia prevalence of 20% among patients treated for at least 3 months. Both the risk of developing the syndrome and the likelihood that it will become irreversible are believed to increase with the duration of treatment and the total cumulative dose.
Metoclopramide therapy should routinely be discontinued in patients who develop signs or symptoms of tardive dyskinesia. It has been suggested that fine vermicular movements of the tongue may be an early sign of the syndrome, and, if the medication is stopped at that time, the syndrome may not develop. Tardive dyskinesia may remit, partially or completely, within several weeks to months after metoclopramide is withdrawn. Metoclopramide itself, however, may suppress (or partially suppress) the signs of tardive dyskinesia, thereby masking the underlying disease process. The effect of this symptomatic suppression upon the long-term course of the syndrome is unknown. Therefore, metoclopramide should not be used for the symptomatic control of tardive dyskinesia.
Care should be exercised in patients being treated with other centrally active drugs.
Since extrapyramidal symptoms may occur with both metoclopramide and neuroleptics such as phenothiazines, care should be exercised in the event of both drugs being prescribed concurrently.
Epilepsy. The frequency and severity of seizures or extrapyramidal reactions may be increased in epileptic patients given metoclopramide.
Dystonia. Dystonic reactions occur in approximately 1% of patients given metoclopramide. These occur more frequently in children and young adults and may occur after a single dose.
Neuroleptic malignant syndrome. Has been reported with metoclopramide in combination with antipsychotics, as well as with metoclopramide monotherapy (see Section 4.8 Adverse Effects (Undesirable Effects)).
Breast cancer and prolactin levels. Metoclopramide elevates prolactin levels and the elevation persists during chronic administration. Tissue culture experiments indicate that approximately one-third of human breast cancers are prolactin dependent in vitro, a factor of potential importance if the prescription of metoclopramide is contemplated in a patient with previously detected breast cancer. Although disturbances such as galactorrhoea, amenorrhoea, gynaecomastia and impotence have been reported with prolactin elevating drugs, the clinical significance of elevated serum prolactin levels is unknown for most patients. An increase in mammary neoplasms has been found in rodents after chronic administration of prolactin stimulating antipsychotic drugs.
Neither clinical studies nor epidemiological studies conducted to date, however, have shown an association between chronic administration of these drugs and mammary tumourigenesis; the available evidence is too limited to be conclusive at this time.
Parkinson's disease. Metoclopramide can exacerbate parkinsonian symptoms, hence should be used with caution, if at all, in patients with parkinsonian syndrome.
Surgery. Following operations such as pyloroplasty or gut anastomosis, metoclopramide therapy should be withheld for three or four days as vigorous muscular contractions may not help healing.
Masking of serious illness. The symptomatic relief provided by metoclopramide may delay recognition of serious disease. This product should not be prescribed or recommended until diagnosis has been established, and should not be substituted for appropriate investigation of the patient's symptoms.
If vomiting persists in a patient receiving Metomax, the patient should be reassessed to exclude the possibility of an underlying disorder, e.g. cerebral irritation.
Other paracetamol containing products. Patients should be warned not to take this product with other paracetamol containing products.
Use in hepatic or renal impairment. Metomax should be administered with caution to patients with renal or hepatic dysfunction.
Plasma concentrations of paracetamol and its conjugates are increased in patients with moderate renal failure. In patients with clinically significant degrees of renal or hepatic impairment, the clearance of metoclopramide is likely to be reduced (see Section 4.2 Dose and Method of Administration).
Use in the elderly. See Section 4.4 Special Warnings and Precautions for Use; Section 4.8 Adverse Effects (Undesirable Effects).
Paediatric use. Metomax is not recommended for use in children and adolescents under 18 years of age.
There is a higher incidence of adverse events from metoclopramide in children (see Section 4.3 Contraindications).
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Paracetamol. Anticoagulants. Anticoagulant dosage may require reduction if treatment with Metomax is prolonged.
Paracetamol absorption is increased by drugs which increase gastric emptying, e.g. metoclopramide, and decreased by drugs which decrease gastric emptying, e.g. propantheline, antidepressants with anticholinergic properties, narcotic analgesics. Paracetamol may increase chloramphenicol concentrations.
Enzyme inducing agents. The likelihood of paracetamol toxicity may be increased by the concomitant use of enzyme inducing agents such as alcohol or anticonvulsant drugs.
Concomitant use with flucloxacillin. Caution should be taken when paracetamol is used concomitantly with flucloxacillin as concurrent intake has been associated with high anion gap metabolic acidosis due to pyroglutamic acidosis, especially in patients with risks factors (see Section 4.4 Special Warnings and Precautions for Use).
Metoclopramide. Anticholinergic drugs and narcotic (opioid-containing) analgesics. The effects of metoclopramide on gastrointestinal motility can be antagonised by these.
CNS depressants. Additive sedative effects can occur when metoclopramide is given with alcohol, sedatives, hypnotics, narcotics or tranquillisers.
Drugs affected by increased gastrointestinal motility. Since metoclopramide accelerates abnormally slow gastric and small bowel peristaltic activity, it may change absorption of orally administered drugs. The absorption of drugs from the small bowel may be accelerated (e.g. paracetamol, tetracycline, levodopa), whereas absorption of drugs from the stomach may be diminished (e.g. digoxin).
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category A)
Pregnancy Category: metoclopramide hydrochloride (Category A), paracetamol (Category A).
Metoclopramide/ paracetamol combination. Insufficient safety data exist to support the use of Metomax (metoclopramide/ paracetamol combination) in pregnancy (see Section 4.3 Contraindications).
Use in lactation. This product should only be used in breastfeeding mothers when the expected benefits to the mother outweigh any potential risk to the baby. Paracetamol and metoclopramide are both excreted in breast milk. The amount available for ingestion by the infant has been reported variously as less than 0.1% of a single dose of paracetamol 500 mg, and as 0.04 to 0.23% of a single 650 mg dose.
Maternal ingestion of paracetamol in usual analgesic doses does not appear to present a risk to the breastfed infant. It is not known whether it has a harmful effect on the newborn. The administration of metoclopramide to breastfeeding mothers may increase the risk of adverse reactions in young children and should be taken into account when making a risk-benefit assessment.
Insufficient safety data exist to support the use of Metomax (metoclopramide/ paracetamol combination) during lactation (see Section 4.3 Contraindications).
4.7 Effects on Ability to Drive and Use Machines
Patients should be cautioned about engaging in activities requiring mental alertness for a few hours after taking this product.
4.8 Adverse Effects (Undesirable Effects)
Paracetamol. Reports of adverse reactions are rare. Although the following reactions have been reported, a causal relationship to the administration of paracetamol has been neither confirmed nor refuted: dyspepsia, nausea, allergic and haematological reactions.
Cases of high anion gap metabolic acidosis due to pyroglutamic acidosis have been observed (frequency cannot be estimated from the available data) in patients with risk factors using paracetamol (see Section 4.4 Special Warnings and Precautions for Use). Pyroglutamic acidosis may occur as a consequence of low glutathione levels in these patients.
Metoclopramide. The most frequent adverse reactions to metoclopramide are restlessness, drowsiness, fatigue and lassitude, which occur in approximately 10% of patients.
Less frequently, insomnia, headache, dizziness, nausea or bowel disturbances may occur. Rare (less than 1 in 1,000) cases of acute depression have been reported. Anxiety or agitation may occur.
Raised serum prolactin levels have been observed during metoclopramide therapy; this effect is similar to that noted with many other compounds.
Although uncommon at normal dosage, various extrapyramidal reactions to metoclopramide, usually of the dystonic type, have been reported. Reactions include spasm of the facial muscles, trismus, rhythmic protrusion of the tongue, a bulbar type of speech, spasm of the extraocular muscles including oculogyric crises, unnatural positioning of the head and shoulders and opisthotonos. There may be a generalised increase in muscle tone. The majority of reactions occur within 36 hours of starting treatment and the effects usually disappear within 24 hours of withdrawal of the drug, however, close observation is required and in cases of more severe reactions, an antiparkinson drug such as benztropine or an anticholinergic antihistamine such as diphenhydramine should be given.
Tardive dyskinesia, which may be persistent, has been reported particularly in elderly patients undergoing long-term therapy with metoclopramide.
Very rare (less than 1 in 10,000) occurrences of the Neuroleptic Malignant Syndrome have been reported. This syndrome is potentially fatal and comprises hyperpyrexia, altered consciousness, muscle rigidity, autonomic instability and elevated levels of creatine phosphokinase (CPK) and must be treated urgently (recognised treatments include dantrolene and bromocriptine). This medicine should be stopped immediately if this syndrome occurs.
Methaemoglobinaemia has also been reported.
Parkinsonian symptoms, including tremor, rigidity, bradykinesia and akinesia, occur rarely in patients receiving metoclopramide but may be associated with usual or excessive doses or with decreased renal function.
There have been isolated reports of hypersensitivity reactions (such as urticaria, maculopapular rash) in patients receiving metoclopramide.
There have been a few cases of neutropenia, leucopenia and agranulocytosis generally without clear cut relationship to metoclopramide.
Sulfhaemoglobinaemia in adults.
Hyperthermia has also been observed.
Raised serum prolactin levels have been observed during metoclopramide therapy; this effect is similar to that noted with many other compounds. Galactorrhoea and breast enlargement have also been observed during metoclopramide therapy.
Respiratory failure, secondary to dystonic reaction, acute asthmatic symptoms of wheezing and dyspnoea may occur.
Urinary incontinence and frequency, sexual dysfunction, priapism and muscle spasm may also occur.
Rarely, cases of hepatotoxicity, characterised by such findings as jaundice and altered liver function tests, when metoclopramide was administered with other drugs with known hepatotoxic potential.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Symptoms. Toxic symptoms of paracetamol overdose include vomiting, abdominal pain, hypotension, sweating, central stimulation with exhilaration and convulsions in children, drowsiness, respiratory depression, cyanosis and coma.
Paracetamol overdose can result in severe liver damage and sometimes acute renal tubular necrosis.
In adults, hepatoxicity may occur after ingestion of a single dose of paracetamol 10 to 15 g (20 to 30 capsules or 10 to 15 times the normal dose); a dose of 25 g (50 capsules) or more is potentially fatal.
Symptoms during the first two days of acute poisoning by paracetamol do not reflect the potential seriousness of the intoxication. Major manifestations of liver failure such as jaundice, hypoglycaemia and metabolic acidosis may take at least three days to develop. Liver damage and death may occur.
Extrapyramidal side effects are the most frequently reported adverse reactions to metoclopramide overdosage. Very rarely AV block has been observed.
Treatment. Prompt treatment is essential even when there are no obvious symptoms.
Treatment consists primarily of management of paracetamol toxicity. Gastric emptying, close observation and supportive therapy is the management plan of choice for metoclopramide intoxication.
In cases of overdosage, methods of reducing absorption of ingested drug are important. Prompt administration of activated charcoal 50 g in 150 mL of water and 150 mL sorbitol 50% solution by mouth may reduce absorption. It is recommended that intravenous fluids such as normal saline be given concurrently. Gastric lavage is indicated if the patient is unwilling or unable to drink an activated charcoal/ sorbitol mixture. Methionine and acetylcysteine may be used as antidotes if given within a few hours of paracetamol overdosage.
If the history suggests that paracetamol 15 g or more has been ingested, administer the following antidote.
Intravenous acetylcysteine 20%. Administer acetylcysteine immediately without waiting for positive urine test or plasma level results if 8 hours or less since overdose ingestion. Initial dose 150 mg/kg over 15 minutes, followed by continuous infusion of 50 mg/kg in glucose 5% 500 mL over four hours and 100 mg/kg in glucose 5% 1 L over 16 hours.
If more than 8 hours have elapsed since the overdosage was taken, the antidote may be less effective.
When treatment for paracetamol toxicity has been initiated; antiparkinson and antihistamine/ anticholinergic drugs such as diphenhydramine hydrochloride can be administered to effectively control extrapyramidal reactions of metoclopramide. Haemodialysis appears ineffective in removing metoclopramide. Similarly, continuous ambulatory peritoneal dialysis does not remove significant amounts of the drug.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Paracetamol is a para-aminophenol derivative that exhibits analgesic and antipyretic activity. It does not possess anti-inflammatory activity.
Metoclopramide stimulates motility of the upper gastrointestinal tract without stimulating gastric, biliary or pancreatic secretions. Its mode of action is unclear. It seems to sensitise tissues to the action of acetylcholine. The effect of metoclopramide on motility is not dependent on intact vagal innervation, but it can be abolished by anticholinergic drugs.
Metoclopramide increases the tone and amplitude of gastric (especially antral) contractions, relaxes the pyloric sphincter and the duodenal bulb, and increases peristalsis of the duodenum and jejunum resulting in accelerated gastric emptying and intestinal transit. It increases the resting tone of the lower oesophageal sphincter. It has little, if any effect on the motility of the colon or gall bladder.
Metoclopramide has dopamine antagonist activity. Like the phenothiazines and related drugs, which are also dopamine antagonists, metoclopramide produces sedation and may produce extrapyramidal reactions (see Section 4.4 Special Warnings and Precautions for Use). Metoclopramide inhibits the central and peripheral effects of apomorphine, induces release of prolactin and causes a transient increase in circulating aldosterone levels.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Paracetamol. Absorption. After oral administration, paracetamol is absorbed rapidly and completely from the gastrointestinal tract; peak plasma concentrations occur 20 to 120 minutes after administration, with a mean value of about 51 minutes.
Distribution. Paracetamol is uniformly distributed throughout most body fluids; the apparent volume of distribution is 1 to 1.2 L/kg.
Paracetamol can cross the placenta and is excreted in breast milk. Plasma protein binding is negligible at usual therapeutic concentrations but increases with increasing concentrations.
Metabolism. Paracetamol is metabolised by the hepatic microsomal enzyme system. In adults at therapeutic doses, paracetamol is mainly conjugated with glucuronide (45 to 55%) or sulfate (20 to 30%). A minor proportion (less than 20%) is metabolised to catechol derivatives and mercapturic acid compounds via oxidation.
Excretion. Paracetamol is excreted in the urine mainly as the glucuronide and sulfate conjugates. Less than 5% is excreted as unchanged paracetamol with 85 to 90% of the administered dose eliminated in the urine within 24 hours of ingestion. The elimination half-life varies from 1.9 to 5.7 hours, with a mean value of about 3.2 hours. Food intake delays paracetamol absorption.
Metoclopramide. The onset of pharmacological action is 30 to 60 minutes following an oral dose.
Absorption. After oral administration, the peak plasma concentrations occur in 38 minutes to 2.5 hours, with a mean value of about 1 hour.
Distribution. Metoclopramide is excreted in breast milk. Plasma protein binding is 13 to 22%.
Metabolism. About 80% of the drug is excreted in the urine in the first 24 hours, approximately half as the glucuronide and sulfate conjugates and half as unchanged drug.
Excretion. The elimination half-life varies from 3.2 to 14 hours, with a mean value of about 6.7 hours. Impaired renal function results in reduced clearance of metoclopramide and an increased half-life (15 hours).
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Colloidal anhydrous silica, magnesium stearate, purified talc, sodium starch glycollate, black ink (ID 2328, ID 2343), gelatin capsule size 0 (ID 11306).
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C.
6.5 Nature and Contents of Container
Metomax capsules. Container type: PVC/PVDC/Al blisters.
Pack size: 10 capsules.
Australian register of therapeutic goods (ARTG). AUST R 121343 - Metomax capsule blister pack.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking it to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure. Paracetamol.
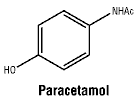
CAS number. 103-90-2.
Paracetamol is a white crystalline powder, sparingly soluble in water, freely soluble in alcohol, very slightly soluble in ether and in methylene chloride.
Metoclopramide hydrochloride monohydrate.
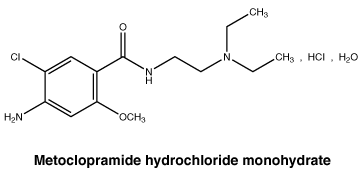
CAS number. 54143-57-6.
Metoclopramide hydrochloride is a white or almost white, crystalline powder or crystals, very soluble in water, freely soluble in alcohol, sparingly soluble in methylene chloride, practically insoluble in ether.
7 Medicine Schedule (Poisons Standard)
S3 (Pharmacist Only Medicine).
Date of First Approval
16 August 2005
Date of Revision
17 November 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.