Metopirone
Brand Information
| Brand name | Metopirone |
| Active ingredient | Metyrapone |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Metopirone.
Summary CMI
METOPIRONE®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using METOPIRONE?
METOPIRONE contains the active ingredient metyrapone. METOPIRONE is used as a test to check if ACTH (adrenocorticotropic hormone), a natural body hormone acting on corticosteroid secretion), is being produced properly.
For more information, see Section 1. Why am I using METOPIRONE? in the full CMI.
2. What should I know before I use METOPIRONE?
Do not use if you have ever had an allergic reaction to metyrapone or any of the ingredients listed at the end of the CMI.
Do not use if you suffer from a condition whereby your adrenal glands do not produce enough steroid hormones (cortisol or aldosterone) known as Addison's disease.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use METOPIRONE? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with METOPIRONE and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use METOPIRONE?
- Your doctor will tell you how many capsules to take. This depends on the type of test you are having and your bodyweight.
- Swallow the capsules whole after meals or with milk or yoghurt. Do not chew them.
More instructions can be found in Section 4. How do I use METOPIRONE? in the full CMI.
5. What should I know while using METOPIRONE?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using METOPIRONE? in the full CMI.
6. Are there any side effects?
Less serious side effects may include tiredness, headache, being short of breath when exercising, dizziness and looking pale which may be symptoms of anaemia; bleeding or bruising more easily than normal; high or low blood pressure; feeling or being sick; pain in the stomach area; weight loss, salt craving, muscle weakness, mood changes, low blood sugar or menstrual irregularities which may be symptoms of low levels of steroid hormones; fever, severe chills, sore throat or mouth ulcers which may be symptoms of infection; feeling drowsy or light-headed. Serious side effects may include allergic symptoms (such as a rash, itching or hives on the skin, wheezing or other problems breathing, swollen face, lips, tongue or other parts of the body), shortness of breath or fever which may be a symptom of a serious lung infection. For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
METOPIRONE®
Active ingredient: metyrapone
Consumer Medicine Information (CMI)
This leaflet provides important information about using METOPIRONE. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using METOPIRONE.
Where to find information in this leaflet:
1. Why am I using METOPIRONE?
2. What should I know before I use METOPIRONE?
3. What if I am taking other medicines?
4. How do I use METOPIRONE?
5. What should I know while using METOPIRONE?
6. Are there any side effects?
7. Product details
1. Why am I using METOPIRONE?
METOPIRONE contains the active ingredient metyrapone. METOPIRONE is a diagnostic agent. It acts by reducing the production of the adrenal gland hormones, cortisol and corticosterone (which are also called corticosteroids).
METOPIRONE is used to check if ACTH (adrenocorticotropic hormone), a natural body hormone acting on corticosteroid secretion, is being produced properly.
Changes in the production of ACTH may happen for several reasons, for example, due to changes in the function of the adrenal or pituitary gland, or due to treatment with certain medicines.
2. What should I know before I use METOPIRONE?
Warnings
Do not use METOPIRONE if you:
- suffer from a condition whereby your adrenal glands do not produce enough steroid hormones (cortisol or aldosterone) known as Addison's disease
- are allergic to metyrapone or any of the ingredients listed at the end of this leaflet
Always check the ingredients to make sure you can use this medicine.
Check with your doctor if you:
- take any medicines for any other condition
- have any other medical conditions, such as:
- impaired liver function or cirrhosis of the liver (a chronic liver disorder leading to progressive loss of liver function)
- an underactive thyroid gland (which can cause weight gain, dry brittle hair, cold intolerance, constipation, forgetfulness, or personality changes)
- an underactive pituitary gland (which can cause an imbalance of some hormones)
- high blood pressure
- Cushing's syndrome (a syndrome that can occur if your body has too much cortisol for a long time)
The results of the test may be affected by some of these medical conditions or you may be at greater risk of developing certain side effects.
It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
There is not enough information to recommend its use during pregnancy. If there is an urgent need for you to have the METOPIRONE test, your doctor can advise you about the risks and benefits.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
You should not breastfeed while you are having the METOPIRONE test. There is not enough information to recommend breast-feeding while you are having the test.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may affect the results of the METOPIRONE test. These include:
- corticosteroid medicines (medicines used to reduce inflammation and affect your immune system)
- any hormone treatments (e.g. tetracosactide, growth hormone, treatments for infertility, the birth control pill and hormone replacement therapy (HRT))
- medicines to treat epilepsy (e.g. phenytoin or barbiturates)
- medicines to treat mental health problems such as anxiety and depression (e.g. amitriptyline, alprazolam and chlorpromazine)
- medicines for your thyroid gland (e.g. levothyroxine, carbimazole, propylthiouracil)
- cyproheptadine, a medicine used for treating allergies
The medicine below could be affected by METOPIRONE:
- paracetamol, a medicine used to treat fever and pain
The medicines below could affect METOPIRONE:
- insulin or oral medicines for the treatment of diabetes
You may need to stop taking some of your medicines during the test. Your doctor will advise you.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect METOPIRONE.
4. How do I use METOPIRONE?
How much to use
Your doctor will decide how many METOPIRONE capsules you need depending on the type of test you are having and your body weight.
How to take METOPIRONE
- Swallow the capsules whole after meals or with milk or yoghurt.
- Do not chew the capsules.
How the test is done
There are two types of METOPIRONE tests. Your doctor will advise you which one you will have.
- Short single dose test:
- This test can either be done at home or overnight in hospital.
- At around midnight you will the number of METOPIRONE capsules your doctor tells you to. This may be up to maximum 3 grams (12 capsules).
- Eight hours later, a blood sample will be taken.
- After the test you may also have to take a dose of 50 mg cortisone to prevent any unwanted effects on your adrenal glands.
- Multiple dose test:
- This test is always done in hospital.
- First, your urine is collected for 24 hours.
- Then you will take 500 to 750 mg (2 to 3 capsules) of METOPIRONE every 4 hours for the next 24 hours.
- Your urine will be collected again for the following 24 hours and the results of the urine tests will be compared.
If you use too much METOPIRONE
If you think that have used too much METOPIRONE, you may need urgent medical attention.
You should immediately:
- contact your doctor, or
- phone the Poisons Information Centre
(by calling 13 11 26), or - go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using METOPIRONE?
Things you should do
- take any supplementary hormone medicine that your doctor prescribes. METOPIRONE may temporarily lower the amount of hormones produced by your adrenal gland and the medicine will help correct this.
- Remind any doctor, dentist or pharmacist you visit that you have had or are about to have METOPIRONE.
Call your doctor straight away if you:
- Develop shortness of breath and fever over hours or days. You may be developing a serious lung infection.
- Have skin rashes and breathing difficulties that may be an allergic reaction.
Things you should not do
- Do not take more capsules than you are meant to.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how METOPIRONE affects you.
METOPIRONE may cause dizziness and drowsiness in some people.
Your child should not ride bicycles, climb trees or play on playground equipment while taking METOPIRONE.
Looking after your medicine
- Store the capsules in their original container in a place where the temperature stays below 25°C.
- Protect from moisture.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
When to discard your medicine
If it is damaged or the expiry date has passed.
Getting rid of any unwanted medicine
Take it to any pharmacy for safe disposal.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Blood
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Lungs
| Call your doctor straight away or go straight to the Emergency Department at your nearest hospital. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What METOPIRONE contains
| Active ingredient (main ingredient) | metyrapone |
| Other ingredients (inactive ingredients) |
|
| Potential allergens | This medicine contains hydroxybenzoates |
Do not take this medicine if you are allergic to any of these ingredients.
What METOPIRONE looks like
METOPIRONE capsules are white to yellowish white oblong soft gelatin capsules marked "HRA" on one side in red ink.
Each capsule contains 250 mg metyrapone.
Each HDPE bottle contains 50 capsules.
ARTG number: AUST R 11043
Who distributes METOPIRONE
Chiesi Australia Pty Ltd
Level 7, Suite 1, 500 Bourke Street,
Melbourne, VIC 3000
Email: medinfo.au@chiesi.com
Website: www.chiesi.com.au
This leaflet was prepared in December 2025
®= Registered Trademark
Brand Information
| Brand name | Metopirone |
| Active ingredient | Metyrapone |
| Schedule | S4 |
MIMS Revision Date: 01 August 2023
1 Name of Medicine
Metyrapone.
2 Qualitative and Quantitative Composition
Metopirone is supplied as soft gelatin capsules each containing 250 mg of metyrapone.
Excipients with known effects. Sodium ethyl hydroxybenzoate, sodium propyl hydroxybenzoate.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Metopirone capsules 250 mg are white to yellowish white oblong soft gelatin capsules marked HRA on one side in red ink.
4 Clinical Particulars
4.1 Therapeutic Indications
Diagnostic use. For the diagnosis of latent ACTH deficiency such as in cases of known pituitary dysfunction or of a suspected pituitary tumour, as well as before and after surgical intervention in the region of the pituitary; and, to assess the degree of ACTH suppression during or after glucocorticoid therapy.
For the differential diagnosis of states of adrenocortical hyperfunction in Cushing's syndrome.
4.2 Dose and Method of Administration
Dose as a diagnostic agent. i) Short single dose test (which can be carried out in ambulant patients) for the diagnosis of latent ACTH deficiency. Adults. In the short single dose test, 11-desoxycortisol (compound S) and/or ACTH are determined in the plasma following a single dose of Metopirone. At around midnight, the patient is given 30 mg/kg (maximum 3 g metyrapone).
The blood sample for the assay is taken early in the morning (7:30 - 8:00 hours). The plasma should be frozen as soon as possible. The patient is then given a prophylactic dose of 50 mg cortisone acetate.
Paediatric population. The same dose as in adults is recommended in children.
Assessment. The accepted normal values employed will depend on the method used for assaying ACTH and 11-desoxycortisol and may vary in different laboratories. A rise in plasma ACTH to at least 44 picomol/L (200 nanogram/L), or in 11-desoxycortisol to over 0.2 micromol/L (70 microgram/L), usually indicates a normal ACTH reserve.
Patients in whom adrenocortical insufficiency is suspected, and who cannot be adequately supervised at home, should be hospitalised for the night as a precautionary measure.
ii) Multiple dose test (which can only be carried out in hospital) for the diagnosis of latent ACTH deficiency and the differential diagnosis of states of adrenocortical hyperfunction in Cushing's syndrome. Adults. The patient must be hospitalised. The urinary excretion of steroids is measured. First, baseline values are determined for the 24 hours preceding the test. Then, on the second day, 500 to 750 mg metyrapone is administered every 4 hours for 24 hours, to a total of 3.0 to 4.5 g.
The effect is evaluated in two consecutive 24-hour urinary samples. The effect on the urinary steroid values can be expected to reach its maximum within this 24-hour period.
Paediatric population. The paediatric dosage recommendation is based on limited data. In children the dosage should be 15 mg/kg body weight, with a minimum dose of 250 mg every 4 hours for 6 doses.
Assessment. ACTH deficiency. When the anterior pituitary is functioning normally Metopirone causes a pronounced increase (to double or more) in the urinary excretion of 11-desoxycortisol and other 11-desoxycorticosteroids. The absence of such an increase indicates secondary adrenocortical insufficiency.
Cushing's syndrome. If the urinary excretion of 11-desoxycorticosteroids increases in response to Metopirone, this indicates that excessive production of ACTH has led to adrenocortical hyperplasia (Cushing's disease). Such an increase can be taken as a sign that there is no adrenocortical tumour producing cortisol autonomously.
Method of administration. Oral administration. It is recommended that the capsules be taken together with yoghurt or milk or after meals to minimise nausea and vomiting which can lead to impaired absorption.
4.3 Contraindications
Adrenocortical insufficiency.
Hypersensitivity to metyrapone or to any of the excipients listed in Section 6.1 List of Excipients.
4.4 Special Warnings and Precautions for Use
The metyrapone multiple dose diagnostic test should be restricted to the hospital setting.
Identified precautions. Patients with reduced adrenal secretory capacity and serious hypopituitarism. The ability of the adrenal cortex to respond to exogenous ACTH should be demonstrated before Metopirone is employed as a test, because Metopirone may induce acute adrenal insufficiency in patients with reduced adrenal secretory capacity as well as in patients with global pituitary insufficiency. The test should be performed in hospital with close monitoring in case of suspected adrenocortical insufficiency.
Patients taking drugs affecting the hypothalamo-pituitary adrenal axis. Before the Metopirone test is carried out, drugs affecting pituitary or adrenocortical function should be discontinued (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
In cases where adrenocortical or anterior pituitary function is more severely impaired, Metopirone may provoke transient adrenocortical insufficiency. This can be rapidly overcome by administering a corticosteroid.
Patients with hypothyroidism. In cases of thyroid hypofunction, the urinary steroid excretion may rise very slowly or not at all, in response to Metopirone.
Opportunistic infections. Patients with severe Cushing's syndrome are at risk for opportunistic infections such as Pneumocystis jirovecii pneumonia during Metopirone treatment. Generally, infection must be anticipated in such patients and careful management is warranted. Initiation of an appropriate prophylactic treatment may be considered.
Hypertension. Long-term treatment with Metopirone can cause hypertension or worsen existing hypertension due to excessive secretion of desoxycorticosterone.
Excipients. The presence of the excipients sodium ethyl parahydroxybenzoate and sodium propyl parahydroxybenzoate can cause allergic reactions, which might be delayed. This medicine contains less than 1 mmol sodium (23 mg) per capsule. It is essentially 'sodium free'.
Use in hepatic impairment. Patients with liver cirrhosis often show a delayed response to Metopirone, because the liver damage results in prolonging the plasma elimination half-life of cortisol.
Use in the elderly. Clinical studies of Metopirone did not include sufficient numbers of subjects aged 65 years and over to determine whether they respond differently from younger subjects.
Clinical evidence would indicate that no special dosage regimen is required. In general, dose selection for an elderly patient should be cautious, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Paediatric use. See Section 4.2 Dose and Method of Administration.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
The interaction potential of metyrapone is partly unknown and therefore caution is advised when initiating and discontinuing treatment with other medicinal products. If changes to the effect and/or safety profile of metyrapone or the concomitant drug are seen, suitable action should be taken.
Observed interactions. Anticonvulsants (e.g. phenytoin, barbiturates), psychoactive drugs (e.g. amitriptyline, chlorpromazine, and alprazolam), hormone preparations that affect the hypothalamo-pituitary axis, corticosteroids, cyproheptadine and antithyroid agents may exert an influence on the results of the Metopirone test.
If these drugs cannot be withdrawn, the necessity of carrying out the Metopirone test should be reviewed.
Anticipated interactions. Metopirone may potentiate paracetamol (acetaminophen) toxicity in humans.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. The effect of Metopirone on human fertility has not been investigated in clinical studies. No dedicated animal fertility studies have been conducted with metyrapone. In repeat-dose toxicity studies, metyrapone has been shown to cause adverse effects on spermatogenesis (loss of spermatogonia, spermatocytes and spermatozoa in dogs at 20 mg/kg/day intraperitoneally [IP]) and ovarian follicular development (reduced ovary and uterus weights, underdeveloped uterus in mice at 100 mg/kg/day IP). Metyrapone can decrease reproductive hormones by targeting adrenal androgenesis.
Use in pregnancy. (Category B3)
There are no adequate data on the use of Metopirone in pregnant women.
Metopirone is not recommended during pregnancy when used as a diagnostic test unless the potential benefit outweighs the risks clearly necessary (in this case, blood pressure should be monitored and hypertension managed appropriately to avoid complications such as pre-eclampsia) and in women of childbearing potential not using contraception.
Human data. The Metopirone test was administered to pregnant women in their second and third trimester of pregnancy and evidence was found that the fetal pituitary responded to the enzymatic block. Transplacental passage of Metopirone has been shown in humans. There are a few published reports of low cortisol levels at birth in infants exposed in utero following chronic use of metyrapone in pregnant females.
Animal data. Maternal administration of metyrapone during pregnancy caused reduction in implantations and increased fetal malformation in mice (40 mg/kg/day SC). Embryofetal effects were seen in rats at a cumulative dose of at least 60 mg/animal SC over 1 or 2 days (adrenal hypertrophy, reduced serum corticosterone levels) during the period of organogenesis. Transplacental passage of metyrapone has been shown in rabbits.
Use in lactation. There is insufficient information on the presence of metyrapone in human milk, the effects on the breastfed infant, or the effects on milk production. A risk to newborns/infants cannot be excluded. Breast-feeding should be discontinued during treatment with Metopirone.
4.7 Effects on Ability to Drive and Use Machines
Since Metopirone may cause dizziness and sedation, patients should not drive or operate machinery until these effects have passed.
4.8 Adverse Effects (Undesirable Effects)
Adverse drug reactions are ranked under heading of frequency, the most frequent first, using the following convention: very common (≥ 1/10); common (≥ 1/100, < 1/10); uncommon (≥ 1/1,000, < 1/100); rare (≥ 1/10,000, < 1/1,000); very rare (< 1/10,000), not known (cannot be estimated from the available data). Within each frequency grouping, adverse reactions are ranked in order of decreasing seriousness.
Blood and the lymphatic system disorders. Not known: leukopenia, anaemia, thrombocytopenia.
Endocrine disorders. Rare: adrenal insufficiency.
Nervous system disorders. Common: dizziness, sedation, headache.
Not known: lightheadedness.
Vascular disorders. Common: hypotension.
Not known: hypertension.
Gastrointestinal disorders. Common: nausea, vomiting.
Rare: abdominal pain.
Skin and subcutaneous tissue disorders. Rare: allergic skin reactions.
Reporting of suspected adverse reactions. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Signs and symptoms. The clinical picture of overdosage with Metopirone is characterised by gastrointestinal symptoms and signs of acute adrenocortical insufficiency.
Laboratory findings: hyponatraemia, hypochloraemia, hyperkalaemia.
In patients under treatment with insulin or oral antidiabetics, the signs and symptoms of acute poisoning with Metopirone may be aggravated or modified.
Treatment. There is no specific antidote. Immediate treatment is essential in the management of metyrapone overdose, patients should be referred to hospital urgently for immediate medical attention. Treatment with activated charcoal may be considered if the overdose has been taken within 1 hour.
In addition to general measures, a large dose of hydrocortisone should be administered at once, together with IV saline and glucose. This should be repeated as necessary in accordance with the patient's clinical condition.
For a few days: blood pressure and fluid and electrolyte balance should be monitored.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
Pharmacotherapeutic group: Diagnostic agent, test for pituitary function. ATC code: V04CD01.
5.1 Pharmacodynamic Properties
Mechanism of action. Metopirone inhibits reversibly the biosynthesis of cortisol, corticosterone, and aldosterone in the adrenal cortex by blocking enzymatic 11-beta-hydroxylation in the steroid ring. In the normal person, a compensatory increase in ACTH release follows and the secretion of 11-desoxycortisol, 11-desoxycorticosterone and 17-hydroxycorticoids is markedly accelerated.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. Metyrapone is rapidly absorbed after administration by mouth and is also rapidly eliminated from the plasma. Peak concentrations are usually attained in plasma 1 hour after ingestion of Metopirone.
Distribution. Following a dose of 750 mg, the mean peak concentration is 3.7 microgram/mL and decreases to a mean value of 0.5 microgram/mL 4 hours after ingestion.
Metabolism. The plasma elimination half-life of metyrapone is about 2 hours after oral administration. Metyrapol (reduced metyrapone) is the principal active metabolite. The metyrapone/ metyrapol ratio in the plasma 8 hours after a single oral dose is 1/1.5.
Excretion. Following a total dosage of 4.5 g metyrapone (750 mg every 4 hours), the quantities excreted in the urine 72 hours after the first dose averaged 5.3% of the total dosage in the form of metyrapone (9.2% in free form and 90.8% conjugated with glucuronic acid) and 38.5% in the form of metyrapol (8.1% in free form and 91.9% conjugated with glucuronic acid).
5.3 Preclinical Safety Data
Genotoxicity. No studies for genotoxicity have been performed with Metopirone.
Carcinogenicity. No studies for carcinogenicity have been performed with Metopirone.
Teratogenicity. Animal reproduction studies, adequate to evaluate teratogenicity and postnatal development, have not been conducted.
6 Pharmaceutical Particulars
6.1 List of Excipients
It also contains sodium ethyl hydroxybenzoate, ethyl vanillin, gelatin, glycerol, acetanisole, Macrogol 400, Macrogol 4000, sodium propyl hydroxybenzoate, titanium dioxide, purified water and Edible ink Red (PI 3115) as excipients.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Protect from moisture. Keep out of the reach and sight of children.
6.5 Nature and Contents of Container
Each HDPE bottle with polypropylene child resistant closure with a liner for induction seal contains 50 capsules.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
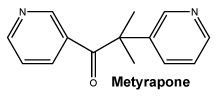
Chemical structure. Chemical name: 2-methyl-1,2-di-3-pyridyl-1-propanone.
Molecular formula: C14H14N2O.
Molecular weight: 226.27 g/mol.
Structural formula:
7 Medicine Schedule (Poisons Standard)
Schedule 4 (S4) Prescription Only Medicine.
Date of First Approval
02 August 1991
Date of Revision
04 July 2023
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.