Metrogyl
Brand Information
| Brand name | Metrogyl |
| Active ingredient | Metronidazole |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Metrogyl.
Summary CMI
METROGYL®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using METROGYL?
METROGYL contains the active ingredient metronidazole. METROGYL is used to treat certain infections caused by bacteria and other organisms in different parts of the body. It is also used to prevent or treat certain infections that may occur during surgery.
For more information, see Section 1. Why am I using METROGYL? in the full CMI.
2. What should I know before I use METROGYL?
Do not use if you have ever had an allergic reaction to METROGYL or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use METROGYL? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with METROGYL and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use METROGYL?
Follow the instructions provided and use METROGYL until your doctor tells you to stop.
More instructions can be found in Section 4. How do I use METROGYL? in the full CMI.
5. What should I know while using METROGYL?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using METROGYL? in the full CMI.
6. Are there any side effects?
Serious side effects: Allergic reaction: Some symptoms of an allergic reaction include skin rash, itching, shortness of breath or swelling of the face, lips or tongue, which may cause difficulty in swallowing or breathing. If you have been on prolonged METROGYL therapy, you may experience unusual numbness of the feet or hands. Posterior reversible encephalopathy syndrome (PRES) - A brain condition with symptoms such as headache, confusion, visual disturbance and seizures. Drug Reaction with Eosinophilia and Systemic Symptoms (DRESS) - A serious condition with symptoms such as flu-like symptoms, rash with a fever, enlarged lymph nodes, increase in a type of white blood cell (eosinophilia) or increased levels of liver enzymes.
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
METROGYL®
Active ingredient(s): metronidazole
Consumer Medicine Information (CMI)
This leaflet provides important information about using METROGYL. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using METROGYL.
Where to find information in this leaflet:
1. Why am I using METROGYL?
2. What should I know before I use METROGYL?
3. What if I am taking other medicines?
4. How do I use METROGYL?
5. What should I know while using METROGYL?
6. Are there any side effects?
7. Product details
1. Why am I using METROGYL?
METROGYL contains the active ingredient metronidazole. METROGYL is an antibiotic that belongs to a group of medicines called nitroimidazoles.
METROGYL is used to treat certain infections caused by bacteria and other organisms in different parts of the body.
It is also used to prevent or treat certain infections that may occur during surgery.
This medicine works by killing or stopping the growth of bacteria and other organisms causing these infections.
Your doctor may have prescribed METROGYL for another reason.
Ask your doctor if you have any questions about why METROGYL has been prescribed for you.
This medicine is available only with a doctor's prescription.
There is no evidence that this medicine is addictive.
2. What should I know before I use METROGYL?
Warnings
Do not use METROGYL if:
- you are allergic to metronidazole, or any of the ingredients listed at the end of this leaflet.
Always check the ingredients to make sure you can use this medicine. - you have evidence of, or have a history of, a blood disorder.
- you have, or have ever had, a disease of the brain, spinal cord or nerves.
- you have Cockayne syndrome or being treated in combination with other antibiotic drugs.
- the expiry date on the pack has passed. If you use this medicine after the expiry date has passed it may have no effect at all, or worse, an entirely different effect.
- the packaging is torn or shows signs of tampering.
Check with your doctor if you:
- have any allergies to any other medicines or any other substances such as foods, preservatives or dyes.
- have or have ever had any health problems/ medical conditions including:
- a blood disorder
- disease of the brain, spinal cord or nerves
- liver or kidney disease
- an inflammatory disease of the small intestine (e.g., Crohn's disease)
- Cockayne syndrome - drink alcohol. Do not drink alcohol during (and for 24 hours after stopping) treatment with METROGYL.
- have experienced sensitivity to metronidazole, or any of the other ingredients in this medicine.
Tell your doctor immediately and stop taking metronidazole if you develop:
- stomach pain, anorexia, nausea, vomiting, fever, malaise, fatigue, jaundice, dark urine, putty or mastic coloured stools or itching.
If you have not told your doctor or pharmacist about any of the above, tell them before you start using METROGYL.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with METROGYL and affect how it works. These include:
- any medication that may cause ECG disturbances (certain anti-arrhythmics (medicines for heart rhythm disorders), certain antibiotics, psychotropic medicines)
- warfarin or other medicines used to prevent blood clots
- Antabuse® (disulfiram), a medicine used to treat chronic alcohol dependence
- medications containing alcohol (ethanol), e.g., some cough syrups
- some anticancer drugs, such as carmustine, cyclophosphamide monohydrate, 5-fluorouracil or busulfan
- phenytoin, a drug used to treat convulsions
- phenobarbital (phenobarbitone), a medicine for convulsions or sedation
- cimetidine, a medicine used to treat gastric reflux and ulcers
- lithium, a medicine used to treat manic depressive illness and some other types of depression
- cyclosporin, a medicine used to prevent organ transplant rejection or to treat immune responses
- vecuraonium, a medicine used is used to relax your muscles during an operation as part of the general anesthetic
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect METROGYL.
You may need different amounts of your medicine or you may need to take different medicines. Your doctor will advise you.
Your doctor and pharmacist may have more information on medicines to be careful with or avoid while taking METROGYL.
4. How do I use METROGYL?
Follow all directions given to you by your doctor and pharmacist carefully. They may differ from the information contained in this CMI.
If you do not understand the instructions on the box, ask your doctor or pharmacist for help.
How much to take
- The dose will vary from patient to patient. Your doctor will decide the right dose for you.
- Follow the instructions provided when METROGYL was prescribed, including the number of days it should be taken.
How long to use
- For treating infection, METROGYL tablets are usually taken for 7 days, however, your doctor may decide to reduce or extend your treatment. Your doctor will tell you how much METROGYL to take.
How to take METROGYL
Swallow METROGYL tablets whole with a glass of water, preferably during or after a meal.
Do not chew the tablets.
This may help reduce the possibility of stomach upset.
If you forget to take METROGYL
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to. Otherwise, take it as soon as you remember, then go back to taking it as you would normally.
Do not take a double dose to make up for the dose you missed.
If you have trouble remembering to take METROGYL, ask your pharmacist for some hints.
If you use too much METROGYL
If you think that you have used too much METROGYL, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(Australia telephone 13 11 26) for advice, or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
If you take too many tablets you may experience vomiting and a feeling of disorientation.
5. What should I know while using METROGYL?
Things you should do
Call your doctor straight away if you:
- the symptoms of your infection do not improve or become worse
- you become pregnant
- you are about to start taking any new medicines
Some people being treated with metronidazole can experience mental health problems such as irrational thoughts, hallucinations, feeling confused or feeling depressed, including thoughts of self-harm or suicide.
These symptoms can occur even in people who have never had similar problems before. If you or others around you notice any of these side effects stop taking this medicine and seek medical advice straight away.
If you experience abdominal pain, cramps, diarrhoea with possible bloody stools (which may be signs of inflammatory bowel disease).
If you experience symptoms such as headache, confusion, visual disturbance and seizures (which may be signs of a brain condition namely Posterior Reversible Encephalopathy Syndrome or PRES).
If you get a sore, white mouth or tongue while taking or soon after stopping METROGYL treatment, tell your doctor. Also tell your doctor if you get vaginal itching or discharge.
This may mean you have a fungal/yeast infection called thrush. Sometimes the use of METROGYL allows fungi/yeast to grow and the above symptoms to occur. METROGYL does not work against fungi/yeast.
If you experience flu-like symptoms, a rash with a fever, enlarged lymph nodes and an increase in a type of white blood cell (eosinophilia), stop treatment immediately and contact your doctor or healthcare professional. Other abnormal blood test results may include (but are not limited to) increased levels of liver enzymes.
If you are using METROGYL for 10 days or longer, make sure you have any tests of your blood and nervous system that your doctor may request.
If you need to have a blood test while using METROGYL, tell your doctor as METROGYL may affect the results of some laboratory tests.
Remind any doctor, dentist or pharmacist you visit that you are using METROGYL.
Things you should not do
- Do not give METROGYL to anyone else, even if they have the same condition as you.
- Do not use METROGYL to treat any other medical complaints unless your doctor tells you to.
- Do not stop taking your tablets if you feel better.
- If you do not complete the full course prescribed by your doctor, all of the bacteria/organisms causing your infection may not be killed. These bacteria/organisms may continue to grow and multiply so that your infection may not clear completely or may return.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how METROGYL affects you.
While using METROGYL you may feel dizzy or experience vertigo (spinning sensation), confused, hallucinations (see or hear things that are not there), convulsions (have fits) or temporary eyesight problems (such as blurred or double vision). If this happens, do not drive or use any machinery or tools.
Drinking alcohol
Tell your doctor if you drink alcohol.
Alcohol may make you feel very sick, vomit, have stomach cramps, headaches and flushing.
Do not drink alcohol during (and for 24 hours after stopping) treatment with METROGYL.
Looking after your medicine
- Keep METROGYL tablets in the bottle until it is time to use them. If you take tablets out of the bottle, they may not keep well.
- Keep METROGYL tablets in a cool dry place where the temperature stays below 30°C.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
General
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Stop using METROGYL, and tell your doctor immediately |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What METROGYL contains
| Active ingredient (main ingredient) | Metronidazole 200 mg or 400 mg |
| Other ingredients (inactive ingredients) |
|
| Potential allergens |
|
Do not take this medicine if you are allergic to any of these ingredients.
What METROGYL looks like
METROGYL 200 (AUST R 17654) is a 11 mm, normal convex, white tablet debossed “MZ/200” on one side and “G” on the other side. Available in pack sizes of 21 tablets or 250 tablets (hospital use only)
METROGYL 400 (AUST R 17655) is a 12.5 mm, normal convex, pale yellow tablet debossed “MZ/400” on one side and “G” on the other side. Available in pack size of 21 tablets.
Who distributes METROGYL
Arrotex Pharmaceuticals Pty Ltd
15 – 17 Chapel Street
Cremorne VIC 3121
www.arrotex.com.au
This leaflet was prepared in March 2026.
Brand Information
| Brand name | Metrogyl |
| Active ingredient | Metronidazole |
| Schedule | S4 |
MIMS Revision Date: 01 May 2026
1 Name of Medicine
Metronidazole.
2 Qualitative and Quantitative Composition
Each Metrogyl 200 and Metrogyl 400 contains the active ingredient metronidazole 200 mg and metronidazole 400 mg respectively.
Excipients with known effect. Sulfites, galactose and sugars as lactose.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Metrogyl 200 (metronidazole) 200 mg: 11 mm, normal convex, white tablet debossed "MZ/200" on one side and "G" on the other side.
Metrogyl 400 (metronidazole) 400 mg: 12.5 mm, normal convex, pale yellow tablet debossed "MZ/400" on one side and "G" on the other side.
4 Clinical Particulars
4.1 Therapeutic Indications
Metronidazole is indicated in the oral treatment of:
1. Urogenital trichomoniasis in the female (trichomonal vaginitis) and in the male. The male consort of females suffering from urogenital trichomoniasis should be treated concurrently.
2. Bacterial vaginosis.
3. All forms of amoebiasis (intestinal and extraintestinal disease).
4. Giardiasis.
5. Acute ulcerative gingivitis.
6. Anaerobic infections including septicaemia, bacteraemia, brain abscess, necrotising pneumonia, osteomyelitis, puerperal sepsis, pelvic abscess, pelvic cellulitis and postoperative wound infections, in which the pathogens have been identified as Bacteroides fragilis and other species of Bacteroides, and other species such as Fusobacteria, Eubacteria, Clostridia and anaerobic Streptococci.
Metronidazole may be used prophylactically to prevent infection by anaerobic organisms of the surgical site following appendicectomy, colonic surgery, vaginal hysterectomy, abdominal surgery in the presence of anaerobes in the peritoneal cavity and surgery performed in the presence of anaerobic septicaemia.
4.2 Dose and Method of Administration
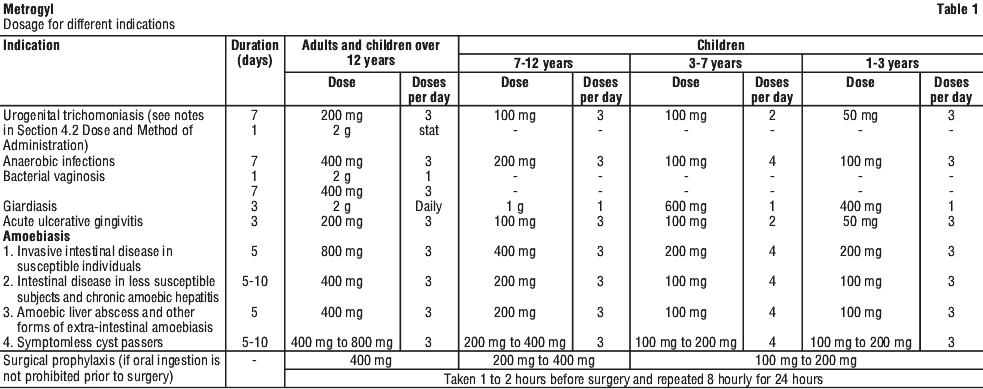
Summarised in Table 1.
A maximum of 4 g should not be exceeded during 24 hour period.
Oral. The tablets should be swallowed, without chewing, with half a glass of water. Treatment for seven days should be satisfactory for most patients but, depending on clinical and bacteriological assessment, the clinician might decide to prolong treatment.
In patients with impaired liver function, dosage should be reduced or dosage intervals increased. Plasma metronidazole levels should be monitored (see Section 4.4 Special Warnings and Precautions for Use).
In elderly patients, the pharmacokinetics of metronidazole may be altered and therefore monitoring of serum levels may be necessary to adjust the metronidazole dosage accordingly. Plasma metronidazole levels should be monitored (see Section 4.4 Special Warnings and Precautions for Use).
If treated during the second or third trimester, the one day course of therapy should not be used as it results in higher serum levels which reach the foetal circulation (see Section 4.6 Fertility, Pregnancy and Lactation).
When repeat courses of the drug are required, it is recommended that an interval of four to six weeks elapse between courses and the presence of the trichomonad be reconfirmed by appropriate laboratory measures. Total and differential leucocyte counts should be made before and after retreatment.
Surgical prophylaxis. Note. Prevention of infection at the surgical site requires that adequate tissue concentration of the drug should have been achieved at the time of surgery. The doses and route of administration should be selected in this case to achieve this objective.
As an oral ingestion is often prohibited 12 hours or longer before surgery, and it may not be practical for a variable period following surgery, tablets are not considered to be an appropriate formulation for prophylactic use. However, if oral intake is not contraindicated and is feasible following surgery, 400 mg may be taken one to two hours before surgery and repeated every eight hours for 24 hours.
4.3 Contraindications
1. Patients with active organic disease of the central nervous system.
2. Patients with evidence of, or history of blood dyscrasias should not receive the drug since upon occasion a mild leucopenia has been observed during its administration. However, no persistent haematological abnormalities have been observed in animals or clinical studies.
3. Hypersensitivity to metronidazole and other imidazoles.
4. Metronidazole is contraindicated in patients with Cockayne syndrome. Severe irreversible hepatotoxicity/acute liver failure with fatal outcomes have been reported after initiation of metronidazole in patients with Cockayne syndrome (see Section 4.8 Adverse Effects (Undesirable Effects)).
4.4 Special Warnings and Precautions for Use
Alcohol. Alcoholic beverages and drugs containing alcohol, should not be consumed by patients being treated with metronidazole, or for at least 24 hours afterwards, as nausea, vomiting, abdominal cramps, headaches, tachycardia, and flushing may occur. There is the possibility of a disulfiram-like (Antabuse) effect reaction.
Candidiasis. Candida overgrowth in the gastrointestinal or genital tract may occur during metronidazole therapy and require treatment with a candidacidal drug.
Posterior reversible encephalopathy syndrome (PRES). Patients treated with metronidazole have been reported to develop posterior reversible encephalopathy syndrome (PRES). If patients taking metronidazole present with symptoms indicating PRES such as headache, altered mental status, seizures, and visual disturbances, a radiological procedure (e.g. MRI) should be performed. If PRES is diagnosed, adequate blood pressure control and immediate discontinuation of metronidazole is advised. Most patients completely recover after appropriate measures are taken.
Gonococcal infection. Underlying gonococcal infection may persist after elimination of Trichomonas vaginalis.
Severe bullous skin reactions. Cases of severe bullous skin reactions such as Stevens Johnson syndrome (SJS), toxic epidermal necrolysis (TEN) or acute generalised exanthematous pustulosis (AGEP) and drug reaction with eosinophilia and systemic symptoms (DRESS) have been reported with metronidazole (see Section 4.8 Adverse Effects (Undesirable Effects)). If symptoms or signs of SJS, TEN, AGEP or DRESS are present, metronidazole treatment must be immediately discontinued.
Long-term therapy. If metronidazole is to be administered for more than 10 days, it is recommended that haematological tests, especially total and differential leucocyte counts, be carried out regularly and that patients be monitored for adverse reactions such as peripheral neuropathy or central neuropathy (such as paresthesia, ataxia, dizziness, vertigo, convulsive seizures). If leucopenia or abnormal neurological signs occur, the drug should be discontinued immediately.
Inflammatory bowel disease (IBD). Use of metronidazole may increase the risk of subsequent inflammatory bowel disease (IBD).
Surgical drainage. Use of metronidazole does not obviate the need for aspiration of pus whenever indicated.
Nervous system. Metronidazole should be used with caution in patients with active or chronic severe peripheral and central nervous system diseases due to the risk of neurological damage.
Patients should be warned about the potential for confusion, dizziness, hallucinations, convulsions or transient visual disorders and advised not to drive or use machinery if these symptoms occur.
Treatment with metronidazole should be discontinued if ataxia or any other symptom of central nervous system (CNS) involvement occurs.
A rare case of reversible but profound neurological deterioration has been reported following a single oral dose of metronidazole; it is therefore advisable that a patient taking metronidazole for the first time is not left unattended for a period of two hours. The appearance of abnormal neurologic signs demands prompt discontinuation of metronidazole therapy and, when severe, immediate medical attention. Activated charcoal may be administered to aid in the removal of unabsorbed drug, if no more than two or three hours have elapsed since administration of the drug.
Suicidal ideation. Cases of suicidal ideation with or without depression have been reported during treatment with metronidazole. Patients should be advised to discontinue treatment and contact their healthcare provider immediately if they experience psychiatric symptoms during treatment.
Use in hepatic impairment. No information available. As metronidazole is partly metabolised in the liver, caution should be exercised in patients with impaired liver function or hepatic encephalopathy.
Metronidazole may interfere with certain chemical analysis of serum aspartate transaminase (AST), alanine transaminase (ALT), lactate dehydrogenase (LDH), triglycerides and hexokinase glucose to give abnormally low values.
Dosage should be reduced or dosage intervals increased (see Section 4.2 Dose and Method of Administration).
Use in renal impairment. In patients on twice weekly haemodialysis, metronidazole and its major active metabolite are rapidly removed during an 8 hour period of dialysis, resulting in plasma concentration falling below the therapeutic range. Hence a further dose of metronidazole would be needed after dialysis to restore an adequate plasma concentration. In patients with renal failure, the half-life of metronidazole is unchanged but those of its major metabolites are prolonged 4-fold or greater. The accumulation of the hydroxy metabolite could be associated with side effects and measurement of its plasma concentrations by high pressure liquid chromatography (HPLC) has been recommended.
General. Treatment with metronidazole should be discontinued should pancreatitis occur once other causes of this disease are excluded.
Where there is clinical evidence of a trichomonal infection in the sexual partner, they should be treated concomitantly to avoid reinfection.
Patients should be warned that metronidazole may darken urine. This is probably due to a metabolite of metronidazole and seems to have no clinical significance.
Susceptibility/ resistance. Development of drug-resistant bacteria. Prescribing metronidazole in the absence of a proven or strongly suspected bacterial infection is unlikely to provide benefit to the patient and risks the development of drug-resistant bacteria.
Use in the elderly. The pharmacokinetics of metronidazole may be altered and therefore monitoring of serum levels may be necessary to adjust the metronidazole dosage accordingly (see Section 4.2 Dose and Method of Administration).
Paediatric use. No data available.
Effects on laboratory tests. Metronidazole may interfere with certain types of blood test determinations in blood (aminotransferase (ALT), aspartate aminotransferase (AST), lactate dehydrogenase (LDH), triglycerides, glucose) which may lead to false negative or an abnormally low result. These analytical determinations are based on a decrease in ultraviolet absorbance, a fact that occurs when nicotinamide adenine dinucleotide hydrogen (NADH) is oxidized to nicotinamide adenine dinucleotide (NAD). The interference is due to the similarity in the absorption peaks of NADH (340 nanometer) and metronidazole (322 nanometer) at pH 7.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Warfarin. Metronidazole enhances the activity of warfarin, therefore if Metrogyl is to be given to patients receiving this or other anticoagulants, the dosages of anticoagulants should be recalibrated. There is an increased haemorrhagic risk caused by decreased hepatic metabolism. Prothrombin times and anticoagulant activity should be monitored.
Alcohol. Alcoholic beverages and drugs containing alcohol should not be consumed during metronidazole therapy and for at least one day afterwards because the possibility of a disulfiram-like (Antabuse effect) reaction (vomiting, tachycardia, and flushing).
Carmustine (BCNU) or cyclophosphamide. Metronidazole should be used with caution in patients receiving these drugs.
Lithium. Plasma levels of lithium may be increased by metronidazole. Lithium retention accompanied by evidence of possible renal damage has been reported in patients treated simultaneously with lithium and metronidazole. Lithium treatment should be tapered or withdrawn before administering metronidazole. Plasma concentration of lithium, creatinine, urine osmolality and electrolytes should be monitored in patients under treatment with lithium while they receive metronidazole.
Disulfiram. Administration of disulfiram and metronidazole has been associated with acute psychoses and confusion in some patients; therefore, these drugs should not be used concomitantly. Metronidazole should not be given to patients who have taken disulfiram within the last two weeks.
Hepatic enzyme inducers. The simultaneous administration of drugs that induce microsomal hepatic enzymes, such as phenytoin or (phenobarbital) phenobarbitone, may accelerate the elimination of Metrogyl, resulting in reduced plasma levels; impaired clearance of phenytoin has also been reported.
In single dose studies, metronidazole injection did not interfere with the biotransformation of diazepam, antipyrine or phenytoin in man. However, patients maintained on phenytoin were found to have toxic blood levels after oral metronidazole administration. Phenytoin concentration returned to therapeutic blood level after discontinuance of metronidazole. The metabolism of metronidazole has been reported to be increased by concurrent administration of phenobarbital or phenytoin.
Hepatic enzyme inhibitors. The simultaneous administration of drugs that decrease microsomal hepatic enzyme activity, such as cimetidine, may prolong the half-life and decrease plasma clearance of metronidazole.
Ciclosporin. There is a risk of ciclosporin serum levels increasing when it is used in combination with metronidazole. Serum ciclosporin and serum creatinine should be closely monitored when coadministration is necessary.
5-Fluorouracil. Metronidazole used in combination with 5-fluorouracil may lead to reduced clearance of 5-fluorouracil, resulting in increased toxicity.
Busulfan. Plasma levels of busulfan may be increased by metronidazole, which may lead to severe busulfan toxicity.
QT interval. QT prolongation has been reported, particularly when metronidazole was administered with drugs with the potential for prolonging the QT interval.
Vecuronium. A slight potentiation of the neuromuscular blocking activity of vecuronium has been reported in patients administered metronidazole.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category B2)
The safety of use of metronidazole in pregnancy has not been established and its use should be avoided; if essential, short high-dose regimes should not be used. Metronidazole should not be given in the first trimester of pregnancy as it crosses the placenta and enters fetal circulation rapidly. As its effects on human fetal organogenesis are not known, its use in pregnancy should be carefully evaluated. Although it has not been shown to be teratogenic in either human or animal studies, such a possibility cannot be excluded.
Use of metronidazole for trichomoniasis in the second and third trimesters should be restricted to those in whom local palliative treatment has been inadequate to control symptoms.
If a patient is treated during the 2nd or 3rd trimesters of pregnancy for urogenital trichomoniasis, the 2 g stat dose therapy should not be used as it results in higher serum levels which reach the fetal circulation.
Use in lactation. Metronidazole is secreted in breast milk (see Section 5.2 Pharmacokinetic Properties). In view of its tumorigenic and mutagenic potential, breastfeeding is not recommended (see Section 5.3 Preclinical Safety Data).
4.7 Effects on Ability to Drive and Use Machines
Patients should be warned about the potential for confusion, dizziness, drowsiness, vertigo, hallucinations or convulsions or transient visual disorders and advised not to drive or use machinery if these symptoms occur. See Section 4.4 Special Warnings and Precautions for Use; Section 4.8 Adverse Effects (Undesirable Effects).
4.8 Adverse Effects (Undesirable Effects)
Frequency, type and severity of adverse reactions in children are the same as in adults.
Gastrointestinal. Metronidazole when given orally is well tolerated. Common adverse reactions refer to the gastrointestinal tract, particularly nausea, sometimes accompanied by headache, anorexia and occasionally vomiting, diarrhoea, epigastric pain or distress, dyspepsia abdominal cramping and constipation. Oral mucositis, dry mouth and taste disorders have also been reported. A metallic, sharp, unpleasant taste is not unusual. Cases of pancreatitis which abated on withdrawal of the drug, have been reported. Patients with Crohn's disease are known to have an increased incidence of gastrointestinal and certain extraintestinal cancers. If patients receiving metronidazole drink alcoholic beverages, they may experience abdominal distress, nausea, vomiting, flushing or headache. A modification of the taste of alcoholic beverages has also been reported.
Furry tongue, tongue discolouration, coated tongue, glossitis and stomatitis have occurred; these may be associated with a sudden overgrowth of Candida which may occur during effective therapy.
Body as a whole. Hypersensitivity reactions include rash, pruritus, flushing, urticaria, fever, angioedema and anaphylactic shocks. Nasal congestion and dryness of the mouth have been reported. Mild erythematous eruptions have been experienced as have fleeting joint pains sometimes resembling serum sickness. Pustular eruptions and acute generalised exanthematous pustulosis have been reported. Fixed drug eruption has been reported. Stevens-Johnson syndrome and toxic epidermal necrolysis have also been reported. Drug reactions with eosinophilia and systemic symptoms (DRESS) have been reported.
Liver. Increase in liver enzymes (AST, ALT, alkaline phosphatase), cholestatic or mixed hepatitis and hepatocellular liver injury, sometimes with jaundice, have been reported.
Cases of liver failure requiring liver transplant have been reported in patients treated with metronidazole in combination with other antibiotic drugs; all spiramycin except one case of tetracycline.
Cases of severe irreversible hepatotoxicity/acute liver failure, including cases with fatal outcomes with very rapid onset after initiation of systemic use of metronidazole, have been reported in patients with Cockayne syndrome (latency from drug start to signs of liver failure as short as 2 days) (see Section 4.3 Contraindications).
Haematology. A moderate leucopenia may be observed occasionally. If this occurs, the total leucocyte count may be expected to return to normal after the course of medication is completed. One case of bone marrow depression has been reported. If profound bone marrow suppression occurs, use of metronidazole should be ceased and appropriate supportive therapy instituted. Cases of agranulocytosis, neutropenia or thrombocytopenia have been reported.
Psychiatric/central nervous system disorders. Dizziness, drowsiness, vertigo, incoordination, headache and convulsive seizures have been reported. Psychotic disorders such as confusion and hallucinations have been reported. Depression, depressed mood, insomnia, irritability, weakness have been experienced, as has peripheral neuropathy, characterised mainly by numbness or paraesthesia of an extremity. There have been reports of encephalopathy (e.g. confusion, vertigo) and subacute cerebellar syndrome (e.g. ataxia, dysarthria, gait impairment, nystagmus and tremor), which may resolve with the discontinuation of the drug. Since persistent peripheral neuropathy has been reported in some patients receiving prolonged administration of metronidazole, such subjects should be specifically warned about these reports and should be told to stop the drug and report immediately if any neurological symptoms occur. Aseptic meningitis has been reported.
Frequency not known: vertigo, posterior reversible encephalopathy syndrome (PRES).
Reproductive system and breast disorders. A single case of gynecomastia has been reported which resolved on discontinuing metronidazole administration.
Eye disorders. Optic neuropathy/ neuritis and transient vision disorders such as diplopia, myopia, blurred vision, decreased visual acuity and changes in colour vision have been reported.
Ear and labyrinth disorders. Impaired hearing/ hearing loss (including sensorineural) and tinnitus have been reported.
Genitourinary tract. Proliferation of Candida also may occur in the vagina. Dryness and burning of the vagina or vulva, pruritus, dysuria, cystitis and a sense of pelvic pressure have been reported. Very rarely dyspareunia, fever, polyuria, incontinence, decrease of libido, proctitis and pyuria have occurred in patients receiving the drug.
Instances of a darkened urine have been reported and this manifestation has been the subject of special investigation. Although the pigment which is probably responsible for this phenomenon has not been positively identified, it is almost certainly a metabolite of metronidazole. It seems certain that it is of no clinical significance and may be encountered only when metronidazole is administered in higher than recommended doses.
Cardiovascular. Flattening of the T wave may be seen in ECG tracings.
Frequency not known: QT prolongation has been reported, particularly when metronidazole was administered with drugs with the potential for prolonging the QT interval.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Symptoms. Overdosage with metronidazole appears to be associated with very few abnormal signs or symptoms.
Disorientation, ataxia and vomiting may occur, especially after ingestion of large amounts. In case of suspected massive overdosages, a symptomatic and supportive treatment should be instituted.
Single oral doses of metronidazole, up to 12 g, have been reported in suicide attempts and accidental overdoses.
Treatment. There is no specific antidote for metronidazole overdosage. In cases of suspected overdosage, a symptomatic and supportive treatment should be instituted.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. No data available.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Pharmacokinetic. Metronidazole is readily absorbed, peak serum concentration is reached approximately 1 to 2 hours after an oral dose. Food does not significantly affect absorption and the bioavailability of the dose approaches 100% when compared with intravenous administration. Traces of metronidazole are detectable after 24 hours. The biological half-life of oral metronidazole has been determined as 6 to 7 and 7.3 hours respectively.
Metronidazole is widely distributed in body tissues and fluids. It crosses the blood brain barrier and the placenta. The concentration in breast milk of nursing mothers is similar to those in serum. The serum half-life of unchanged metronidazole is about 8 to 10 hours. Metronidazole is excreted in the urine as unchanged drug and its metabolites including acid oxidation products and glucuronides. Metronidazole is not protein bound to any significant degree. Most of the dose is excreted in the urine as metronidazole and its metabolites, including acid oxidation products and glucuronides.
Microbiology. Metronidazole is active against a wide range of pathogenic microorganisms, notably Trichomonas vaginalis and other trichomonads, Entamoeba histolytica, Giardia lamblia, Balantidium coli and the causative organisms of acute ulcerative gingivitis. Metronidazole also displays antibacterial activity in vitro against several species of anaerobic bacteria including Bacteroides fragilis and other species of Bacteroides, and other species such as Fusobacteria, Eubacteria, Clostridia and anaerobic Streptococci. The minimum inhibitory concentration (MIC) for most susceptible anaerobes is < 6.2 microgram/mL.
Metronidazole is inactive against aerobic and facultative anaerobic bacteria.
5.3 Preclinical Safety Data
Genotoxicity. In studies on the mutagenic potential of metronidazole, the Ames test was positive, while several nonbacterial tests in animals were negative. In patients with Crohn's disease, metronidazole increased the chromosome abnormalities in circulating lymphocytes. The use of metronidazole for longer treatment than usually required should be carefully weighed (see Section 4.4 Special Warnings and Precautions for Use) and the benefit/risk ratio should therefore be carefully assessed in each case particularly in relation to the severity of the disease and the age of the patient.
Carcinogenicity. Metronidazole has been shown to be tumorigenic and carcinogenic in rodents.
6 Pharmaceutical Particulars
6.1 List of Excipients
Metrogyl 200 and Metrogyl 400 contain the following excipients: lactose monohydrate, disodium edetate, ethylcellulose, sodium starch glycollate, colloidal anhydrous silica, guar gum, magnesium stearate, quinoline yellow aluminium lake (Metrogyl 400 only).
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C. Protect from light.
6.5 Nature and Contents of Container
Metrogyl 200. Container type: HDPE bottle.
Pack sizes: 21 or 250 tablets.
Metrogyl 400. Container type: HDPE bottle.
Pack sizes: 21 tablets.
Some pack sizes may not be marketed.
Australian register of therapeutic goods (ARTG). AUST R 17654 - Metrogyl 200 metronidazole 200 mg tablet bottle.
AUST R 17655 - Metrogyl 400 metronidazole 400 mg tablet bottle.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking it to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure. Structural formula:
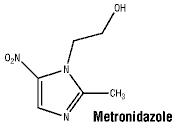
CAS number. 443-48-1.
Metronidazole crystals are white to brownish in colour.
Metronidazole has a melting point of 159-162°C. A saturated solution of aqueous metronidazole has a pH of between 6 and 7.5. Solubility (g/100 mL) of metronidazole at 20°C: 1 in water; 0.5 in ethanol; 0.4 in chloroform; slightly soluble in ether; soluble in dilute acids.
7 Medicine Schedule (Poisons Standard)
S4 (Prescription Only Medicine).
Date of First Approval
20 September 1991
Date of Revision
06 March 2026
Summary Table of Changes
Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.