Miacalcic
Brand Information
| Brand name | Miacalcic |
| Active ingredient | Calcitonin salmon (salcatonin) |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Miacalcic.
Summary CMI
MIACALCIC®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using MIACALCIC?
MIACALCIC contains the active ingredient calcitonin salmon. MIACALCIC is used to treat Paget's disease (a bone disorder) when other treatments have not worked or can't be used. It is also used to help to reduce the level of calcium in the blood when it becomes too high (hypercalcaemia). For more information, see Section 1. Why am I using MIACALCIC? in the full CMI.
2. What should I know before I use MIACALCIC?
Do not use if you have ever had an allergic reaction to MIACALCIC or any of the ingredients listed at the end of the CMI.
Do not use MIACALCIC if you are pregnant or breastfeeding.
Talk to your doctor if you have any other medical conditions or take any other medicines.
For more information, see Section 2. What should I know before I use MIACALCIC? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with MIACALCIC and affect how it works. A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use MIACALCIC?
- Your doctor will decide the dose and provide you with instructions on how MIACALCIC should be used.
- For Paget's disease the usual dose is 80 to 100 I.U. (International Units) of MIACALCIC each day or every second day, given by intramuscular, or subcutaneous injection which you may give yourself.
- For hypercalcaemia the usual dose is 5 to 10 I.U. of MIACALCIC per kg of body weight per day, given intravenously or by intramuscular injection by the doctor or nurse, or subcutaneous injection which you may give yourself.
More instructions can be found in Section 4. How do I use MIACALCIC? in the full CMI.
5. What should I know while using MIACALCIC?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using MIACALCIC? in the full CMI.
6. Are there any side effects?
Less serious side effects include feeling sick, diarrhoea, stomach pain, dizziness, headache, changes in taste, feeling tired, pain in your joints. The serious side effects that you need urgent attention for include serious reaction at the injection site and sudden allergic reaction (skin rash or hives, problems breathing, swollen face and/or body, fast heartbeat, fainting).
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
MIACALCIC®
Active ingredient: calcitonin salmon
Consumer Medicine Information (CMI)
This leaflet provides important information about using MIACALCIC. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using MIACALCIC.
Where to find information in this leaflet:
1. Why am I using MIACALCIC?
2. What should I know before I use MIACALCIC?
3. What if I am taking other medicines?
4. How do I use MIACALCIC?
5. What should I know while using MIACALCIC?
6. Are there any side effects?
7. Product details
1. Why am I using MIACALCIC?
MIACALCIC contains the active ingredient calcitonin salmon. It is similar to calcitonin, a hormone produced by your body.
MIACALCIC is used to treat Paget's disease when other treatments have not worked or can't be used. Paget's disease is a long-term disorder of the bones. MIACALCIC protects the bones from being weakened and may also relieve bone pain.
MIACALCIC is also used to treat hypercalcaemia (high calcium level in the blood). It helps to reduce the level of calcium in the blood when it becomes too high.
2. What should I know before I use MIACALCIC?
Warnings
Do not use MIACALCIC if:
- you are pregnant or breastfeeding
- you are allergic to calcitonin salmon, or any of the ingredients listed at the end of this leaflet. Your doctor may have performed a skin test to check for an allergy before giving you this medicine.
Always check the ingredients to make sure you can use this medicine.
Check with your doctor if you:
- have any other medical conditions, especially kidney problems. Your doctor may want to prescribe a lower dose of MIACALCIC than usual if you have a kidney problem
- take any medicines for any other condition
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Do not use MIACALCIC if you are pregnant.
Talk to your doctor if you are pregnant or intend to become pregnant.
Do not use MIACALCIC if you are breastfeeding.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
There is not enough experience with MIACALCIC in pregnant women or nursing mothers to recommend its use.
Children and adolescents
MIACALCIC is not recommended for children except in special cases, since experience with using MIACALCIC in this age group is limited.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with MIACALCIC and affect how it works. This includes:
- Lithium (a medicine used to mental illness). Your dosage may need to be adjusted.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect MIACALCIC.
4. How do I use MIACALCIC?
How much to use
The dose of MIACALCIC depends on your medical condition.
- For Paget's disease:
- The usual dose is 80 to 100 I.U. (international units) of MIACALCIC each day or every second day.
- The dose is given by intramuscular or subcutaneous injection.
- The dose of MIACALCIC may be adjusted depending on how your body responds to it.
- You may need to have it anywhere from a short time to a few years. - For hypercalcaemia:
- The usual dose is 5 to 10 I.U. of MIACALCIC per kg of body weight per day.
- It can be given intravenously or by intramuscular or subcutaneous injection. - Follow the instructions provided by your doctor and use MIACALCIC until your doctor tells you to stop.
When to use MIACALCIC
For Paget's disease MIACALCIC should be used daily or every second day.
For hypercalcaemia: MIACALCIC should be used every day.
How to use MIACALCIC
MIACALCIC is given as an injection. It can be injected intravenously (into the vein) or intramuscularly (into the muscle of the thigh or buttock). This will be done by a doctor or nurse.
It can also be injected subcutaneously (into the layer of fat just under the skin).
If you will be giving the subcutaneous injections yourself, your doctor or nurse will teach you how to inject yourself properly.
If you forget to use MIACALCIC
If it is more than 4 hours until your next dose is due, have the dose as soon as you remember and then go back to having it as you would normally.
If it is less than 4 hours until your next dose is due, wait and have your dose at the usual time.
Do not have a double dose to make up for the one that you missed.
It won't do any harm if you miss a dose but some of your symptoms may come back temporarily until you get back on schedule.
If you have too much MIACALCIC
If you think that you have had too much MIACALCIC, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using MIACALCIC?
Things you should do
Keep all of your doctor's appointments so that your progress can be checked.
If you have to take MIACALCIC for a long time (for example, to treat Paget's disease), your doctor may want you to have X-rays or blood and urine tests from time to time to make sure MIACALCIC is working and to prevent unwanted side effects from happening.
Using MIACALCIC for a long time might increase the risk of cancer. Your doctor will keep assessing the benefits against the possible risks of using this medicine.
See your doctor if your symptoms reappear.
Sometimes after using MIACALCIC for a while, your symptoms may reappear (this is called a "rebound" effect). If this happens, you may need to stop MIACALCIC for a short time and then start it again. This helps to restore the good effects of the medicine.
Call your doctor straight away if you:
- become pregnant while taking this medicine
- feel sick or have other digestive problems. These may be short-lived problems and your doctor may give you another medicine to help with these or change the dose of MIACALCIC or how often it is given.
- have a serious reaction at the site of the injection.
- have an allergic reaction that affects other parts of your body
Remind any doctor, dentist or pharmacist you visit that you are using MIACALCIC.
Things you should not do
- Do not dilute MIACALCIC when having it for intramuscular or subcutaneous injection.
- When having it as an intravenous infusion (which must be diluted), MIACALCIC must not be diluted in anything other than 0.9% sodium chloride solution.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how MIACALCIC affects you.
MIACALCIC may cause dizziness in some people. Make sure you know how you react to MIACALCIC before you drive a car, operate machinery or do anything else that could be dangerous.
Looking after your medicine
- Keep the ampoules in the carton until it is time to use them.
- Keep your ampoules in the refrigerator (between 2°C and 8°C). Do not freeze.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills
Keep it where young children cannot reach it.
When to discard your medicine
Throw out any MIACALCIC left in the ampoule once the dose has been injected.
MIACALCIC does not contain any preservatives.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, or has been left out of the fridge, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention. See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Blood and urine tests
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Infusion/injection-related reaction
| Call your doctor straight away or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What MIACALCIC contains
| Active ingredient (main ingredient) |
|
| Other ingredients (inactive ingredients) |
|
Do not take this medicine if you are allergic to any of these ingredients.
What MIACALCIC looks like
MIACALCIC is a clear, colourless solution that contains no particles. It comes in 1 mL clear glass ampoules.
Each carton contains 5 ampoules.
There are two strengths:
50 IU of calcitonin salmon in 1 mL
and
100 IU calcitonin salmon in 1 mL
Not all strengths may be marketed.
ARTG numbers:
MIACALCIC 50 IU/1 mL: AUST R 13364
MIACALCIC 100 IU/1 mL: AUST R 13363
Who distributes MIACALCIC
Chiesi Australia Pty Ltd
Level 7, Suite 1, 500 Bourke Street,
Melbourne, VIC 3000
Email: medinfo.au@chiesi.com
W: www.chiesi.com.au
This leaflet was prepared in March 2026
® = Registered Trademark
Brand Information
| Brand name | Miacalcic |
| Active ingredient | Calcitonin salmon (salcatonin) |
| Schedule | S4 |
MIMS Revision Date: 01 May 2026
1 Name of Medicine
Calcitonin salmon.
2 Qualitative and Quantitative Composition
Each 1 mL solution for injection contains 50 or 100 IU calcitonin salmon (present as polyacetate polyhydrate).
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Clear, colourless solution, practically free of visible foreign particles.
4 Clinical Particulars
4.1 Therapeutic Indications
Active Paget's disease in patients who do not respond to alternative treatments or for whom such treatments are not suitable.
Hypercalcaemia.
4.2 Dose and Method of Administration
Note. 1 IU = 1 MRC unit. One unit corresponds to 0.2 microgram of the pure peptide.
Miacalcic may be administered subcutaneously (s.c.), intramuscularly (i.m.) or intravenously (i.v.); local and systemic tolerance is generally good with all 3 routes of administration at recommended dosages.
Due to the association between occurrence of malignancies and long-term calcitonin use (see Section 4.4 Special Warnings and Precautions for Use), the treatment duration in all indications should be limited to the shortest period of time possible and using the lowest effective dose.
Dosage. Hypercalcaemia. Treatment should be limited to the shortest duration possible. The recommended dose is 5-10 IU per kg daily, administered by slow i.v. infusion in 500 mL normal saline over at least six hours, or by slow i.v. injection in 2 to 4 divided doses spread over the day.
Alternatively, the same daily dose may be given by one or more s.c. or i.m. injections. If the volume of Miacalcic for injection exceeds 2 mL, i.m. injection is preferable and multiple sites of injection should be used.
Rehydration should be considered. Emergency treatment is followed by specific treatment of the underlying disease, if required.
Paget's disease. 80-100 IU daily by s.c. or i.m. injection. In some cases the injections may be given only every second day. In particular, after improvement of the objective and subjective symptoms, an injection of 50 IU per day may be sufficient.
The duration of treatment depends on the therapeutic indication and the patient's response. The need for ongoing therapy should be assessed by a health practitioner on a regular basis.
Following cessation of chronic treatment, return of biochemical values to pretreatment levels may take weeks or years.
Method of administration. For intramuscular or subcutaneous use. The solution requires no further dilution.
Patients who are instructed in the self administration of s.c. injections must receive precise directions from the physician or the nurse.
For intravenous infusion. Intravenous infusion is the most effective method of administration and should always be used in emergency or severe cases of hypercalcaemia. Dilute the required amount of calcitonin salmon in 500 mL of 0.9% sodium chloride and infuse over at least 6 hours.
Elderly. Use adult dosage with care. It should be noted that most patients with Paget's disease are elderly.
Hepatic impairment. No information available.
Renal impairment. A smaller dose may be required in renal impairment, as calcitonin salmon is metabolised and excreted predominantly by the kidneys.
4.3 Contraindications
Pregnancy and lactation (see Section 4.6 Fertility, Pregnancy and Lactation).
Hypersensitivity to calcitonin salmon or to any of the excipients in the formulation.
4.4 Special Warnings and Precautions for Use
Hypersensitivity reactions. Being a polypeptide, calcitonin salmon may give rise in rare cases to localised or generalised hypersensitivity reactions. Allergic type reactions, including single cases of anaphylactic shock, have been reported. If such symptoms are observed and can definitely be ascribed to the effect of the drug, treatment should be discontinued.
Malignancies. Meta-analyses of randomised controlled trials conducted in patients with osteoarthritis and osteoporosis have shown that long-term calcitonin salmon use is associated with a small but statistically significant increase in the incidence of malignancies compared to placebo (see Section 4.8 Adverse Effects (Undesirable Effects)). These meta-analyses demonstrated an increase in the absolute rate of occurrence of malignancies for patients treated with calcitonin compared to placebo which varied between 0.7% and 2.36%. Numerical imbalances between calcitonin and placebo were observed after 6 to 12 months of therapy. The increased malignancy risk with the meta-analysis was heavily influenced by a single large 5 year trial, which had an observed risk difference of 3.4%. Imbalances in risk were still observed when analyses excluded basal cell carcinoma. There were several limitations with the meta-analysis data and it is not clear how these limitations affect the results. A mechanism for this observation has not been identified. Patients in these trials were treated with oral or intranasal formulations however it cannot be excluded that an increased risk also applies when calcitonin is administered s.c., i.m. or i.v. The benefits for the individual patient should be carefully evaluated against possible risks (see Section 4.8 Adverse Effects (Undesirable Effects)).
Sensitivity testing. It is advisable to perform a scratch or intradermal skin test to determine sensitivity before administration, as calcitonin is a protein. A 1 in 100 dilution should be used.
Escape phenomena. Escape phenomena seen sometimes in long-term therapy are usually due to a saturation of the binding sites rather than to the development of antibodies. After an interruption of treatment, the therapeutic response to Miacalcic is restored (see Section 4.8 Adverse Effects (Undesirable Effects)).
Use in the elderly. See Section 4.2 Dose and Method of Administration.
Paediatric use. Long-term safety and efficacy have not been established in children, and therefore calcitonin is not recommended for paediatric use except in exceptional circumstances. Calcitonin salmon has been used in familial hyperphosphatasaemia. Unless the physician considers that prolonged treatment is indicated on compelling medical grounds, prolonged treatment should be avoided as calcitonin may interfere with bone growth. In the absence of specific dosage experience in children, the doses relating to bodyweight should be used cautiously. Dosage should be adjusted to desired effect.
Effects on laboratory tests. Calcitonin decreases the rate of bone turnover in conditions with an increased rate of bone resorption and formation, such as active Paget's disease, malignant osteolysis and some forms of osteoporosis characterised by a high bone turnover. This can be measured biochemically as a decrease in urine hydroxyproline excretion and a decrease in serum alkaline phosphatase levels.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Concomitant use of calcitonin salmon and lithium may lead to a reduction in plasma concentrations. The dose of lithium may need to be adjusted.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category B2)
There is no information on the drug's use in pregnancy and therefore the drug should not be used in pregnant women or those likely to become pregnant unless the expected benefits outweigh any potential risk.
Use in lactation. Animal studies suggest that calcitonin might suppress lactation in nursing mothers. Treatment during lactation is not recommended.
4.7 Effects on Ability to Drive and Use Machines
Miacalcic may cause fatigue, dizziness and visual disturbances (see Section 4.8 Adverse Effects (Undesirable Effects)), which may impair the reactions of the patient. Patients should be warned that these effects may occur, in which case they should not drive or use machines.
4.8 Adverse Effects (Undesirable Effects)
For Miacalcic ampoules no recent frequency estimations based on clinical trials are available. Estimations based on the number of post-marketing reports received lead to frequencies lower than those reported in controlled clinical trials with Miacalcic nasal spray. For events attributed to the systemic administration of calcitonin salmon therefore the same (higher) frequency categories as used for the nasal spray was also used for Miacalcic ampoules.
Adverse reactions (Table 1) are listed by MedDRA system organ class. Within each system organ class, the adverse drug reactions are ranked by frequency, with the most frequent reactions first. Within each frequency grouping, adverse drug reactions are presented in order of decreasing seriousness. In addition, the corresponding frequency category for each adverse drug reaction is based on the following convention (CIOMS III): very common (≥ 1/10); common (≥ 1/100 to < 1/10); uncommon (≥ 1/1,000 to < 1/100); rare (≥ 1/10,000 to < 1/1,000); very rare (< 1/10,000).
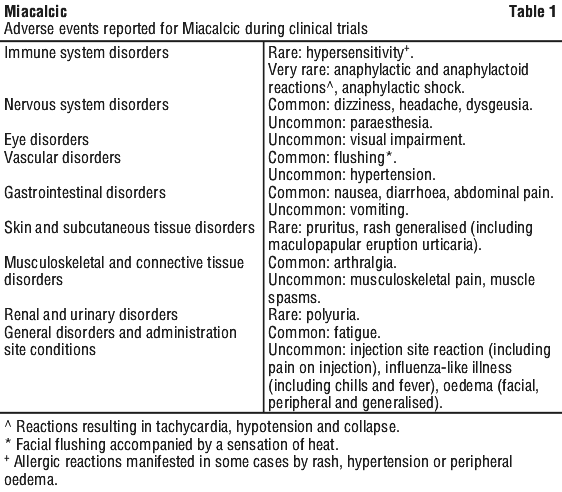
Immunological. Calcitonin salmon binding antibodies may develop in some patients after several months (generally of low titre and more likely to occur with higher doses). However, the development of antibodies does not necessarily cause clinical resistance but may do so in a small number of cases.
Malignancies. Meta-analyses of randomised controlled trials conducted in patients with osteoarthritis and osteoporosis have shown that long-term calcitonin use is associated with a small but statistically significant increase in the incidence of malignancies compared to patients treated with placebo. A mechanism for this observation has not been identified (see Section 4.4 Special Warnings and Precautions for Use).
Post-marketing experience. The following reactions have been identified through post-marketing reporting and literature review. Because this adverse drug reaction has been reported voluntarily from a population of uncertain size, it is not possible to reliably estimate its frequency which is therefore categorised as not known.
Central and peripheral nervous system. Tremor.
Metabolism and nutrition disorders. Hypocalcaemia.
Skin and subcutaneous tissue disorders. Urticaria.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
Nausea, vomiting, flushing and dizziness are known to be dose dependent when Miacalcic is administered parenterally. Nausea and vomiting have occurred following administration of Miacalcic as a parenteral overdose, but severe adverse reactions due to overdosage have so far not been reported.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group, ATC code: Regulator of calcium homeostasis, H05BA01.
The secretion and biosynthesis of calcitonin in both animals and man are regulated by the concentration of calcium in plasma. When the calcium concentration is high the amount of the hormone increases.
The pharmacological activity of salmon calcitonin is the same as that of mammalian calcitonin. In man, data on relative potency are sparse, but calcitonin salmon is thought to be at least 10-40 times as potent by weight as porcine or human calcitonin in producing hypocalcaemia depending on methodology. Presumably due to its greater affinity for receptor binding sites in bone and kidney, and slower rate of metabolism, calcitonin salmon has a longer duration of action.
Calcitonin inhibits osteoclastic bone resorption, altering both the number and/or resorptive activity of osteoclasts. There is suggestive evidence in animals and man that calcitonin may promote bone and collagen formation via an increase in osteoblastic activity. However, the exact role of calcitonin on osteoblastic activity has not been fully established.
Calcitonin decreases the rate of bone turnover in conditions with an increased rate of bone resorption and formation, such as active Paget's disease, malignant osteolysis and some forms of osteoporosis characterised by a high bone turnover. This can be measured biochemically as a decrease in urine hydroxyproline excretion and a decrease in serum alkaline phosphatase levels.
Calcitonin treatment of Paget's disease may relieve bone pain, lower skin temperature over involved bone, decrease excessive cardiac output, stabilise hearing and allow radiographic and histological regression of bone lesions. Clinical experience demonstrates that calcitonin salmon possesses analgesic activity. Investigations have shown binding sites specific to calcitonin salmon in some areas of the central nervous system.
Calcitonin increases the excretion of phosphate, calcium and sodium by decreasing their tubular reabsorption.
Calcitonin is effective in diminishing hypercalcaemia in patients with hyperparathyroidism, vitamin D intoxication and osteolytic bone metastases.
The gastrointestinal effects attributed to calcitonin include the inhibition of gastric acid secretion, stimulation of the intestinal secretion of water and electrolytes, inhibition of pancreatic enzyme secretion and modifications of glucose insulin relationships. Calcitonin probably has no major effects on the intestinal absorption of calcium and does not affect gastrointestinal motility.
Mechanism of action. It has been postulated that cyclic AMP is involved in the secretion of calcitonin, which binds specifically to the membrane receptors of the target tissue and stimulates cyclic AMP accumulation.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. Due to its polypeptide nature, calcitonin salmon is not administered by the oral route as intestinal proteases inactivate the drug. It is administered by s.c., i.m., or i.v. routes. The onset of action is immediate after i.v. administration and occurs in about 15 minutes following i.m. or s.c. administration, with peak plasma levels being attained within one hour.
After s.c. administration, peak plasma levels are reached in about 23 minutes. The bioavailability of calcitonin salmon is about 70% following both i.m. and s.c. administration.
Distribution. Protein binding: 30-40%.
Volume of distribution: 0.15-0.30 litres/kg.
Metabolism. Studies suggest that calcitonin salmon is rapidly metabolised to unidentified and inactive metabolites primarily in the kidneys, but also in the blood and peripheral tissues. The metabolic clearance rate of calcitonin salmon appears to be lower than either porcine or synthetic human calcitonin.
Excretion. Up to 95% of calcitonin salmon and its metabolites are excreted by the kidney, of which less than 2% is unchanged drug.
The absorption half-life is reported to be 8-22 minutes. The elimination half-life is about 60 minutes following i.m. administration and 60-90 minutes following s.c. or i.v. administration. The apparent biological half-life is several hours.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Glacial acetic acid, sodium acetate, sodium chloride, water for injections.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store at 2-8°C. Refrigerate. Do not freeze.
6.5 Nature and Contents of Container
1 mL clear Type 1 glass ampoule with a one-point-cut (OPC) for ease of opening. Packs of 5 ampoules per carton.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure.
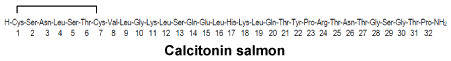
7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Date of First Approval
21 August 1991
Date of Revision
11 March 2026
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.