Minims Phenylephrine
Brand Information
| Brand name | Minims Phenylephrine |
| Active ingredient | Phenylephrine hydrochloride |
| Schedule | S2 | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Minims Phenylephrine.
Summary CMI
Minims® Phenylephrine
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Minims Phenylephrine?
Minims Phenylephrine contains the active ingredient phenylephrine hydrochloride. Minims Phenylephrine is used to dilate (enlarge) the pupil of the eye so that your doctor can examine your eye(s). It is usually used at the beginning of an eye examination.
For more information, see Section 1. Why am I using Minims Phenylephrine? in the full CMI.
2. What should I know before I use Minims Phenylephrine?
Do not use if you have ever had an allergic reaction to phenylephrine or any of the ingredients listed at the end of the CMI. Do not use the 10% strength of Minims Phenylephrine in children or the elderly. Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Minims Phenylephrine? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Minims Phenylephrine and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Minims Phenylephrine?
Your doctor will normally instil the drops into your eyes for you. For adults, the usual dose is one drop instilled into the eye(s) and this may be repeated once if necessary, at least one hour after the first drop. For children and the elderly, the usual dose is one drop of the 2.5% solution to the eye(s). If your doctor instructs you to instil the drops, more instructions can be found in Section 4. How do I use Minims Phenylephrine? in the full CMI.
5. What should I know while using Minims Phenylephrine?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Minims Phenylephrine? in the full CMI.
6. Are there any side effects?
All medicines can have side effects. If they do occur, they are usually minor and temporary. Do not be alarmed by this list. You may not experience any of them. Less serious side effects include eye pain or stinging, blurred vision for a short time, sensitivity to light, eye redness or swelling, itchy or red or watery eyes, small or constricted pupils and pale skin around the eyes. Serious side effects include allergic reaction and effects to the body as a whole.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Minims® Phenylephrine
Active ingredient: Phenylephrine hydrochloride
Consumer Medicine Information (CMI)
This leaflet provides important information about using Minims Phenylephrine. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Minims Phenylephrine.
Where to find information in this leaflet:
1. Why am I using Minims Phenylephrine?
2. What should I know before I use Minims Phenylephrine?
3. What if I am taking other medicines?
4. How do I use Minims Phenylephrine?
5. What should I know while using Minims Phenylephrine?
6. Are there any side effects?
7. Product details
1. Why am I using Minims Phenylephrine?
Minims Phenylephrine contains the active ingredient phenylephrine hydrochloride. Minims Phenylephrine belongs to a class of medicines called sympathomimetic agents.
Minims Phenylephrine is used to dilate (enlarge) the pupil of the eye so that your doctor can examine your eye(s). It is usually used at the beginning of an eye examination.
2. What should I know before I use Minims Phenylephrine?
Warnings
Do not use Minims Phenylephrine if:
- You are allergic to phenylephrine, or any of the ingredients listed at the end of this leaflet. Always check the ingredients to make sure you can use this medicine.
Do not use the 10% strength of Minims Phenylephrine in children or the elderly. Children and the elderly may be more susceptible to the effects of phenylephrine.
Check with your doctor if:
- You have had an allergy to any other medicines, foods, preservatives or dyes.
- You have any other medical conditions, especially:
- Glaucoma (high pressure in the eye)
- Asthma
- Diabetes
- High blood pressure
- Fast heart beat
- Hyperthyroidism (overactive thyroid gland)
- Arteriosclerosis (thickening or hardening of the arteries)
- Heart disease
- Aneurysms (abnormal swelling of a blood vessel)
- Eye redness. - You take any medicines for any other condition.
- You are a senior person.
- Your eyes have a narrow drainage angle.
Check with your doctor if Minims Phenylephrine is to be given to a child.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
This medicine has been developed for use in the eye only. It should not be injected.
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and Minims Phenylephrine may interfere with each other. These medicines may be affected by Minims Phenylephrine or may affect how well it works. These include:
- Medicines to lower blood pressure
- Medicines to treat depression
- Medicines to treat Parkinson's disease
- Medicines to treat heart disease
- Halothane (a medicine that makes you sleep and not feel pain during surgery)
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Minims Phenylephrine.
4. How do I use Minims Phenylephrine?
Your doctor will normally instil the drops into your eyes for you. If your doctor instructs you to instil the drops, please follow the directions below.
How much to use
- Use Minims Phenylephrine only when prescribed by your doctor. Your doctor will tell you how often to use the eye drops and how many drops to use each time.
- Adults: The usual dose is one drop instilled into the eye(s). This may be repeated once if necessary, at least one hour after the first drop.
- Children and Elderly: The usual dose is one drop of the 2.5% solution to the eye(s).
- Sometimes a drop of local anaesthetic may be used before using Minims Phenylephrine Eye Drops. This is to help prevent the stinging effect of phenylephrine eye drops.
How to use Minims Phenylephrine
If you are wearing contact lenses, remove them before putting the drops in your eye.
You may find it easier to put drops in your eye while you are sitting or lying down.
If you are instilling your own drops, then follow these steps to use the eye drops:
- Wash your hands well with soap and water.
- Shake the container.
- Break the seal.
- Hold the container upside down in one hand between your thumb and forefinger.
- Using your other hand, gently pull down your lower eyelid to form a pouch or pocket.
- Tilt your head back and look up.
- Put the tip of the container close to your lower eyelid. Do not let it touch your eye.
- Release the required number of drops into the pouch or pocket formed between your eye and eyelid by gently squeezing the container.
- Close your eye. Do not blink or rub your eye.
- While your eye is closed, place your index finger against the inside corner of your eye and press against your nose for about two minutes. This is very important when administering to children. This will help to stop the medicine from draining through the tear duct to the nose and throat, from where it can be absorbed into other parts of your body.
- Repeat steps 4-10 for the other eye if needed.
- Throw away any eye drops left in the container.
- Wait at least 15 minutes before replacing your contact lenses.
If you use too much Minims Phenylephrine
Accidental swallowing of the solution is unlikely to cause any bad side effects due to the low content of Phenylephrine.
If you think that you or anyone else may have swallowed Minims Phenylephrine or used too much Minims Phenylephrine, you should immediately:
- phone the Poisons Information Centre
(in Australia phone 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using Minims Phenylephrine?
Things you should do
- If you experience any irritation, pain, swelling, excessive tear production or light-sensitivity while using the eye drops, flush the treated eye(s) with lots of water (room temperature) for at least 15 minutes. Stop using the eye drops and contact your doctor if these symptoms persist.
Things you should not do
- Do not use this medicine to treat any other complaints unless your doctor tells you to.
- Do not give this medicine to anyone else, even if you think they have the same condition as you.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Minims Phenylephrine affects you.
Minims Phenylephrine may cause blurred vision for a short time or stinging when it is instilled into the eye. Allow enough time to regain clear vision before driving or using machines.
Looking after your medicine
Keep the unopened eye drops in a refrigerator (2-8°C) where children cannot reach them. Do not freeze the eye drops or expose them to strong light.
When to discard your medicine
Discard each single dose unit and any remaining solution in it immediately after use. Do not save unused contents.
Getting rid of any unwanted medicine
If your doctor tells you to stop using the eye drops or it is out of date or damaged, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date or if the packaging is torn or shows signs of tampering.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Eyes:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Symptoms of an allergic reaction, such as:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects online (in Australia, go to www.tga.gov.au/reporting-problems). By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
Minims Phenylephrine 2.5% eye drops are available from a pharmacy. Minims Phenylephrine 10% eye drops are only available with a doctor's prescription.
What Minims Phenylephrine contains
| Active ingredient (main ingredient) | Phenylephrine hydrochloride (2.5% w/v or 10% w/v) |
| Other ingredients (inactive ingredients) |
|
Do not take this medicine if you are allergic to any of these ingredients.
The eye drops do not contain any preservatives.
What Minims Phenylephrine looks like
Minims Phenylephrine is available in two strengths:
- Minims Phenylephrine 2.5% eye drops (AUST R 46174)
- Minims Phenylephrine 10% eye drops (AUST R 32276).
Minims Phenylephrine comes in single dose units (tubes) containing 0.5mL of a clear, colourless solution. Each tube is wrapped in a polyester/paper blister. The blisters are packed in cartons of 20 units.
Who distributes Minims Phenylephrine
In Australia, Minims Phenylephrine is supplied by:
Bausch & Lomb (Australia) Pty Ltd
Level 2, 12 Help Street
Chatswood, NSW 2067
Phone: 1800 251 150
This leaflet was prepared in July 2025.
Brand Information
| Brand name | Minims Phenylephrine |
| Active ingredient | Phenylephrine hydrochloride |
| Schedule | S2 | S4 |
MIMS Revision Date: 01 March 2021
1 Name of Medicine
Phenylephrine hydrochloride.
2 Qualitative and Quantitative Composition
Minims Phenylephrine Eye Drops containing phenylephrine hydrochloride 2.5% w/v (25 mg/mL) or 10% w/v (100 mg/mL). No preservatives are contained in the formulation.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
A single-use eye drops, solution.
Minims Phenylephrine Eye Drops are clear, colourless sterile solutions, reasonable free from visible particulate matter.
4 Clinical Particulars
4.1 Therapeutic Indications
Phenylephrine is a directly acting sympathomimetic agent used topically in the eye as a mydriatic. Minims Phenylephrine Hydrochloride Eye Drops are indicated to dilate the pupil for diagnostic or therapeutic procedures.
4.2 Dose and Method of Administration
The use of a drop of topical anaesthetic a few minutes before instillation of Minims Phenylephrine Eye Drops is recommended to prevent stinging.
Adults. Instil one drop topically to each eye. If necessary, this dose may be repeated once only, at least one hour after the first drop.
Children and the elderly. Instil one drop of the 2.5% solution topically to the eye. It is not usually necessary to exceed this dose. The use of phenylephrine 10% is contraindicated in children (see Section 4.3 Contraindications; Section 4.4 Special Warnings and Precautions for Use) and the elderly because of the increased risks of systemic toxicity.
Systemic absorption of phenylephrine may be reduced by compressing the lacrimal sac at the medial canthus for a minute during and following the instillation of the drops. (This blocks the passage of the drops via the nasolacrimal duct to the wide absorptive area of the nasal and pharyngeal mucosa. It is especially advisable in children.)
Each Minims Phenylephrine Eye Drops unit should be discarded after a single use.
4.3 Contraindications
Minims Phenylephrine Eye Drops are contraindicated in:
Patients with hypersensitivity to any of the components of the preparation.
Children and the elderly with Minims Phenylephrine Hydrochloride Eye Drops 10% because of the increased risk of systemic toxicity.
4.4 Special Warnings and Precautions for Use
Identified precautions. Minims Phenylephrine Eye Drops are for topical ophthalmic use only. The solution should not be injected.
Caution must be exercised when using Minims Phenylephrine Hydrochloride Eye Drops in the following patient groups:
Patients with cardiac disease, hypertension, aneurysms, long-standing insulin dependent diabetes mellitus and tachycardia.
Patients on monoamine oxidase inhibitors, tricyclic antidepressants and antihypertensive agents (including beta blockers).
Patients with closed angle glaucoma (unless previously treated with iridectomy) and patients with a narrow angle prone to glaucoma precipitated by mydriatics.
The use of phenylephrine 10% solution is contraindicated in children and the elderly because of the increased risks of systemic toxicity (see Paediatric use).
Patients treated with:
Antihypertensive agents. Topical phenylephrine may reverse the action of many antihypertensive agents with possibly fatal consequences.
Monoamine oxidase inhibitors. There is an increased risk of adrenergic reactions when used simultaneously with, or up to three weeks after, the administration of MAOIs.
Tricyclic antidepressants. The pressor response to adrenergic agents and the risk of cardiac arrhythmia may be potentiated in patients receiving tricyclic antidepressants (or within several days of their discontinuation).
Halothane. Because of the increased risk of ventricular fibrillation, phenylephrine should be used with caution during general anaesthesia with anaesthetic agents which sensitise the myocardium to sympathomimetics.
Cardiac glycosides or quinidine. There is an increased risk of arrhythmias if phenylephrine is used in patient taking cardiac glycosides or quinidine.
There have been rare reports associating the use of phenylephrine hydrochloride 2.5% and 10% ophthalmic solutions with the development of serious cardiovascular reactions, including ventricular arrhythmias and myocardial infarctions. These episodes, some ending fatally, have usually occurred in elderly patients with pre-existing cardiovascular diseases.
A significant elevation in blood pressure is rare but has been reported following conjunctival instillation of recommended doses of phenylephrine hydrochloride 10% ophthalmic solutions. Caution should be exercised in children, the elderly, and patients with diabetes, hypertension, hyperthyroidism, generalised arteriosclerosis or cardiovascular disease.
Minims Phenylephrine Eye Drops should be used with caution in the presence of long standing bronchial asthma.
To reduce the risk of precipitating an attack of narrow angle glaucoma the anterior chamber angle should be evaluated before use.
Ocular hyperaemia can increase the absorption of phenylephrine given topically. Corneal clouding may occur if phenylephrine 10% is instilled when the corneal epithelium has been denuded or damaged.
Due to a strong action of the drug on the dilator muscle, older individuals may also develop transient pigment floaters in the aqueous humor 30 to 45 minutes following the administration of the eye drops. The appearances may be confused with anterior uveitis or to a microscopic hyphema.
Systemic absorption of phenylephrine may be reduced by compressing the lacrimal sac at the medial canthus for a minute during and following the instillation of the drops. (This blocks the passage of the drops via the nasolacrimal duct to the wide absorptive area of the nasal and pharyngeal mucosa.)
Use in the elderly. The use of phenylephrine 10% is contraindicated in the elderly because of the increased risks of systemic toxicity. Where phenylephrine eye drops are indicated for use in this group, the 2.5% solution should be used.
Paediatric use. Use of phenylephrine 10% solution is contraindicated in children. Serious systemic adverse reactions have been reported with ophthalmic products containing phenylephrine in children below 12 years of age.
There is inadequate clinical experience in children aged 12 to 18 years. Where phenylephrine eye drops are indicated for use in this group, the 2.5% solution should be used.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Although negligible phenylephrine passes into the bloodstream after ocular instillation, drug interactions are nevertheless possible. The interactions observed with phenylephrine administered by any route should therefore be taken into account (see Section 4.4 Special Warnings and Precautions for Use).
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Studies have not been performed in either animals or humans to evaluate the potential for phenylephrine to impair fertility.
Use in pregnancy. (Category B2)
Safety for use in pregnancy has not been established. Minims Phenylephrine Eye Drops should only be used during pregnancy if it is considered by the physician to be essential.
Use in lactation. Safety for use in lactation has not been established. Minims Phenylephrine Eye Drops should only be used during lactation if it is considered by the physician to be essential.
4.7 Effects on Ability to Drive and Use Machines
Minims Phenylephrine Eye Drops may cause stinging and transient blurring of vision. Patients should be advised not to drive or operate hazardous machinery until vision is clear.
4.8 Adverse Effects (Undesirable Effects)
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
Local. Eye pain and stinging on instillation, temporary blurred vision, photophobia, conjunctival allergy, reactive hyperaemia and transient punctuate keratitis may occur. Other local adverse effects reported include: lacrimation, corneal oedema, pigmented aqueous floaters, rebound miosis, and rebound conjunctival vasoconstriction.
Systemic. Palpitations, tachycardia, extrasystoles, cardiac arrhythmias and hypertension, headache, subarachnoid haemorrhage, reflex bradycardia, blanching of the skin, trembling or tremors, and increased perspiration.
Serious cardiovascular reactions including significant hypertension, aneurisms, coronary artery spasm, ventricular arrhythmias and myocardial infarctions have occurred following topical use of 2.5% and 10% phenylephrine. These sometimes fatal reactions have usually occurred in patients with pre-existing cardiovascular disease.
Undesirable effects. Paediatric population. Phenylephrine 2.5% eye drops. Periorbital pallor in preterm patients: frequency not known (cannot be estimated from the available data).
Phenylephrine 10% eye drops. Respiratory, thoracic and mediastinal disorders. Pulmonary oedema: frequency not known (cannot be estimated from the available data).
4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
Because severe toxic reaction to phenylephrine is of rapid onset and short duration, treatment is primarily supportive. Prompt injection of a rapidly acting alpha-adrenergic blocking agent such as phentolamine (dose 2 to 5 mg IV) has been recommended.
Reversal of mydriasis is possible with 0.1% thymoxamine.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Phenylephrine is a direct acting sympathomimetic agent. It causes mydriasis via the stimulation of alpha-adrenergic receptors. There is almost no cycloplegic effect.
Phenylephrine is an alpha-agonist, with both alpha-1A and alpha-1B effects.
Alpha-adrenergic receptors are unimportant in the aqueous humour outflow response, hence there is no effect on intraocular pressure in open angle glaucoma.
The phenylephrine molecule differs from adrenaline only by the substitution of a hydrogen atom for a hydroxyl group on position 4 of the benzene ring.
Maximal mydriasis occurs in 10-90 minutes with recovery after 5-7 hours.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. Phenylephrine is a weak base at physiological pH. The extent of ocular penetration is determined by the condition of the cornea. A healthy cornea presents a physical barrier, in addition to which, some metabolic activity may occur. Where the corneal epithelium is damaged, the effect of the barrier and the extent of metabolism are reduced, leading to greater absorption.
5.3 Preclinical Safety Data
Genotoxicity. Phenylephrine was negative in tests for bacterial mutagenicity and did not cause chromosomal aberrations in Chinese hamster ovary cells.
Carcinogenicity. There are no studies on the carcinogenicity of phenylephrine by the topical ocular route. No carcinogenic activity was noted in mice or rats receiving oral doses of up to 270 and 50 mg/kg/day, respectively, for 2 years.
6 Pharmaceutical Particulars
6.1 List of Excipients
Sodium metabisulphite, disodium edetate and purified water.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store at 2°C to 8°C. (Refrigerate. Do not freeze.) Do not expose to strong light.
6.5 Nature and Contents of Container
Minims Phenylephrine Eye Drops are supplied in a single use polypropylene tube (unit) overwrapped in a polyester/paper blister. The blisters are packed in cartons of 20 units. Each unit contains approximately 0.5 mL solution.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure.
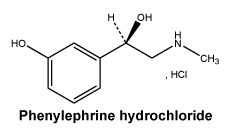
Molecular formula: C9H13NO2.HCl.
Molecular weight: 203.7.
CAS number. 61-76-7.
7 Medicine Schedule (Poisons Standard)
Minims Phenylephrine Eye Drops 2.5% - S2 - Pharmacy Medicine.
Minims Phenylephrine Eye Drops 10% - S4 - Prescription Only Medicine.
Date of First Approval
17 June 2009
Date of Revision
27 January 2021
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.