Minims Prednisolone
Brand Information
| Brand name | Minims Prednisolone |
| Active ingredient | Prednisolone sodium phosphate, Prednisolone sodium phosphate |
| Schedule | S4 |
Consumer medicine information (CMI) leaflet
Please read this leaflet carefully before you start using the Minims Prednisolone
Summary CMI
Minims® Prednisolone
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Minims Prednisolone?
Minims Prednisolone contains the active ingredient prednisolone sodium phosphate. Minims Prednisolone is used to treat inflammation of the eye(s) that is not caused by an infection.
For more information, see Section 1. Why am I using Minims Prednisolone? in the full CMI.
2. What should I know before I use Minims Prednisolone?
Do not use if you have ever had an allergic reaction to prednisolone or any of the ingredients listed at the end of the CMI. Do not use if you have or suspect you have an eye infection or glaucoma (high pressure in the eye). Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Minims Prednisolone? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Minims Prednisolone and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Minims Prednisolone?
- Your doctor will normally instil the drops into your eyes for you.
- The usual dose is one drop instilled into the eye.
If your doctor instructs you to instil the drops, more instructions can be found in Section 4. How do I use Minims Prednisolone? in the full CMI.
5. What should I know while using Minims Prednisolone?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Minims Prednisolone? in the full CMI.
6. Are there any side effects?
All medicines can have side effects. If they do occur, they are usually minor and temporary. Do not be alarmed by this list. You may not experience any of them. Less serious side effects include blurred vision and/or problems seeing clearly, cataract, short-term eye discomfort, slower healing of the eye's surface, dilated pupil and droopy eyelid. Serious side effects that require urgent medical attention include uveitis (a type of eye inflammation), eye infection and allergic reaction.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
1. Why am I using Minims Prednisolone?
Minims Prednisolone contains the active ingredient prednisolone sodium phosphate. Minims Prednisolone belongs to a class of medicines called corticosteroids.
Minims Prednisolone is used to treat inflammation of the eye(s) that is not caused by an infection.
2. What should I know before I use Minims Prednisolone?
Warnings
Do not use Minims Prednisolone if:
- You are allergic to prednisolone, or any of the ingredients listed at the end of this leaflet. Always check the ingredients to make sure you can use this medicine.
- You have or suspect you have an eye infection.
- You have or suspect you have glaucoma (high pressure in the eye).
Do not use Minims Prednisolone in children for continuous, long periods of time.
Check with your doctor if:
- You have had an allergy to any other medicines, foods, preservatives or dyes.
- You have any other medical conditions, especially:
- eye infection
- history of herpes simplex keratitis (an eye infection caused by herpes simplex virus)
- thin cornea (clear front section of the eye) or sclera (the white of the eye that wraps around the eyeball)
- glaucoma (high pressure in the eye). - You take any medicines for any other condition.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
This medicine has been developed for use in the eye only. It should not be swallowed or injected.
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and Minims Prednisolone may interfere with each other. These medicines may be affected by Minims Prednisolone or may affect how well it works. These include:
- Barbiturates (medicines to treat seizure disorders, help sleep and relieve anxiety)
- Sedative hypnotics (medicines to help sleep and relieve anxiety)
- Medicines to treat depression
- Medicines called anticholinesterases (used to treat Alzheimer's disease)
- Medicines to treat viral infections of the eye
- Salicylates e.g. aspirin
- Medicines containing cobicistat (used to treat HIV/AIDS).
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Minims Prednisolone.
4. How do I use Minims Prednisolone?
Your doctor will normally instil the drops into your eyes for you. If your doctor instructs you to instil the drops, please follow the directions below.
How much to use
- Use Minims Prednisolone only when prescribed by your doctor. Your doctor will tell you how often to use the eye drops and how many drops to use each time.
- The usual dose is one drop instilled into the eye as required.
How to use Minims Prednisolone
If you are wearing contact lenses, remove them before putting the drops in your eye.
You may find it easier to put drops in your eye while you are sitting or lying down.
If you are instilling your own drops, then follow these steps to use the eye drops:
- Wash your hands well with soap and water.
- Shake the container.
- Break the seal.
- Hold the container upside down in one hand between your thumb and forefinger.
- Using your other hand, gently pull down your lower eyelid to form a pouch or pocket.
- Tilt your head back and look up.
- Put the tip of the container close to your lower eyelid. Do not let it touch your eye.
- Release the required number of drops into the pouch or pocket formed between your eye and eyelid by gently squeezing the container.
- Close your eye. Do not blink or rub your eye.
- While your eye is closed, place your index finger against the inside corner of your eye and press against your nose for about two minutes. This is very important when administering to children. This will help to stop the medicine from draining through the tear duct to the nose and throat, from where it can be absorbed into other parts of your body.
- Repeat steps 4-10 for the other eye if needed.
- Throw away any eye drops left in the container.
- Wait at least 15 minutes before replacing your contact lenses.
If you use too much Minims Prednisolone
If you think that you have used too much Minims Prednisolone, you may need urgent medical attention.
Accidental swallowing of the solution is unlikely to cause any bad side effects due to the low content of prednisolone.
If you think that you or anyone else may have swallowed Minims Prednisolone or used too much Minims Prednisolone, you should immediately:
- phone the Poisons Information Centre
(in Australia phone 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using Minims Prednisolone?
Things you should do
- If you have any irritation, pain, swelling, excessive tear production or light-sensitivity while using the eye drops, flush the treated eye(s) with lots of water (room temperature) for at least 15 minutes. Stop using the eye drops and contact your doctor if symptoms persist.
- If you experience blurred vision or other visual disturbances, contact your doctor.
- If you become pregnant while using this medicine, tell your doctor immediately.
Things you should not do
- Do not use this medicine to treat any other complaints unless your doctor tells you to.
- Do not give this medicine to anyone else, even if you think they have the same condition as you.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Minims Prednisolone affects you.
Minims Prednisolone generally does not cause any problems with your ability to drive a car or use machines. However, it may cause blurred vision for a short time when it is instilled into the eye. Allow enough time to regain clear vision before driving or using machines.
Looking after your medicine
Keep the unopened eye drops in a refrigerator (2-8°C) where children cannot reach them. Do not freeze the eye drops or expose them to strong light.
When to discard your medicine
Discard each single dose unit and any remaining solution in it immediately after use. Do not save unused contents.
Getting rid of any unwanted medicine
If your doctor tells you to stop using the eye drops or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Eyes:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Symptoms of uveitis (a type of eye inflammation), such as:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Some side effects (for example, an increase in the pressure of the eye) can only be found when your doctor or optometrist does tests from time to time.
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects online (in Australia, go to www.tga.gov.au/reporting-problems). By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Minims Prednisolone contains
| Active ingredient (main ingredient) | Prednisolone sodium phosphate (0.5% w/v) |
| Other ingredients (inactive ingredients) |
|
Do not take this medicine if you are allergic to any of these ingredients.
The eye drops do not contain any preservatives.
What Minims Prednisolone looks like
Minims Prednisolone comes in single dose units (tubes) containing 0.5mL of a clear, colourless solution. Each tube is wrapped in a polyester/paper blister. The blisters are packed in cartons of 20 units (AUST R 32233).
Who distributes Minims Prednisolone
In Australia, Minims Prednisolone is supplied by:
Bausch & Lomb (Australia) Pty Ltd
Level 2, 12 Help Street
Chatswood, NSW 2067
Phone: 1800 251 150
This leaflet was prepared in September 2025.
Brand Information
| Brand name | Minims Prednisolone |
| Active ingredient | Prednisolone sodium phosphate, Prednisolone sodium phosphate |
| Schedule | S4 |
MIMS Revision Date: 01 November 2018
1 Name of Medicine
Prednisolone Sodium Phosphate.
2 Qualitative and Quantitative Composition
Minims Prednisolone Eye Drops contain prednisolone sodium phosphate 0.5% w/v as well as disodium edetate, monobasic sodium phosphate, sodium chloride, sodium hydroxide and purified water. No preservatives are contained in the formulation.
3 Pharmaceutical Form
Minims Prednisolone Eye Drops are clear, colourless sterile eye drops. Each unit contains approximately 0.5 mL solution in a container that has a twist and pull cap. Each unit should be discarded after a single use. The solution has a neutral pH.
4 Clinical Particulars
4.1 Therapeutic Indications
Minims Prednisolone Eye Drops are indicated for non-infected inflammatory conditions of the eye.
4.2 Dose and Method of Administration
Adults and the elderly. One drop applied topically to the eye as required.
Children. At the discretion of the physician.
Systemic absorption may be reduced by compressing the lacrimal sac at the medial canthus for a minute during and following the instillation of the drops. (This blocks the passage of the drops via the naso-lacrimal duct to the wide absorptive area of the nasal and pharyngeal mucosa.)
Each Minims Prednisolone Eye Drops unit should be discarded after a single use.
4.3 Contraindications
Minims Prednisolone Eye Drops are contraindicated in the following patient groups/conditions.
Patients with hypersensitivity to any of the components of the preparation.
Presence of viral, fungal, tuberculous or other bacterial infection.
In children, long-term, continuous topical corticosteroid therapy should be avoided due to possible adrenal suppression.
Glaucoma.
4.4 Special Warnings and Precautions for Use
Minims Prednisolone Eye Drops are for topical ophthalmic use only. The solution should not be injected.
Care should be taken to ensure that the eye is not infected before Minims Prednisolone Eye Drops is used. Corticosteroids may reduce resistance to and aid in the establishment of bacterial, viral, or fungal infections and mask the clinical signs of infection, preventing recognition of ineffectiveness of the antibiotic, or may suppress hypersensitivity reactions to substances in the product. Fungal infection should be suspected in patients with persistent corneal ulceration who have been or are receiving these drugs and corticosteroid therapy should be discontinued if fungal infection occurs. If bacterial infection is present, appropriate anti-bacterial therapy should be used and if the infection does not respond promptly, the corticosteroid should be discontinued and other appropriate therapy initiated.
Steroid medication in the treatment of patients with a history of herpes simplex keratitis requires great caution; frequent slit microscopy is mandatory.
Prolonged application to the eye of preparations containing corticosteroids has caused increased intraocular pressure. If the treating physician determines the clinical need outweighs the risks in patients with glaucoma (see Section 4.3 Contraindications), intraocular pressure should be closely monitored and any undesirable elevation treated promptly.
Various ocular diseases and long term use of topical corticosteroids have been known to cause corneal and scleral thinning. Use of topical corticosteroids in the presence of thin corneal or scleral tissues may lead to perforation.
Systemic absorption of prednisolone may be reduced by compressing the lacrimal sac at the medial canthus for a minute during and following the instillation of the drops. (This blocks the passage of the drops via the naso-lacrimal duct to the wide absorptive area of the nasal and pharyngeal mucosa.)
Visual disturbance. Visual disturbance may be reported with systemic and topical corticosteroid use. If a patient presents with symptoms such as blurred vision or other visual disturbances, the patient should be considered for referral to an ophthalmologist for evaluation of possible causes which may include cataract, glaucoma or rare diseases such as central serous chorioretinopathy (CSCR) which have been reported after use of systemic and topical corticosteroids.
Use in the elderly. No data available.
Paediatric use. In children, long-term, continuous topical corticosteroid therapy should be avoided due to possible adrenal suppression (see Section 4.3 Contraindications).
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Although negligible prednisolone passes into the bloodstream after ocular instillation, drug interactions are nevertheless possible. The interactions observed with prednisolone administered by any route should therefore be taken into account.
Corticosteroids are known to increase the effects of barbiturates, sedative hypnotics and tricyclic antidepressants.
Corticosteroids will, however, decrease the effects of anticholinesterases, antiviral eye preparations and salicylates.
Co-treatment with CYP3A inhibitors, including cobicistat-containing products, is expected to increase the risk of systemic side-effects. The combination should be avoided unless the benefit outweighs the increased risk of systemic corticosteroid side-effects, in which case patients should be monitored for systemic corticosteroid side-effects.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Studies have not been performed in either animals or humans to evaluate the potential for prednisolone to impair fertility.
Use in pregnancy. (Category B3)
Topical administration of corticosteroids to pregnant animals can cause abnormalities of foetal development and although the relevance of this finding to human beings has not been established, the use of Minims Prednisolone Eye Drops during pregnancy should be avoided.
Use in lactation. Systemically absorbed prednisolone is excreted in breast milk, therefore, use of Minims Prednisolone Eye Drops to breastfeeding mothers is not recommended.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
Prolonged treatment with corticosteroids in high dosage is occasionally associated with cataract development.
Other ocular effects of corticosteroid therapy reported include: transient ocular discomfort, posterior subcapsular cataracts, ocular hypertension or glaucoma, defects in visual acuity and field of vision, optic nerve damage, decreased resistance to ocular infection, corneal epithelial healing impairment, uveitis, mydriasis and ptosis.
Following long term therapy, systemic effects of steroids are rarely reported but possible following the use of Minims Prednisolone Eye Drops.
Post marketing. Eye disorders: vision blurred.
4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
Overdosage with Minims Prednisolone Eye Drops is unlikely to occur.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. The actions of corticosteroids are mediated by the binding of the corticosteroid molecules to receptor molecules located within sensitive cells. Corticosteroid receptors are present in human trabecular meshwork cells and in rabbit iris ciliary body tissue.
Corticosteroids are thought to act by the induction of phospholipase A2 inhibitory proteins, collectively called lipocortins. It is postulated that these proteins control the biosynthesis of potent mediators of inflammation such as prostaglandins and leukotrienes by inhibiting the release of their common precursor arachidonic acid. Arachidonic acid is released from membrane phospholipids by phospholipase A2.
The activation and migration of leucocytes will be affected by prednisolone. A 1% solution of prednisolone acetate has been demonstrated to cause a 51% reduction in polymorphonuclear leucocyte mobilisation to an inflamed rabbit cornea.
Corticosteroids also lyse and destroy lymphocytes. These actions of prednisolone all contribute to its anti-inflammatory effect.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Aqueous humour levels have been reported in 93 human eyes, dosed with 50 microL of a 0.5% prednisolone sodium phosphate solution, prior to undergoing cataract extraction. Detectable levels were noted at the 90-240 minute interval. Levels were still detectable up to 8 hours after dosing, but not after 10 hours.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. There are no studies on the carcinogenicity of prednisolone by the topical ocular route. No carcinogenic activity was noted in the mouse at oral doses up to 5 mg/kg/day for 18 months. In male rats, administration of prednisolone in the drinking water at a dose level of 0.4 mg/kg/day for two years caused an increased incidence of hepatocellular tumours. This carcinogenic response does not appear to be related to genotoxic activity.
6 Pharmaceutical Particulars
6.1 List of Excipients
See Section 2 Qualitative and Quantitative Composition.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store at 2°C to 8°C. (Refrigerate. Do not freeze.) Do not expose to strong light.
6.5 Nature and Contents of Container
Minims Prednisolone Eye Drops are supplied as a clear colourless sterile eye drops in a single use polypropylene tube (unit) overwrapped in a polyester/paper blister. The blisters are packed in cartons of 20 units. Each unit contains approximately 0.5 mL solution.
6.6 Special Precautions for Disposal
Each Minims Prednisolone Eye Drops unit should be discarded after a single use.
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical name: 11β,17-Dihydroxy- 3,20-dioxopregna-1,4-dien-21-yl disodium phosphate. Molecular formula: C21H27Na2O8P. Molecular weight: 484.4.
Chemical structure.
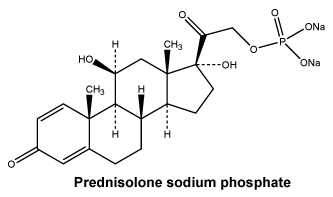
7 Medicine Schedule (Poisons Standard)
S4.
Date of First Approval
18 June 2009
Date of Revision
05 September 2018
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. NPS MedicineWise disclaims all liability (including for negligence) for any loss, damage or injury resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.