Minims Tropicamide
Brand Information
| Brand name | Minims Tropicamide |
| Active ingredient | Tropicamide, Tropicamide |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Minims Tropicamide.
Summary CMI
Minims® Tropicamide
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Minims Tropicamide?
Minims Tropicamide contains the active ingredient tropicamide. Minims Tropicamide is used to temporarily dilate (enlarge) the pupil of the eye and to stop the movement of the muscles that change the shape of the lens. It is usually used at the beginning of an eye examination so that your doctor can examine your eye(s).
For more information, see Section 1. Why am I using Minims Tropicamide? in the full CMI.
2. What should I know before I use Minims Tropicamide?
Do not use if you have ever had an allergic reaction to Tropicamide or any of the ingredients listed at the end of the CMI. Do not use if you have or suspect you have narrow angle glaucoma (high pressure in the eye). Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Minims Tropicamide? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Minims Tropicamide and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Minims Tropicamide?
Your doctor will normally instil the drops into your eyes for you. The usual dose is one drop instilled into the eye followed by a second drop after an interval of 5 minutes. If necessary, a further one drop may be instilled after 30 minutes.
If your doctor instructs you to instil the drops, more instructions can be found in Section 4. How do I use Minims Tropicamide? in the full CMI.
5. What should I know while using Minims Tropicamide?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Minims Tropicamide? in the full CMI.
6. Are there any side effects?
All medicines can have side effects. If they do occur, they are usually minor and temporary. Do not be alarmed by this list. You may not experience any of them. Less serious side effects include: blurred vision and/or problems seeing clearly, local irritation or stinging or feeling of having something in the eye, discomfort in the eye(s) due to sensitivity to light. This may also involve some discolouration of the front of the eye. Serious side effects include: allergic reaction, significant increases in eye pressure and effects to the body as a whole.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Minims® Tropicamide
Active ingredient: Tropicamide
Consumer Medicine Information (CMI)
This leaflet provides important information about using Minims Tropicamide. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Minims Tropicamide.
Where to find information in this leaflet:
1. Why am I using Minims Tropicamide?
2. What should I know before I use Minims Tropicamide?
3. What if I am taking other medicines?
4. How do I use Minims Tropicamide?
5. What should I know while using Minims Tropicamide?
6. Are there any side effects?
7. Product details
1. Why am I using Minims Tropicamide?
Minims Tropicamide contains the active ingredient tropicamide. Minims Tropicamide belongs to a class of medicines called antimuscarinic agents.
Minims Tropicamide is used to temporarily dilate (enlarge) the pupil of the eye and to stop the movement of the muscles that change the shape of the lens. It is usually used at the beginning of an eye examination so that your doctor can examine your eye(s).
2. What should I know before I use Minims Tropicamide?
Warnings
Do not use Minims Tropicamide if:
- You are allergic to tropicamide, or any of the ingredients listed at the end of this leaflet. Always check the ingredients to make sure you can use this medicine.
- You have or suspect you have narrow angle glaucoma (high pressure in the eye). Tropicamide can increase the pressure in the eye.
Check with your doctor if:
- You have had an allergy to any other medicines, foods, preservatives or dyes.
- You have any other medical conditions, especially:
- glaucoma (high pressure in the eye)
- fast heartbeat or any heart condition or had heart surgery
- inflamed eye - You take any medicines for any other condition.
- You are a senior person.
- You wear contact lenses. You should not wear contact lenses while using this medicine.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
This medicine has been developed for use in the eye only. It should not be injected.
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Use in children
Minims Tropicamide is not recommended for use in children.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and Minims Tropicamide may interfere with each other. These medicines may be affected by Minims Tropicamide or may affect how well it works. These include:
- Procaine (a numbing medicine)
- Cisapride (a medicine used to increase the movement of the gut)
- Procainamide (a medicine used to treat irregular heartbeats).
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Minims Tropicamide.
4. How do I use Minims Tropicamide?
Your doctor will normally instil the drops into your eyes for you. If your doctor instructs you to instil the drops, please follow the directions below.
How much to use
- Use Minims Tropicamide only when prescribed by your doctor. Your doctor will tell you how often to use the eye drops and how many drops to use each time.
- The usual dose is one drop instilled into the eye followed by a second drop after an interval of 5 minutes. If necessary, a further one drop may be instilled after 30 minutes.
How to use Minims Tropicamide
If you are wearing contact lenses, remove them before putting the drops in your eye.
You may find it easier to put drops in your eye while you are sitting or lying down.
If you are instilling your own drops, then follow these steps to use the eye drops:
- Wash your hands well with soap and water.
- Shake the container.
- Break the seal.
- Hold the container upside down in one hand between your thumb and forefinger.
- Using your other hand, gently pull down your lower eyelid to form a pouch or pocket.
- Tilt your head back and look up.
- Put the tip of the container close to your lower eyelid. Do not let it touch your eye.
- Release the required number of drops into the pouch or pocket formed between your eye and eyelid by gently squeezing the container.
- Close your eye. Do not blink or rub your eye.
- While your eye is closed, place your index finger against the inside corner of your eye and press against your nose for about two minutes. This will help to stop the medicine from draining through the tear duct to the nose and throat, from where it can be absorbed into other parts of your body.
- Repeat steps 4-10 for the other eye if needed.
- Throw away any eye drops left in the container.
- Wait at least 15 minutes before replacing your contact lenses.
If you use too much Minims Tropicamide
Accidental swallowing of the solution is unlikely to cause any bad side effects due to the low content of tropicamide.
If you think that you or anyone else may have swallowed Minims Tropicamide or used too much Minims Tropicamide, you should immediately:
- phone the Poisons Information Centre
(in Australia phone 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using Minims Tropicamide?
Things you should do
- If you experience any irritation, pain, swelling, excessive tear production or light-sensitivity while using the eye drops, flush the treated eye(s) with lots of water (room temperature) for at least 15 minutes. Stop using the eye drops and contact your doctor if these symptoms persist.
- Your eyes may be sensitive to light while using this medicine. It is recommended that you protect your eyes from light e.g. by wearing sunglasses.
Things you should not do
- Do not use this medicine to treat any other complaints unless your doctor tells you to.
- Do not give this medicine to anyone else, even if you think they have the same condition as you.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Minims Tropicamide affects you.
Minims Tropicamide may cause blurred vision for a short time when it is instilled into the eye. Your eyes may be sensitive to light while using this medicine. Allow enough time to regain clear vision before driving or using machines.
Looking after your medicine
Keep the unopened eye drops in a refrigerator (2-8°C) where children cannot reach them. Do not freeze the eye drops or expose them to strong light.
When to discard your medicine
Discard each single dose unit and any remaining solution in it immediately after use. Do not save unused contents.
Getting rid of any unwanted medicine
If your doctor tells you to stop using the eye drops or it is out of date or damaged, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date or if the packaging is torn or shows signs of tampering.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Eyes:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Symptoms of an allergic reaction, such as:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Some side effects (for example, an increase in the pressure of the eye) might only be found when your doctor or optometrist does tests from time to time.
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects online (in Australia, go to www.tga.gov.au/reporting-problems). By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Minims Tropicamide contains
| Active ingredient (main ingredient) | Tropicamide (0.5% w/v or 1% w/v) |
| Other ingredients (inactive ingredients) |
|
Do not take this medicine if you are allergic to any of these ingredients.
The eye drops do not contain any preservatives.
What Minims Tropicamide looks like
Minims Tropicamide is available in two strengths:
- Minims Tropicamide 0.5% eye drops (AUST R 32288)
- Minims Tropicamide 1% eye drops (AUST R 32289).
Minims Tropicamide comes in single dose units (tubes) containing 0.5mL of a clear, colourless solution. Each tube is wrapped in a polyester/paper blister. The blisters are packed in cartons of 20 units.
Who distributes Minims Tropicamide
In Australia, Minims Tropicamide is supplied by:
Bausch & Lomb (Australia) Pty Ltd
Level 2, 12 Help Street
Chatswood, NSW 2067
Phone: 1800 251 150
This leaflet was prepared in May 2025.
Brand Information
| Brand name | Minims Tropicamide |
| Active ingredient | Tropicamide, Tropicamide |
| Schedule | S4 |
MIMS Revision Date: 01 July 2020
1 Name of Medicine
Tropicamide.
2 Qualitative and Quantitative Composition
Minims Tropicamide Eye Drops contain tropicamide 0.5% and 1% w/v. No preservatives are contained in the formulation.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
A single-use eye drops, solution.
Minims Tropicamide are clear, colourless sterile ophthalmic solution. No preservatives are contained in the formulation.
4 Clinical Particulars
4.1 Therapeutic Indications
Minims Tropicamide Eye Drops is one of the antimuscarinics used to produce mydriasis and cycloplegia.
4.2 Dose and Method of Administration
Adults (including the elderly). To produce mydriasis, 1 or 2 drops of a 0.5% solution are instilled 15 to 20 minutes before examination of the eye.
To produce cycloplegia, 1 or 2 drops of a 1% solution are required, repeated after 5 minutes. A further drop may be necessary to prolong the effect after 20 to 30 minutes.
Systemic absorption can be minimised by pressure on the tear duct immediately after application.
4.3 Contraindications
Hypersensitivity to tropicamide or any other component listed in the formulation.
Narrow angle glaucoma.
4.4 Special Warnings and Precautions for Use
Identified precautions. For topical ophthalmic use only. Not for injection.
Tropicamide should not be used in patients with a narrow angle glaucoma since it may raise intraocular pressure and precipitate an acute attack.
As with other antimuscarinic agents, tropicamide should be used with caution in conditions characterised by tachycardia, such as thyrotoxicosis, heart failure and in cardiac surgery where it may further accelerate the heart rate. Care is required in patients with acute myocardial infarction as ischaemia and infarction may be made worse.
Tropicamide may significantly increase intraocular pressure.
Use with caution in an inflamed eye as the hyperaemia greatly increases the rate of systemic absorption through the conjunctiva.
Avoid contact with soft contact lenses. Patients must be instructed to remove contact lenses prior to the application of tropicamide eye drops and wait at least 15 minutes before reinsertion.
Use in the elderly. In the elderly and others where increased ocular pressure may be encountered, mydriatics and cycloplegics should be used with caution.
Paediatric use. Tropicamide has been reported to be inadequate for cycloplegia in children. Avoid use in children.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
The effects of antimuscarinic agents such as tropicamide may be enhanced by the concomitant administration of other drugs with antimuscarinic properties. Prolonged mydriatic and cycloplegic effects of tropicamide were reported with preinstillation of procaine.
Other drugs interactions. Drug interactions have been reported with co-administration of an anticholinergic agent cisapride counteracting gastromotility inducement; procainamide coadministered with tropicamide may result in additive antivagal effects on the anterioventricular nodal conduction.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Studies in animals have not been conducted to evaluate these effects.
Use in pregnancy. (Category B2)
Animal reproduction studies have not been conducted with tropicamide. It is not known whether tropicamide can cause foetal harm when administered to pregnant women or can affect reproductive capacity.
Use in lactation. It is not known whether this drug is excreted in human milk.
4.7 Effects on Ability to Drive and Use Machines
This preparation may cause transient blurring of vision on application. Patients should be advised not to drive or operate hazardous machinery until vision is clear. Patients may experience sensitivity to light and should protect their eyes from bright illumination during dilation.
4.8 Adverse Effects (Undesirable Effects)
Body. Anaphylaxis.
CVS. Transient bradycardia followed by tachycardia, with palpitations and arrhythmias.
CNS. Drowsiness and sedation, inability to concentrate and fatigue, psychotic reactions and behavioural disturbances, more common in children than adults.
Resp. Dryness of the mouth with difficulty in swallowing and talking, thirst, reduced bronchial secretions, bronchospasm.
GIT. Difficulty in micturition, as well as reduction in the tone and motility of the gastrointestinal tract leading to constipation. Occasionally, vomiting may occur.
Skin. Rash erythematous and pruritus. Flushing and dryness of the skin, increased sweating.
Ocular. Photophobia with or without corneal staining, significant increases in intraocular pressure, corneal irritation, smarting eyes, severe oedema of the eyelids and rhinitis. Dilation of pupils (mydriasis) with loss of accommodation (cycloplegia) and photophobia.
Paediatric population. Tropicamide may cause central nervous system disturbances, which may be dangerous in infants and children. Psychotic reactions, behavioural disturbances and cardio-respiratory collapse have been reported with this class of drug, especially in children.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
As Minims Tropicamide are single dose units of 0.5 mL overdose is unlikely to occur.
Should overdosage occur causing local effects, e.g. sustained mydriasis, physostigmine 0.25% w/v should be applied.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Minims Tropicamide is a topical antimuscarinic ophthalmic preparation that blocks the responses of the sphincter muscle of the iris and the accommodative muscle of the ciliary body to cholinergic stimulation producing pupillary dilation (mydriasis) and paralysis of accommodation (cycloplegia).
Tropicamide has an action similar to atropine but its cycloplegic and mydriatic effects have a more rapid onset and a shorter duration of effect. Tropicamide has the quickest recovery time of all the antimuscarinics.
Mydriasis is produced within 20 to 40 minutes of instillation and usually lasts for about 6 hours; cycloplegia is maximal within about 30 minutes and is short lasting with complete recovery of accommodation normally within 6 hours.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
No data available.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Sodium hydroxide, hydrochloric acid and purified water.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store at 2°C to 8°C. (Refrigerate. Do not freeze.) Protect from light.
6.5 Nature and Contents of Container
Minims Tropicamide Eye Drops are supplied as a clear colourless sterile eye drops in a single use polypropylene tube (unit) overwrapped in a polyester/paper blister. The blisters are packed in cartons of 20 units. Each unit contains approximately 0.5 mL.
Minims Tropicamide Eye Drops are available in two strengths 0.5% (5 mg/mL) and 1% (10 mg/mL).
6.6 Special Precautions for Disposal
Each Minims unit should be discarded after a single use.
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure.
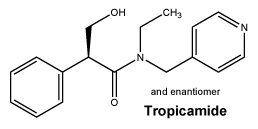
Molecular formula: C17H20N2O2.
Molecular weight: 284.4.
CAS number. 1508-75-4.
Tropicamide is a white or almost white, crystalline powder, slightly soluble in water, freely soluble in ethanol and in methylene chloride.
7 Medicine Schedule (Poisons Standard)
S4 - Prescription Only Medicine.
Date of First Approval
05 June 2000
Date of Revision
02 June 2020
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.