Minirin Nasal Spray
Brand Information
| Brand name | Minirin Nasal Spray |
| Active ingredient | Desmopressin acetate |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Minirin Nasal Spray.
Summary CMI
MINIRIN® NASAL SPRAY
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using MINIRIN Nasal Spray?
MINIRIN Nasal Spray contains the active ingredient desmopressin acetate, which is a synthetic version of a naturally occurring substance produced in the brain called vasopressin. It is used for several different conditions including primary nocturnal enuresis (bedwetting), cranial diabetes insipidus and as a diagnostic test to establish the kidneys ability to concentrate urine in adults. For more information, see Section 1. Why am I using MINIRIN Nasal Spray? in the full CMI.
2. What should I know before I use MINIRIN Nasal Spray?
Do not use if you have ever had an allergic reaction to desmopressin acetate or any of the ingredients listed at the end of the CMI. (see Section 7. Product details in the full CMI.) Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding. For more information, see Section 2. What should I know before I use MINIRIN Nasal Spray? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with MINIRIN Nasal Spray and affect how it works. A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use MINIRIN Nasal Spray?
MINIRIN Nasal Spray is meant to be sprayed into the nostrils. Do not inhale (sniff the spray).
- Bedwetting (nocturnal enuresis) - The usual starting dose is one spray (10 micrograms) into each nostril at bedtime. The dose will then be adjusted by your doctor according to your response. The maximum dose is two sprays in each nostril (40 micrograms).
- Cranial diabetes insipidus - For children the daily doses vary up to two sprays (20 micrograms). For adults the daily doses vary up to four sprays (40 micrograms). The daily dose is usually given as two divided doses. Sometimes a single daily dose is sufficient to control your condition.
- Test the ability of the kidneys to concentrate urine - If you are an adult, you will require a single dose of up to 40 micrograms at any one time. If you are a child, you will require a single dose of up to 10 micrograms at any one time.
More instructions can be found in Section 4. How do I use MINIRIN Nasal Spray? in the full CMI.
5. What should I know while using MINIRIN Nasal Spray?
| Things you should do |
|
| Things you should not do |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using MINIRIN Nasal Spray? in the full CMI.
6. Are there any side effects?
All medicines can have side effects. Most of them are minor and temporary but some may need medical attention.
Tell your doctor if you experience any side effects, including headache, stomach pain, nausea or vomiting, rapid weight gain, confusion or drowsiness. These are signs and symptoms of hyponatraemia (low sodium levels in the blood), an uncommon, but serious possible side effect of MINIRIN Nasal Spray. For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
MINIRIN® NASAL SPRAY
Active ingredient(s): desmopressin acetate
Consumer Medicine Information (CMI)
This leaflet provides important information about using MINIRIN Nasal Spray. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using MINIRIN Nasal Spray.
Where to find information in this leaflet:
1. Why am I using MINIRIN Nasal Spray?
2. What should I know before I use MINIRIN Nasal Spray?
3. What if I am taking other medicines?
4. How do I use MINIRIN Nasal Spray?
5. What should I know while using MINIRIN Nasal Spray?
6. Are there any side effects?
7. Product details
1. Why am I using MINIRIN Nasal Spray?
MINIRIN Nasal Spray contains the active ingredient desmopressin acetate. MINIRIN Nasal Spray is a synthetic version of the naturally occurring substance produced in the brain called vasopressin.
MINIRIN Nasal Spray has several different actions on the body including an action on the kidney to reduce the amount of urine produced. It is used for several different conditions including:
- primary nocturnal enuresis (bedwetting) in patients over 6 years of age, who have a normal ability to concentrate urine and who have not responded to treatment with an enuresis alarm or in patients in whom an enuresis alarm is contraindicated or inappropriate and where the oral administration of desmopressin is not feasible
- cranial diabetes insipidus (large amounts of urine being produced day and night and constant thirst)
- as a diagnostic test to establish if the kidneys have the ability to concentrate urine.
2. What should I know before I use MINIRIN Nasal Spray?
Warnings
Do not use MINIRIN Nasal Spray if:
- you are allergic to desmopressin, or any of the ingredients listed at the end of this leaflet
- you suffer from polydipsia (excessive thirst and increased fluid intake) or psychogenic polydipsia (psychologically caused increased thirst and increased fluid intake) or where you are in the habit of drinking large amounts of fluid
- you have cardiac insufficiency (shortness of breath, swelling of the feet or legs due to fluid build-up)
- you are taking diuretics (water or fluid tablets)
- you have moderately or severely reduced kidney function where you pass little or no urine
- you have low levels of sodium in your blood (hyponatraemia)
- you have a disorder which causes excessive release of anti-diuretic hormone (SIADH), resulting in weakness, tiredness or confusion
- you are breast-feeding
- for a child under the age of 6 years. Safety and effectiveness in children younger than 6 years have not been established
- the expiry date printed on the pack has passed
- the package is torn or shows signs of tampering.
Always check the ingredients to make sure you can use this medicine.
MINIRIN Nasal Spray should only be used when oral forms of MINIRIN are inappropriate.
This is because a side-effect that can change the fluid and salt balance in your body is less common with oral forms. For further information, please discuss this with your doctor.
Check with your doctor if you:
- have any other medical conditions especially the following:
- a known allergy to anti-diuretic hormone
- too little or too much fluid in the body
- a nasal infection, blocked or runny nose
- heart or blood vessel disease or any other disease for which you take diuretics
- low blood pressure
- cystic fibrosis or any medical condition causing fluid and/or electrolyte imbalance
- any medical condition that could be made worse by fluid and/or electrolyte disturbance
- any disease of the blood clotting cells (platelets)
- serious problems with bladder function or with passing urine.
- raised pressure within your head (increased intracranial pressure) - take any medicines for any other condition
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
MINIRIN Nasal Spray should only be used in a pregnant woman if necessary. Your doctor can discuss with you the risks and benefits involved.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
The active ingredient in MINIRIN Nasal Nasal Spray passes into breast milk.
Do not breastfeed if you are using this medicine.
If you have not told your doctor about any of the above, tell him/her before you start using MINIRIN Nasal Spray.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with MINIRIN Nasal Spray and affect how it works.
Medicines that may increase the effect of MINIRIN Nasal Spray include:
- medications which are known to release antidiuretic hormone (e.g. tricyclic antidepressants, selective serotonin reuptake inhibitors (SSRIs) chlorpromazine, carbamazepine) or some medications (such as chlorpropamide) which are known to treat high blood sugar (diabetes), as they can increase the risk of fluid buildup in the body
- medicines used to treat high blood pressure and some other conditions (ACE inhibitors or angiotensin receptor blockers e.g. enalapril, perindopril, irbesartan etc.)
- non-steroidal anti-inflammatory drugs (NSAIDs), medicines used to relieve pain and inflammation. NSAIDs may induce water retention/low sodium levels in the blood (hyponatraemia).
These medicines may be affected by MINIRIN Nasal Spray or may affect how well it works. You may need different amounts of your medicines, or you may need to take different medicines.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect MINIRIN Nasal Spray.
4. How do I use MINIRIN Nasal Spray?
Follow all directions given to you by your doctor or pharmacist carefully.
They may differ from the information contained in this leaflet.
The instructions on how to use MINIRIN Nasal Spray are included in the carton.
Prime the spray before using it for the first time by pressing it at least four times, or until an even spray is obtained. If the spray has not been used during the last 7 days, it is necessary to prime it again by pressing it a couple of times until an even spray is obtained before placing the nozzle in the nostril.
It is very important for you to follow these instructions closely to make sure that you will always receive the correct dose of your medication. MINIRIN Nasal Spray is meant to be sprayed into the nostrils. Do not inhale (sniff the spray).
If you do not understand the instructions in the leaflet, ask your doctor or pharmacist for help.
How much to use
Some of the details for MINIRIN Nasal Spray will vary depending on the purpose for which it is used.
Bedwetting (nocturnal enuresis)
- The usual starting dose is one spray (10 micrograms) into each nostril at bedtime. The dose will then be adjusted by your doctor according to your response. The maximum dose is two sprays in each nostril (40 micrograms).
- Fluid intake should be limited in preparation for your dose of MINIRIN Nasal Spray (see 'Things to be careful of').
Cranial diabetes insipidus
- The dose you need will be adjusted to suit your personal requirements. The daily dose is usually given as two divided doses. Sometimes a single daily dose is sufficient to control your condition.
- For children the daily doses vary up to two sprays (20 micrograms). For adults the daily doses vary up to four sprays (40 micrograms).
- Since MINIRIN Nasal Spray cannot deliver doses less than 10 micrograms per spray, the MINIRIN Intranasal Solution and the rhinyle delivery system will need to be used, if lower doses are needed.
Follow the instructions provided and use MINIRIN Nasal Spray until your doctor tells you to stop.
When to use MINIRIN Nasal Spray
Bedwetting (nocturnal enuresis)
- MINIRIN Nasal Spray should be used at bedtime.
Cranial diabetes insipidus
- MINIRIN Nasal Spray should be used at times specified by your doctor.
How long to use MINIRIN Nasal Spray
Continue using your medicine for as long as your doctor tells you.
This medicine helps to control your condition but does not cure it. It is important to keep using your medicine even if you feel well.
Bedwetting (nocturnal enuresis)
- MINIRIN Nasal Spray for bedwetting is usually taken for periods up to three months.
- After this period, you should have at least one week without MINIRIN Nasal Spray to check if your bedwetting has stopped. If not, continue for another three months.
Cranial diabetes insipidus
- Your doctor will advise you.
Test the ability of the kidneys to concentrate urine
- Your doctor will advise you.
If you forget to use MINIRIN Nasal Spray
Bedwetting (nocturnal enuresis)
- Skip the missed dose and take the next dose when you are meant to.
Cranial diabetes insipidus
- Take the next dose as soon as you remember. You will then have to adjust the time of the following dose. When this is done, go back to taking your medicine as you would normally. You should talk to your doctor about this situation. Do not take a double dose.
- If you are not sure what to do, ask your doctor or pharmacist.
- If you have trouble remembering to take your medicine, ask your pharmacist for some hints.
Test the ability of the kidneys to concentrate urine
- If you are an adult, you will require a single dose of up to 40 micrograms at any one time.
- If you are a child, you will require a single dose of up to 10 micrograms at any one time.
- Children who are less than one year of age and need to be tested for the ability of the kidney to concentrate urine should require this test under careful supervision in hospital.
If you use too much MINIRIN Nasal Spray
If you think that you have used too much MINIRIN Nasal Spray, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
Symptoms of an overdose may include confusion, drowsiness, continuing headache, nausea or vomiting, rapid weight gain due to a build-up of water in the body, or, in severe cases, convulsions.
5. What should I know while using MINIRIN Nasal Spray?
Things you should do
- If you are about to start any new medicine, remind your doctor and pharmacist that you are using MINIRIN Nasal Spray.
- If you are going to have surgery, tell the surgeon or anaesthetist that you are using this medicine.
- It may affect other medicines used during surgery.
- If you become pregnant while using this medicine, tell your doctor immediately.
- If you are about to have any blood tests, tell your doctor that you are using this medicine.
- It may interfere with the results of some tests.
- Keep all your doctor's appointments so that your progress can be checked.
- Your doctor may do some tests from time to time to make sure the medicine is working and to prevent unwanted side effects.
- Remind any doctor, dentist or pharmacist you visit that you are using MINIRIN Nasal Spray.
Things you should not do
- Do not use MINIRIN Nasal Spray to treat any other complaints unless your doctor tells you to do so.
- Do not give your medicine to anyone else, even if they have the same condition as you.
- Do not stop using your medicine or change the dosage without checking with your doctor.
- If you stop taking it suddenly, your condition may worsen.
Things to be careful of
All patients on treatment with this medicine should avoid excessive fluid intake as this may lead to a build-up of water in the body.
Bedwetting (nocturnal enuresis)
- You must avoid drinking fluids from one hour before using MINIRIN Nasal Spray until at least eight hours after administration of the nasal spray.
- Over this period, drink no more than a few sips of water or other fluids. This is because a high fluid intake during this period can increase the chance that you will feel unwell (e.g. headache, nausea, dizziness).
- Remember to drink normally throughout the day.
- This is very important to prevent dehydration.
Cranial diabetes insipidus
- Carefully follow your doctor's instruction about fluid intake.
- It is very important to keep your body water in balance, so that you do not let yourself get too thirsty or drink too much fluid.
Test the ability of the kidneys to concentrate urine
- You must avoid drinking fluids from one hour before using MINIRIN Nasal Spray until at least eight hours after administration of the nasal spray.
- Over this period, drink no more than a few sips of water or other fluids. This is because a high fluid intake during this period can increase the chance that you will feel unwell (e.g. headache, nausea, dizziness).
Driving or using machines
This medicine is not expected to affect your ability to drive a car or operate machinery.
Looking after your medicine
Keep MINIRIN Nasal Spray in the original container in upright position to protect from moisture and light until it is time to use it.
If you store the medicine out of its original packaging it may not keep well.
Store it in a cool dry place, where the temperature stays below 25°C, away from moisture, heat or sunlight.
For example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Heat and dampness can destroy some medicines.
Follow the instructions in the carton on how to take care of your medicine properly.
Keep it where young children cannot reach it.
A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date. The expiry date refers to the last day of that month.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
If you are an elderly patient, you may have an increased chance of getting side effects.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Side effects
| Side effects | What to do |
Very common side effects (affects 1 or more in 10 users):
| These side effects are not usually serious but can become serious. Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are using MINIRIN Nasal Spray.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What MINIRIN Nasal Spray contains
| Active ingredient (main ingredient) | desmopressin acetate |
| Other ingredients (inactive ingredients) |
|
MINIRIN Nasal Spray: Contains 10 micrograms desmopressin acetate per one actuation/spray.
Do not use this medicine if you are allergic to any of these ingredients.
What MINIRIN Nasal Spray looks like
MINIRIN Nasal Spray 6 mL is supplied in a brown, glass bottle equipped with a pre-compression spray pump delivering 60 doses of 10 micrograms desmopressin acetate. (AUST R 59320)
Who distributes MINIRIN Nasal Spray
Ferring Pharmaceuticals Pty Ltd
Suite 2, Level 1, Building 1
20 Bridge Street
Pymble NSW 2073
Toll free: 1800 337 746
This leaflet was prepared in July 2025.
AU-MN-2500001_v.1.0
MINIRIN® is a trademark of Ferring.
Brand Information
| Brand name | Minirin Nasal Spray |
| Active ingredient | Desmopressin acetate |
| Schedule | S4 |
MIMS Revision Date: 01 October 2019
1 Name of Medicine
Desmopressin acetate.
2 Qualitative and Quantitative Composition
Minirin Nasal Spray also contains sodium chloride, citric acid monohydrate, dibasic sodium phosphate-dihydrate, benzalkonium chloride solution 50% as preservative and water-purified.
Desmopressin free base represents 89% of the desmopressin acetate content. This is due to the difference in molecular weight as well as the presence of acetic acid/ acetate, water and impurities.
3 Pharmaceutical Form
Nasal spray, solution.
4 Clinical Particulars
4.1 Therapeutic Indications
Diabetes insipidus. The treatment of ADH sensitive cranial diabetes insipidus, including treatment of posthypophysectomy polydipsia and polyuria.
Nocturnal enuresis. Minirin Nasal Spray is indicated for the symptomatic treatment of primary nocturnal enuresis in patients who have normal ability to concentrate urine. Minirin Nasal Spray should be used only in patients who are refractory to the enuresis alarm or in patients in whom enuresis alarm is contraindicated or inappropriate, and where the oral administration of desmopressin is not feasible.
Renal concentrating capacity. By intranasal administration to adults and children as a diagnostic test to establish renal concentrating capacity.
4.2 Dose and Method of Administration
Note. Minirin Nasal Spray is for intranasal administration only. Administration of desmopressin acetate by intravenous or intramuscular injection may be used when the intranasal route is inconvenient.
Caution. The intravenous or intramuscular dose is about one tenth of the intranasal dose.
For ADH-sensitive cranial diabetes insipidus. Adult. The average daily dose is 10 to 40 microgram intranasally.
Paediatric. Intranasal. 2.5 to 20 micrograms daily.
The daily dose is usually given as two divided doses. The dosage must be determined for each individual patient and adjusted according to the diurnal pattern of response. Response should be estimated by two parameters: adequate duration of sleep and adequate, but not excessive, water turnover. In the event of signs of water retention/ hyponatraemia, treatment should be interrupted and the dose adjusted. A single daily dose may be appropriate if it is tolerated and also satisfactorily controls the diabetes insipidus. About one third of patients may be controlled on a small daily dose. For immediate postoperative polyuria and polydipsia, the dose should be controlled by measurement of the urine osmolality. Monitoring in a high dependency setting is recommended. If there is doubt that a dose has been administered, a second dose should not be given until diuresis has occurred.
Primary nocturnal enuresis. Dosage should be adjusted according to the individual. The recommended initial dose for those 6 years of age and older is 20 microgram or 0.2 mL solution intranasally at bedtime. Adjustment up to 40 microgram is suggested if the patient does not respond. Some patients may respond to 10 and a downward adjustment to 10 microgram can be made if the patient responds to 20 microgram. Note that each actuation of the spray contains 10 microgram of desmopressin acetate. It is recommended that one-half of the dose be administered per nostril. Since the spray cannot deliver less than 10 microgram, smaller doses should be delivered by the rhinyle delivery system. A restricted fluid intake is recommended overnight after administration (see Section 4.4 Special Warnings and Precautions for Use, fluid intake). Patients should be treated for an initial period of 1-3 months followed by a withdrawal of 1 week to assess cure rate. Relapsed patients should be continued for a further 1-3 months at the standard dose.
As a diagnostic test of renal concentrating capacity. See Section 4.4 Special Warnings and Precautions for Use.
Intranasal. Adults. Single dose of up to 40 micrograms.
Children. Single dose of up to 20 micrograms.
Infants. Single dose of up to 10 microgram (see Section 4.4 Special Warnings and Precautions for Use).
Instructions to be given to patients. The physician should carefully explain the use of the spray device and advise the patient not to inhale. Patients should be instructed to read accompanying directions on use of the spray pump carefully before use. Prime the spray before using it for the first time by pressing it at least four times, or until an even spray is obtained. If the spray has not been used during the last 7 days it is necessary to prime it again by pressing it a couple of times until an even spray is obtained before placing the nozzle in the nostril.
4.3 Contraindications
Habitual and psychogenic polydipsia (resulting in a urine production exceeding 40 mL/kg/24 hours).
A history of known or suspected cardiac insufficiency and other conditions requiring treatment with diuretics.
Moderate and severe renal insufficiency (creatinine clearance below 50 mL/min).
Known hyponatraemia.
Syndrome of inappropriate antidiuretic hormone secretion (SIADH).
Hypersensitivity to desmopressin acetate or any of the excipients in Minirin Nasal Spray.
4.4 Special Warnings and Precautions for Use
Desmopressin acetate is ineffective for the treatment of nephrogenic diabetes insipidus.
Only use Minirin Nasal Spray in patients where orally administered formulations are not feasible (see Section 4.8 Adverse Effects (Undesirable Effects), Postmarketing experience).
Hyponatraemia and hydration. Hyponatraemia in the context of the use of desmopressin is generally due to fluid overload, thus careful attention to fluid balance is needed. Other causes of hyponatraemia which may need excluding depending on the clinical situation include renal salt wasting due to central lesions, renal disorders or adrenal disorders. There is some evidence from postmarketing data for the occurrence of severe hyponatraemia in association with the nasal spray formulation of desmopressin when it is used in the treatment of central diabetes insipidus.
Central diabetes insipidus. The aim of fluid therapy is to replace urinary fluid loss.
Children, patients with cognitive impairment, and patients with inadequate thirst sensation need close monitoring of fluid intake.
Regular monitoring of serum and urinary sodium and osmolality is recommended at the discretion of the clinician.
Primary nocturnal enuresis. When used for the treatment of primary nocturnal enuresis the fluid intake must be limited to a minimum from 1 hour before until 8 hours after administration.
Check serum electrolytes at least once if therapy is continued beyond 7 days.
Testing of renal concentrating capacity. When used for diagnostic purposes the fluid intake must be limited to a maximum of 0.5 L to satisfy thirst from 1 hour before until at least 8 hours after administration. Renal concentrating capacity testing in children below the age of 1 year should only be performed under carefully supervised conditions in hospital.
When Minirin Nasal Spray is prescribed it is recommended to start at the lowest dose, ensure compliance with fluid restriction instructions; increase dose progressively, with caution; ensure adult supervision when a child is administering the drug in order to control the dose intake.
Desmopressin acetate should not be administered to dehydrated or overhydrated patients until water balance has been adequately restored.
Nasal infections/ rhinorrhoea. Intranasal administration may be ineffective and unreliable absorption may result in the presence of local infection or rhinorrhoea. In patients being treated for enuresis, treatment should cease until the nasal condition resolves. Bodyweight should be regularly monitored.
Myocardial ischaemia. Desmopressin acetate should be used with caution in patients with cardiovascular disease and the elderly.
Hypersensitivity. Patients with a known hypersensitivity to ADH should be tested for sensitivity to desmopressin acetate before the full dose is given.
Postoperative use. The use of desmopressin in a postoperative setting should only occur after the diagnosis of diabetes insipidus has been confirmed. Small doses should be administered with strict fluid balance and regular clinical assessment.
Minirin Nasal Spray should be used with caution in patients with cystic fibrosis because of impaired water handling and increased risk of hyponatraemia.
Precautions to prevent fluid overload must be taken in patients at risk of increased intracranial pressure.
Severe bladder dysfunction and outlet obstruction should be considered before starting treatment for primary nocturnal enuresis.
Treatment with desmopressin should be interrupted or carefully adjusted during acute intercurrent illnesses characterised by fluid and/or electrolyte imbalance (such as systemic infections, fever, gastroenteritis.
Additional information. High doses of desmopressin, such as those used to treat bleeding, are contraindicated in patients with type IIB von Willebrand's disease. Use of Minirin Nasal Spray in this patient group is not approved or recommended. At high doses, intravenously administered desmopressin has a vasodilatory effect and may cause a minor decrease in systolic or diastolic blood pressure. In haemophilia where high doses are given extreme care is paid to water balance.
Due to the presence of benzalkonium chloride, Minirin Nasal Spray may cause bronchospasm.
Use in elderly. See Section 4.4 Special Warnings and Precautions for Use.
Paediatric use. See Section 4.4 Special Warnings and Precautions for Use.
Effects on laboratory tests. See Section 4.4 Special Warnings and Precautions for Use.
4.5 Interactions with Other Medicines and Other Forms of Interactions
See Table 1.
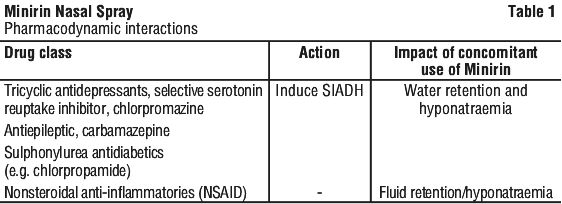
It is unlikely that desmopressin will interact with drugs affecting hepatic metabolism, since desmopressin has been shown not to undergo significant liver metabolism in in vitro studies with human microsomes. However, formal in vivo interaction studies have not been performed.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category B1)
Caution should be exercised when prescribing to pregnant women.
Data on a limited number (n = 53) of exposed pregnancies in women with diabetes insipidus as well as data on a limited number of exposed pregnancies in women with bleeding complications (n = 216) indicate no adverse effects of desmopressin on pregnancy or on the health of the fetus/ newborn child. To date, no other relevant epidemiological data are available. Animal studies do not indicate direct or indirect harmful effects with respect to pregnancy, embryonic/ fetal development, parturition or postnatal development.
Embryofetal development studies performed with desmopressin in rats and rabbits given subcutaneous doses up to 50 nanogram/kg/day and 200 microgram/kg/day, respectively, and in rats given intravenous doses up to 241 microgram/kg/day, revealed no evidence for a harmful effect on the fetus.
Use in lactation. Subtherapeutic levels of desmopressin acetate have been detected in the breast milk of lactating women. Until further evidence of its safe use during lactation is available, it is not to be administered to lactating women.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Clinical trials. Primary nocturnal enuresis. Table 2 lists the percentage of patients having adverse experiences without regard to relationship to study drug from the pooled pivotal study data for primary nocturnal enuresis.
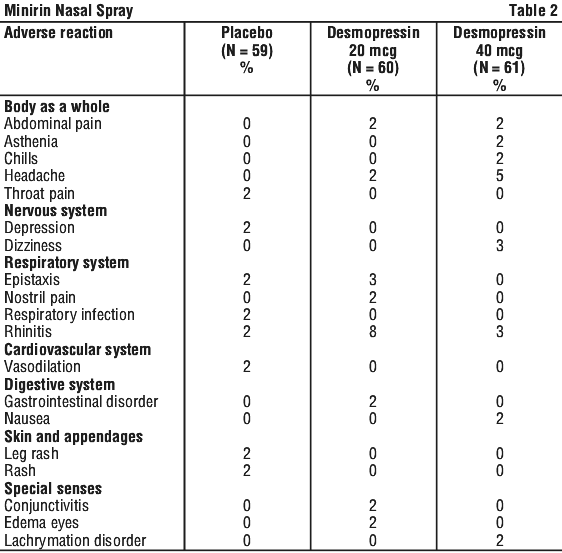
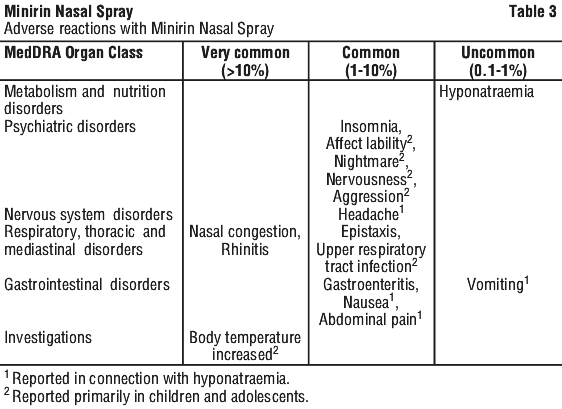
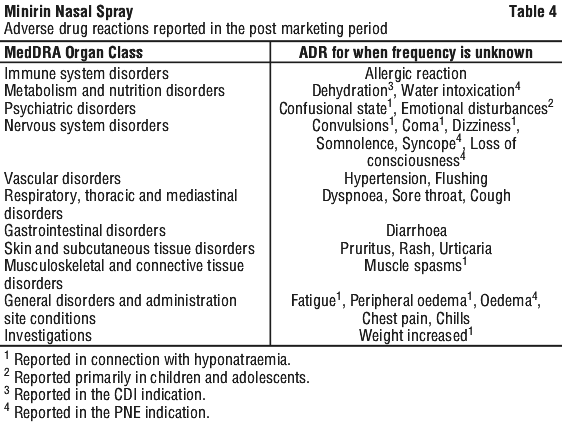
4.9 Overdose
Overdose of Minirin Nasal Spray leads to a prolonged duration of action with an increased risk of water retention and hyponatraemia.
Treatment. Treatment of hyponatraemia should be individualised. Treatment should include discontinuing desmopressin treatment, instigation fluid restriction and symptomatic treatment, if needed.
For information on the management of overdose, contact the Poisons Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: vasopressin and analogues.
ATC code: H01B A02.
Minirin Nasal Spray contains desmopressin, a structural analogue of the natural pituitary hormone arginine vasopressin, also known as antidiuretic hormone (ADH). Early treatment of central diabetes insipidus used a more or less purified extract from bovine or porcine posterior pituitaries. These caused unpleasant complications of use. When vasopressin became known, two forms were found: arginine vasopressin (found in humans) and lysine vasopressin (found in pig pituitaries).
Two chemical changes have been made to the natural hormone to form desmopressin: a) desamination of the N-terminal of cysteine-1; b) substitution of 8-D-arginine for 8-L-arginine.
According to results from antidiuretic and pressor tests in rats these changes increase antidiuretic activity three to fivefold, while pressor activity is reduced to 0.1% of that of ADH.
Mechanism of action. The actions of Minirin can be summarised as follows.
Antidiuretic action. Minirin acts at a receptor site in the renal collecting tubule to increase permeability to water reabsorption.
Effect on factor VIII. High doses (0.3 micrograms/kg intravenously) of desmopressin acetate produce marked and sustained increases of factor VIII coagulant activity (VIII:C) as well as of the von Willebrand factor (vWF). At the same time plasminogen activator is released.
Effect on bleeding time. At doses of 0.3 to 0.4 microgram/kg intravenously, desmopressin acetate results in a normalisation of, or marked reduction in, the prolonged skin (template) bleeding time. The exact mechanism of this effect is not known.
It is not known whether the effects of Minirin are direct or act through a mediator or second messenger.
There is a temporal correlation between a reduction in bleeding time and the presence in plasma of high molecular weight monomers of the von Willebrand factor which are thought to be released from storage sites.
It is thought likely that Minirin exerts its effect through its V2-receptor agonist activity.
Desmopressin acetate is thought to be resistant to the inactivation that occurs with ADH. Intravenous or intramuscular doses should be about one tenth the intranasal dose for equivalent efficacy.
In some patients, the duration of effect may be sufficiently long to permit once daily dosage if the single dose can be tolerated.
Other effects. Oxytocic effect. A slight in vitro oxytocic effect has been reported in animals. A slight stimulatory effect on uterine activity in nonpregnant women has been noted at doses of 15 and 20 micrograms intranasally (see Section 4.6 Fertility, Pregnancy and Lactation, Use in pregnancy).
Clinical trials. Relevant data not documented in this Product Information.
5.2 Pharmacokinetic Properties
Absorption. Using i.v. or i.m. doses, 100% of desmopressin is systematically available. Used intranasally, it is estimated that 10% is available. Thus, i.v. or i.m. doses are one tenth that of the intranasal route. The extent of absorption is similar for the spray and the rhinyle, with a trend towards higher absorption associated with the spray. Mean Cmax and AUC values are approximately 40% higher with the spray than with the rhinyle; however, there is considerable intra and inter individual variability in plasma levels of desmopressin.
Distribution. It is believed to be similar to ADH. No information is available on protein binding.
Metabolism. It is thought that the presence of the D-isomer in position eight protects desmopressin acetate from the enzyme which inactivates ADH.
Excretion. The excretion of desmopressin acetate is similar to that of ADH but considerably slower. Clinically intranasal desmopressin acetate is effective for approximately 10-12 hours.
Half-life. No information is available for intranasal administration. For i.v. administration of labelled desmopressin acetate, biexponential half-lives of 7.8 minutes and 75.5 minutes were recorded. The duration of drug effect is 8-20 hours, with much individual variation.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
See Section 2 Qualitative and Quantitative Composition.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Protect from light. Do not freeze.
6.5 Nature and Contents of Container
Spray pump of 5 mL delivering 50 doses of 10 micrograms desmopressin acetate.
Spray pump of 6 mL delivering 60 doses of 10 micrograms desmopressin acetate. Not all pack sizes are being distributed in Australia.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Synonyms of desmopressin. DDAVP; 1-desamino-8-D-arginine vasopressin; desamino-cys-1-D-arginine-8-vasopressin.
Chemical structure.
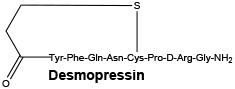
Molecular weight (base): 1069.22.
Desmopressin is a white, fluffy powder, soluble in water, alcohol and glacial acetic acid.
7 Medicine Schedule (Poisons Standard)
S4.
Date of First Approval
21 July 1997
Date of Revision
28 August 2019
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.