Neosynephrine
Brand Information
| Brand name | Neosynephrine |
| Active ingredient | Phenylephrine hydrochloride |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Neosynephrine.
Summary CMI
NEOSYNEPHRINE™
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I being treated with NEOSYNEPHRINE?
NEOSYNEPHRINE contains the active ingredient phenylephrine hydrochloride. NEOSYNEPHRINE is used to help increase your blood pressure and slow down your heart, for example, during surgery or in states of shock. It does this by causing constriction (narrowing) of your blood vessels.
For more information, see Section 1. Why am I being treated with NEOSYNEPHRINE? in the full CMI.
2. What should I know before treatment with NEOSYNEPHRINE?
Do not use if you have ever had an allergic reaction to phenylephrine hydrochloride, sodium metabisulfite or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before treatment with NEOSYNEPHRINE? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with NEOSYNEPHRINE and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How is NEOSYNEPHRINE given?
- NEOSYNEPHRINE must only be given by a doctor or nurse.
- It is given to you in three ways: as a slow injection or infusion (drip) into a vein, as a deep injection into a muscle or as an injection just under the skin.
More instructions can be found in Section 4. How is NEOSYNEPHRINE given? in the full CMI.
5. What should I know during treatment with NEOSYNEPHRINE?
| Things you should do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine | NEOSYNEPHRINE will be stored in the hospital, where the temperature stays below 25°C. |
For more information, see Section 5. What should I know during treatment with NEOSYNEPHRINE? in the full CMI.
6. Are there any side effects?
Side-effects may include headache or irritability, restlessness, tingling in the hands or feet and changes to heart rate such as slowing down, or increasing heartbeat.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
NEOSYNEPHRINE™ (nee-o-SIN-eh-frin)
Active ingredient(s): Phenylephrine Hydrochloride (Fen-nel-EF-reen Hi-dro-KLOR-ide)
Consumer Medicine Information (CMI)
This leaflet provides important information about using NEOSYNEPHRINE. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using NEOSYNEPHRINE.
Where to find information in this leaflet:
1. Why am I being treated with NEOSYNEPHRINE?
2. What should I know before treatment with NEOSYNEPHRINE?
3. What if I am taking other medicines?
4. How is NEOSYNEPHRINE given?
5. What should I know during treatment with NEOSYNEPHRINE?
6. Are there any side effects?
7. Product details
1. Why am I being treated with NEOSYNEPHRINE?
NEOSYNEPHRINE contains the active ingredient phenylephrine hydrochloride. It belongs to a group of medicines called vasoconstrictors.
NEOSYNEPHRINE is used to help increase your blood pressure and slow down your heart, for example, during surgery or in states of shock. It does this by causing constriction (narrowing) of your blood vessels.
2. What should I know before treatment with NEOSYNEPHRINE?
Warnings
Do not use NEOSYNEPHRINE if:
- You are allergic to:
- phenylephrine hydrochloride.
- sodium metabisulfite.
- any of the ingredients listed at the end of this leaflet. - You have very high blood pressure.
- You have a very fast heart beat (palpitations).
Symptoms of an allergic reaction may include:
- shortness of breath, wheezing or difficulty breathing.
- swelling of the face, lips, tongue or other parts of the body.
- rash, itching or hives on the skin.
Always check the ingredients to make sure you can use this medicine.
Check with your doctor if you:
- have any other medical conditions:
- thyroid disease.
- slow heart rate.
- other heart disease.
- severe arteriosclerosis (a disease of the arteries). - take any medicines for any other condition.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Your doctor will discuss the risks and benefits of being given NEOSYNEPHRINE during pregnancy.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Your doctor will discuss the risks and benefits of being given NEOSYNEPHRINE when breast-feeding.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with NEOSYNEPHRINE and affect how it works. These include:
- certain medicines to treat depression (such as monoamine oxidase inhibitors and tricyclic antidepressants).
- halothane anaesthetics (a type of medicine used to keep you asleep during an operation).
- certain medicines used to stimulate labour in pregnant women.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect NEOSYNEPHRINE.
4. How is NEOSYNEPHRINE given?
How it is given
- NEOSYNEPHRINE must only be given by a doctor or nurse.
- NEOSYNEPHRINE can be given to you in three ways:
- as a slow injection or infusion (drip) into a vein,
- as a deep injection into a muscle or,
- as an injection just under the skin. - Sometimes it is diluted before use and given with fluids into a vein.
- Your doctor will determine how you will be given this medicine. It may depend on why you need to use it.
How much to use
Your doctor will decide what dose and how long you will receive NEOSYNEPHRINE. The dose will be different, depending on how your doctor gives you NEOSYNEPHRINE and why you are being given it.
If you are given too much NEOSYNEPHRINE
As NEOSYNEPHRINE is given to you in hospital under the supervision of your doctor, it is very unlikely that you will receive too much.
If you think that you have been given too much NEOSYNEPHRINE, you may need urgent medical attention.
Symptoms of overdose include sensation of fullness in the head, tingling in the hands or feet and irregular or fast heartbeat.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know during treatment with NEOSYNEPHRINE?
Things you should do
Your doctor will monitor your blood pressure closely while you are being given NEOSYNEPHRINE.
Remind any doctor, dentist or pharmacist you visit that you are being treated with NEOSYNEPHRINE, especially if you are about to be started on any new medicine.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how NEOSYNEPHRINE affects you.
Drinking alcohol
No information is available.
Looking after your medicine
NEOSYNEPHRINE will be stored in the pharmacy or kept on the hospital ward.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or if it is out of date, the hospital will dispose of it safely.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Side effects
| Side effects | What to do |
General:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Heart rate and pulse related symptoms:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What NEOSYNEPHRINE contains
| Active ingredient (main ingredient) | phenylephrine hydrochloride |
| Other ingredients (inactive ingredients) |
|
| Allergens |
|
Do not use this medicine if you are allergic to any of these ingredients.
NEOSYNEPHRINE does not contain lactose, sucrose, gluten, tartrazine or any other azo dyes.
What NEOSYNEPHRINE looks like
NEOSYNEPHRINE is a clear, colourless, aqueous solution in glass ampoules presented in amber clamshells each containing 5 ampoules.
Five packs of 5 ampoules are shrink-wrapped to form packs of 25 ampoules (Aust R 15472).
Who distributes NEOSYNEPHRINE
Pfizer Australia Pty Ltd
Sydney NSW
Toll Free Number: 1800 675 229
www.pfizermedicalinformation.com.au
This leaflet was prepared in September 2024.
Brand Information
| Brand name | Neosynephrine |
| Active ingredient | Phenylephrine hydrochloride |
| Schedule | S4 |
MIMS Revision Date: 01 August 2019
1 Name of Medicine
Phenylephrine hydrochloride.
2 Qualitative and Quantitative Composition
Each ampoule contains 10 mg of phenylephrine hydrochloride per one mL (1% solution) as well as 3.5 mg of sodium chloride for isotonicity, 4 mg of sodium citrate dihydrate and 1 mg of citric acid as buffering agents, 2 mg of sodium metabisulfite as an antioxidant and water for injections. It is for use in one patient on one occasion only. Discard any residue.
Excipient with known effect. Sodium metabisulfite.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Neo-Synephrine, brand of phenylephrine hydrochloride injection, is a vasoconstrictor and pressor drug chemically related to adrenaline and ephedrine. Phenylephrine hydrochloride is a synthetic sympathomimetic agent. It is a white or almost white, crystalline powder, freely soluble in water and alcohol.
Neo-Synephrine is a clear, colorless, aqueous solution, free from visible particulates, in sterile form for parenteral injection.
Solution for injection.
It has a pH of between 4.5 and 6.5.
Neo-Synephrine contains no antimicrobial preservative.
4 Clinical Particulars
4.1 Therapeutic Indications
Phenylephrine hydrochloride is intended for the maintenance of an adequate level of blood pressure during spinal and inhalation anaesthesia and for the treatment of vascular failure in shock, shock-like states, and drug induced hypotension, or hypersensitivity. It is also employed to overcome paroxysmal supraventricular tachycardia, to prolong spinal anaesthesia, and as a vasoconstrictor in regional analgesia.
4.2 Dose and Method of Administration
Dosage. Mild or moderate hypotension. Subcutaneously or intramuscularly. Usual dose, from 2 mg to 5 mg. Range, from 1 mg to 10 mg. Initial dose should not exceed 5 mg.
Intravenously. Usual dose, 0.2 mg. Range, from 0.1 mg to 0.5 mg. Initial dose should not exceed 0.5 mg.
Injections should not be repeated more often than every 10 to 15 minutes. A 5 mg intramuscular dose should raise blood pressure for one to two hours. A 0.5 mg intravenous dose should elevate the pressure for about 15 minutes.
Severe hypotension and shock (including medicine related hypotension). Blood volume depletion should always be corrected as fully as possible before any vasopressor is administered. When, as an emergency measure, intra-aortic pressures must be maintained to prevent cerebral or coronary artery ischemia, phenylephrine hydrochloride can be administered before and concurrently with blood volume replacement.
Hypotension and occasionally severe shock may result from overdosage or idiosyncrasy following the administration of certain drugs, especially adrenergic and ganglion blocking agents, rauwolfia and veratrum alkaloids, and phenothiazine tranquilizers. Patients who receive a phenothiazine derivative as preoperative medication are especially susceptible to these reactions. As an adjunct in the management of such episodes, phenylephrine hydrochloride is a suitable agent for restoring blood pressure.
Higher initial and maintenance doses of phenylephrine hydrochloride are required in patients with persistent or untreated severe hypotension or shock. Hypotension produced by powerful peripheral adrenergic blocking agents, chlorpromazine or phaeochromocytomectomy may also require more intensive therapy.
Continuous infusion. Add 10 mg of the drug (1 mL of 1% solution) to 500 mL of dextrose 5% injection or sodium chloride 0.9% injection (providing a 1:50,000 solution equivalent to 20 microgram per mL). To raise the blood pressure rapidly, start the infusion at about 100 microgram to 180 microgram per minute (based on 20 drops per mL this would be 100 to 180 drops per minute). When the blood pressure is stabilized (at a low normal level for the individual), a maintenance rate of 40 microgram to 60 microgram per minute usually suffices (based on 20 drops per mL this would be 40 to 60 drops per minute). If the drop size of the infusion system varies from the 20 drops per mL, the dose must be adjusted accordingly.
If a prompt initial pressor response is not obtained, additional increments of phenylephrine hydrochloride (10 mg or more) are added to the infusion bottle. The rate of flow is then adjusted until the desired blood pressure level is obtained. (In some cases, a more potent vasopressor, such as norepinephrine bitartrate, may be required.) Hypertension should be avoided. The blood pressure should be checked frequently. Headache and/or bradycardia may indicate hypertension. Arrhythmias are rare.
Spinal anaesthesia (hypotension). Routine parenteral use of phenylephrine hydrochloride has been recommended for the prophylaxis and treatment of hypotension during spinal anaesthesia. It is best administered subcutaneously or intramuscularly three or four minutes before injection of the spinal anaesthetic. The total requirement for high anaesthetic levels is usually 3 mg and, for lower levels, 2 mg. For hypotensive emergencies during spinal anaesthesia, phenylephrine hydrochloride may be injected intravenously, using an initial dose of 0.2 mg. Any subsequent dose should not exceed the previous dose by more than 0.1 mg to 0.2 mg and no more than 0.5 mg should be administered in a single dose. To combat hypotension during spinal anaesthesia in children, a dose of 0.5 mg to 1 mg per 25 pounds (approx 11 kg) body weight, administered subcutaneously or intramuscularly, is recommended.
Prolongation of spinal anaesthesia. The addition of 2 mg to 5 mg of phenylephrine hydrochloride to the anaesthetic solution increases the duration of motor block by as much as approximately 50 percent without any increase in the incidence of complications such as nausea, vomiting, or blood pressure disturbances.
Vasoconstrictor for regional analgesia. Concentrations about ten times those employed when adrenaline is used as a vasoconstrictor are recommended. The optimum strength is 1:20,000 (equivalent to 50 microgram per mL) made by adding 1 mg of phenylephrine hydrochloride to every 20 mL of local anaesthetic solution. Some pressor responses can be expected when 2 mg or more are injected.
Paroxysmal supraventricular tachycardia. Rapid intravenous injection (within 20 to 30 seconds) is recommended; the initial dose should not exceed 0.5 mg, and subsequent doses, which are determined by the initial blood pressure response, should not exceed the preceding dose by more than 0.1 mg to 0.2 mg, and should never exceed 1 mg.
Paediatric use. To combat hypotension during spinal anaesthesia in children, a dose of 0.5 mg to 1 mg per 25 pounds (approx. 11 kg) body weight, administered subcutaneously or intramuscularly, is recommended.
Method of administration. Phenylephrine hydrochloride is generally injected subcutaneously, intramuscularly, slowly intravenously, or in dilute solution as a continuous intravenous infusion. In patients with paroxysmal supraventricular tachycardia and, if indicated, in case of emergency, phenylephrine hydrochloride is administered directly intravenously. The dose should be adjusted according to the pressor response. (See Table 1.)


4.3 Contraindications
Phenylephrine hydrochloride should not be used in patients with severe hypertension, ventricular tachycardia, or in patients who are hypersensitive to it.
4.4 Special Warnings and Precautions for Use
If used in conjunction with oxytocic medicines, the pressor effect of sympathomimetic pressor amines is potentiated (see Section 4.6 Fertility, Pregnancy and Lactation). The obstetrician should be warned that some oxytocic drugs may cause severe persistent hypertension and that even a rupture of a cerebral blood vessel may occur during the postpartum period.
Contains sodium metabisulfite, a sulfite that may cause allergic type reactions including anaphylactic symptoms and life threatening or less severe asthmatic episodes in certain susceptible people. The overall prevalence of sulfite sensitivity in the general population is unknown and probably low. Sulfite sensitivity is seen more frequently in asthmatic than in nonasthmatic people.
General. Phenylephrine hydrochloride should be employed only with extreme caution in elderly patients or in patients with hyperthyroidism, bradycardia, partial heart block, myocardial disease, or severe arteriosclerosis.
Use in hepatic impairment. No data available.
Use in renal impairment. No data available.
Use in the elderly. No data available.
Paediatric use. No data available.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Vasopressors, particularly metaraminol, may cause serious cardiac arrhythmias during halothane anaesthesia and therefore should be used only with great caution or not at all.
Oxytocic medicines. The pressor effect of sympathomimetic pressor amines is potentiated (see Section 4.4 Special Warnings and Precautions for Use).
MAO inhibitors. The pressor effect of sympathomimetic pressor amines is markedly potentiated in patients receiving monoamine oxidase inhibitors (MAOI). Therefore, when initiating pressor therapy in these patients, the initial dose should be small and used with due caution. The pressor response of adrenergic agents may also be potentiated by tricyclic antidepressants.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category B2)
Animal reproduction studies have not been conducted with phenylephrine hydrochloride. It is also not known whether phenylephrine hydrochloride can cause foetal harm when administered to a pregnant woman or can affect reproduction capacity. Phenylephrine hydrochloride should be given to a pregnant woman only if clearly needed.
Studies in animals are inadequate or may be lacking, but available data show no evidence of an increased occurrence of fetal damage.
Use in lactation. It is not known whether this medicine is excreted in human milk. Because many are excreted in human milk, caution should be exercised when phenylephrine hydrochloride is administered to a nursing woman.
Use in labour and delivery. If vasopressor drugs are either used to correct hypotension or added to the local anaesthetic solution, the obstetrician should be cautioned that some oxytocic drugs may cause severe persistent hypertension and that even a rupture of a cerebral blood vessel may occur during the postpartum period (see Section 4.4 Special Warnings and Precautions for Use).
4.7 Effects on Ability to Drive and Use Machines
No data available.
4.8 Adverse Effects (Undesirable Effects)
Headache, reflex bradycardia, excitability, restlessness and, rarely, arrhythmias.
Reported suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Overdosage may induce ventricular extrasystoles and short paroxysms of ventricular tachycardia, a sensation of fullness in the head and tingling of the extremities. Should an excessive elevation of blood pressure occur, it may be immediately relieved by an α-adrenergic blocking agent, e.g. phentolamine.
The oral LD50 in the rat is 350 mg/kg, in the mouse 120 mg/kg.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Phenylephrine hydrochloride produces vasoconstriction that lasts longer than that of adrenaline and ephedrine. Responses are more sustained than those to adrenaline, lasting 20 minutes after intravenous and as long as 50 minutes after subcutaneous injection. Its action on the heart contrasts sharply with that of adrenaline and ephedrine, in that it slows the heart rate and increases the stroke output, producing no disturbance in the rhythm of the pulse.
Phenylephrine hydrochloride is a powerful postsynaptic alpha-receptor stimulant with little effect on the beta-receptors of the heart. In therapeutic doses, it produces little if any stimulation of either the spinal cord or cerebrum. A singular advantage of this drug is the fact that repeated injections produce comparable effects.
The predominant actions of phenylephrine hydrochloride are on the cardiovascular system. Parenteral administration causes a rise in systolic and diastolic pressures in man and other species. Accompanying the pressor response to phenylephrine hydrochloride is a marked reflex bradycardia that can be blocked by atropine; after atropine, large doses of phenylephrine hydrochloride increase the heart rate only slightly. In man, cardiac output is slightly decreased and peripheral resistance is considerably increased. Circulation time is slightly prolonged, and venous pressure is slightly increased; venous constriction is not marked. Most vascular beds are constricted; renal, splanchnic, cutaneous, and limb blood flows are reduced but coronary blood flow is increased. Pulmonary vessels are constricted and pulmonary arterial pressure is raised.
The drug is a powerful vasoconstrictor, with properties very similar to those of norepinephrine but almost completely lacking the chronotropic and inotropic actions on the heart. Cardiac irregularities are seen only very rarely even with large doses.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. No data available.
Distribution. No data available.
Metabolism. No data available.
Excretion. No data available.
5.3 Preclinical Safety Data
No long-term animal studies have been done to evaluate the potential of phenylephrine hydrochloride in these areas.
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Sodium chloride, sodium metabisulfite, sodium citrate dihydrate, citric acid, water for injections, nitrogen.
6.2 Incompatibilities
No data available.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C.
Protect from light if removed from the packaging.
6.5 Nature and Contents of Container
Neo-Synephrine is presented in glass ampoules. The air in all ampoules has been displaced by nitrogen gas. The product is presented in amber clamshells each containing 5 ampoules. Five packs of five (5) ampoules are shrink-wrapped to form packs of 25 ampoules.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
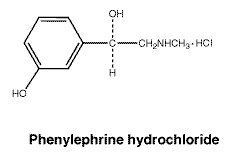
Chemical name: (-)-m-hydroxy-α-[(methylamino)methyl] benzyl alcohol hydrochloride.
Molecular formula: C9H13NO2.HCl.
Molecular weight: 203.7.
Chemical structure.
7 Medicine Schedule (Poisons Standard)
S4.
Date of First Approval
10 September 1991
Date of Revision
07 June 2019
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.