Neoral
Brand Information
| Brand name | Neoral |
| Active ingredient | Ciclosporin |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Neoral.
Summary CMI
Neoral®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using Neoral?
Neoral contains the active ingredient ciclosporin. Neoral is used to prevent organ rejection in people who have had kidney, liver or heart transplant. It is also used to treat different diseases that occur due to issues with your immune system such as nephrotic syndrome (a kidney disease), severe active rheumatoid arthritis (a joint disease having pain, swelling and stiffness in joints), severe psoriasis (a skin disease that causes rash with itchy scaly patches), severe atopic dermatitis (a skin disease that causes dry itchy and inflamed skin).
For more information, see Section 1. Why am I using Neoral? in the full CMI.
2. What should I know before I use Neoral?
Do not use if you have ever had an allergic reaction to Neoral or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Neoral? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Neoral and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Neoral?
More instructions can be found in Section 4. How do I use Neoral? in the full CMI.
5. What should I know while using Neoral?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Neoral? in the full CMI.
6. Are there any side effects?
Neoral may increase your risk of infections, cause headache, tiredness, tremors, burning sensation in the hands or feet, excess hair growth, vomiting, feeling sick, swollen or puffy gums, allergic reactions such as hives, rash, itchy skin.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Neoral®
Active ingredient(s): ciclosporin
Consumer Medicine Information (CMI)
This leaflet provides important information about using Neoral. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Neoral.
Where to find information in this leaflet:
1. Why am I using Neoral?
2. What should I know before I use Neoral?
3. What if I am taking other medicines?
4. How do I use Neoral?
5. What should I know while using Neoral?
6. Are there any side effects?
7. Product details
1. Why am I using Neoral?
Neoral contains the active ingredient ciclosporin. Neoral belongs to group of medicines known as immunosuppressive agents. These medicines help to control your body's immune system.
Neoral is used for people who have had a kidney, liver or heart transplant, to prevent the body from rejecting a new organ. It does this by blocking the development of special cells which would normally attack the transplanted tissue.
It is also used to treat other conditions which are thought to be caused by a problem with your immune system:
- Nephrotic syndrome (a kidney disease)
- Severe active rheumatoid arthritis (a disease affecting the joints with pain and swelling)
- Severe psoriasis (a skin disease with thickened patches of red skin, often with silvery scales)
- Severe atopic dermatitis (a skin allergy causing dry itchy and inflamed skin).
2. What should I know before I use Neoral?
Warnings
Do not use Neoral if:
- you are allergic to ciclosporin or any of the ingredients listed at the end of this leaflet.
Symptoms of allergic reactions may include flushing of face or area between neck and tummy area, shortness of breath or difficulty in breathing, coughing up blood, shortness of breath, wheezing, blood pressure changes and fast heartbeat.
Your doctor may discontinue if you develop allergic reactions.
Always check the ingredients to make sure you can use this medicine.
Do not use Neoral if you are taking it for non-transplant related conditions and you have:
- uncontrolled high blood pressure
- any uncontrolled infection.
Use in children
Do not give Neoral to patients under 16 years of age to treat severe rheumatoid arthritis, psoriasis or atopic dermatitis. However, it can be used in children younger than 16 years of age who have had an organ transplant or who have a kidney disease (nephrotic syndrome).
Check with your doctor if you have:
- Kidney problems - Neoral should not be taken by some patients with impaired kidney function.
- Any type of cancer or conditions that may develop into cancer.
- Vaccinations planned as these may be less effective during treatment with Neoral. Use of live attenuated vaccines should be avoided.
- Liver problems
- Severe heart, lung or blood vessel disease.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
There is an increased risk of problems in the mother and unborn child with the use of Neoral or any immuno-suppressive therapy in general.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Breast-feeding is not recommended if you are on treatment with Neoral. Ciclosporin, the main ingredient of the medicine, passes into the breast milk and can cause serious problems for the baby.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Neoral and affect how it works. Let your doctor know if you are using any of the following drugs:
- Methotrexate, a medicine used to treat rheumatoid arthritis
- Antibiotics, medicines used to treat infections, e.g. erythromycin, clarithromycin, azithromycin, rifampicin, ciprofloxacin, trimethoprim, gentamicin, tobramycin, vancomycin
- Antifungal medicines, e.g. fluconazole, ketoconazole, itraconazole, voriconazole, amphotericin B
- Tacrolimus, everolimus, sirolimus, mycophenolate sodium, other medicines which are used to suppress the immune system
- Diuretics (water tablets), a medicine that increases your amount of urine passed
- Heart or blood pressure medicines, e.g. lercanidipine, nifedipine, amlodipine, verapamil, diltiazem, nicardipine
- Digoxin, a medicine used to treat heart failure
- Statins, medicines used to treat high cholesterol, e.g. simvastatin, atorvastatin, pravastatin
- Fenofibrate, a medicine used to treat high cholesterol
- Certain medicines used to prevent fits or seizures e.g. carbamazepine, phenytoin
- Methylprednisolone, a medicine used to treat inflammatory diseases such as arthritis, asthma, colitis
- Colchicine, a medicine used to treat gout
- NSAIDs (non-steroid anti-inflammatory drugs), used to treat pain e.g. diclofenac, naproxen, indomethacin
- Recombinant human erythropoietin (rhEPO), a medicine used to treat specific types of anaemia
- Repaglinide, a medicine used to treat diabetes
- Cannabidiol (medicinal cannabis)
- Certain medicines used to decrease the amount of acid in your stomach, e.g. cimetidine, ranitidine
- Some oral contraceptives medicines, e.g. levonorgestrel and norethisterone
- Etoposide, imatinib, doxorubicin, medicines used to treat certain cancers
- Octreotide, a medicine used to treat a condition called acromegaly, and certain types of cancer
- Metoclopramide, a medicine used to treat nausea and vomiting
- Orlistat, a medicine used to help with weight loss
- St John's wort, a herbal supplement used to treat conditions such as depression.
Consult your doctor or pharmacist if you are taking any other medicines.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Neoral.
4. How do I use Neoral?
How much to take
Neoral is available as capsules and as an oral solution.
- Your doctor will determine the correct dose for you. It will depend on how much you weigh, what condition you are taking it for, how well Neoral works for you, and whether you have any side effects from this medicine.
- Your doctor may adjust your dose based on your response to the medicine.
- Follow the instructions provided. Check with your doctor or pharmacist if you are not sure how much to take.
When to take Neoral
- Take Neoral in the morning and at night.
How to take Neoral:
Capsules
- Do not remove the capsules from the foil blister pack until you are ready to take them.
- Swallow the capsules whole with a full glass of water.
- Do not chew them.
Oral solution
The oral solution is provided with two syringes for measuring the doses. The 1-mL syringe is used to measure doses less than or equal to 1 mL (each graduation of 0.05 mL corresponds to 5 mg of cyclosporin). The 4-mL syringe is used to measure doses greater than 1 mL and up to 4 mL (each graduation of 0.1 mL corresponds to 10 mg of cyclosporin). The oral solution should be diluted in a glass, not plastic container.
- Measure out the dose using the syringe provided.
- Add the dose to a glass of orange juice, apple juice or soft drink just before you take it. Do not use grapefruit juice.
- Make sure the syringe does not touch the liquid in the glass.
- Stir the liquid well immediately before drinking it.
- Rinse the container with more juice or soft drink to make sure that the whole dose has been taken.
- Wipe the outside of the syringe with a clean, dry tissue and put it back into its case. Do not wash the syringe with water, alcohol or any other liquid.
5. What should I know while using Neoral?
Things you should do
Keep all your medical appointments.
Your doctor will do routine tests to monitor your blood levels, to check how well your kidneys and liver are working, and to decide if your dose needs to be changed.
Limit your exposure to sunlight and UV light.
If you go out in the sun, wear a hat, appropriate protective clothing and use sunscreen with a high protection factor.
This will help prevent the risk of developing skin cancer.
Avoid eating large amounts of foods that are high in potassium content.
Taking Neoral can increase the amount of potassium in your blood. Too much potassium will cause unwanted side effects. Check with your doctor or pharmacist if you are not sure what foods contain high amounts of potassium.
Avoid taking this medicine with grapefruit, grapefruit juice or a fatty meal as it may cause unwanted side effects.
Take special care of your teeth and gums and see your dentist regularly.
Neoral may increase your risk of gum disease.
Call your doctor straight away if you:
- develop any lumps anywhere on/in your body, or develop any moles, or you notice changes in existing moles.
- become pregnant while having Neoral
- experience any symptoms of infection (e.g. fever, sore throat).
Remind any doctor, dentist or pharmacist you visit that you are using Neoral.
Things you should not do
- Do not stop treatment unless your doctor or healthcare professional tells you.
- Do not have any vaccinations without first consulting with your doctor.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Neoral affects you.
Neoral may make you feel sleepy, dizzy, have blurred vision or headaches.
If your child is on Neoral, check how the medicine affects them before they ride a bike or do activities that need coordination and concentration.
Drinking alcohol
Tell your doctor if you drink alcohol.
Drinking alcohol while on treatment with Neoral may lead to unwanted effects. Your doctor may ask you to limit your alcohol intake.
Looking after your medicine
Capsules
10 mg capsules: Store below 25°C.
25 mg, 50 mg, 100 mg capsules: Store below 30°C.
Oral solution
Store between 15-30°C. Do not refrigerate. Throw away 2 months after opening the bottle.
Store the capsules and oral solution in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on windowsills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
General problems:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Some side effects may not give you any symptoms and can only be found when tests are done.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Neoral contains
| Active ingredient (main ingredient) | ciclosporin |
| Other ingredients (inactive ingredients) | Capsules and Oral solution:
|
| Potential allergens | Contains ethanol |
Do not take this medicine if you are allergic to any of these ingredients.
What Neoral looks like
Neoral 10mg capsules: yellow-white, oval soft gelatin capsules printed in red ink with "NVR" and "10". Supplied in foil blister packs of 60 capsules.
Neoral 25mg capsules: blue-grey, oval soft gelatin capsules imprinted in red ink with "NVR" and "25". Supplied i in foil blister packs of 30 capsules.
Neoral 50mg capsules: yellow-white, oval soft gelatin capsules imprinted in red ink with "NVR" and "50". Supplied in foil blister packs of 30 capsules.
Neoral 100mg capsules: blue-grey, oval soft gelatin capsules imprinted in red ink with "NVR" and "100". Supplied in foil blister packs of 30 capsules.
Neoral 100mg/ml oral solution: Clear, faintly yellow-brownish liquid in a 50 mL glass bottle, with a 1mL and 4mL syringe for measuring the dose.
Australian Registration Numbers:
Neoral 10mg capsule AUST R 59614
Neoral 25mg capsule AUST R 49722
Neoral 50mg capsule AUST R 49723
Neoral 100mg capsule AUST R 49724
Neoral 100mg/ml oral solution AUST R 49725
Who distributes Neoral
Neoral is supplied in Australia by:
NOVARTIS Pharmaceuticals Australia Pty Limited
ABN 18 004 244 160
54 Waterloo Road
Macquarie Park NSW 2113
Telephone: 1 800 671 203
Website: www.novartis.com.au
® = Registered Trademark
This leaflet was prepared in December 2025.
Internal Document Code: neo210324c_v2 based on PI neo210324i
Brand Information
| Brand name | Neoral |
| Active ingredient | Ciclosporin |
| Schedule | S4 |
MIMS Revision Date: 01 May 2024
1 Name of Medicine
Ciclosporin.
2 Qualitative and Quantitative Composition
Neoral ciclosporin capsules. Each Neoral 10 mg capsule contains 10 mg ciclosporin.
Each Neoral 25 mg capsule contains 25 mg ciclosporin.
Each Neoral 50 mg capsule contains 50 mg ciclosporin.
Each Neoral 100 mg capsule contains 100 mg ciclosporin.
Neoral ciclosporin oral solution. Neoral oral solution contains 100 mg/mL of ciclosporin.
Excipients with known effect. Alcohol (as ethanol 11.8% v/v in the capsules and ethanol 12% v/v in the oral solution).
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Neoral ciclosporin capsules. 10 mg: yellow-white oblong soft gelatin capsules, imprinted in red with "NVR 10 mg".
25 mg: blue-grey oval soft gelatin capsules imprinted in red with "NVR 25 mg".
50 mg: yellow-white oblong soft gelatin capsules imprinted in red with "NVR 50 mg".
100 mg: blue-grey oblong soft gelatin capsules imprinted in red with "NVR 100 mg".
Neoral ciclosporin oral solution. 100 mg/mL: clear, faintly yellow to faintly brownish-yellow liquid.
4 Clinical Particulars
4.1 Therapeutic Indications
Neoral is indicated:
As an immunosuppressive agent for the prevention of graft rejection following kidney, liver and heart allogeneic transplantation.
For induction and/or maintenance of remission in the nephrotic syndrome. Ciclosporin is not a first line agent. Its use should be restricted to occasions when steroids and cytostatic drugs have failed, or are not tolerated, or are considered inappropriate, and when renal function is unimpaired (see Section 4.4 Special Warnings and Precautions for Use).
For the treatment of severe, active rheumatoid arthritis in patients for whom classical slow acting antirheumatic agents (including methotrexate) are inappropriate or ineffective.
In patients with severe psoriasis in whom conventional therapy is ineffective or inappropriate and the disease has caused a significant interference with quality of life.
For the treatment of severe atopic dermatitis when other treatment is ineffective or inappropriate.
Careful monitoring of all ciclosporin treated patients is mandatory. Ciclosporin should only be used by medical practitioners who are experienced in the use of immunosuppressive therapy (see Section 4.4 Special Warnings and Precautions for Use).
4.2 Dose and Method of Administration
The dose ranges given below are intended to serve as guidelines only. Neoral should always be given in two divided doses. Capsules should be swallowed whole.
Organ transplantation. Treatment with Neoral should be initiated within 12 hours before surgery at a dose of 10 to 15 mg/kg given in two divided doses. This dose should be maintained as a daily dose for one to two weeks post-operatively before being gradually reduced until a maintenance dose of about 2-6 mg/kg per day, given in two divided doses, is reached. Routine monitoring of ciclosporin blood concentrations is required (see Section 4.2 Dose and Method of Administration, Clinical blood level monitoring). The results obtained will serve as a guide for determining the actual dosage required in individual patients.
When Neoral is given together with other immunosuppressants (e.g. with corticosteroids or as part of a triple or quadruple drug therapy) lower doses may be used. Some renal transplant patients may need less than 5 mg/kg per day even at one month after transplantation when Neoral is given together with corticosteroids.
Paediatric. Experience with ciclosporin in young children is limited. Children from one year of age have received the drug in standard dosage. In several studies, paediatric patients required and tolerated higher doses of ciclosporin per kg bodyweight than those used in adults.
Nephrotic syndrome. For inducing remission, the recommended dose is 5 mg/kg per day for adults and 6 mg/kg per day for children if, with the exception of proteinuria, renal function is normal. In patients with impaired renal function, the initial dose should not exceed 2.5 mg/kg per day (see Section 4.3 Contraindications).
The combination of ciclosporin with low doses of oral corticosteroids has been used by some practitioners, but data on this combination are insufficient at present to allow a recommendation for such use to be made.
If no improvement has been observed after 3 months treatment, Neoral therapy should be discontinued.
The doses need to be adjusted individually according to efficacy (proteinuria) and safety (primarily serum creatinine), but should not exceed 5 mg/kg per day in adults and 6 mg/kg per day in children.
For maintenance treatment, the dose should be slowly reduced to the lowest effective level.
Rheumatoid arthritis. For the first 6 weeks of treatment the recommended dose is 3 mg/kg per day orally given in two divided doses. To achieve full effectiveness, up to 12 weeks of Neoral therapy may be required. However, if there is no clinical response in 4 to 8 weeks, the dose of Neoral can be increased at 1 to 2 month intervals by 0.5 to 1.0 mg/kg per day up to a maximum dose of 5.0 mg/kg per day.
For maintenance treatment, the dose has to be titrated individually according to tolerability. If a patient is on an effective maximum tolerable dose with no further improvements expected, and has been stable for at least 3 months, the dose of Neoral should be decreased at 0.5 mg/kg per day increments monthly or bimonthly to the lowest effective dose.
If there is essentially no clinical response by 6 months and the maximal tolerable dose has been administered for 3 months, Neoral should be discontinued. (After 3 months of Neoral therapy without response, blood level monitoring of ciclosporin may be of value to evaluate patient compliance and/or drug absorption.)
Dose adjustment based on creatinine values. If the serum creatinine remains increased by more than 30% above creatinine concentrations recorded before starting ciclosporin therapy at more than one measurement, the dosage of Neoral should be reduced. If the serum creatinine increases by more than 50%, a dosage reduction by 50% is mandatory. These recommendations apply even if the patient's values still lie within the laboratory normal range. If the dose reduction is not successful in reducing levels within one month, Neoral treatment should be discontinued.
See Section 4.5 Interactions with Other Medicines and Other Forms of Interactions for information on the concomitant use of Neoral and nonsteroidal anti-inflammatory drugs.
Psoriasis. For inducing remission, the recommended initial dose is 2.5 mg/kg per day orally given in two divided doses. If there is no improvement after one month, the daily dose may be increased gradually, but should not exceed 5 mg/kg. Treatment should be discontinued in patients in whom sufficient response of psoriatic lesions cannot be achieved within six weeks on 5 mg/kg per day, or in whom the effective dose is not compatible with the safety guidelines (see Section 4.4 Special Warnings and Precautions for Use).
An initial dose of 5 mg/kg per day is justified in patients whose condition requires rapid improvement.
For maintenance treatment, doses have to be titrated individually to the lowest effective level, and should not exceed 5 mg/kg per day.
The benefits of treatment can only be expected to continue while treatment is being given.
Atopic dermatitis. Due to the variability of this condition, treatment must be individualised. The recommended dose range is 2.5 to 5 mg/kg per day given in two divided oral doses. If a starting dose of 2.5 mg/kg per day does not achieve a satisfactory response within two weeks of therapy, the daily dose may be rapidly increased to a maximum of 5 mg/kg. In very severe cases, rapid and adequate control of the disease is more likely to occur with a starting dose of 5 mg/kg per day. Once a satisfactory response is achieved, the dose should be reduced gradually and, if possible, Neoral should be discontinued. Subsequent relapse may be managed with a further course of Neoral.
Although a course of 8 weeks' therapy may be sufficient to achieve clearing, up to one year's therapy has been shown to be effective and well tolerated, provided the monitoring guidelines are followed.
Pharmaceutical recommendations. Administration of Neoral solution. The oral solution is provided with two syringes for measuring the doses. The 1 mL syringe is used to measure doses less than or equal to 1 mL (each graduation of 0.05 mL corresponds to 5 mg of ciclosporin). The 4 mL syringe is used to measure doses greater than 1 mL and up to 4 mL (each graduation of 0.1 mL corresponds to 10 mg of ciclosporin).
Neoral solution should be diluted with preferably orange or apple juice; however, other drinks such as soft drinks can be used according to individual taste. Immediately before taking the solution, it should be stirred well. Owing to its possible interference with the pre-hepatic metabolism of ciclosporin by the cytochrome P450 enzyme system in the wall of the gastrointestinal tract, grapefruit juice should be avoided as a diluent.
The syringe should not come in contact with the diluent. If the syringe is to be cleaned, do not rinse it but wipe the outside with a dry tissue.
Neoral solution should be used within 2 months of opening the bottle and be stored between 15 and 30°C, preferably not below 20°C for prolonged periods, as it contains oily components of natural origin which tend to solidify at low temperatures. A jelly-like formation may occur below 20°C, which is however reversible at temperatures up to 30°C. Minor flakes or a slight sediment may still be observed. These phenomena do not affect the efficacy and safety of the product, and the dosing by means of the pipette remains accurate.
Conversion from Sandimmun to Neoral. The available data indicate that after a 1:1 conversion from the conventional oral forms of Sandimmun to Neoral, the trough concentrations of ciclosporin in whole blood are comparable. In many patients, however, higher peak concentrations (Cmax) and an increased exposure to the drug (AUC) may occur. In a small percentage of patients these changes are more marked and may be of clinical significance. Their magnitude depends largely on the individual variance in the absorption of ciclosporin from the originally used Sandimmun, which is known to be highly variable in its bioavailability. Patients with variable trough concentrations or very high doses of Sandimmun may be poor or inconsistent absorbers of ciclosporin (e.g. liver transplant patients with cholestasis or poor bile secretion, children or some kidney transplant recipients) who may, on conversion to Neoral, become good absorbers. Therefore, in this population, the increase in bioavailability of ciclosporin following a 1:1 conversion from Sandimmun to Neoral might be greater than usually observed. The dose of Neoral should therefore be down titrated individually according to their target trough concentration range. It needs to be emphasised that the absorption of ciclosporin from Neoral is less variable and the correlation between ciclosporin trough concentrations and exposure (in terms of AUC) is much stronger than with Sandimmun. This makes ciclosporin blood trough concentrations a more robust and reliable parameter for therapeutic monitoring.
Since the conversion from Sandimmun to Neoral may result in increased drug exposure, the following rules must be observed:
In transplant patients, Neoral should be started with the same daily dose as was previously used with Sandimmun. Ciclosporin trough concentrations in whole blood should be monitored initially within 4 to 7 days after the conversion to Neoral. In addition, clinical safety parameters such as serum creatinine and blood pressure are to be monitored during the first 2 months after the conversion. If the ciclosporin trough blood concentrations are beyond the therapeutic range and/or worsening of the clinical safety parameters occur, the dosage must be adjusted accordingly.
In patients treated for nontransplant indications, Neoral should be started with the same daily dose as was used with Sandimmun. Two, 4 and 8 weeks after the conversion, serum creatinine concentrations and blood pressure should be monitored. If serum creatinine concentrations or blood pressure significantly exceed the pre-conversion levels, or if serum creatinine concentrations increase to more than 30% above creatinine concentrations prior to Sandimmun therapy at more than one measurement, the dose should be reduced (see Section 4.2 Dose and Method of Administration; Section 4.4 Special Warnings and Precautions for Use). In case of unexpected toxicity or inefficacy of ciclosporin, blood trough concentrations should also be monitored.
Conversion between oral ciclosporin formulations. Switching from one oral ciclosporin formulation to another should be made with caution and under physician supervision. The introduction of the new formulation must be made with monitoring of blood levels of ciclosporin to ensure that pre-conversion levels are attained.
Clinical blood level monitoring. The monitoring of ciclosporin blood concentration is of value in the management of patient dosage. It must be remembered, however, that the concentration of ciclosporin in the blood is only one of many factors contributing to the clinical status of the patient (see Section 4.4 Special Warnings and Precautions for Use). Results should therefore serve only as a guide to dosing in the context of other clinical and laboratory parameters.
Detailed recommendations on therapeutic monitoring of ciclosporin in transplantation are presented in the paper by Morris RG, Tett SE and Ray JE entitled "Ciclosporin A Monitoring in Australia: Consensus Recommendations" (Ther. Drug Monit. 1994; 16: 570-576). The general consensus outlined in this paper is that ciclosporin parent drug concentrations be measured only in whole blood using an analytical technique exhibiting minimal or no cross reactivity with ciclosporin metabolites. The blood sample should be taken immediately before the next ciclosporin dose and the blood collection time recorded and standardised to prior to either the morning or evening dose.
Usually, routine monitoring of ciclosporin blood concentrations need not be performed in patients receiving ciclosporin for nontransplant indications. However, monitoring may be indicated in some patients; for example, where there is unexpected treatment failure or relapse, where patients may be at high risk of an adverse reaction or a drug interaction involving ciclosporin, or where there is an urgent need to establish ciclosporin exposure, for example in some life threatening situations.
4.3 Contraindications
All indications. Known hypersensitivity to ciclosporin and/or any excipients of Neoral.
Nontransplant indications. Uncontrolled hypertension, uncontrolled infection. Primary or secondary immunodeficiency excluding autoimmune diseases and selective IgA deficiency.
The use of ciclosporin in nephrotic syndrome is contraindicated in patients with impaired baseline renal function (serum creatinine > 200 micromol/L in adults and > 140 micromol/L in children). In other nontransplant indications, ciclosporin is contraindicated in patients with impaired renal function of any degree of severity.
4.4 Special Warnings and Precautions for Use
General precautions. Patient management. Only physicians experienced in immunosuppressive therapy and the management of kidney, heart and liver transplant patients and/or in the management of nephrotic syndrome, severe rheumatoid arthritis, severe psoriasis or severe atopic dermatitis should use ciclosporin. Patients receiving the drug should be managed in facilities equipped and staffed with adequate laboratory and supportive medical resources. The physician responsible for maintenance therapy should receive complete information for the follow-up of the patient.
Monitoring ciclosporin levels in transplant patients. When Sandimmun is used in transplant patients, routine monitoring of ciclosporin blood levels is an important safety measure (see Section 4.2 Dose and Method of Administration).
Development of malignancies. Like other immunosuppressants, ciclosporin increases the risk of developing lymphomas and other malignancies, particularly those of the skin. The increased risk appears to be related to the degree and duration of immunosuppression rather than to the use of specific agents. Hence, a treatment regimen containing multiple immunosuppressants (including ciclosporin) should be used with caution as this could lead to lymphoproliferative disorders and solid organ tumours, some with reported fatalities (see Section 4.8 Adverse Effects (Undesirable Effects)).
Because of the significantly increased risk over time of developing skin cancers, patients taking ciclosporin should be strongly advised to avoid excessive unprotected exposure to ultraviolet light or the sun.
There is limited long-term information on the development of possible malignancy and chronic nephrotoxicity following the use of ciclosporin.
Development of infections. Like other immunosuppressants, ciclosporin predisposes patients to the development of a variety of bacterial, fungal, parasitic and viral infections, often with opportunistic pathogens. Activation of latent polyomavirus infections that may lead to polyomavirus associated nephropathy (PVAN), especially to BK virus nephropathy (BKVN), or to JC virus associated progressive multifocal leukoencephalopathy (PML) have been observed in patients receiving ciclosporin. These conditions are often related to a high total immunosuppressive burden and should be considered in the differential diagnosis in immunosuppressed patients with deteriorating renal function or neurological symptoms. Serious and/or fatal outcomes have been reported. Effective pre-emptive and therapeutic strategies should be employed, particularly in patients on multiple long-term immunosuppressive therapy (see Section 4.8 Adverse Effects (Undesirable Effects)).
Hypertension. Hypertension induced by ciclosporin has been reported in up to 50% of post-transplant patients and 8.5% of patients being treated for nontransplant indications. The pathophysiology of ciclosporin induced nephrotoxicity and hypertension are closely related. Regular monitoring of blood pressure is required during ciclosporin therapy; if hypertension develops, appropriate antihypertensive treatment must be instituted (see Section 4.8 Adverse Effects (Undesirable Effects)). Diuretics (especially thiazide and loop diuretics) are not recommended. Concomitant use of diuretics and ciclosporin could predispose the patient to prerenal azotaemia, worsening of hyperuricaemia, glucose intolerance or hyperlipidaemia. For treatment of hypertension due to ciclosporin, if calcium channel blockers are indicated, only those which do not interfere with ciclosporin pharmacokinetics are recommended (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
As both recombinant human erythropoietin and ciclosporin are reported to increase blood pressure in a significant number of patients, caution should be exercised when administering these agents concomitantly (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Biochemical changes. Hyperkalaemia. Hyperkalaemia, which may become life threatening, can occur with ciclosporin treatment, especially in patients with renal dysfunction (see Section 4.8 Adverse Effects (Undesirable Effects)). It can be treated successfully and has also been found to disappear spontaneously. Patients receiving ciclosporin should avoid high dietary potassium intake and not be given potassium containing medication or potassium sparing diuretics (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions). Caution is also required when ciclosporin is coadministered with angiotensin converting enzyme inhibitors or angiotensin II receptor antagonists. Monitoring of serum potassium is recommended, especially in patients with marked renal dysfunction.
Calcium metabolism. Although apparently not observed so far in clinical use, physicians should be aware that in various animal studies using doses comparable with those used clinically, there were several changes indicative of a drug related disturbance in calcium metabolism.
Hypomagnesaemia. Hypomagnesaemia may increase the risk of ciclosporin related neurotoxicity. Ciclosporin enhances the clearance of magnesium. This can lead to symptomatic hypomagnesaemia, especially in the peritransplant period (see Section 4.8 Adverse Effects (Undesirable Effects)). Therefore, control of serum magnesium levels is recommended in the peritransplant period, particularly in the presence of neurological symptoms/ signs. If considered necessary, magnesium supplementation should be given.
Hyperuricaemia. Increased incidences of hyperuricaemia and acute gout have been reported after ciclosporin treatment (see Section 4.8 Adverse Effects (Undesirable Effects)). Special monitoring of serum uric acid in high risk patients is recommended.
Lipoprotein abnormalities. Recent reports suggest that ciclosporin may increase by 15 to 20% total cholesterol and low density lipoprotein cholesterol concentrations, as well as increase triglyceride levels, in renal and cardiac post-transplant patients. This effect does not seem to relate to total ciclosporin dose or ciclosporin plasma levels and may be associated with risk factors other than immunosuppressive treatment. It is advisable to perform lipid determinations before treatment and after the first month of therapy. In the event of increased lipids being found, restriction of dietary fat and, if appropriate, a dose reduction should be considered (see Section 4.8 Adverse Effects (Undesirable Effects)). Drug treatment of ciclosporin associated hyperlipidaemia is usually complicated by drug interactions between ciclosporin and some lipid lowering agents (e.g. nicotinic acid and bile acid sequestrants). Special care in combining lipid lowering agents with ciclosporin is recommended.
P-glycoprotein (Pgp). Ciclosporin may increase blood levels of concomitant medications that are substrates for the multidrug efflux transporter P-glycoprotein (Pgp) or the organic anion transporter proteins (OATP) such as aliskiren, dabigatran or bosentan. Coadministration of ciclosporin with aliskiren is not recommended. Coadministration of ciclosporin together with dabigatran or bosentan should be avoided. These recommendations are based on the potential clinical impact of these interactions (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Variable bioavailability. Due to the interindividual variations in absorption and elimination of ciclosporin, doses should be individually titrated according to clinical response, adverse reactions (especially renal or hepatic dysfunction) and trough blood concentrations for transplant patients in particular. Patients with malabsorption may have difficulty in achieving therapeutic levels with the oral forms of ciclosporin.
Vaccination. During treatment with ciclosporin, vaccination may be less effective and the use of live attenuated vaccines should be avoided (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Special excipient: ethanol. Neoral oral solution and soft gelatin capsules contain 12% v/v ethanol and 11.8% v/v ethanol respectively. The Neoral ethanol content should be taken into account when administered to pregnant or breastfeeding women, patients presenting with liver disease or epilepsy, alcoholic patients, or children.
Use in special patient groups. Use in organ transplantation. Ciclosporin should not be used concurrently with other immunosuppressive agents except adrenal corticosteroids. However, some centres use ciclosporin along with azathioprine and corticosteroids or other immunosuppressive agents (all in low doses) with the aim of reducing the possible risk of ciclosporin induced renal side effects (see below). Immunosuppression can lead to increased susceptibility to infection and the possible development of malignancies or lymphoproliferative disorders. Infections are most likely to occur in the first year after transplantation, with their incidence declining substantially thereafter. Bacterial infections, primarily involving the urinary tract, lungs, i.v. line related sepsis and/or wound sites, and viral infections, typically herpes or cytomegalovirus, tend to be the most frequent.
Acute and chronic nephrotoxicity. A frequent and potentially serious complication, an increase in serum creatinine and urea may occur during the first few weeks of ciclosporin therapy. These functional changes are dose dependent and reversible, usually responding to dose reduction. During long-term treatment, some patients may develop structural changes in the kidney (e.g. arteriolar hyalinosis, tubular atrophy, and interstitial fibrosis) which, in renal transplant patients, have to be differentiated from changes due to chronic rejection (see Section 4.8 Adverse Effects (Undesirable Effects)). Ciclosporin may also cause dose dependent, reversible increases in serum bilirubin and occasionally in liver enzymes. Close monitoring of parameters adequate for assessing renal and hepatic function is required. Abnormal values may necessitate dose reduction (see Section 4.2 Dose and Method of Administration; Section 5.1 Pharmacodynamic Properties, Clinical trials).
Post-transplant lymphomas, both polyclonal and monoclonal, and other lymphoproliferative lesions often associated with Epstein-Barr virus infections have been reported in immunosuppressed patients including those on ciclosporin, although no causal relationship has been established. Reduction or discontinuation of immunosuppression has caused regression of the lesions, often without subsequent rejection of the graft. The clinical incidence of lymphoma with ciclosporin does not appear to be greater than with other immunosuppressives.
Care should be taken in using ciclosporin with systemic antibiotics or other compounds that have nephrotoxic effects (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Use in nephrotic syndrome. Since ciclosporin can impair renal function, it is necessary to assess renal function frequently and to reduce the dose by 25-50% when serum creatinine increases by more than 30% above creatinine concentrations recorded before starting ciclosporin therapy. If the increase from baseline exceeds 50%, further reductions should be considered. These recommendations apply even if the patient's values still lie within the laboratory's normal range (see Section 4.2 Dose and Method of Administration).
Ciclosporin associated structural changes on renal biopsy have been observed without consistent alteration in serum creatinine in patients treated with ciclosporin for some months. Therefore renal biopsy should be considered if, in the treatment of nephrotic syndrome, ciclosporin therapy has been maintained for more than one year.
Patients with nephrotic syndrome who have any kind of malignancy should not be treated with ciclosporin. In patients with nephrotic syndrome treated with immunosuppressants (including ciclosporin) the occurrence of malignancies (including Hodgkin's lymphoma) has occasionally been reported.
Use in rheumatoid arthritis. Patients with impaired renal function, abnormal liver function, uncontrolled hypertension, uncontrolled infections or any kind of malignancy, or premalignant conditions such as leukoplakia, monoclonal paraproteinaemia, myelodysplastic syndrome and dysplastic naevi, should not receive ciclosporin. Ciclosporin should not be used in patients where severe complications of the heart, peripheral blood vessels or lungs are involved.
Patients with rheumatoid arthritis seem particularly susceptible to the nephrotoxic effects of ciclosporin. It appears that both the underlying disease process and the various therapies used contribute to the increased susceptibility to nephropathy. Clinical trials using lower doses of ciclosporin have reported less serious changes in renal function. However, moderate to severe and/or irreversible changes have been observed in patients receiving low doses of ciclosporin when the drug is administered long-term. The risk of nephrotoxicity further increases in patients being treated with a combination of low dose ciclosporin and nonsteroidal anti-inflammatory drugs (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Since ciclosporin can impair renal function, a reliable baseline concentration of serum creatinine should be established by at least two measurements prior to treatment, and serum creatinine should be monitored at 2 weekly intervals during the first 3 months of therapy. Thereafter, measurements can be made every 4 weeks, but more frequent checks are necessary when the ciclosporin dose is increased or concomitant treatment with a nonsteroidal anti-inflammatory drug is initiated or its dosage increased (see Section 4.2 Dose and Method of Administration).
Discontinuation of the drug may become necessary if hypertension developing during ciclosporin therapy cannot be controlled by appropriate antihypertensive therapy (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
As with other long-term immunosuppressive treatments (including ciclosporin), an increased risk of lymphoproliferative disorders must be borne in mind. Special caution should be observed if Neoral is used in combination with methotrexate (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Use in psoriasis. Patients with abnormal renal function, uncontrolled hypertension, uncontrolled infections (see Section 4.3 Contraindications) or any kind of malignancy other than of the skin (see below) should not receive ciclosporin.
Since ciclosporin can impair renal function, a reliable baseline concentration of serum creatinine should be established by at least two measurements prior to treatment and serum creatinine should be monitored at two-weekly intervals for the first three months of therapy. Thereafter, if creatinine remains stable, measurements should be made at monthly intervals. If the serum creatinine remains increased by more than 30% above creatinine concentrations recorded before starting ciclosporin therapy at more than one measurement, the dosage of ciclosporin must be reduced by 25-50%. These recommendations apply even if the patient's values still lie within the laboratory normal range (see Section 4.2 Dose and Method of Administration). If a dose reduction is not successful in reducing levels within one month, ciclosporin treatment should be discontinued.
Discontinuation of ciclosporin therapy is recommended if hypertension developing during ciclosporin therapy cannot be controlled with appropriate therapy (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Elderly patients should be treated only in the presence of disabling psoriasis and renal function should be monitored with particular care.
In psoriatic patients on ciclosporin, as in those on conventional therapy, development of malignancies (in particular of the skin) has been reported. It was noted in an epidemiological study of clinical trials on the use of ciclosporin in psoriasis (n = 1439) that 32 cases of malignancies were reported during these trials, 17 cases of which were skin cancers; almost all of the patients who developed skin cancers had previously been exposed to PUVA. No case of melanoma was reported. In the same study, it was estimated that the risk of developing skin cancer, solid malignant tumours or lymphomas in psoriatic patients treated with ciclosporin increased by 12.40, 3.19 or 5.05 respectively over a reference population. (These estimates were corrected for the risk of developing skin or other cancers in the untreated psoriatic population, but not for the increased risk of skin and other cancers associated with previous therapy with PUVA or immunosuppressive agents other than ciclosporin.) It is noteworthy that most of the data used in the study were obtained from trials conducted in the USA and northern Europe, where the baseline incidence of skin cancer is significantly lower than that in Australia. Therefore, patients undergoing ciclosporin therapy who have been exposed to excessive Australian sunlight may be expected to have a higher risk of developing skin cancers than American or European patients.
In view of the potential risk of skin cancer, patients being treated with ciclosporin should not receive concomitant ultraviolet B irradiation or PUVA photochemotherapy.
Skin lesions not typical for psoriasis but suspected to be malignant or premalignant should be biopsied before ciclosporin treatment is started. Patients with malignant or premalignant alterations of the skin should be treated with ciclosporin only after appropriate treatment of such lesions and if no other option for successful therapy exists.
In a few psoriatic patients treated with ciclosporin, lymphoproliferative disorders have occurred. These were responsive to prompt drug discontinuation.
Long-term safety data on the use of ciclosporin in psoriasis are at present limited. There is only limited experience with the use of ciclosporin in children with psoriasis.
Use in atopic dermatitis. Patients with abnormal renal function, uncontrolled hypertension, uncontrolled infections (see Section 4.3 Contraindications) or any kind of malignancy should not receive ciclosporin.
Since ciclosporin can impair renal function, a reliable baseline level of serum creatinine should be established by at least 2 measurements prior to treatment, and serum creatinine should be monitored at 2 weekly intervals for the first 3 months of therapy. Thereafter, if creatinine remains stable, measurements should be made at monthly intervals. If the serum creatinine increases and remains increased to more than 30% above baseline at more than one measurement, the dosage of ciclosporin must be reduced by 25 to 50%. These recommendations apply even if the patient's values still lie within the laboratory's normal range (see Section 4.2 Dose and Method of Administration). If dose reduction is not successful in reducing levels within one month, ciclosporin should be discontinued.
Discontinuation of ciclosporin therapy is also recommended if hypertension developing during ciclosporin therapy cannot be controlled with appropriate therapy (see Section 4.8 Adverse Effects (Undesirable Effects)).
As the experience with ciclosporin in children with atopic dermatitis is still limited to date, its use in this patient population is not recommended.
Elderly patients should be treated only in the presence of disabling atopic dermatitis and renal function should be monitored with particular care.
Benign lymphadenopathy is commonly associated with flares in atopic dermatitis and invariably disappears spontaneously or with general improvement in the disease. Lymphadenopathy observed on treatment with ciclosporin should be regularly monitored. Lymphadenopathy which persists despite improvement in disease activity should be examined by biopsy as a precautionary measure to ensure the absence of lymphoma.
Active herpes simplex infections should be allowed to clear before treatment with ciclosporin is initiated, but are not a reason for drug withdrawal if they occur during treatment, unless infection is severe.
Skin infections with Staphylococcus aureus are not an absolute contraindication for ciclosporin therapy, but should be controlled with appropriate antibacterial agents. Oral erythromycin, known to have the potential to increase the blood concentration of ciclosporin (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions) should be avoided or, if there is no alternative, it is recommended to closely monitor blood levels of ciclosporin, renal function, and for side effects of ciclosporin.
In view of the potential risk of skin cancer, patients on ciclosporin should be warned to avoid excess unprotected sun exposure and should not receive concomitant ultraviolet B irradiation or PUVA photochemotherapy.
Use in hepatic impairment. Ciclosporin may cause increases in serum bilirubin and liver enzymes (see Section 4.8 Adverse Effects (Undesirable Effects)). There have been solicited and spontaneous postmarketing reports of hepatotoxicity and liver injury including cholestasis, jaundice, hepatitis and liver failure in patients treated with ciclosporin. Most reports included patients with significant co-morbidities, underlying conditions and other confounding factors including infectious complications and comedications with hepatotoxic potential. In some cases, mainly in transplant patients, fatal outcomes have been reported (see Section 4.8 Adverse Effects (Undesirable Effects)). These changes appear to be dose related and reversible. Hepatic dysfunction, as measured by rises in serum bilirubin, has been found to be associated with proportional increases in serum ciclosporin concentrations in some cases.
Use in renal impairment. Ciclosporin may cause increases in serum creatinine and urea concentrations, even at recommended doses, as a result of a reduced glomerular filtration rate. The mechanism leading to these changes is not fully understood. These changes are usually dose-dependent and reversible with reduction of ciclosporin dosage. Structural changes to the kidney (e.g. interstitial fibrosis) may also occur, usually at higher ciclosporin dose levels. Although these renal changes are less common than functional changes, they may be irreversible. In non-transplant indications, the risk of renal structural changes is greater if the serum creatinine concentration increases by more than 30% from the patient's own baseline value. Thus regular measurements of serum creatinine must be made. In renal transplant patients who have received long-term treatment with ciclosporin, structural changes in the kidney must be differentiated from organ rejection.
Close monitoring of all parameters is required, with dose adjustments when indicated.
Use in the elderly. Experience in the elderly is limited, but no particular problems have been reported following use of the drug at the recommended dose. However, factors sometimes associated with aging, in particular impaired renal function, make careful supervision essential and may necessitate dosage adjustment.
In rheumatoid arthritis clinical trials with ciclosporin, 17.5% of patients were aged 65 or older. These patients were more likely to develop systolic hypertension on therapy, and more likely to show serum creatinine rises ≥ 50% above the baseline after 3-4 months of therapy. Clinical studies in transplant and psoriasis patients did not include a sufficient number of subjects aged 65 and over to determine whether they respond differently from younger subjects. Other reported clinical experiences have not identified differences in response between the elderly and younger patients.
In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal or cardiac function, and of concomitant disease or other drug therapy.
Paediatric use. Except for use in transplantation and in the treatment of nephrotic syndrome, there is no adequate experience available with ciclosporin; its use in children under 16 years of age for nontransplant indications other than nephrotic syndrome cannot be recommended. Also see Section 4.4 Special Warnings and Precautions for Use, Special excipient: ethanol.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Live attenuated vaccines. During treatment with ciclosporin, vaccination may be less effective and the use of live attenuated vaccines should be avoided (see Section 4.4 Special Warnings and Precautions for Use).
Potassium sparing drugs. Caution is required for concomitant use of potassium sparing drugs (e.g. potassium sparing diuretics, angiotensin converting enzyme inhibitors, angiotensin II receptor antagonists) or potassium containing drugs, since they may lead to significant increases in serum potassium (see Section 4.4 Special Warnings and Precautions for Use, Hypertension).
Methotrexate. Care should be taken when using ciclosporin together with methotrexate in rheumatoid arthritis patients due to the risk of nephrotoxic synergy (see Section 4.4 Special Warnings and Precautions for Use).
Antibiotics. Care should be taken when using ciclosporin in conjunction with systemic antibiotics or other compounds known to have nephrotoxic effects, e.g. aminoglycosides (including gentamicin, tobramycin), amphotericin B, ciprofloxacin, melphalan, colchicine, trimethoprim (plus sulphamethoxazole), as additive nephrotoxicity has been reported to occur. Close monitoring of renal function (in particular serum creatinine) should be performed. If a significant impairment of renal function occurs, the dosage of the coadministered drug should be reduced or alternative treatment considered.
In oncology patients, the intravenous coadministration of anthracycline antibiotics and very high doses of ciclosporin has resulted in significant increased exposure in the anthracycline antibiotics (e.g. doxorubicin, mitoxantrone, daunorubicin).
Tacrolimus. Concomitant use with tacrolimus should be avoided due to increased potential for nephrotoxicity.
Lercanidipine. Caution should be observed when coadministering lercanidipine with ciclosporin as the AUCs of both drugs increased with concomitant administration (see Section 4.4 Special Warnings and Precautions for Use).
Everolimus, sirolimus. Elevations in serum creatinine were observed in the studies using everolimus or sirolimus in combination with full dose ciclosporin microemulsion (Neoral). This effect is often reversible with ciclosporin dose reduction. Everolimus and sirolimus had only a minor influence on ciclosporin pharmacokinetics. Coadministration of ciclosporin significantly increases blood levels of everolimus and sirolimus.
Fibric acid derivatives. In graft recipients there have been isolated reports of considerable but reversible impairment of kidney function (with corresponding increase in serum creatinine) following concomitant administration of fibric acid derivatives (e.g. bezafibrate, fenofibrate). Kidney function must therefore be closely monitored in these patients. In the event of significant impairment of kidney function the comedication should be withdrawn.
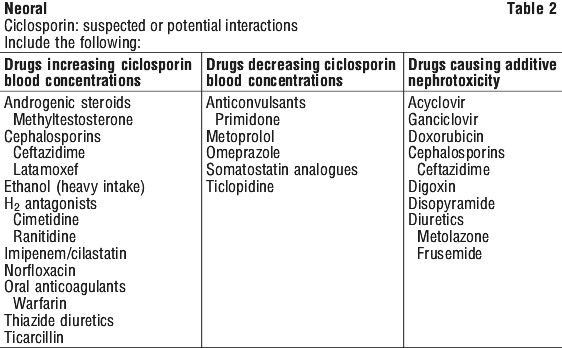
Cytochrome P450 isozymes, P-glycoprotein (Pgp). Various agents, as listed in Tables 1 and 2, are known to either increase or decrease the serum or whole blood concentrations of ciclosporin by competitive inhibition or induction of those hepatic enzymes involved in the metabolism or excretion of ciclosporin, in particular CYP3A4. Ciclosporin is a substrate of P-gp, hence inhibitors or inducers of P-gp may alter the concentrations of ciclosporin. Ciclosporin is also an inhibitor of CYP3A4 and of the multidrug efflux transporter P-glycoprotein and may increase plasma levels of comedications that are substrates of this enzyme and/or transporter. Thus it is recommended that coadministration of such drugs with ciclosporin be avoided. In situations where combined administration is unavoidable, the following basic recommendations should be observed:
In transplant patients: frequent monitoring of ciclosporin blood concentrations and appropriate modification of ciclosporin dosage are essential (see Section 4.2 Dose and Method of Administration, Clinical blood level monitoring). This is especially important during the introduction or withdrawal of the coadministered drug.
In nontransplant patients, where the relationship between blood level of ciclosporin and clinical effects is less well established, frequent assessment of renal function and careful monitoring for ciclosporin related side effects may be more appropriate.
Aliskiren, dabigatran, ambrisentan, bosentan. Ciclosporin may reduce the clearance of aliskiren, bosentan or dabigatran. Following concomitant administration of ciclosporin and aliskiren, the Cmax of aliskiren was increased by approximately 2.5-fold and the AUC by approximately 5-fold. However, the pharmacokinetic profile of ciclosporin was not significantly altered (see Section 4.4 Special Warnings and Precautions for Use).
Concomitant administration of dabigatran and ciclosporin leads to increased plasma levels of dabigatran due to the Pgp inhibitory activity of ciclosporin (see Section 4.4 Special Warnings and Precautions for Use). Dabigatran has a narrow therapeutic index and an increase in plasma level may be associated with an increased risk of bleeding.
Multiple dose administration of ambrisentan and ciclosporin in healthy volunteers resulted in an increase in ambrisentan exposure whilst ciclosporin exposure was marginally increased. Coadministration of bosentan and ciclosporin in healthy volunteers resulted in an increase in bosentan exposure and a decrease in ciclosporin exposure.
Food. The bioavailability of ciclosporin has been shown to increase when the drug is taken concomitantly with grapefruit juice or a fat rich meal. It has been suggested that grapefruit juice inhibits prehepatic metabolism of ciclosporin by the cytochrome P450 enzyme system in the wall of the gastrointestinal tract.
Nifedipine. The concurrent administration of nifedipine with ciclosporin may result in an increased rate of gingival hyperplasia compared with that observed when ciclosporin is given alone. The concomitant use of nifedipine should be avoided in patients in whom gingival hyperplasia develops as a side effect of ciclosporin (see Section 4.8 Adverse Effects (Undesirable Effects)). As nifedipine and amlodipine can cause gingival hyperplasia it is advised that these agents should be avoided in patients who develop gingival hypertrophy under ciclosporin.
Corticosteroids. It has been found that prednisolone clearance is reduced in patients treated with ciclosporin and that plasma concentrations of ciclosporin increase following the administration of high dose methylprednisolone.
Digoxin. Ciclosporin may reduce the clearance of digoxin, thereby causing digoxin toxicity. Severe digitalis toxicity has been seen within days of starting ciclosporin in several patients taking digoxin. The concomitant use of digoxin with ciclosporin should be carefully considered. If coadministration is necessary, close clinical observation is required in order to enable early detection of toxic manifestations of the drug, followed by reduction in dosage or drug withdrawal.
Colchicine, HMG-CoA reductase inhibitors, etoposide. Ciclosporin may also reduce the clearance of colchicine and HMG-CoA reductase inhibitors (statins) and etoposide, thereby enhancing the potential of these drugs to induce muscular toxicity, including muscle pain and weakness, myositis and occasionally rhabdomyolysis. There are reports on the potential of ciclosporin to enhance the toxic effects of colchicine such as myopathy and neuropathy, especially in patients with renal dysfunction. Literature and postmarketing cases of myotoxicity, including muscle pain and weakness, myositis and rhabdomyolysis, have been reported with concomitant administration of ciclosporin with lovastatin, simvastatin, atorvastatin, pravastatin and, rarely, fluvastatin. The concomitant use of these drugs with ciclosporin should be carefully considered. If coadministration is necessary, close clinical observation is required in order to enable early detection of toxic manifestations of the drug, followed by reduction in dosage or drug withdrawal. When concurrently administered with ciclosporin, the dosage of statins should be reduced according to label recommendations. Statin therapy needs to be temporarily withheld or discontinued in patients with signs and symptoms of myopathy or those with risk factors predisposing to severe renal injury, including renal failure, secondary to rhabdomyolysis.
Nonsteroidal anti-inflammatory drugs (NSAIDs). As nonsteroidal anti-inflammatory drugs alone can have an adverse effect on renal function, addition of these drugs to ciclosporin therapy or an increase in their dosage should initially be accompanied by particularly close monitoring of renal function. The concomitant use of diclofenac was found to result in a significant increase in the bioavailability of diclofenac, with a possible consequence of reversible renal function impairment. The increase in bioavailability of diclofenac is most probably caused by a reduction of its first-pass metabolism. If diclofenac is started during ciclosporin therapy, a dose of diclofenac at the lower end of the therapeutic range should be used initially. For most nonsteroidal anti-inflammatory drugs there is a lack of direct data on whether or not they interact with ciclosporin. However, if nonsteroidal anti-inflammatory drugs with low first-pass effect (e.g. aspirin) are given together with ciclosporin, no increase in their bioavailability is expected.
Recombinant human erythropoietin (rhEPO). As both recombinant human erythropoietin and ciclosporin are reported to increase blood pressure in a significant number of patients, caution should be exercised when administering these agents concomitantly.
Repaglinide. Ciclosporin may increase the plasma concentrations of repaglinide and thereby increase the risk of hypoglycaemia.
Cannabidiol. There have been reports of increased blood levels of another calcineurin inhibitor during concomitant use with cannabidiol. This interaction may occur due to inhibition of intestinal P-glycoprotein, leading to increased bioavailability of the calcineurin inhibitor. Ciclosporin and cannabidiol should therefore be co-administered with caution, closely monitoring for side effects. In transplant recipients, monitor ciclosporin whole blood trough concentrations and adjust the ciclosporin dose if needed. In non-transplant patients, monitoring of ciclosporin blood levels, with dose adjustment if needed, should be considered.
Interactions resulting in decrease of other drug levels. Concomitant administration of ciclosporin and mycophenolate sodium or mofetil in transplant patients may decrease the exposure of mycophenolic acid by 20-50% when compared with other immunosuppressants.
The coadministration of a single dose of ciclosporin (200 mg or 600 mg) with a single dose of eltrombopag (50 mg) decreased plasma eltrombopag AUCinf by 18% to 24% and Cmax by 25% to 39%. This decrease in exposure is not considered clinically meaningful.
This information should be taken into consideration when coadministering these drugs.

4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category C)
Ciclosporin was shown to be embryo and fetotoxic in rats and rabbits at doses toxic to dams (rat at 30 mg/kg per day and rabbit at 100 mg/kg per day orally). Toxicity was indicated by increased pre and postnatal mortality and reduced foetal weight together with related skeletal retardations. In the well tolerated dose range (rats up to 17 mg/kg per day and rabbits up to 30 mg/kg per day orally) ciclosporin did not demonstrate embryolethal or teratogenic effects.
In two published research studies, rabbits exposed to ciclosporin in utero (10 mg/kg/day subcutaneously) had reduced numbers of nephrons. These rabbits exhibited renal hypertrophy, systemic hypertension and progressive renal insufficiency when examined between 11 and 35 weeks of age in one study. The relevance of these findings for humans is unknown but cannot be dismissed.
The experience with using ciclosporin in pregnancy is still limited. Although there is no evidence that ciclosporin has a direct teratogenic effect in man, the use of immunosuppressive therapy in general is associated with a higher risk of complications in the mother and the infant. Pregnant women receiving immunosuppressive therapies after transplantation, including ciclosporin and ciclosporin containing regimens, are at risk of premature delivery (< 37 weeks). It appears that the risk of foetal growth retardation is increased in mothers taking immunosuppressants, including ciclosporin. In addition, a few cases of congenital abnormalities, within the normal range, have been reported in association with ciclosporin use during pregnancy. No causal relationship has been established.
Ciclosporin may cause immunosuppression in the infant. The long-term effects on the offspring of mothers who have been treated with ciclosporin have not been assessed. Limited observations in 34 children exposed to ciclosporin in utero are available, up to a median age of 3 years (range 6 months to 7 years). Renal function was normal in all children and blood pressure was normal in the 12 children in whom it was measured.
Female transplant recipients wishing to conceive should be informed of the above mentioned risks and, like all drugs, ciclosporin should be used in pregnancy only if the potential benefit justifies the potential risk to the foetus. In nontransplant patients, ciclosporin is not recommended in pregnancy where alternative treatments are available.
The ethanol content of Neoral formulations should also be taken into account in pregnant women (see Section 4.4 Special Warnings and Precautions for Use, Special excipient: ethanol).
Males treated with ciclosporin have been reported to have fathered normal children.
Use in lactation. Ciclosporin passes into the breast milk. Mothers receiving treatment with ciclosporin should not, therefore, breastfeed their infants. Because of the potential of Neoral to cause serious adverse drug reactions in breastfed newborns/ infants, a decision should be made whether to abstain from breastfeeding or to abstain from using the medicinal drug, taking into account the importance of the medicinal product to the mother.
The ethanol content of Neoral formulations should also be taken into account (see Section 4.4 Special Warnings and Precautions for Use, Special excipient: ethanol).
4.7 Effects on Ability to Drive and Use Machines
Neoral may cause neurological and visual disturbances (see Section 4.8 Adverse Effects (Undesirable Effects)). Caution should be exercised when driving a motor vehicle or operating machines. No studies on the effects of Neoral on the ability to drive and use machines have been performed.
4.8 Adverse Effects (Undesirable Effects)
The following adverse reactions observed with Sandimmun are also likely to occur with Neoral. They are usually dose dependent and responsive to dose reduction.
Evidence of renal or hepatic dysfunction warrants close monitoring of blood concentrations and possibly reduction in dose. In the various indications, the overall spectrum of side effects is essentially the same. However, there are differences in incidence and severity. As a consequence of the higher initial doses and longer maintenance therapy required after transplantation, side effects are more frequent in transplant patients than in patients treated for other indications.
Infections and infestations. Patients receiving immunosuppressive therapies, including ciclosporin and ciclosporin containing regimens, are at increased risk of infections (viral, bacterial, fungal, parasitic) (see Section 4.4 Special Warnings and Precautions for Use). Both generalised and localised infections can occur. Pre-existing infections may also be aggravated and reactivation of polyomavirus infections may lead to polyomavirus associated nephropathy (PVAN) or to JC virus associated progressive multifocal leukoencephalopathy (PML). Serious and/or fatal outcomes have been reported.
Neoplasms benign, malignant and unspecified (including cysts and polyps). Patients receiving immunosuppressive therapies, including ciclosporin and ciclosporin containing regimens, are at increased risk of developing lymphomas or lymphoproliferative disorders and other malignancies, particularly of the skin. The frequency of malignancies increases with the intensity and duration of therapy (see Section 4.4 Special Warnings and Precautions for Use). Some malignancies may be fatal.
Tabulated summary of adverse drug reactions. The adverse reactions (Table 3) are listed by MEDRA system organ class. Within each class, the adverse drug reactions are ranked by frequency, with the most frequent reactions first. Within each frequency grouping, adverse drug reactions are presented in order of decreasing seriousness. In addition the corresponding frequency category for each adverse drug reaction is based, using the following convention (CIOMS III):
Very common ≥ 10%; common ≥ 1% and < 10%; uncommon ≥ 0.1% and < 1%; rare ≥ 0.01% and < 0.1%; very rare < 0.01%.

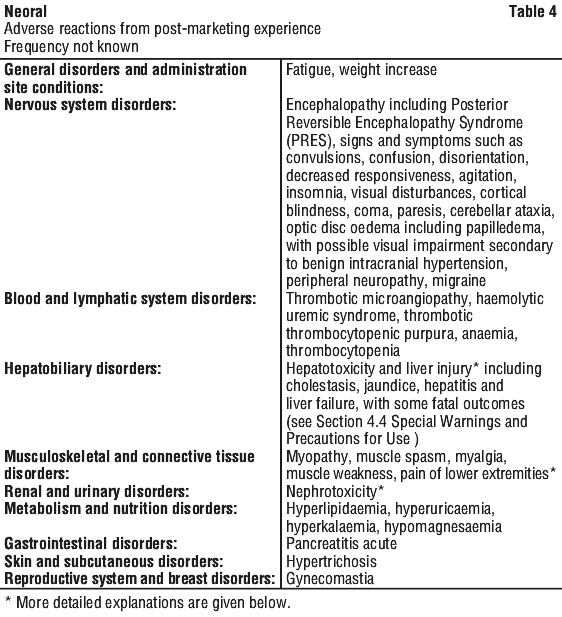
Postmarketing experience. In addition to the adverse events reported in the clinical trials, the following adverse reactions (Table 4) have been reported in postmarketing surveillance. These adverse drug reactions are obtained from spontaneous case reports and literature cases. Because these reactions are reported voluntarily from a population of uncertain size, it is not possible to reliably estimate their frequency, which is therefore categorized as frequency not known.
Acute and chronic nephrotoxicity. Patients receiving calcineurin inhibitors (CNIs) therapies, including ciclosporin and ciclosporin containing regimens are at increased risk of acute or chronic nephrotoxicity. There have been reports from clinical trials and from the postmarketing setting associated with the use of Neoral. Cases of acute nephrotoxicity reported disorders of ion homeostasis, such as hyperkalemia, hypomagnesemia, hyperuricemia. Cases reporting chronic morphological changes included arteriolar hyalinosis, tubular atrophy and interstitial fibrosis (see Section 4.4 Special Warnings and Precautions for Use).
Pain of lower extremities. Isolated cases of pain in lower extremities have been reported in association with ciclosporin. Pain of lower extremities has also been noted as part of calcineurin inhibitor induced pain syndrome (CIPS) as described in literature.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Symptoms. Experience on acute overdosage with oral ciclosporin is limited. Children up to the age of 4 years who had taken oral doses of up to 600 mg, and adults who had taken oral doses of up to 10 g (about 150 mg/kg), presented with only transient, nonserious adverse effects such as hypertension, increased serum creatinine, abnormal liver function test results and gastrointestinal disorders. A serious event has, however, been reported in a 11 year old liver transplant recipient who experienced a seizure associated with hypomagnesaemia following an oral dose of ciclosporin of 2600 mg.
Treatment. If indicated, symptomatic treatment and general supportive measures should be followed in cases of overdosage. Ciclosporin is not dialysable to any great extent, nor is it cleared well by charcoal haemoperfusion.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
Ciclosporin (BAN, AAN), ciclosporin (prop. INN), ciclosporine (USAN), also known as ciclosporin A. A cyclic polypeptide consisting of 11 amino acids.
It is produced as a metabolite by the fungus species Beauveria nivea (formerly Tolypocladium inflatum Gams).
Neoral is a microemulsion pre-concentrate formulation of ciclosporin. When mixed with water (either in the form of gastric fluid or a beverage), Neoral immediately forms a microemulsion which facilitates the gastrointestinal absorption of ciclosporin. Pharmacokinetic studies have demonstrated that Neoral provides more consistent ciclosporin absorption than the conventional oral forms of Sandimmun (see Section 5.2 Pharmacokinetic Properties). Also, in a small number of patients, conversion from Sandimmun to Neoral will result in a marked increase in ciclosporin exposure. It is therefore important when converting patients to Neoral to adhere to the recommendations given, see Section 4.2 Dose and Method of Administration, Conversion from Sandimmun to Neoral section.
5.1 Pharmacodynamic Properties
Pharmacotherapeutic group: immunosuppressive agents, calcineurin inhibitors.
ATC Code: L04A D01.
Mechanism of action. Ciclosporin is a potent immunosuppressive agent which prevents or delays rejection of solid organ allografts or xenografts in various animal models including skin, heart, kidney, pancreas, small intestine and lung.
It delays the onset of graft-versus-host disease after bone marrow transplantation in rodents. Successful kidney, pancreas, liver, heart, bone marrow and heart-lung allogeneic transplants have been performed in man using ciclosporin.
Ciclosporin may be used alone or with low dose corticosteroids in the prophylaxis of organ rejection following solid organ transplants. It may also be used in the treatment of transplant rejection in patients previously receiving other immunosuppressive agents.
Beneficial effects of ciclosporin therapy have also been shown in some cases of nephrotic syndrome, rheumatoid arthritis, psoriasis and atopic dermatitis (see Section 4.1 Therapeutic Indications). Ciclosporin is thought to be effective in these diseases since they are known or appear to be of autoimmune origin.
Studies in animals suggest that ciclosporin inhibits the development of cell mediated reactions, including allograft immunity, delayed cutaneous hypersensitivity, experimental allergic encephalomyelitis, Freund's adjuvant arthritis, graft versus host disease and also T cell dependent antibody production. It also inhibits lymphokine production and release including interleukin 2 or T cell growth factor (TCGF). Ciclosporin appears to block the resting lymphocytes in the G0 or G1 phase of the cell cycle, and inhibits the antigen triggered release of lymphokines by activated T cells.
All available evidence suggests that ciclosporin acts specifically and reversibly on lymphocytes. Unlike cytostatic agents, it does not depress haemopoiesis and has no effect on the function of phagocytic cells. Therefore, it may be expected that patients treated with ciclosporin may be less prone to infection than those receiving other immunosuppressive therapy. This was reported in two randomised trials of ciclosporin versus standard immunotherapy but was not found in a third.
Clinical trials. The clinical data submitted in Australia in support of the registration of Neoral included the results of multiple dose studies that compared ciclosporin pharmacokinetics for Neoral and Sandimmun in de novo and stable renal transplant recipients and in sufferers of severe psoriasis and rheumatoid arthritis, and studies that compared the safety and tolerability of Neoral and Sandimmun in de novo and stable renal transplant recipients over a period of 12 months. Data have also been generated on the use of Neoral in liver and heart transplantation, although these were not included in the Australian Neoral registration application.
Clinical trials on use in severe atopic dermatitis. The use of ciclosporin in severe atopic dermatitis was supported primarily by the results of two prospective, double blind, placebo controlled, crossover trials conducted over an 8 week treatment period (studies SIM 79 and SIM 80) and a double blind, placebo controlled, parallel group study conducted over a 6 week treatment period (study SIM 24). A dose of 5 mg/kg per day was used throughout these three studies. In addition, five open labelled studies were performed to examine relapse rates following ciclosporin withdrawal or the effects of long-term therapy and different dosing strategies. In one of these studies (SIM SF04), 5 mg/kg per day of the drug was given for 6 weeks, then the relapse rates were observed over a further 6 weeks; patients who relapsed were then given a second course of ciclosporin and monitored again for relapse. In the long-term studies, the dose of ciclosporin was adjusted according to response and side effects. In several of the open studies, patients were commenced on a low ciclosporin dose (2.5-3.0 mg/kg per day), which was then adjusted if necessary. In all of the clinical studies, the Sandimmun formulation of ciclosporin was used.
A total of 86 patients were treated with ciclosporin in the placebo controlled studies and about 250 patients were entered into the open labelled studies, of which 178 were involved in long-term studies (98 of whom were treated for at least 12 months). Most of the patients treated in the clinical programme were adults who had severe, long standing atopic dermatitis that was resistant to conventional therapy and/or caused significant suffering and disability. In the controlled studies and in most of the open label studies, the primary measures of efficacy were the area of skin involvement and the severity of the skin disease. Other measures included itch and loss of sleep scores, the extent of topical steroid use, and a patient assessment of symptoms.
The results of the placebo controlled studies demonstrated that ciclosporin is highly effective in the majority of patients with severe atopic dermatitis; only 5 of the 86 patients treated in these studies failed to respond to therapy. The results of the long-term studies showed that efficacy could be maintained at doses less than 5 mg/kg per day over the duration of these studies, although, due to their uncontrolled nature, it is difficult to assess the effects of the natural course of the disease on the long-term results. In study SIM SF04, 43% and 52% of patients relapsed 2 weeks after cessation of the first and second course of ciclosporin therapy, respectively; the relapse rate climbed to 71 and 87%, respectively, after 6 weeks. As expected, patients who commenced on a low ciclosporin dose followed by a dose adjustment did not respond as well initially as those who started on a high dose, but eventually the response rate for both groups was similar. While adverse events were frequent in the controlled studies, these were nevertheless consistent with the known side effect profile of ciclosporin. Nephrotoxicity and hypertension, normally a concern with ciclosporin use, were observed only in few patients in the short-term studies.
5.2 Pharmacokinetic Properties
Absorption. When Neoral is given, it provides dose linearity in ciclosporin exposure (AUCblood) and more consistent ciclosporin absorption from the gastrointestinal tract than Sandimmun. Neoral provides increased within-day consistency in whole blood trough concentrations and peak-trough fluctuations when compared with Sandimmun. Also, the dosing schedule of Neoral is less influenced by diurnal rhythm and food than that of Sandimmun. These properties combined yield a lower within-patient variability in pharmacokinetics of ciclosporin and a stronger correlation between trough concentration and total exposure (AUCblood). As a consequence of these additional advantages, the time schedule of Neoral administration need no longer take that of meals into account. In addition, Neoral produces a more uniform exposure to ciclosporin throughout the day and from day to day on a maintenance regimen.
Neoral capsules and Neoral oral solution are bioequivalent. The data available indicate that following a 1:1 conversion from the conventional oral forms of Sandimmun to Neoral, trough concentrations in whole blood are comparable, thereby remaining in the desired therapeutic trough concentration range (see Section 4.2 Dose and Method of Administration, Conversion from Sandimmun to Neoral). Compared to the conventional oral forms of Sandimmun, Neoral is more quickly absorbed (resulting in a 1 hour earlier mean tmax and a 59% higher mean Cmax) and exhibits, on average, a 29% higher bioavailability.
Distribution. Ciclosporin is distributed largely outside the blood volume with an apparent volume of distribution of 3.5 L/kg (average). Within the blood, distribution is concentration dependent, with 33-47% present in plasma, 4-9% in lymphocytes, 5-12% in granulocytes and 41-58% in erythrocytes. At higher concentrations the leucocytes and erythrocytes become saturated. In plasma, approximately 90% is bound to proteins, mostly lipoproteins.
Metabolism. Ciclosporin is extensively biotransformed to approximately 15 metabolites. There is no single major metabolic pathway. All metabolites identified so far contain the intact cyclic peptide structure of the parent compound. Major pathways consist of mono and dihydroxylation and N-demethylation at various positions. Hepatic dysfunction, as measured by a rise in serum bilirubin, may be associated with a proportional rise in ciclosporin blood concentrations.
Excretion. There is a high variability in the data reported on the terminal half-life of ciclosporin depending on the assay applied and on the target population. The terminal half-life ranged from 6.3 hours in healthy volunteers to 20.4 hours in patients with severe liver disease. Elimination is primarily biliary with only 6% of the oral dose excreted in the urine, of which only 0.1% is unchanged drug.
5.3 Preclinical Safety Data
Genotoxicity. Ciclosporin was not genotoxic in a series of assays for gene mutations and chromosomal damage. However, an assay for sister chromatid exchange in human lymphocytes in vitro gave indications of a positive effect at high concentrations.
Carcinogenicity. Carcinogenicity studies were carried out in rats and mice. A 78 week mouse study, at oral doses of 1, 4 and 16 mg/kg per day, revealed a trend towards increased incidences of lymphomas at the highest dose studied. In another study with ARK mice treated with 150 mg/kg ciclosporin in the diet, ciclosporin accelerated the development of lymphomas. In a 24 month rat study, conducted at oral doses of 0.5, 2 and 8 mg/kg per day, no significant increase in tumour incidence was reported, though the study had limited sensitivity. Ciclosporin enhanced the development of lymphomas induced in two strains of male mice by single whole body irradiation or N-methyl-N-nitrosourea. The increased incidence of lymphomas observed clinically in immunosuppressed patients may possibly be related to the degree of immunosuppression (see Section 4.4 Special Warnings and Precautions for Use, General). Malignancies including Kaposi's sarcoma have also been reported in ciclosporin treated patients.
6 Pharmaceutical Particulars
6.1 List of Excipients
Neoral soft gelatin capsules and oral solution contain dl-α-tocopherol, ethanol (see Section 4.4 Special Warnings and Precautions for Use), propylene glycol, corn glycerides, polyoxyl 40 hydrogenated castor oil. Neoral capsule shells contain gelatin, propylene glycol, glycerol, titanium dioxide, iron oxide black CI 77499 (25 and 100 mg capsules only). The printing ink contains carminic acid CI 75470, aluminium chloride, sodium hydroxide, hydroxypropyl methylcellulose 2910.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
For information on interactions with other medicines and other forms of interactions, see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Oral solution. Store between 15-30°C. Do not refrigerate.
Capsules. 10 mg: Store below 25°C.
25 mg, 50 mg, 100 mg: Store below 30°C.
6.5 Nature and Contents of Container
Neoral ciclosporin capsules. 10 mg: blister packs of 30 and 60 capsules.
25 mg: blister packs of 30, 50, and 60 capsules.
50 mg: blister packs of 30, 50, and 60 capsules.
100 mg: blister packs of 30, 50, and 60 capsules.
Neoral ciclosporin oral solution. 100 mg/mL: bottle of 50 mL with 1 mL and 4 mL dose dispensers.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Ciclosporin is poorly soluble in water (0.004% w/w) and n-hexane, but is very soluble in other organic solvents and in lipids.
Chemical structure.
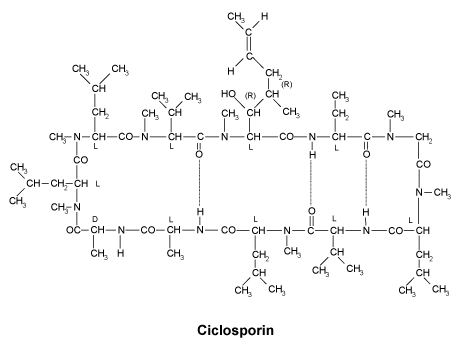
Chemical formula: C62H111N11O12.
Molecular weight: 1202.635.
CAS number. 59865-13-3.
7 Medicine Schedule (Poisons Standard)
Schedule 4.
Date of Revision
21 March 2024
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.