Noradrenaline BNM
Brand Information
| Brand name | Noradrenaline BNM |
| Active ingredient | Noradrenaline (norepinephrine) |
| Schedule | S4 |
Consumer medicine information (CMI) leaflet
Please read this leaflet carefully before you start using the Noradrenaline BNM
Summary CMI
Noradrenaline BNM
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I being treated with Noradrenaline BNM?
Noradrenaline BNM contains the active ingredient noradrenaline (norepinephrine) acid tartrate monohydrate.
Noradrenaline BNM is used to restore blood pressure back to normal.
For more information, see Section 1. Why am I being treated with Noradrenaline BNM? in the full CMI.
2. What should I know before I am given Noradrenaline BNM?
Do not use if you have ever had an allergic reaction to noradrenaline or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I am given Noradrenaline BNM? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Noradrenaline BNM and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How is Noradrenaline BNM given?
Your doctor will decide what dose you will receive. This depends on your condition and how you respond to Noradrenaline BNM often, the dose is adjusted according to how the medicine is affecting your blood pressure.
More instructions can be found in Section 4. How is Noradrenaline BNM given? in the full CMI.
5. What should I know while being given Noradrenaline BNM?
| Things you should do | Remind any doctor, dentist or pharmacist you visit that you are being given Noradrenaline BNM. |
| Driving or using machines | Do not drive or use any machines or tools until you know how Noradrenaline BNM affects you. |
For more information, see Section 5. What should I know while being given Noradrenaline BNM? in the full CMI.
6. Are there any side effects?
Side effects include skin and tissue damage close to the site of injection, anxiety, headache, dizziness, low or irregular heartbeat, tingling or numbness in the feet, and coldness, numbness or discolouration of your limbs.
Serious side effects include allergic reaction symptoms (shortness of breath, wheezing or difficulty breathing, swelling of the face, lips, mouth, throat or tongue or other parts of the body and, rash, itching or hives on the skin), Very high blood pressure symptoms (headache, dizziness, sensitivity to light, chest pain, slow heart-beat, sweating and pale skin, vomiting, and trouble breathing and shortness of breath).
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
1. Why am I being treated with Noradrenaline BNM?
Noradrenaline BNM contains the active ingredient noradrenaline (norepinephrine) acid tartrate monohydrate.
Noradrenaline BNM belongs to a group of medicines called sympathomimetic amines. It works by making the heartbeat more forcefully and constricting blood vessels. This results in a higher blood pressure and greater blood flow to the heart and other organs of the body. Noradrenaline BNM is used to restore blood pressure back to normal.
Noradrenaline BNM is given in emergency situations because of medical conditions which cause a dramatic drop in blood pressure. Such situations/conditions could be:
- associated with surgical procedures
- infections caused by poliovirus
- a heart attack
- infection in the blood
- severe reaction to a medicine
- severe reaction to a blood transfusion
- allergic reactions.
It is given only by a doctor or nurse, usually in hospital.
Ask your doctor if you have any questions about why it has been prescribed for you.
Noradrenaline BNM is only available with a doctor's prescription.
There is no evidence that Noradrenaline BNM is addictive.
2. What should I know before I am given Noradrenaline BNM?
Warnings
Noradrenaline BNM is not suitable for everyone. You must not be given Noradrenaline BNM if:
- You are allergic to noradrenaline (norepinephrine), or any other similar medicines such as adrenaline, phenylephrine, pseudoephedrine, or any of the ingredients listed at the end of this leaflet.
- You have
- low blood pressure due to low blood volume
- blood clots in certain vessels.
If given in these cases, it can further reduce blood supply to important organs.
- Always check the ingredients to make sure you can use this medicine.
Check with your doctor if you:
- have allergies to any other medicines and/or substances, such as foods, preservatives or dyes.
- develop a rash or other symptoms of an allergic reaction. These symptoms may be:
- shortness of breath, wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin. - have any other medical conditions
- have or have had an overactive thyroid gland.
- you are over 65 years old. Elderly persons may be more sensitive to the effects of the medicine.
- take any medicines for any other condition
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Noradrenaline BNM may be given to pregnant women and women who are breastfeeding, but your doctor must be told if you are pregnant or breastfeeding.
Your doctor will discuss the possible risks and benefits of you being given Noradrenaline BNM during pregnancy and breastfeeding.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Noradrenaline BNM and affect how it works.
These include:
- antidepressants including moclobemide, imipramine and amitriptyline.
- Guanethidine
The above medicines may be affected by Noradrenaline BNM or may affect how well it works. You may need different amounts of Noradrenaline BNM, or you may need to use different medicines.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Noradrenaline BNM.
Your doctor and pharmacist may have more information on medicines to be careful with or avoid while being given Noradrenaline BNM.
4. How is Noradrenaline BNM given?
How much is given
Your doctor will decide how many infusions you need, and how often you should receive them. The need for more doses will depend on how the medicine is affecting your blood pressure.
How Noradrenaline BNM is given
Noradrenaline BNM must be diluted before use and will be given by slow intravenous infusion (drip) into a large vein. This medicine must only be given by a doctor or a nurse.
How long it will be given
This medicine will be given until blood pressure and blood circulation to all organs is back to normal and remains normal even when the medicine is no longer given.
If you are given too much Noradrenaline BNM
As Noradrenaline BNM is given to you in hospital under the supervision of your doctor, it is very unlikely that you will receive an overdose.
Symptoms of an overdose may include headache, dizziness, sensitivity to light, slow heartbeat, chest pain, sweating and pale skin, vomiting, or trouble breathing and shortness of breath.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
Immediately tell your doctor or nurse if you think that you or anyone else may have been given too much Noradrenaline BNM. They have information on how to recognise and treat an overdose.
5. What should I know while being given Noradrenaline BNM?
Things you should do
If you are about to be started on any new medicine, tell your doctor and pharmacist that you are being given Noradrenaline BNM. Likewise, tell any other doctors, dentists and pharmacists who are treating you that you are being given this medicine.
Call your doctor straight away if you:
develop a rash or other symptoms of an allergic reaction.
These symptoms may be:
- shortness of breath, wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Noradrenaline BNM affects you.
Looking after your medicine
- Noradrenaline BNM will be stored in the pharmacy or on the ward under the recommended storage conditions.
- Do not refrigerate or freeze. Store it in a cool dry place away from moisture, heat or sunlight. Keep where the temperature stays below 25°C.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Ask your doctor or pharmacist to answer any questions you may have.
Less serious side effects
| Less serious side effects | What to do |
Administration site conditions
| Speak to your doctor or nurse if you have any of these side effects. |
Serious side effects
| Serious side effects | What to do |
| serious allergic reaction related symptoms of an allergic reaction
| Call your doctor straight away or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. This is a very serious side effect. you may need urgent medical attention. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects, you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Noradrenaline BNM contains
| Active ingredient (main ingredient) | noradrenaline (norepinephrine) |
| Other ingredients (inactive ingredients) |
|
Noradrenaline BNM is sulfite free and does not contain any preservatives.
Do not take this medicine if you are allergic to any of these ingredients.
What Noradrenaline BNM looks like
Noradrenaline BNM is a clear sterile concentrated solution for injection.
It is available in 1 mL and 4 mL glass ampoules. A box contains 10 ampoules.
Each 1 mL ampoule contains 1 mg of noradrenaline (norepinephrine) base.
Not all presentations may be available.
Australian Registration Number:
1 mg/1 mL: AUST R 248492
4 mg/4 mL: AUST R 248512
Who distributes Noradrenaline BNM
Boucher & Muir Pty Ltd
Level 9, 76 Berry Street
North Sydney NSW 2060
Distributed in New Zealand by:
BNM Group
39 Anzac Road
Browns Bay
Auckland 0753
This leaflet was prepared in October 2025.
Brand Information
| Brand name | Noradrenaline BNM |
| Active ingredient | Noradrenaline (norepinephrine) |
| Schedule | S4 |
MIMS Revision Date: 01 October 2022
1 Name of Medicine
Noradrenaline (norepinephrine) (as acid tartrate monohydrate).
2 Qualitative and Quantitative Composition
Each 1 mL ampoule contains noradrenaline (norepinephrine) 1 mg in 1 mL (1:1000), present as 2 mg of noradrenaline (norepinephrine) acid tartrate monohydrate in 1 mL.
Each 4 mL ampoule contains noradrenaline (norepinephrine) 4 mg in 4 mL (1:1000), present as 8 mg of noradrenaline (norepinephrine) acid tartrate monohydrate in 4 mL.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Noradrenaline BNM is a sterile, clear colourless concentrated solution for injection available in ampoules.
4 Clinical Particulars
4.1 Therapeutic Indications
For the restoration of blood pressure in certain acute hypotensive states (e.g. phaeochromocytomectomy, sympathectomy, poliomyelitis, spinal anaesthesia, myocardial infarction, septicaemia, blood transfusion and drug reactions).
As an adjunct in the treatment of cardiac arrest. To restore and maintain an adequate blood pressure after an effective heartbeat and ventilation have been established by other means.
4.2 Dose and Method of Administration
Noradrenaline BNM is a concentrated solution for injection which must be diluted in glucose containing solutions prior to infusion. An infusion of Noradrenaline BNM should be given into a large vein (see Section 4.4 Special Warnings and Precautions for Use).
Noradrenaline BNM must be administered in 5% glucose solution in distilled water or 5% glucose in saline solution, and must not be administered in saline solution alone. Whole blood or plasma, if indicated to increase blood volume, should be administered separately.
This product contains no antimicrobial preservative; to reduce microbiological hazard use as soon as practicable after preparation. If storage is necessary, hold at 2 to 8°C for not more than 24 hours. In-use storage times and conditions prior to use are the responsibility of the user.
Noradrenaline BNM is for single use in one patient only. Discard any residue. Discoloured solutions or those containing a precipitate should not be used. Avoid contact with iron salts, alkalis or oxidising agents.
Restoration of blood pressure in acute hypotensive states. Blood volume depletion should always be corrected as fully as possible before any vasopressor is administered. When, as an emergency measure, intra-aortic pressures must be maintained to prevent cerebral or coronary artery ischaemia, Noradrenaline BNM can be administered before and concurrently with blood volume replacement.
Average dosage. Add 1 mL of Noradrenaline BNM (1:1000) to 250 mL, or 4 mL of Noradrenaline BNM (1:1000) to 1 litre, of 5% glucose solution. Each 1 mL of this dilution contains 4 microgram of noradrenaline (norepinephrine) (= 8 microgram of noradrenaline (norepinephrine) acid tartrate monohydrate). Give this dilution intravenously via a catheter well advanced centrally into the vein and securely fixed, if possible, avoiding a catheter tie-in technique as it promotes stasis. A drip bulb is necessary to permit an accurate estimation of the rate of flow in drops per minute.
After observing the response to an initial dose of 2 to 3 mL (8 to 12 microgram of base) per minute, adjust the rate of flow to establish and maintain a low normal blood pressure (usually 80 to 100 mmHg systolic) sufficient to maintain the circulation to vital organs. In previously hypertensive patients, it is recommended that the blood pressure should be raised no higher than 40 mmHg below the pre-existing systolic pressure.
The average maintenance dose ranges from 0.5 to 1 mL per minute (2 to 4 microgram of base). Great individual variation occurs in the dose required to attain and maintain an adequate blood pressure. In all cases, dosage of Noradrenaline BNM should be titrated according to the response of the patient. Occasionally much larger daily doses (as high as 68 mg base or 68 x 1 mL ampoules or 17 x 4 mL ampoules) may be necessary if the patient remains hypotensive, but occult blood volume depletion should always be suspected and corrected when present. Dilution can be varied depending on the clinical fluid volume requirement.
Duration of therapy. The infusion should be continued until adequate blood pressure and tissue perfusion are maintained without therapy. The infusion rate should then be reduced gradually avoiding abrupt withdrawal. In some of the reported cases of vascular collapse due to acute myocardial infarction, treatment was required for up to six days.
Adjunctive treatment in cardiac arrest. Infusions of Noradrenaline BNM are usually administered intravenously during cardiac resuscitation to restore and maintain an adequate blood pressure after an effective heartbeat and ventilation have been established by other means. Noradrenaline's (norepinephrine's) beta-adrenergic stimulating action is also thought to increase the strength and effectiveness of systolic contractions once they occur.
Average dosage. To maintain systemic blood pressure during the management of cardiac arrest, Noradrenaline BNM is used in the same manner as described under Restoration of blood pressure in acute hypotensive states.
Paediatric use. Safety and effectiveness in paediatric patients has not been established.
Use in the elderly. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Noradrenaline BNM infusion solutions should not be administered into the veins in the leg in elderly patients.
4.3 Contraindications
Noradrenaline BNM should not be given to patients who are hypotensive from hypovolaemia except as an emergency measure to maintain coronary and cerebral artery perfusion until blood volume replacement therapy can be completed.
If Noradrenaline BNM is continuously administered to maintain blood pressure in the absence of blood volume replacement, the following may occur: severe peripheral and visceral vasoconstriction, decreased renal perfusion and urine output, poor systemic blood flow despite "normal" blood pressure, tissue hypoxia and lactate acidosis.
Noradrenaline BNM should not be given to patients with mesenteric or peripheral vascular thrombosis (because of the risk of increasing ischaemia and extending the area of infarction) unless, in the opinion of the attending physician, the administration of Noradrenaline BNM is necessary as a lifesaving procedure.
Ventricular tachycardia or fibrillation cardiac arrhythmias may result from the use of noradrenaline (norepinephrine) in patients with profound hypoxia or hypercarbia.
Hypersensitivity to noradrenaline (norepinephrine) or any of the excipients.
4.4 Special Warnings and Precautions for Use
Noradrenaline BNM should be used with extreme caution in patients receiving monoamine oxidase (MAO) inhibitors or antidepressants of the triptyline or imipramine types because severe, prolonged hypertension may result.
General. Particular caution should be observed in patients with coronary, mesenteric or peripheral vascular thrombosis because noradrenaline (norepinephrine) may increase the ischemia and extend the area of infarction. Similar caution should be observed in patients with hypotension following myocardial infarction, in patients with Prinzmetal's variant angina, and in patients with diabetes, hypertension or hyperthyroidism.
Noradrenaline (norepinephrine) should only be administered by healthcare professionals who are familiar with its use.
Noradrenaline (norepinephrine) should be used with caution in patients who exhibit profound hypoxia or hypercarbia.
Avoid hypertension. Because of the potency and varying responses to noradrenaline (norepinephrine), the possibility exists that hypertension may be produced with overdoses of this pressor agent. Hence it is desirable to record the blood pressure every two minutes from the time administration is started until the desired blood pressure is obtained, and then every five minutes if administration is to be continued. The rate of flow must be watched constantly, and the patient should not be left unattended whilst receiving Noradrenaline BNM. Headache may be a symptom of hypertension due to overdose.
Hypersensitivity. Certain patients may be hypersensitive to the effects of noradrenaline (norepinephrine), e.g. patients with hyperthyroidism (see Section 4.8 Adverse Effects (Undesirable Effects)).
Site of infusion. Noradrenaline BNM should be given into a large vein, particularly an antecubital vein, because when administered into this vein, the risk of necrosis of the overlying skin from prolonged vasoconstriction is apparently very slight. The femoral vein is also an acceptable route of administration. A catheter tie in technique should be avoided if possible, since the obstruction to blood flow around the tubing may cause stasis and increased local concentration of noradrenaline (norepinephrine). As occlusive vascular diseases are more likely to occur in the lower rather than in the upper extremity, the leg veins in elderly patients or in those suffering from such disorders should be avoided. Gangrene has been reported in a lower extremity when infusions of noradrenaline (norepinephrine) were given in an ankle vein.
Extravasation. The infusion site should be checked frequently for free flow. Care should be taken to avoid extravasation of Noradrenaline BNM into the tissues as local necrosis might ensue due to the vasoconstrictive action of the drug. Blanching along the course of the infused vein, sometimes without obvious extravasation, has been attributed to vasa vasorum constriction with increased permeability of the vein wall, permitting some leakage. This may also progress on rare occasions to superficial slough, particularly during infusion into leg veins in elderly patients or in those suffering from obliterative vascular disease. Hence, if blanching occurs, consideration should be given to changing the infusion site at intervals to allow the effects of local vasoconstriction to subside.
The antidote for extravasation ischaemia is phentolamine. To prevent sloughing and necrosis in areas in which extravasation has occurred, the area should be infiltrated as soon as possible with 10 mL to 15 mL of saline solution containing 5 mg to 10 mg of phentolamine. Using a syringe with a fine hypodermic needle, the solution is infiltrated liberally throughout the area. Sympathetic blockade with phentolamine causes immediate and conspicuous local hyperaemic changes if the area is infiltrated within 12 hours. Therefore, phentolamine should be given as soon as possible after extravasation is noted.
Fluid replacement. Noradrenaline (norepinephrine) should be used only in conjunction with appropriate blood volume replacement. When infusing noradrenaline (norepinephrine), the blood pressure and rate of flow should be checked frequently to avoid hypertension.
Prolonged administration of any potent vasopressor may result in plasma volume depletion which should be continuously corrected by appropriate fluid and electrolyte replacement therapy. If plasma volumes are not corrected, hypotension may recur when the infusion is discontinued, or blood pressure may be maintained at the risk of severe peripheral and visceral vasoconstriction (e.g. decreased renal perfusion) with reduced blood flow and tissue perfusion with subsequent tissue hypoxia and lactic acidosis, and possible ischemic injury.
Use in the elderly. Clinical studies of noradrenaline (norepinephrine) injection 1:1000 did not include sufficient numbers of subjects aged 65 and over to determine whether they respond differently from younger subjects. In general, dose selection for an elderly patient should be cautious, usually starting at the low end of the dosing range, reflecting the greater frequency of decreased hepatic, renal, or cardiac function, and of concomitant disease or other drug therapy.
Noradrenaline BNM infusion solutions should not be administered into the veins in the leg in elderly patients.
Withdrawal of therapy. The noradrenaline (norepinephrine) infusion should be gradually decreased since abrupt withdrawal can result in acute hypotension.
Paediatric use. Safety and effectiveness in paediatric patients has not been established. The use of noradrenaline (norepinephrine) in children is not recommended.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Noradrenaline BNM infusion solutions should not be mixed with other medicines. Extreme caution should be exercised in patients receiving monoamine oxidase (MAO) inhibitors and antidepressants of the triptyline or imipramine types (see Section 4.4 Special Warnings and Precautions for Use). Linezolid, adrenergic-serotonergic drugs or any other cardiac sensitizing agents are not recommended because severe, prolonged hypertension and possible arrhythmias may result.
Guanethidine. The effects of noradrenaline (norepinephrine) may be enhanced by guanethidine.
Please see Section 6.2 Incompatibilities.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Studies have not been performed.
Use in pregnancy. (Category B3)
Noradrenaline BNM should be given to a pregnant woman only if clearly needed.
Animal studies indicate noradrenaline (norepinephrine) may impair placental perfusion and induce foetal bradycardia. It may also exert a contractile effect on the pregnant uterus and lead to foetal asphyxia in late pregnancy. However, the clinical significance of these changes to a human foetus is unknown. These possible risks to the foetus should therefore be weighed against the potential benefit to the mother.
Use in lactation. It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when Noradrenaline BNM is administered to a nursing woman.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration. Patients should refrain from driving or using machines until they know that the medicinal product does not negatively affect these abilities.
4.8 Adverse Effects (Undesirable Effects)
Prolonged administration of any potent vasopressor may result in plasma volume depletion which should be continuously corrected by appropriate fluid and electrolyte replacement therapy. If plasma volumes are not corrected, hypotension may recur when Noradrenaline BNM is discontinued, or blood pressure may be maintained at the risk of severe peripheral and visceral vasoconstriction (e.g. decreased renal perfusion) with diminution in blood flow and tissue perfusion with subsequent tissue hypoxia and lactic acidosis and possible ischaemic injury. Gangrene of extremities has been rarely reported. Bradycardia sometimes occurs, probably as a reflex result of a rise in blood pressure. Overdoses or conventional doses in hypersensitive persons (e.g. hyperthyroid patients) cause severe hypertension with violent headache, photophobia, stabbing retrosternal pain, pallor, intense sweating and vomiting.
The following reactions can occur.
Body as a whole. Ischaemic injury due to potent vasoconstrictor action and tissue hypoxia. Gangrene, hypertension, plasma depletion.
Cardiovascular system. Bradycardia, probably as a reflex of a rise in blood pressure, cardiogenic shock, arrhythmias and stress cardiomyopathy.
Nervous system. Anxiety, transient headache.
Respiratory system. Respiratory difficulty, dyspnea.
Skin and appendages. Extravasation necrosis at injection site.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
Overdosage with Noradrenaline BNM may result in severe hypertension, reflex bradycardia, marked increase in peripheral resistance and decreased cardiac output. Headache may indicate severe hypertension. Pulmonary oedema, photophobia, retrosternal pain, pallor, intense sweating and vomiting may occur. In the event of overdose, treatment with noradrenaline (norepinephrine) should be withdrawn and appropriate corrective measures initiated.
For information on the management of overdose, contact the Poison Information Centre in Australia on 13 11 26; in New Zealand on 0800 764 766.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Noradrenaline (norepinephrine), a sympathomimetic amine, acts predominantly on α-receptors and on β-receptors in the heart. It therefore causes peripheral vasoconstriction (α-adrenergic action), and a positive inotropic effect on the heart and dilation of coronary arteries (β-adrenergic action). These actions result in an increase in systemic blood pressure and coronary artery blood flow. In myocardial infarction accompanied by hypotension, noradrenaline (norepinephrine) usually increases aortic blood pressure, coronary artery blood flow, and myocardial oxygenation, thereby helping to limit the area of myocardial ischaemia and infarction. Venous return is increased and the heart tends to resume a more normal rate and rhythm than in the hypotensive state. In hypotension that persists after correction of blood volume deficits, noradrenaline (norepinephrine) helps raise the blood pressure to an optimal level and establish a more adequate circulation.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
No data available.
5.3 Preclinical Safety Data
Genotoxicity. Studies have not been performed.
Carcinogenicity. Studies have not been performed.
6 Pharmaceutical Particulars
6.1 List of Excipients
Sodium chloride and water for injections.
6.2 Incompatibilities
Infusion solutions containing noradrenaline (norepinephrine) acid tartrate monohydrate have been reported to be incompatible with iron salts, alkalis and oxidising agents, barbiturates, chlorphenamine maleate, chlorothiazide, nitrofurantoin, phenytoin, sodium bicarbonate, sodium iodide, streptomycin sulfate, sulfadiazine and sulfafurazole (see Section 4.2 Dose and Method of Administration).
Please see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25°C. Do not refrigerate or freeze. Protect from light.
6.5 Nature and Contents of Container
Noradrenaline BNM is available as 1 mg/1 mL and 4 mg/4 mL single use glass ampoules. Both presentations are supplied in packs of 10 ampoules per carton.
Not all presentations may be distributed in Australia and New Zealand.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure.
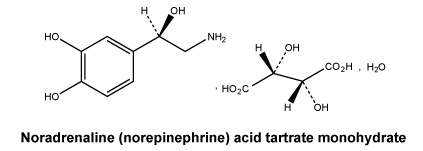
Molecular weight: 337.3.
Chemically, noradrenaline (norepinephrine) acid tartrate monohydrate: (1R)-2-amino-1-(3,4-dihydroxyphenyl) ethanol hydrogen (2R,3R)-2,3-dihydroxybutanedioate monohydrate is a white or almost white crystalline powder. It is freely soluble in water and slightly soluble in ethanol (96%).
Noradrenaline BNM concentrated solution for injection has a pH of 3.0 to 4.5.
CAS number. 69815-49-2.
7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Date of First Approval
20 July 2016
Date of Revision
15 August 2022
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. NPS MedicineWise disclaims all liability (including for negligence) for any loss, damage or injury resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.