Normison
Brand Information
| Brand name | Normison |
| Active ingredient | Temazepam |
| Schedule | S4 |
Consumer medicine information (CMI) leaflet
Please read this leaflet carefully before you start using the Normison
Summary CMI
NORMISON tablets
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using NORMISON?
NORMISON contains the active ingredient temazepam. NORMISON is used to treat sleeping problems, also called insomnia.
For more information, see Section 1. Why am I using NORMISON? in the full CMI.
2. What should I know before I use NORMISON?
Do not use if you have ever had an allergic reaction to temazepam or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use NORMISON? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with NORMISON and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use NORMISON?
- The dose of NORMISON varies from patient to patient.
- Your doctor will tell you how many tablets you need to take each day and when to take them.
- Swallow NORMISON with a glass of water. It can be taken with or without food.
More instructions can be found in Section 4. How do I use NORMISON? in the full CMI.
5. What should I know while using NORMISON?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using NORMISON? in the full CMI.
6. Are there any side effects?
Tell your doctor or pharmacist a soon as possible if you do not feel well while you are taking NORMISON. Some mild side effects include dizziness, drowsiness, light-headedness, headache, nausea, stomach pain and dry mouth. Some serious side effects which may require medical attention include confusion, loss of memory, disorientation, behavioural or mood changes, signs of frequent infections such as fever, and yellowing of the eyes and skin (jaundice).
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
1. Why am I using NORMISON?
NORMISON contains the active ingredient temazepam, a benzodiazepine which is thought to work by acting on the brain chemicals.
NORMISON is used to treat sleeping problems, also called insomnia.
Ask your doctor if you have any questions about why NORMISON has been prescribed for you.
Your doctor may have prescribed it for another reason.
2. What should I know before I use NORMISON?
In general, benzodiazepines such as NORMISON should be taken for short periods only (for example 2 to 4 weeks). Continuous long-term use is not recommended unless advised by your doctor.
The use of benzodiazepines may lead to dependence on the medicine.
Warnings
Do not use NORMISON if:
- you are allergic to temazepam, any other benzodiazepine medicines or any of the ingredients listed at the end of this leaflet.
- you have severe and chronic lung disease or breathing difficulties
- you have sleep apnoea.
- you suffer from depression with or without anxiety problems. Temazepam can increase thoughts of death or suicide.
- the packaging shows signs of tampering or the tablets do not look quite right.
- the expiry date printed on the pack has passed.
Check with your doctor if you:
- have any medical conditions, especially the following:
- liver, kidney or lung problems
- epilepsy, fits or convulsions
- severe muscle weakness known as myasthenia gravis
- low blood pressure as it may lead to heart or brain complications.
- any blood disorder
- glaucoma (increased pressure in the eye)
- depression, psychosis or schizophrenia
- drug or alcohol dependence or a past history of these problems. - take any medicines for any other condition
- are allergic to any other medicines, foods, dyes or preservatives
- plan to have surgery.
NORMISON is not recommended for use in children under 16 years of age, as its safety and effectiveness have not been established in this age group.
Suddenly stopping NORMISON in patients with epilepsy can cause a temporary increase in the number and severity of seizures.
Your doctor may have prescribed NORMISON for depression or psychosis. NORMISON is not recommended as the first choice of treatment for depression and psychosis. It may increase depression, worsen mental illness, suicidal thoughts and actions.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
NORMISON should not be used during pregnancy.
It should be avoided during the first trimester of pregnancy as it may increase the risk of defects present at birth.
It may cause unwanted effects in the newborn baby if taken during the late phase of pregnancy or during childbirth, such as low muscle strength, shallow breathing and feeding problems.
Your doctor will discuss the risks and benefits of taking NORMISON during pregnancy.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
NORMISON may pass into breast milk and cause drowsiness and feeding difficulties in the baby. Your doctor will discuss the risks and benefits of taking NORMISON when breastfeeding.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with NORMISON and affect how it works. These include:
- other sleeping tablets, sedatives or tranquillisers
- medicines for depression, schizophrenia and other mental illnesses
- medicines to treat epilepsy and fits
- medicines for allergies, for example antihistamines or cold tablets
- medicines used to treat Parkinson's disease
- theophylline, a medicine used to treat asthma
- muscle relaxants
- some pain relievers.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect NORMISON.
4. How do I use NORMISON?
How much to take
- The usual starting dose is one 10 mg tablet each night.
- The dose of NORMISON varies from patient to patient.
- Your doctor will tell you how many tablets you need to take each day. This depends on your condition and whether you are taking any other medicines.
- Elderly people over 65 years of age may need smaller doses.
When to take NORMISON
- NORMISON is usually used for short periods only (such as 2 to 4 weeks).
- Your doctor will tell you when to take NORMISON.
This depends on your condition and whether you are taking any other medicines. - Continuous long-term use is not recommended unless advised by your doctor. The use of benzodiazepines may lead to dependence on the medicine.
- Follow the instructions provided and use NORMISON until your doctor tells you to stop.
How to take NORMISON
- Swallow NORMISON with a glass of water.
- NORMISON can be taken with or without food.
If you forget to use NORMISON
NORMISON should be used regularly at the same time each day.
If you forget to take NORMISON before you go to bed and you wake up late in the night or early in the morning, do not take NORMISON as you may have trouble waking in the morning.
If it is almost time for your next dose, skip the dose you missed and take your next dose when you are meant to.
Do not take a double dose to make up for the dose you missed.
This may increase the chance of you getting an unwanted side effect.
If you are unsure about whether to take your next dose, speak to your doctor or pharmacist.
If you take too many NORMISON tablets
If you think that you have taken too many NORMISON tablets, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
Symptoms of an overdose include feeling drowsy, confused, tired, dizzy, breathing difficulty, feeling weak, low blood pressure, lacking mobility and coordination, being unconscious, or rarely it may be fatal.
5. What should I know while using NORMISON?
Things you should do
- Take NORMISON exactly as your doctor has prescribed.
- If you have to have any blood tests, tell your doctor that you are taking NORMISON.
It may affect the result of some tests. - Visit your doctor regularly so they can check on your progress.
Your doctor will check your condition to see whether you should continue to take NORMISON. - Tell your doctor if you feel this medicine is not helping your condition.
- Tell your doctor or a mental health professional immediately if you have any suicidal thoughts or other mental/mood changes.
All thoughts of suicide must be taken seriously. - Keep enough NORMISON to last weekends and holidays.
Be careful if you are elderly, unwell or taking other medicines.
Some people may experience side effects such as drowsiness, confusion, dizziness and unsteadiness, which may increase the risk of a fall.
Remind any doctor, dentist or pharmacist you visit that you are using NORMISON.
Call your doctor straight away if you:
- feel this medicine is not helping your condition.
- become pregnant while you are taking NORMISON.
Things you should not do
- Do not stop taking your medicine or change the dose, without first checking with your doctor.
- Do not take NORMISON for a longer time than your doctor has prescribed.
- Do not use this medicine to treat any other complaints unless your doctor tells you to.
- Do not give it to anyone else, even if they have the same condition as you.
Stopping NORMISON suddenly may cause some unwanted withdrawal effects. It is more common in patients that have received high doses over longer periods of time.
Withdrawal symptoms include insomnia, anxiety, unusual mood, panic attacks, dizziness, light sensitivity, confusion, (seeing or hearing things that are not real (hallucinations), vomiting, sweating, fits (convulsions), a feeling of loss of identity/feeling detached from yourself (depersonalisation or derealisation) and loss of short-term memory.
Your doctor may want you to gradually reduce the amount you are taking before stopping completely. This may help reduce the possibility of unwanted side effects.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how NORMISON affects you.
NORMISON may cause drowsiness or dizziness in some people. If you take NORMISON at night, you may still be drowsy or dizzy the next day.
Make sure you know how you react to NORMISON before you drive a car, operate machinery, or do anything else that could be dangerous if you are drowsy, dizzy or not alert.
Drinking alcohol
Tell your doctor if you drink alcohol.
Combining NORMISON and alcohol can make you more sleepy, dizzy or light-headed or increase the risk of sleep walking and some other related sleep behaviours which may include sleep-driving, making phone calls, or preparing and eating food while asleep.
Your doctor may suggest that you avoid alcohol or reduce the amount of alcohol you drink while you are taking NORMISON.
Looking after your medicine
Keep this medicine protected from moisture, where the temperature stays below 30°C
Store it in a cool dry place, away from heat or sunlight.
For example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Heat and dampness can destroy some medicines.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor immediately or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
| Go straight to the Emergency Department at your hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What NORMISON contains
| Active ingredient (main ingredient) |
|
| Other ingredients (inactive ingredients) |
|
| Potential allergens |
|
Do not take this medicine if you are allergic to any of these ingredients.
What NORMISON looks like
NORMISON tablets are white to off-white, round, biconvex tablets with one side plain and the other with a stylised “S”. Available in blister packs of 25 tablets.
Australian Registration Number: AUST R 41637
Who distributes NORMISON
In Australia:
Aspen Pharmacare Australia Pty Ltd
34-36 Chandos Street
St Leonards NSW 2065
In New Zealand:
Pharmacy Retailing (NZ) Ltd t/a
Healthcare Logistics
58 Richard Pearse Drive
Airport Oaks
Mangere, Auckland
Telephone: (09) 9185 100
Fax: (09) 9185 101
This leaflet was revised in October 2025.
Brand Information
| Brand name | Normison |
| Active ingredient | Temazepam |
| Schedule | S4 |
MIMS Revision Date: 01 October 2024
1 Name of Medicine
Temazepam.
2 Qualitative and Quantitative Composition
Normison tablets contain temazepam 10 mg as the active ingredient.
Excipients with known effect. Sugars as lactose.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Normison 10 mg are white to off-white, round, biconvex tablet, with stylised "S" on one side and plain on the other side.
4 Clinical Particulars
4.1 Therapeutic Indications
Adjunctive therapy in the short term management of insomnia in adults.
4.2 Dose and Method of Administration
Dosage should be individualised for maximum beneficial effect. For use as a hypnotic, the usual adult dose is 10 - 30 mg taken one-half hour before retiring. In elderly or debilitated patients, 10 mg Normison is the initial recommended dosage.
The need for continued therapy with Normison in patients who have been taking medication for several weeks should be evaluated periodically.
Normison is not recommended for children.
4.3 Contraindications
Patients with a known hypersensitivity to benzodiazepines or to any of the formulation.
Patients with chronic obstructive airways disease with incipient respiratory failure.
Patients with sleep apnoea.
Temazepam should not be used as monotherapy to treat depression, or symptoms of anxiety associated with depression, due to a risk of suicide (see Section 4.4 Special Warnings and Precautions for Use).
4.4 Special Warnings and Precautions for Use
Hypotension. Although hypotension has occurred rarely, Normison should be administered with caution to patients in whom a drop in blood pressure might lead to cardiac or cerebral complications. This is particularly important in elderly patients.
Amnesia. Transient amnesia or memory impairment has been reported in association with the use of benzodiazepines.
Myasthenia gravis. Normison could increase the muscle weakness in myasthenia gravis and should be used with caution in this condition.
Acute narrow angle glaucoma. Caution should be used in the treatment of patients with acute narrow angle glaucoma (because of atropine-like side effects).
Depression, psychosis and schizophrenia. Normison is not recommended as primary therapy in patients with depression and psychosis. In such conditions, psychiatric assessment and supervision are necessary if benzodiazepines are indicated. Benzodiazepines may increase depression in some patients, and may contribute to deterioration in severely disturbed schizophrenics with confusion and withdrawal. Suicidal tendencies may be present or uncovered, and protective measures may be required.
CNS and/or paradoxical reactions. As with other benzodiazepines and CNS active drugs, three idiosyncratic symptom clusters, which may overlap, have been described.
Amnestic symptoms: anterograde amnesia with appropriate or inappropriate behaviour;
Confusional states: disorientation, derealisation, depersonalization and/or clouding of consciousness; and
Agitational states: sleep disturbances, restlessness, irritability, aggression and excitation.
Temazepam should be discontinued if confusion or agitation occurs.
Paradoxical reactions such as acute rage, stimulation or excitement may occur; should such reactions occur, Normison should be discontinued. Such reactions may be more likely to occur in children and the elderly.
Impaired respiratory function. Use of benzodiazepines, including temazepam, may lead to potentially fatal respiratory depression. Caution in the use of Normison is recommended in patients with respiratory depression. In patients with chronic obstructive pulmonary disease, benzodiazepines can cause increased arterial carbon dioxide tension and decreased arterial oxygen tension.
Epilepsy. Abrupt withdrawal of benzodiazepines in patients with convulsive disorders may be associated with a temporary increase in the frequency and/or severity of seizures.
Abuse. Caution must be exercised in administering Normison to individuals known to be addiction prone or those whose history suggests they may increase the dosage on their own initiative. It is desirable to limit repeat prescription without adequate medical supervision.
Dependence. The use of benzodiazepines, including temazepam, may lead to physical and psychological dependence as defined by the presence of a withdrawal syndrome on discontinuation of the drug. The risk of dependence increases with higher doses and longer term use and is further increased in patients with a history of alcoholism or drug abuse or in patients with significant personality disorders. Temazepam may have abuse potential, especially in patients with a history of drug and/or alcohol abuse.
Duration of treatment. In general, benzodiazepines should be prescribed for short periods only (e.g. 2-4 weeks). Continuous long-term use of Normison is not recommended. There is evidence that tolerance develops to the sedative effects of benzodiazepines. After as little as one week of therapy withdrawal symptoms can appear following the cessation of recommended doses (e.g. rebound insomnia following cessation of a hypnotic benzodiazepine).
Tolerance. Tolerance, as defined by a need to increase the dose in order to achieve the same therapeutic effect, seldom occurs in patients receiving recommended doses under medical supervision. Tolerance to sedation may occur with benzodiazepines especially in those with drug seeking behaviour.
Withdrawal. Withdrawal symptoms similar in character to those noted with barbiturates and alcohol have occurred following abrupt discontinuation of benzodiazepines. These symptoms can range from headache, nausea, diarrhoea, loss of appetite, insomnia, anxiety, tensions, depression, restlessness, irritability, rebound phenomena, dysphoria, dizziness, abdominal cramps, agitation, palpitations, tachycardia, panic attacks, vertigo, myoclonus, akinesia, hypersensitivity to light, sound and touch, abnormal body sensations (e.g. feelings of motion, metallic taste), depersonalisation, derealisation, hyperacusis, delusional beliefs, hyperreflexia, numbness/ tingling extremities and loss of short-term memory, to a major syndrome which may include convulsions/ seizures, tremor, abdominal and muscle cramps, confusional states, delirium, hallucinations, hyperthermia, psychosis, vomiting and sweating. Convulsions/ seizures may be more common in patients with pre-existing seizure disorders or who are taking other drugs that lower the seizure threshold, such as antidepressants. Such manifestations of withdrawal, especially the more serious ones, are more common in those patients who have received excessive doses over a prolonged period. However, withdrawal symptoms have also been reported following abrupt discontinuation of benzodiazepines taken continuously at therapeutic levels. Accordingly, Normison should be terminated by tapering the dose to minimise occurrence of withdrawal symptoms. Patients should be advised to consult with their physician before either increasing the dose or abruptly discontinuing the medication.
Rebound phenomena have been described in the context of benzodiazepine use. Rebound insomnia and anxiety mean an increase in the severity of these symptoms beyond pretreatment levels following cessation of benzodiazepines. Rebound phenomena in general possibly reflect re-emergence of pre-existing symptoms combined with withdrawal symptoms described earlier. Some patients prescribed benzodiazepines with very short half-lives (in the order of 2 to 4 hours) may experience relatively mild rebound symptoms in between their regular doses. Withdrawal/rebound symptoms may follow high doses taken for relatively short periods.
Dose tapering. Following the prolonged use of Normison at therapeutic doses, withdrawal from the medication should be gradual. An individualised withdrawal timetable needs to be planned for each patient in whom dependence is known or suspected. Periods from four weeks to four months have been suggested. As with other benzodiazepines, when treatment is suddenly withdrawn, a temporary increase of sleep disturbance can occur after use of Normison (see Section 4.4, Dependence).
Somnambulism and associated behaviours. Complex behaviours such as "sleep-driving" (i.e. driving while not fully awake after taking a sedative-hypnotic, with amnesia for the event) have been reported with sedative hypnotics. These events can occur in sedative-hypnotics naïve as well as in sedative-hypnotic experienced persons. These events can occur at normal therapeutic doses, and the risk appears to be increased when sedative-hypnotics are combined with alcohol or other CNS depressants or used at doses exceeding the maximum recommended dose. Due to the risk to the patient and the community, discontinuation of sedative-hypnotics should be strongly considered for patients who report a "sleep-driving" episode. Other complex behaviours (e.g. preparing and eating food, making phone calls, or having sex) have been reported in patients who are not fully awake after taking a sedative-hypnotic. As with "sleep-driving", patients usually do not remember these events.
Angioedema. Angioedema involving the tongue, glottis and larynx has been reported in some patients after taking the first or subsequent doses of sedative-hypnotics. These cases of angioedema may cause airway obstruction and be fatal; this has required medical therapy in emergency departments for some patients. Additional symptoms have been reported in some patients including dyspnea, throat closing, or nausea and vomiting suggesting anaphylaxis.
Concomitant use with alcohol/CNS depressants. The concomitant use of temazepam with alcohol or/and CNS depressants should be avoided. Such concomitant use has the potential to increase the clinical effects of temazepam which may include severe sedation, clinically relevant respiratory and/or cardio-vascular depression (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
Impaired renal/liver function and blood dyscrasia. Patients with impaired renal or hepatic function should use benzodiazepine medication with caution and dosage reduction may be advisable. In rare instances some patients taking benzodiazepines have developed blood dyscrasias, and some have had elevations of liver enzymes. As with other benzodiazepines, periodic blood counts and liver function tests are recommended. The use of temazepam may worsen hepatic encephalopathy; therefore, temazepam should be used with caution in patients with severe hepatic insufficiency and/or encephalopathy.
Use in the elderly or debilitated patients. Such patients may be particularly susceptible to the sedative effects of benzodiazepines and associated giddiness, ataxia and confusion, which may increase the possibility of a fall. Normison 10 mg is the recommended starting dose for these patients.
Paediatric use. The safety and effectiveness of temazepam has not been established in children less than 16 years of age.
Effects on laboratory tests. No interference with laboratory tests have been identified or reported with the use of temazepam.
Minor EEG changes, usually low voltage fast activity, of no known clinical significance, have been reported with benzodiazepine administration.
4.5 Interactions with Other Medicines and Other Forms of Interactions
CNS depressants. The benzodiazepines, including Normison, produce additive CNS depressant effects when co-administered with other medications which themselves produce CNS depression, e.g. barbiturates, alcohol, sedatives, tricyclic antidepressants, non-selective MAO inhibitors, phenothiazines and other antipsychotics, skeletal muscle relaxants, antihistamines, or narcotic analgesics and anaesthetics.
Cytochrome P450. The cytochrome P450 system has not been shown to be involved in the disposition of Normison and, unlike many benzodiazepines, pharmacokinetic interactions involving the P450 system have not been observed with Normison.
Anticonvulsants. Interactions have been reported between some benzodiazepines and anticonvulsants, with changes in the serum concentration of the benzodiazepine or anticonvulsant. It is recommended that patients be observed for altered responses when benzodiazepines and anticonvulsants are prescribed together, and that serum level monitoring of the anticonvulsant be performed more frequently.
Theophylline/aminophylline. Administration of theophylline or aminophylline may reduce the sedative effects of benzodiazepines.
Potentiation of anticholinergic effects. The anticholinergic effects of other drugs including atropine and similar drugs, antihistamines and antidepressants may be potentiated.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Fertility in male and female rats was not adversely affected by temazepam.
Use in pregnancy. (Category C)
Temazepam should not be used during pregnancy.
Benzodiazepines cross the placenta and may cause hypoactivity, hypotonia, reduced respiratory function, apnoea, feeding problems, hypothermia and impaired metabolic response to cold stress in the newborn infant of mothers who have received benzodiazepines during the late phase of pregnancy or at delivery. Continuous treatment during pregnancy and administration of high doses in connection with delivery should be avoided. Withdrawal symptoms in newborn infants have been reported with this class of drugs.
The use of benzodiazepines during the first trimester of pregnancy should almost always be avoided. An increased risk of congenital malformations associated with the use of benzodiazepines during the first trimester of pregnancy has been suggested in several studies. In humans, umbilical cord blood samples indicate placental transfer of benzodiazepines and their glucuronide metabolites. If the drug is prescribed to a woman of child-bearing potential, she should be warned to contact her physician regarding discontinuation of the drug if she intends to become, or suspects that she is pregnant.
Non-teratogenic effects. The use of benzodiazepines during the last phase of pregnancy or at delivery may require ventilation of the infant at birth.
In animal studies, an increased perinatal mortality has been seen following concomitant administration of temazepam and diphenhydramine to rabbits in the later stages of gestation compared with rabbits that received either drug alone. It is recommended that the use of temazepam be avoided in pregnant women receiving antihistamines.
Use in lactation. Caution should be exercised when Normison is given to breast feeding women.
Normison is believed to be excreted in human breast milk, and may cause drowsiness and feeding difficulties in the infant.
4.7 Effects on Ability to Drive and Use Machines
As with all patients taking CNS depressant medications, patients receiving Normison should be warned not to operate dangerous machinery or motor vehicles until it is known that they do not become drowsy or dizzy from Normison therapy. Abilities may be impaired on the day following use. In sleep laboratory studies in volunteers, doses of 10 and 20 mg did not significantly affect morning performance, however the 30 mg dose produced impairment of psychomotor behaviour on the morning following night time administration. Discontinuation of Normison is highly recommended for patients who report a "sleep-driving" episode (see Section 4.4, Somnambulism and associated behaviours).
4.8 Adverse Effects (Undesirable Effects)
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
All adverse reactions reported with temazepam are common with other benzodiazepine compounds. See Table 1.

4.9 Overdose
Overdosage of benzodiazepines is usually manifested by degrees of central nervous system depression ranging from drowsiness to coma. In mild cases, symptoms include drowsiness, mental confusion, lethargy, dysarthria and paradoxical reactions. In more serious cases, symptoms may include ataxia, CNS depression, hypotonia, hypotension, respiratory depression, cardiovascular depression, coma, and very rarely proves fatal.
Treatment. In the management of overdosage with any medication, it should be borne in mind that multiple agents may have been taken.
Following overdosage with oral benzodiazepines, activated charcoal may be given to reduce absorption, if given within one or two hours after ingestion. In patients who are not fully conscious or have impaired gag reflex, consideration should be given to administering activated charcoal via a nasogastric tube, once the airway is protected. Hypotension and respiratory depression should be managed according to general principles.
Haemoperfusion and haemodialysis are not useful in benzodiazepine intoxication. The benzodiazepine antagonist flumazenil may be used in hospitalised patients for the reversal of acute benzodiazepine effects. Please consult the flumazenil product information prior to usage.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Normison hastens the onset of sleep and increases total sleeping time in short-term use.
The exact mechanism of action of benzodiazepines has not yet been elucidated; however, benzodiazepines appear to work through several mechanisms. Benzodiazepines presumably exert their effects by binding to specific receptors at several sites within the central nervous system either by potentiating the effects of synaptic or pre-synaptic inhibition mediated by gamma-aminobutyric acid, or by directly affecting the action potential generating mechanisms.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Absorption. Pharmacokinetic studies have shown that Normison is well absorbed and has a relatively short elimination half-life of approximately 10 hours (range 5-15 hours).
Peak plasma levels of the drug occur 30 to 120 minutes after administration of the tablets.
Metabolism and excretion. With multiple dosing, steady state is obtained by the third day, and there is little or no accumulation of parent drug or metabolites.
Normison is metabolised principally in the liver where most drug is directly conjugated to the glucuronide and excreted in the urine. Some drug is demethylated to oxazepam and eliminated as the glucuronide. The glucuronides of Normison have no demonstrable CNS activity. Following a single oral dose, 80% of the dose appears in the urine, mostly as the conjugates, and 12% of the dose appears in the faeces.
Less than 2% of the dose is excreted unchanged in the urine. Approximately 96% of unchanged drug is bound to plasma proteins.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Lactose monohydrate, microcrystalline cellulose, maize starch and magnesium stearate.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C. Protect from moisture.
6.5 Nature and Contents of Container
Pack sizes of 5, 25, 30 and 100 tablets in PVC/PVdC/Al blisters.
(Note. Not all pack sizes may be available.)
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
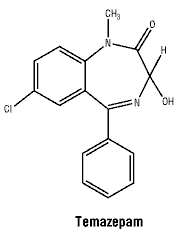
Temazepam is a white, odourless crystalline powder; it is sparingly soluble in alcohol and freely soluble in chloroform, but insoluble in water. The molecular weight is 300.8.
Temazepam is a 1, 4 benzodiazepine with the chemical name 7-chloro-1, 3-dihydro-3-hydroxy-1-methyl-5-phenyl-2H-1-4-benzodiazepin-2-one.
Chemical structure.
7 Medicine Schedule (Poisons Standard)
S4.
Date of First Approval
01 October 1992
Date of Revision
06 September 2024
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. NPS MedicineWise disclaims all liability (including for negligence) for any loss, damage or injury resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.