Nurofen Plus
Brand Information
| Brand name | Nurofen Plus |
| Active ingredient | Ibuprofen + Codeine phosphate hemihydrate |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Nurofen Plus.
Summary CMI
Nurofen® Plus Tablets
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
WARNING: Important safety information is provided in a boxed warning in the full CMI. Read before using this medicine.
1. Why am I using Nurofen Plus?
Nurofen Plus contains the active ingredients Ibuprofen and codeine phosphate. Nurofen Plus is used to provide temporary relief of acute moderate pain and inflammation. For more information, see Section 1. Why am I using Nurofen Plus? in the full CMI.
2. What should I know before I use Nurofen Plus?
Do not use if you have ever had an allergic reaction to Nurofen Plus or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use Nurofen Plus? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Nurofen Plus and affect how it works. A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use Nurofen Plus?
- The usual dose of Nurofen Plus is 2 tablets then, if necessary 1 or 2 tablets every 4 to 6 hours.
More instructions can be found in Section 4. How do I use Nurofen Plus? in the full CMI.
5. What should I know while using Nurofen Plus?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using Nurofen Plus? in the full CMI.
6. Are there any side effects?
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
WARNING:
Limitations of use
Nurofen Plus should only be used when your doctor decides that other treatment options are not able to effectively manage your pain or you cannot tolerate them.
Hazardous and harmful use
Nurofen Plus poses risks of abuse, misuse and addiction which can lead to overdose and death. Your doctor will monitor you regularly during treatment.
Life threatening respiratory depression
Nurofen Plus can cause life-threatening or fatal breathing problems (slow, shallow, unusual or no breathing) even when used as recommended. These problems can occur at any time, but the risk is higher when first starting Nurofen Plus and after a dose increase, if you are older, or have an existing problem with your lungs. Your doctor will monitor you and change the dose as appropriate.
Use of other medicines while taking Nurofen Plus
Using Nurofen Plus with other medicines that can make you feel drowsy such as sleeping tablets (e.g. benzodiazepines), other pain relievers, antihistamines, antidepressants, antipsychotics, gabapentinoids (e.g. gabapentin and pregabalin), cannabis oil and alcohol may result in severe drowsiness, decreased awareness, breathing difficulties, coma and death. Your doctor will minimise the dose and duration of use and monitor you for signs and symptoms of breathing difficulties and sedation. You must not drink alcohol while using Nurofen Plus
Nurofen® Plus Tablets
Active ingredients: Ibuprofen 200mg and Codeine phosphate hemihydrate 12.8mg
Consumer Medicine Information (CMI)
This leaflet provides important information about using Nurofen Plus. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using Nurofen Plus.
Where to find information in this leaflet:
1. Why am I using Nurofen Plus?
2. What should I know before I use Nurofen Plus?
3. What if I am taking other medicines?
4. How do I use Nurofen Plus?
5. What should I know while using Nurofen Plus?
6. Are there any side effects?
7. Product details
1. Why am I using Nurofen Plus?
Nurofen Plus contains the active ingredients Ibuprofen and codeine phosphate. Ibuprofen belongs to a family of medicines called non-steroidal anti-inflammatory drugs (NSAIDS). These medicines work by relieving pain and/or inflammation (swelling, redness, soreness) and fever. Codeine is an opioid analgesic that works in the brain and spinal cord to relieve pain.
Nurofen Plus is used to provide temporary relief of acute moderate pain and inflammation in patients over the age of 12 years.
After taking codeine, the body changes codeine into the active morphine by a special liver enzyme. However, about 8% of people may experience less pain relief compared to others, as their bodies do not change codeine to morphine as well as others.
2. What should I know before I use Nurofen Plus?
Warnings
Do not give Nurofen Plus to children under the age of 12 years.
Do not use Nurofen Plus if:
- you are allergic to ibuprofen or codeine phosphate, or any of the ingredients listed at the end of this leaflet.
- many medicines used to treat headache, period pain and other aches and pains contain aspirin or NSAIDs. If you are not sure if you are taking any of these medicines, ask your pharmacist.
- always check the ingredients to make sure you can use this medicine.
- symptoms of an allergic reaction to these medicines may include:
- asthma, wheezing or shortness of breath.
- swelling of the face, lips or tongue which may cause difficulty in swallowing or breathing.
- hives, itching or skin rash.
- fainting.
If you are allergic to aspirin or NSAIDs and take Nurofen Plus these symptoms may be severe. - you are pregnant or are breast-feeding.
- you are vomiting blood or material that looks like coffee grounds, or have a history of this.
- you have or have had bleeding from the rectum (back passage), black sticky bowel motions (stool) or bloody diarrhoea
- you have peptic ulcer (i.e. stomach or duodenal ulcer) or have had one before.
- you are using other NSAIDS
- you have chronic constipation or shallow breathing, or severe diarrhoea.
- You consume regular and heavy amounts of alcohol.
- You are a CYP2D6 ultra-rapid metaboliser.
- If you have severe liver impairment
- If you have heart or kidney problems
- If you are currently taking a Monoamine oxidase inhibitors (MAOIs) or within 14 days of stopping treatment with a MAOI.
Do not take Nurofen Plus if the packaging is torn or shows signs of tampering.
Do not take Nurofen Plus if the expiry date (EXP) printed on the pack has passed.
If you take this medicine after the expiry date has passed, it may not work as well.
Do not give Nurofen Plus to children under 12 years or to those aged 12 -18 years in whom respiratory function may be compromised.
Check with your doctor if you:
Experience signs of an allergic reaction to this medicine, including breathing problems, swelling of the face and neck region (angioedema), and chest pain. Stop Nurofen Plus immediately and contact your doctor or medical emergencies straight away if you notice any of these signs.
- have any other medical conditions.
- take any medicines for any other condition.
- difficulty breathing, wheezing, chronic cough, allergies, asthma or other breathing conditions.
- a history of drug dependence, including alcohol dependence.
- skin rash (dermatitis).
- skin irritation.
- a history of stomach ulcer.
- liver disease.
- kidney disease.
- heart problems/failure, including. swelling of ankles or feet.
- thyroid problems or low blood pressure.
- a head injury or intracranial pressure.
- prostatic problems.
- stomach problems or recent stomach surgery.
- gave central sleep apnoea (a sleep-related breathing disorder which can cause pauses in breathing during sleep), as a reduced dose of Nurofen Plus may be required.
Tell your doctor if you experience severe upper abdominal pain radiating to the back, nausea, vomiting, or fever, as this could be symptoms associated with inflammation of the pancreas and biliary tract system.
Stop use and tell your doctor if you experience side effects including difficulty swallowing, regurgitation, and chest pain, as these may be signs of a problem with your oesophagus.
Serious skin reactions including exfoliative dermatitis, erythema multiforme, Stevens-Johnson syndrome, toxic epidermal necrolysis, drug reaction with eosinophilia and systemic symptoms (DRESS), acute generalised exanthematous pustulosis (AGEP) have been reported in association with ibuprofen treatment. Stop using Nurofen Plus and seek medical attention immediately if you notice any of the symptoms related to these serious skin reactions described in section 6.
Stop use and tell your doctor if you experience symptoms such as nausea, vomiting, weight loss, fatigue, weakness, dizziness or feeling faint. These may be signs of adrenal insufficiency.
Nurofen Plus may cause changes in levels of hormones which may manifest as problems with sexual intercourse including reduced sex drive, as well as changes in the menstrual cycle and reduced fertility.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Nurofen Plus is should not be used during pregnancy.
Prolonged use of Nurofen Plus at the end of pregnancy may result in withdrawal syndrome in the neonate which may be delayed for several hours to several days after birth. Symptoms in the neonate include stiff muscles, shaking, agitation, convulsions, breathing problems, and slow heartbeat.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
Nurofen Plus should not be taken while breastfeeding.
Addiction
You can become addicted to Nurofen Plus even if you take it exactly as prescribed. Nurofen Plus may become habit forming causing mental and physical dependence. If abused it may become less able to reduce pain.
Dependence
As with all other opioid-containing products, your body may become used to you taking Nurofen Plus. Taking it may result in physical dependence. Physical dependence means that you may experience withdrawal symptoms if you stop taking Nurofen Plus suddenly, so it is important to take it exactly as directed by your doctor.
If you are taking Nurofen Plus for longer than the recommended time or at higher than recommended doses you are at risk of serious harms. These include serious harms to the stomach/gut and kidneys, as well as very low levels of potassium in your blood. These can be fatal (see section 6).
Tolerance
Tolerance to Nurofen Plus may develop, which means that the effect of the medicine may decrease. If this happens, more may be needed to maintain the same effect.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with Nurofen Plus and affect how it works.
These include:
- medicines used to help you relax, sleep or relieve anxiety, such as barbiturates and sedatives.
- aspirin, salicylates or other NSAID medicines.
- Warfarin or other medicines used to prevent blood clots.
- medicines that are used to treat high blood pressure including diuretics (fluid tablets)
- methotrexate.
- zidovudine.
- lithium.
- Mifeprisone, quinolone antibiotics
- medicines used to relieve stomach cramps, prevent travel sickness and to treat Parkinson's disease.
- medicines used to treat diarrhoea (e.g. kaolin, pectin, loperamide).
- metoclopramide, a medicine used to treat nausea and vomiting.
- medicines that affect serotonin levels (serotonergic medicines such as triptans, selective serotonin- and norepinephrine-reuptake inhibitors (SNRIs) and tricyclic antidepressants).
- selective serotonin reuptake inhibitors (SSRIs), and monoamine oxidase inhibitors (MAOIs), medicines used to treat some types of depression.
- quinidine, a medicine used to treat abnormal or irregular heartbeat
- phenothiazines and antipsychotic agents, medicines used to treat mental disorders.
- medicines used to treat neuropathic pain (gabapentinoids such as gabapentin and pregabalin), as they may increase the depressant effects of codeine.
- medicines such as cyclosporin, tacrolimus, prednisone, prednisolone and cortisone which reduce the activity of your immune system.
- probenecid-medicines used to treat gout
- phenytoin – medicine used to treat epilepsy
- other opioid pain killers.
These medicines may be affected by Nurofen Plus or may affect how well it works. You may need to take different amounts of your medicine, or you may need to take different medicines. Your doctor will advise you.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Nurofen Plus.
4. How do I use Nurofen Plus?
How much to take and when to take it
- The usual dose of Nurofen Plus is 2 tablets then, if necessary 1 or 2 tablets every 4 to 6 hours. Do not take more than six tablets in 24 hours.
- Follow the instructions provided and use Nurofen Plus until your doctor tells you to stop.
How to take it
- Take Nurofen Plus by mouth with fluid. It may also be taken with or immediately after food.
How long to take it
- You should not take Nurofen Plus for more than three days at a time.
If your symptoms persist, worsen or new symptoms develop, talk to your doctor.
Talk to your doctor about pain control if Nurofen Plus is not helping.
Depending on your body's individual ability to break down codeine, you may be getting reduced benefit or experience signs of overdose even when you take Nurofen Plus as recommended by your doctor. If overdose symptoms occur, seek immediate medical advice.
If you use too much Nurofen Plus
If you or someone else receive too much (overdose), and experience one or more of the symptoms below, immediately and call triple zero (000) for an ambulance. Keep the person awake by talking to them or gently shaking them every now and then. You should follow the above steps even if someone other than you have accidentally used Nurofen Plus that was prescribed for you. If someone takes an overdose they may experience one or more of the following symptoms:
- Slow, unusual or difficult breathing
- drowsiness, dizziness or unconsciousness.
- slow or weak heartbeat.
- nausea or upset stomach, vomiting and/or gastric irritation.
- convulsions, fits or unconsciousness.
- confusion, loss of coordination, tremors, weakness, seizures, headaches, vision impairment.
- excitability.
- blurred vision, ringing in the ears, or rapid uncontrollable movements of the eyes.
If you think that you have used too much Nurofen Plus, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre (by calling 13 11 26), or
- contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using Nurofen Plus?
When seeking medical attention, take this leaflet and remaining medicine with you to show the doctor. Also tell them about any other medicines or alcohol which have been taken.
Things you should do
- if you are about to be started on any new medicine tell your doctor and pharmacist that you are taking Nurofen Plus.
- remind any doctor, dentist or pharmacist you visit that you are using Nurofen Plus.
- if you are going to have surgery, tell your doctor you are taking Nurofen Plus.
Talk to doctor if your symptoms do not improve.
Your doctor will assess your condition and decide if you should continue to take the medicine.
Call your doctor straight away if you:
- Become pregnant while taking Nurofen Plus
Remind any doctor, dentist or pharmacist you visit that you are using Nurofen Plus.
Things you should not do
- do not take high doses of the medicine for long periods of time unless your doctor tells you to. Products containing codeine should not be taken for prolonged periods. Codeine may be habit forming.
- do not give Nurofen Plus to anyone else, even if they have the same condition as you.
- do not use Nurofen Plus to treat any other complaints unless your doctor tells you to.
- do not take more than the recommended dose unless your doctor tells you to.
- excessive use can be harmful and increase the risk of heart attack, stroke or liver damage.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Nurofen Plus affects you.
Nurofen Plus may cause dizziness light-headedness or drowsiness in some people. If this occurs, do not drive or operate machinery. If you drink alcohol, dizziness, light-headedness and/or drowsiness may be worse.
Drinking alcohol
Tell your doctor if you drink alcohol.
Using Nurofen Plus with alcohol may result in severe drowsiness, decreased awareness, breathing difficulties, coma and death.
Withdrawal
Continue taking your medicine for as long as your doctor tells you. If you stop having this medicine suddenly, your pain may worsen and you may experience some or all of the following withdrawal symptoms:
- nervousness, restlessness, agitation, trouble sleeping or anxiety
- body aches, weakness or stomach cramps
- loss of appetite, nausea, vomiting or diarrhoea
- increased heart rate, breathing rate or pupil size
- watery eyes, runny nose, chills or yawning
- increased sweating.
Nurofen Plus given to the mother during labour can cause breathing problems and signs of withdrawal in the newborn.
Looking after your medicine
- Keep the tablets in the original pack until it is time to take the dose. If you take them out they will not keep well.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Heat and dampness can destroy some medicines.
Keep it where young children cannot reach it.
A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Gastrointestinal related:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Gastrointestinal related:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Nurofen Plus contains
| Active ingredient (main ingredient) | Ibuprofen and codeine phosphate hemihydrate |
| Other ingredients (inactive ingredients) | Microcrystalline cellulose, sodium starch glycollate, hypromellose, pregelatinised maize starch, talc, opaspray white colouring |
| Potential allergens | Nurofen Plus is gluten-free and lactose-free |
Do not take this medicine if you are allergic to any of these ingredients.
What Nurofen Plus looks like
Nurofen Plus tablets are white capsule shaped tablets marked ‘N+’ on one side. (AUST R 116620).
Nurofen Plus is available in blister packs of *4, *6, *12, *24 & 30 tablets.
*not available in Australia
Who distributes Nurofen Plus
Reckitt Benckiser, Sydney NSW, Australia
This leaflet was prepared in November 2025.
Brand Information
| Brand name | Nurofen Plus |
| Active ingredient | Ibuprofen + Codeine phosphate hemihydrate |
| Schedule | S4 |
MIMS Revision Date: 01 February 2026
Hazardous and harmful use. Nurofen Plus poses risks of hazardous and harmful use which can lead to overdose and death. Assess the patient's risk of hazardous and harmful use before prescribing and monitor the patient regularly during treatment (see Section 4.4 Special Warnings and Precautions for Use).
Life threatening respiratory depression. Serious, life-threatening or fatal respiratory depression may occur with the use of Nurofen Plus. Be aware of situations which increase the risk of respiratory depression, modify dosing in patients at risk and monitor patients closely, especially on initiation or following a dose increase (see Section 4.4 Special Warnings and Precautions for Use).
Concomitant use of benzodiazepines and other central nervous system (CNS) depressants, including alcohol. Concomitant use of opioids with benzodiazepines, gabapentinoids, antihistamines, tricyclic antidepressants, antipsychotics, cannabis or other central nervous system (CNS) depressants, including alcohol, may result in profound sedation, respiratory depression, coma, and death. Limit dosages and durations to the minimum required; and monitor patients for signs and symptoms of respiratory depression and sedation. Caution patients not to drink alcohol while taking Nurofen Plus.
1 Name of Medicine
Ibuprofen and codeine phosphate hemihydrate.
2 Qualitative and Quantitative Composition
Each tablet contains ibuprofen 200 mg and codeine phosphate hemihydrate 12.8 mg.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
A white capsule-shaped tablet, containing ibuprofen 200 mg and codeine phosphate hemihydrate 12.8 mg.
The white capsule-shaped tablets are marked 'N+' on one side and plain on the other.
4 Clinical Particulars
4.1 Therapeutic Indications
For the temporary relief of acute moderate pain and inflammation in patients over the age of 12 years.
4.2 Dose and Method of Administration
Adults and children 12 years and over. Initial dose two tablets taken with fluid, then one or two tablets every 4 to 6 hours when necessary. Maximum 6 tablets in a 24-hour period.
Nurofen Plus should not be used for more than three days at a time unless on medical advice, in which case the patient should be reviewed regularly with regards to efficacy, risk factors and ongoing need for treatment.
The lowest effective dose should be used for the shortest duration necessary to relieve symptoms.
Pregnancy. See Section 4.3 Contraindications; Section 4.6 Fertility, Pregnancy and Lactation.
Children. Do not give to children under 12 years.
Nurofen Plus is contraindicated for use in children:
younger than 12 years;
aged between 12-18 years in whom respiratory function might be compromised, including post tonsillectomy and/or adenoidectomy for obstructive sleep apnoea (see Section 4.3 Contraindications; Section 4.4 Special Warnings and Precautions for Use, Paediatric use).
4.3 Contraindications
Known hypersensitivity to ibuprofen, codeine or other opioid analgesics, or any of the excipients.
Patients who have previously shown hypersensitivity reactions (e.g. asthma, rhinitis, angioedema, bronchospasm or urticaria) in response to ibuprofen, acetylsalicylic acid (aspirin) or other non-steroidal anti-inflammatory drugs (NSAIDs).
Active, or a history of recurrent peptic ulcer/haemorrhage (two or more distinct episodes of proven ulceration or bleeding). As with other non-steroidal anti-inflammatory agents, ibuprofen should not be used in patients with a history of, or active gastrointestinal bleeding or in the presence of peptic ulceration.
Chronic constipation and active alcoholism.
Severe respiratory disease, acute respiratory disease and respiratory depression.
Diarrhoea caused by pseudomembranous colitis or poisoning (until the causative organism or toxin has been eliminated from the gastrointestinal tract, since codeine may slow down the elimination, thereby prolonging the diarrhoea).
Use with concomitant NSAIDs, including cyclo-oxygenase-2 specific inhibitors - increased risk of adverse reactions.
Heart (see Section 4.4 Special Warnings and Precautions for Use, Cardiovascular and cerebrovascular effects) or renal problems (see Section 4.4 Special Warnings and Precautions for Use, Use in renal impairment).
Severe hepatic impairment (see Section 4.4 Special Warnings and Precautions for Use, Use in hepatic impairment).
During pregnancy (see Section 4.6 Fertility, Pregnancy and Lactation, Use in pregnancy).
Concomitant treatment with monoamine oxidase inhibitors (MAOIs) or within 14 days of stopping treatment.
Use of codeine containing products is contraindicated in women during breast feeding (see Section 4.6 Fertility, Pregnancy and Lactation, Use in lactation).
In patients for whom it is known they are CYP2D6 ultra-rapid metabolisers (see Section 4.4 Special Warnings and Precautions for Use, CYP2D6 metabolism).
In patients younger than 12 years (see Section 4.4 Special Warnings and Precautions for Use, Paediatric use).
In patients aged between 12 - 18 years in whom respiratory function might be compromised, including post tonsillectomy and/or adenoidectomy for obstructive sleep apnoea, due to an increased risk of developing serious and life-threatening adverse reactions (see Section 4.4 Special Warnings and Precautions for Use, Paediatric use).
Treatment of perioperative pain in setting of coronary artery bypass surgery (CABG).
4.4 Special Warnings and Precautions for Use
Use in the elderly. Adverse effects may have more serious consequences in the elderly, and they may be more susceptible to the CNS depressant effects of opioids.
Undesirable effects may be minimised by using the minimum effective dose for the shortest duration necessary to control symptoms. The elderly have an increased frequency of adverse reactions to NSAIDs especially gastrointestinal bleeding and perforation which may be fatal (see below).
Gastrointestinal. NSAIDs should be given with care to patients with a history of gastrointestinal disease (ulcerative colitis, Crohn's disease) as their condition may be exacerbated (see Section 4.8 Adverse Effects (Undesirable Effects)).
Gastrointestinal bleeding, ulceration and perforation which can be fatal, has been reported with all NSAIDs at any time during treatment, with or without warning symptoms or a previous history of serious GI events.
The risk of GI bleeding, ulceration or perforation is higher with increasing NSAID doses and patients with a history of ulcer, particularly if complicated with haemorrhage or perforation (see Section 4.3 Contraindications) and in the elderly. These patients should commence treatment on the lowest dose available.
Patients with a history of GI toxicity, particularly the elderly, patients with a history of gastrointestinal bleeding or perforation or peptic ulcer haemorrhage related to previous NSAID therapy should report any unusual abdominal symptoms (especially GI bleeding) particularly in the initial stages of treatment.
Care is advised in the administration of Nurofen Plus to patients with obstructive bowel disorders, recent gastrointestinal surgery, gallstones, myasthenia gravis, a history of peptic ulcer or convulsions.
Caution should be advised in patients receiving concomitant medications which could increase the risk of ulceration or bleeding, such as oral corticosteroids, anticoagulants such as warfarin, selective serotonin-reuptake inhibitors or anti-platelet agents such as aspirin (see Section 4.5 Interactions with Other Medicines and Other Forms of Interactions).
When GI bleeding or ulceration occurs in patients receiving Nurofen Plus, the treatment should be withdrawn.
Respiratory. Bronchospasm may be precipitated in patients suffering from, or with a history of bronchial asthma or allergic disease.
SLE and mixed connective tissue disease. Use of ibuprofen in patients with systemic lupus erythematosus (SLE) and mixed connective tissue disease can increase the risk of aseptic meningitis.
Use in hepatic impairment. As with other NSAIDs, elevations of one or more liver function tests may occur in up to 15% of patients. These abnormalities may progress, may remain essentially unchanged, or may resolve with continued therapy. Meaningful elevations (three times the upper limit of normal) of ALT or AST occurred in controlled clinical trials in less than 1% of patients.
Nurofen Plus should be administered with caution in patients with mild to moderate hepatic impairment (see Section 4.3 Contraindications for severe hepatic impairment). Patients should be advised to remain alert for hepatotoxicity and be informed about the signs and/or symptoms of hepatotoxicity (e.g. nausea, fatigue, lethargy, pruritus, jaundice, abdominal tenderness in the right upper quadrant and "flu-like" symptoms) and the steps to take should these signs and/or symptoms occur.
Hepatobiliary disorders. Opioids may cause dysfunction and spasm of the sphincter of Oddi, thus raising intrabiliary pressure and increasing the risk of biliary tract symptoms and pancreatitis. Therefore, Nurofen Plus has to be administered with caution in patients with pancreatitis and diseases of the biliary tract.
Use in renal impairment. Renal impairment as renal function may deteriorate, especially in dehydrated paediatric patients (see Section 4.3 Contraindications; Section 4.8 Adverse Effects (Undesirable Effects)).
Cardiovascular and cerebrovascular effects. Cases of Kounis syndrome have been reported in patients treated with ibuprofen containing products such as Nurofen Plus. Kounis syndrome has been defined as cardiovascular symptoms secondary to an allergic or hypersensitive reaction associated with constriction of coronary arteries and potentially leading to myocardial infarction.
Observational studies have indicated that NSAIDs may be associated with an increased risk of serious cardiovascular events, including myocardial infarction and stroke, which may increase with dose or duration of use. Patients with cardiovascular disease, history of atherosclerotic cardiovascular disease or cardiovascular risk factors may also be at greater risk.
Patients should be advised to remain alert for such cardiovascular events, even in the absence of previous cardiovascular symptoms. Patients should be informed about signs and/or symptoms of serious cardiovascular toxicity and the steps to take if they occur.
Fluid retention, hypertension and oedema have been reported in association with NSAID therapy. Patients taking antihypertensives with NSAIDs may have an impaired antihypertensive response.
Nurofen Plus should be administered with caution in patients with hypertension or fluid retention (see Section 4.3 Contraindications, Heart).
Skin and subcutaneous tissue disorders. Serious skin reactions, some of them fatal, including exfoliative dermatitis, Stevens-Johnson syndrome, drug reaction with eosinophilia with systemic symptoms (see Drug reaction with eosinophilia and systemic symptoms (DRESS)) and toxic epidermal necrolysis, have been reported very rarely in association with the use of NSAIDs (see Section 4.8 Adverse Effects (Undesirable Effects)). Patients appear to be at highest risk of these reactions early in the course of therapy, the onset of the reaction occurring in the majority of cases within the first month of treatment. Nurofen Plus use should be discontinued at the first appearance of skin rash, mucosal lesions, or any other sign of hypersensitivity.
Severe skin reactions. Severe skin reactions such as acute generalised exanthematous pustulosis (AGEP) may occur with ibuprofen-containing products. The acute pustular eruption may occur within the first 2 days of treatment, with fever, and numerous, small, mostly non-follicular pustules arising on a widespread oedematous erythema and mainly localised on the skin folds, trunk, and upper extremities. Patients should be carefully monitored. If signs and symptoms such as pyrexia, erythema, or many small pustules are observed, administration of Nurofen Plus should be discontinued and appropriate measures taken if needed.
Drug reaction with eosinophilia with systemic symptoms (DRESS). DRESS has been reported in patients using NSAIDs. Some of these events have been fatal or life-threatening. DRESS typically, although not exclusively, presents with fever, rash, lymphadenopathy, and/or facial swelling. Other clinical manifestations may include hepatitis, nephritis, haematological abnormalities, myocarditis, or myositis. Sometimes symptoms of DRESS may resemble an acute viral infection. Eosinophilia is often present. Because this disorder is variable in its presentation, other organ systems not noted here may be involved. It is important to note that early manifestations of hypersensitivity, such as fever or lymphadenopathy, may be present even though rash is not evident. If such signs or symptoms are present, discontinue the NSAID and evaluate the patient immediately.
CYP2D6 metabolism. Nurofen Plus is contraindicated for use in patients who are CYP2D6 ultra-rapid metabolisers.
Codeine is metabolised by the liver enzyme CYP2D6 into morphine, its active metabolite. If a patient has a deficiency or is completely lacking this enzyme an adequate analgesic effect will not be obtained. However, if the patient is an extensive or ultrarapid metabolizer there is an increased risk of developing side effects of opioid toxicity even at commonly prescribed doses. These patients convert codeine into morphine rapidly resulting in higher than expected serum morphine levels. General symptoms of opioid toxicity include confusion, somnolence, shallow breathing, small pupils, nausea, vomiting, constipation, and lack of appetite. In severe cases this may include symptoms of circulatory and respiratory depression, which may be life-threatening and very rarely fatal. Children are particularly susceptible due to their immature airway anatomy. Deaths have been reported in children with rapid metabolism who were given codeine for analgesia post adenotonsillectomy. Morphine can also be ingested by infants through breast milk, causing risk of respiratory depression to infants of rapid metaboliser mothers who take codeine.
The prevalence of codeine ultra-rapid metabolism by CYP2D6 in children is not known, but is assumed to be similar to that reported in adults. The prevalence of ultra-rapid metabolisers is estimated to be 1% in those of Chinese, Japanese and Hispanic descent, 3% in African Americans and 1%-10% in Caucasians. The highest prevalence (16%-28%) occurs in North African, Ethiopian and Arab populations. Estimates of prevalence of ultra-rapid metabolisers in different populations are summarised in Table 1.
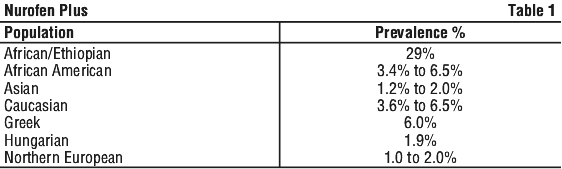
Paediatric use. Nurofen Plus is contraindicated for use in children:
younger than 12 years;
aged between 12 - 18 years in whom respiratory function might be compromised, including post tonsillectomy and/or adenoidectomy for obstructive sleep apnoea. Respiratory depression and death have occurred in some children who received codeine following tonsillectomy and/or adenoidectomy and had evidence of being ultra-rapid metabolisers of codeine due to a CYP2D6 polymorphism.
Post-operative use in children. There have been reports in the published literature that codeine given post-operatively in children after tonsillectomy and/or adenoidectomy for obstructive sleep apnoea, led to rare, but life-threatening adverse events including death (see Section 4.3 Contraindications). All children received doses of codeine that were within the appropriate dose range; however there was evidence that these children were either ultra-rapid or extensive metabolisers in their ability to metabolise codeine to morphine.
Children with compromised respiratory function. Codeine is contraindicated for use in children in whom respiratory function might be compromised including neuromuscular disorders, severe cardiac or respiratory conditions, upper respiratory or lung infections, multiple trauma or extensive surgical procedures. These factors may worsen symptoms of morphine toxicity.
Effects on laboratory tests. Opioid analgesics interfere with a number of laboratory tests including plasma amylase, lipase, bilirubin, alkaline phosphatase, lactate dehydrogenase, alanine aminotransferase and aspartate aminotransferase. Opioids may also interfere with gastric emptying studies as they delay gastric emptying and with hepatobiliary imaging using technetium Tc 99m disofenin as opioid treatment may cause constriction of the sphincter of Oddi and increase biliary tract pressure.
Hazardous and harmful use. Nurofen Plus contains the opioid codeine and is a potential drug of abuse, misuse and addiction. Addiction can occur in patients appropriately prescribed Nurofen Plus at recommended doses.
The risk of addiction is increased in patients with a personal or family history of substance abuse (including alcohol and prescription and illicit drugs) or mental illness. The risk also increases the longer the drug is used and with higher doses. Patients should be assessed for their risks for opioid abuse or addiction prior to being prescribed Nurofen Plus.
All patients receiving opioids should be routinely monitored for signs of misuse and abuse. Opioids are sought by people with addiction and may be subject to diversion. Strategies to reduce these risks include prescribing the drug in the smallest appropriate quantity and advising the patient on the safe storage and proper disposal of any unused drug (see Section 6.4 Special Precautions for Storage; Section 6.6 Special Precautions for Disposal). Caution patients that abuse of oral or transdermal forms of opioids by parenteral administration can result in serious adverse events, which may be fatal.
Patients should be advised not to share Nurofen Plus with anyone else.
Severe hypokalaemia and renal tubular acidosis have been reported due to prolonged use of ibuprofen/codeine at higher than recommended doses, due to the development of codeine dependency. Symptoms may include reduced level of consciousness and generalised weakness.
Respiratory depression. Serious, life-threatening or fatal respiratory depression can occur with the use of opioids even when used as recommended. It can occur at any time during the use of Nurofen Plus but the risk is greatest during initiation of therapy or following an increase in dose. Patients should be monitored closely for respiratory depression at these times.
The risk of life-threatening respiratory depression is also higher in elderly, frail, or debilitated patients, in patients with renal and hepatic impairment and in patients with existing impairment of respiratory function (e.g. chronic obstructive pulmonary disease; asthma). Opioids should be used with caution and with close monitoring in these patients (see Section 4.2 Dose and Method of Administration). The use of opioids is contraindicated in patients with severe respiratory disease, acute respiratory disease and respiratory depression (see Section 4.3 Contraindications).
The risk of respiratory depression is greater with the use of high doses of opioids, especially high potency and modified release formulations, and in opioid naïve patients. Initiation of opioid treatment should be at the lower end of the dosage recommendations with careful titration of doses to achieve effective pain relief. Careful calculation of equianalgesic doses is required when changing opioids or switching from immediate release to modified release formulations, together with consideration of pharmacological differences between opioids. Consider starting the new opioid at a reduced dose to account for individual variation in response.
Risks from concomitant use of benzodiazepines or other CNS depressants, including alcohol. Concomitant use of opioids and benzodiazepines or other CNS depressants, including alcohol, may result in sedation, respiratory depression, coma and death. Because of these risks, concomitant prescribing of Nurofen Plus with CNS depressant medicines, such as other opioid analgesics, benzodiazepines, gabapentinoids, cannabis, sedatives, hypnotics, tricyclic antidepressants, antipsychotics, antihistamines, centrally-active antiemetics and other CNS depressants, should be reserved for patients for whom other treatment options are not possible. If a decision is made to prescribe Nurofen Plus concomitantly with any of the medicines, the lowest effective dose should be used, and the duration of treatment should be as short as possible. Patients should be followed closely for signs and symptoms of respiratory depression and sedation. Patients and their caregivers should be made aware of these symptoms. Patients and their caregivers should also be informed of the potential harms of consuming alcohol while taking Nurofen Plus.
Use of opioids in chronic (long-term) non-cancer pain (CNCP). Opioid analgesics have an established role in the treatment of acute pain, cancer pain and palliative and end-of-life care. Current evidence does not generally support opioid analgesics in improving pain and function for most patients with chronic non-cancer pain. The development of tolerance and physical dependence and risks of adverse effects, including hazardous and harmful use, increase with the length of time a patient takes an opioid. The use of opioids for long-term treatment of CNCP is not recommended.
The use of an opioid to treat CNCP should only be considered after maximised nonpharmacological and non-opioid treatments have been tried and found ineffective, not tolerated or otherwise inadequate to provide sufficient management of pain. Opioids should only be prescribed as a component of comprehensive multidisciplinary and multimodal pain management.
Opioid therapy for CNCP should be initiated as a trial in accordance with clinical guidelines and after a comprehensive biopsychosocial assessment has established a cause for the pain and the appropriateness of opioid therapy for the patient (see Hazardous and harmful use, above). The expected outcome of therapy (pain reduction rather than complete abolition of pain, improved function and quality of life) should be discussed with the patient before commencing opioid treatment, with agreement to discontinue treatment if these objectives are not met.
Owing to the varied response to opioids between individuals, it is recommended that all patients be started at the lowest appropriate dose and titrated to achieve an adequate level of analgesia and functional improvement with minimum adverse reactions.
Immediate-release products should not be used to treat chronic pain, but may be used for a short period in opioid-naïve patients to develop a level of tolerance before switching to a modified-release formulation. Careful and regular assessment and monitoring is required to establish the clinical need for ongoing treatment. Discontinue opioid therapy if there is no improvement of pain and/or function during the trial period or if there is any evidence of misuse or abuse. Treatment should only continue if the trial has demonstrated that the pain is opioid responsive and there has been functional improvement. The patient's condition should be reviewed regularly and the dose tapered off slowly if opioid treatment is no longer appropriate (see Ceasing opioids).
Tolerance, dependence and withdrawal. Neuroadaptation of the opioid receptors to repeated administration of opioids can produce tolerance and physical dependence. Tolerance is the need for increasing doses to maintain analgesia. Tolerance may occur to both the desired and undesired effects of the opioid.
Physical dependence, which can occur after several days to weeks of continued opioid usage, results in withdrawal symptoms if the opioid is ceased abruptly or the dose is significantly reduced. Withdrawal symptoms can also occur following the administration of an opioid antagonist (e.g. naloxone) or partial agonist (e.g. buprenorphine).
Withdrawal can result in some or all of the following symptoms: dysphoria, restlessness/agitation, lacrimation, rhinorrhoea, yawning, sweating, chills, myalgia, mydriasis, irritability, anxiety, increasing pain, backache, joint pain, weakness, abdominal cramps, insomnia, nausea, anorexia, vomiting, diarrhoea, increased blood pressure, increased respiratory rate and increased heart rate.
When discontinuing Nurofen Plus in a person who may be physically-dependent, the drug should not be ceased abruptly but withdrawn by tapering the dose gradually (see Ceasing opioids; see Section 4.2 Dose and Method of Administration).
Neonatal withdrawal syndrome. Chronic use of codeine by the mother at the end of pregnancy may result in a withdrawal syndrome (e.g. hypertonia, neonatal tremor, neonatal agitation, myoclonus, convulsions, apnoea or bradycardia) in the neonate. In many reported cases the withdrawal was serious and required treatment. The syndrome is generally delayed for several hours to several days after birth. (See Section 4.6 Fertility, Pregnancy and Lactation, Use in Pregnancy).
Accidental ingestion/ exposure. Accidental ingestion or exposure of Nurofen Plus especially by children, can result in a fatal overdose of codeine. Patients and their caregivers should be given information on safe storage and disposal of unused Nurofen Plus (see Section 6.4 Special Precautions for Storage; Section 6.6 Special Precautions for Disposal).
Central sleep apnoea. Opioids can cause sleep-related breathing disorders including central sleep apnoea (CSA) and sleep related hypoxemia. Opioid use increases the risk of CSA in a dose-dependent fashion. In patients who present with CSA, consider decreasing the opioid dosage using best practices for opioid taper.
Hyperalgesia. Hyperalgesia may occur with the use of opioids, particularly at high doses. Hyperalgesia may manifest as an unexplained increase in pain, increased levels of pain with increasing opioid dosages or diffuse sensitivity not associated with the original pain. Hyperalgesia should not be confused with tolerance (see Tolerance, dependence and withdrawal). If opioid induced hyperalgesia is suspected, the dose should be reduced and tapered off if possible. A change to a different opioid may be required.
Ceasing opioids. Abrupt discontinuation or rapid decreasing of the dose in a person physically dependent on an opioid may result in serious withdrawal symptoms and uncontrolled pain (see Tolerance, dependence and withdrawal). Such symptoms may lead the patient to seek other sources of licit or illicit opioids. Opioids should not be ceased abruptly in a patient who is physically dependent but withdrawn by tapering the dose slowly. Factors to take into account when deciding how to discontinue or decrease therapy include the dose and duration of the opioid the patient has been taking, the type of pain being treated and the physical and psychological attributes of the patient. A multimodal approach to pain management should be in place before initiating an opioid analgesic taper. During tapering, patients require regular review and support to manage any increase in pain, psychological distress and withdrawal symptoms.
There are no standard tapering schedules suitable for all patients and an individualised plan is necessary. In general, tapering should involve a dose reduction of no more than 10 percent to 25 percent every 2 to 4 weeks (see Section 4.2 Dose and Method of Administration). If the patient is experiencing increased pain or serious withdrawal symptoms, it may be necessary to go back to the previous dose until stable before proceeding with a more gradual taper.
When ceasing opioids in a patient who has a suspected opioid use disorder, the need for medication assisted treatment and/or referral to a specialist should be considered.
Masking of symptoms of underlying infections. Nurofen Plus can mask symptoms of infection, which may lead to delayed initiation of appropriate treatment and thereby worsening the outcome of the infection. This has been observed in bacterial community acquired pneumonia and bacterial complications to varicella. When Nurofen Plus is administered for fever or pain relief in relation to infection, monitoring infection is advised. In non-hospital settings, the patient should consult a doctor if symptoms persist or worsen.
Other precautions. As with other drugs of this class, ibuprofen may mask the usual signs of infection.
Gastrointestinal toxicity. Reports of significant oesophageal dysfunction have been observed via high-resolution manometry in patients taking opioid medicines on a long-term basis. Discontinuation or weaning of opioids should be considered in patients presenting with oesophageal complaints including but not limited to dysphagia, regurgitation, or non-cardiac chest pain.
Codeine may also obscure the diagnosis or the course of gastrointestinal diseases. Nurofen Plus should therefore be administered with caution in such situations.
Nurofen Plus should be administered with caution in patients who have recently had gastrointestinal surgery, paralytic ileus, as codeine may reduce gastrointestinal motility.
Nurofen Plus should be administered with caution in those with hypotension and/or hypothyroidism. The tablets should be used with caution in patients with raised intracranial pressure or head injury.
Nurofen Plus should be administered with caution in patients with prostatic hypertrophy since codeine may cause urinary retention.
Adrenal insufficiency. Adrenal insufficiency has been reported with opioid use, more often following long-term use. Symptoms may include nausea, vomiting, anorexia, fatigue, weakness, dizziness, or low blood pressure. If adrenal insufficiency is suspected, appropriate laboratory testing is recommended and discontinuation of treatment with Nurofen Plus should be considered.
Endocrine effects. Opioids, such as Nurofen Plus, may influence the hypothalamic-pituitary-adrenal or -gonadal axes. Hormonal disturbances that have been observed include an increase in serum prolactin and decreases in plasma cortisol and testosterone. Clinical symptoms may manifest from these hormonal changes.
Androgen deficiency may manifest as low libido, impotence, erectile dysfunction, amenorrhea, or infertility.
Care is advised in the administration of Nurofen Plus to patients with adrenocortical insufficiency.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Nurofen Plus should be avoided in combination with:
Aspirin. Experimental data suggest that ibuprofen may inhibit the effect of low dose aspirin on platelet aggregation when they are dosed concomitantly. However, no clinically relevant effect is considered to be likely for occasional ibuprofen use.
Other NSAIDs: including cyclooxygenase-2-selective inhibitors. Avoid the use of two or more NSAIDs as this may increase the risk of adverse effects.
Nurofen Plus should be used with caution in combination with:
Abiraterone. Abiraterone might reduce analgesic effect of codeine by CYP2D6 inhibition.
Anticholinergics. Concurrent use of codeine and anticholinergic agents may increase the risk of severe constipation and/or urinary retention.
Anticoagulants. Concurrent use of NSAIDs and warfarin has been associated with severe, sometimes fatal haemorrhage. The mechanism of this interaction is not known but may involve increased bleeding from NSAID-induced gastrointestinal ulceration or an additive effect of NSAID inhibition of platelet function with the anticoagulant effect of warfarin. Nurofen Plus should only be used in patients taking warfarin if absolutely necessary. Patients taking this combination must be closely monitored.
Antidiarrhoeal and anti-peristaltic agents. Concurrent use of codeine with antidiarrhoeal and antiperistaltic agents such as loperamide and kaolin may increase the risk of severe constipation.
Antimuscarinics. Concomitant use of antimuscarinics or medications with muscarinic action, e.g. atropine and some antidepressants may result in increased risk of severe constipation which may lead to paralytic ileus and/or urinary retention.
ACE inhibitors, diuretics and other antihypertensives. Ibuprofen, like other NSAIDs can reduce the antihypertensive effect of ACE inhibitors and beta-blockers with possible loss of blood pressure control and can attenuate the natriuretic effects of diuretics.
Hypotensive effects of antihypertensive agents may be potentiated when used concurrently with codeine and lead to orthostatic hypotension. NSAIDs may diminish the effects of antihypertensives and diuretics. Diuretics can increase the risk of nephrotoxicity of NSAIDs.
The combined use of the three classes of drugs, diuretics, an ACE inhibiting drug (ACE inhibitor or angiotensin receptor antagonist) and an anti-inflammatory drug (NSAID or cyclooxygenase-2 (COX-2) inhibitor) all at the same time increases the risk of renal impairment. The combination of drugs from these three classes should be used with caution particularly in elderly patients or those with pre-existing renal impairment.
Antiperistaltic antidiarrhoeals (including kaolin, pectin, loperamide). Concurrent use of these agents with codeine may increase the risk of severe constipation.
Antiplatelet agents and selective serotonin reuptake inhibitors (SSRls). Increased risk of gastrointestinal bleeding (see Section 4.4 Special Warnings and Precautions for Use).
Cardiac glycosides. NSAIDs may exacerbate cardiac failure, reduce GFR and increase plasma glycoside levels. Care should therefore be taken in patients treated with cardiac glycosides.
Central nervous system depressants including alcohol. Codeine may potentiate the effects of CNS depressants, including alcohol and may result in sedation, respiratory depression, coma and death. Because of these risks, concomitant prescribing of Nurofen Plus with CNS depressant medicines, such as other opioid analgesics, benzodiazepines, gabapentinoids, cannabis, sedatives, hypnotics, tricyclic antidepressants, antipsychotics, antihistamines, centrally-active anti-emetics and other CNS depressants, should be reserved for patients for whom other treatment options are not possible (see Section 4.4, Risks from concomitant use of benzodiazepines or other CNS depressants, including alcohol).
Ciclosporin. An increased risk of nephrotoxicity.
Cimetidine. Cimetidine inhibits the metabolism of opioid analgesics resulting in increased plasma concentrations.
Corticosteroids. An increased risk of gastrointestinal ulceration or bleeding may occur with corticosteroids (see Section 4.4 Special Warnings and Precautions for Use).
Drugs that inhibit CYP2D6 such as quinidine, phenothiazines and antipsychotic agents. Can interfere with the metabolism of codeine to morphine, reducing the analgesic effect of codeine.
Gabapentinoids. The concomitant use of codeine with gabapentinoids (gabapentin and pregabalin) may result in respiratory depression, hypotension, profound sedation, coma or death (see Section 4.4 Special Warnings and Precautions for Use).
Hydroxyzine. Concurrent use of hydroxyzine (anxiolytics) with codeine may result in increased analgesia as well as increased CNS depressant, sedative and hypotensive effects.
Lithium. Ibuprofen has been shown to decrease the renal clearance and increase plasma concentrations of lithium. Lithium plasma concentrations should be monitored in patients on concurrent ibuprofen therapy.
Metoclopramide, cisapride and domperidone. Codeine may antagonise the gastrointestinal effects of metoclopramide, cisapride and domperidone.
Methotrexate. NSAIDs inhibit tubular secretion of methotrexate in animals. As a result, reduction in the clearance of methotrexate may occur. Use of high doses of methotrexate concomitantly with NSAIDs should be avoided. At low doses of methotrexate, caution should be used if ibuprofen is administered concomitantly.
Mexiletine. Codeine may delay the absorption of mexiletine and thus reduce the antiarrhythmic effect of the latter.
Mifepristone. NSAIDs should not be used for 8-12 days after mifepristone administration as NSAIDs can reduce the effect of mifepristone.
Moclobemide. Risk of hypertensive crisis.
Monoamine oxidase inhibitors (MAOIs). Concurrent administration or use within 14 days of ceasing monoamine oxidase inhibitors may enhance the potential respiratory depressant effects of codeine. CNS depression or excitation may occur if codeine is given to patients receiving monoamine oxidase inhibitors, or within two weeks of stopping treatment with them.
Naloxone. Naloxone antagonises the analgesic, CNS and respiratory depressant effects of opioid analgesics. Naltrexone also blocks the therapeutic effect of opioids.
NSAIDs and aspirin. Concurrent use of ibuprofen with aspirin or other NSAIDs can lead to increased gastrointestinal adverse effects.
Neuromuscular blocking agents. The respiratory depressant effect caused by neuromuscular blocking agents may be additive to the central respiratory depressant effects of opioid analgesics.
Opioid analgesics. Concurrent use of codeine and other opioid receptor agonists is usually inappropriate as additive CNS depression, respiratory depression and hypotensive effects may occur (see Section 4.4 Special Warnings and Precautions for Use, Risks from concomitant use of benzodiazepines or other CNS depressants, including alcohol).
Probenecid and phenytoin. Interactions may also occur with probenecid, antidiabetic medications and phenytoin.
Quinolone antibiotics. Animal data indicate that NSAIDs can increase the risk of convulsions associated with quinolone antibiotics. Patients taking NSAIDs and quinolone may have an increased risk of developing convulsions.
Quinidine. Quinidine can inhibit the analgesic effect of codeine.
Serotonergic drugs. Serotonin syndrome has been reported during concomitant use of serotonergic drugs including triptans, selective serotonin-reuptake inhibitors (SSRIs), selective serotonin and norepinephrine-reuptake inhibitors (SNRIs), and tricyclic antidepressants, with opioids at recommended dosages.
Tacrolimus. Possible increased risk of nephrotoxicity when NSAIDs are given with tacrolimus.
Tranquillizers, sedatives and hypnotics. Codeine may potentiate the effects of these drugs (see Section 4.4 Special Warnings and Precautions for Use, Risks from concomitant use of benzodiazepines or other CNS depressants, including alcohol).
Zidovudine. Increased risk of haematological toxicity when NSAIDs are given with zidovudine. There is evidence of an increased risk of haemarthroses and haematoma in HIV (+) haemophiliacs receiving concurrent treatment with zidovudine and ibuprofen.
Incompatibilities. Codeine has been reported to be incompatible with phenobarbitone sodium forming a codeine phenobarbitone complex, and with potassium-iodide, forming crystals of codeine periodide. Acetylation of codeine phosphate by acetylsalicylic acid (aspirin) has occurred in solid dosage forms containing the two drugs, even at low moisture levels.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. There is limited evidence that drugs which inhibit cyclo-oxygenase prostaglandin synthesis may cause impairment of female fertility by an effect on ovulation. This is reversible on withdrawal of treatment.
Use in pregnancy. (Category C)
Inhibition of prostaglandin synthesis by ibuprofen may adversely affect pregnancy and/or the embryo/ foetal development.
During the third trimester, all prostaglandin synthesis inhibitors may expose the foetus to cardiopulmonary toxicity (with premature closure of the ductus arteriosus and pulmonary hypertension) and renal dysfunction, which may progress to renal failure with oligohydramnios. At the end of pregnancy, prostaglandin synthesis inhibitors may expose the mother and the neonate to possible prolongation of bleeding time and inhibition of uterine contractions, which may result in delayed or prolonged labour.
Opioid analgesics cross the placenta. Neonatal withdrawal syndrome has been reported in newborn infants with chronic maternal use of codeine during pregnancy. The use of codeine may prolong labour. Administration of codeine during labour may cause respiratory depression in the newborn infant.
Nurofen Plus is therefore contraindicated for use during pregnancy (see Section 4.3 Contraindications).
Oligohydramnios and neonatal renal impairment. Use of NSAIDs from about 20 weeks gestation may cause neonatal renal dysfunction leading to oligohydramnios and, in some cases, neonatal renal impairment.
These adverse outcomes are seen, on average, after days to weeks of treatment, although oligohydramnios has been infrequently reported as soon as 48 hours after NSAID initiation. Oligohydramnios is often, but not always, reversible with treatment discontinuation.
Complications of prolonged oligohydramnios may, for example, include limb contractures and delayed lung maturation. In some post-marketing cases of impaired neonatal renal function, invasive procedures such as exchange transfusion or dialysis were required.
If, after careful consideration of alternative treatment options for pain management, NSAID treatment is necessary from about 20 weeks, limit use to the lowest effective dose and shortest duration possible. Consider ultrasound monitoring of amniotic fluid if treatment extends beyond 48 hours. Discontinue treatment with NSAIDs if oligohydramnios occurs.
Use in lactation. The use of Nurofen Plus during breastfeeding is contraindicated (also see Section 4.4 Special Warnings and Precautions for Use, CYP2D6 metabolism) due to risk of respiratory depression in the infant. Ibuprofen appears in breast milk in very low concentrations. Analgesic doses excreted in breast milk are generally low. However, infants of breastfeeding mothers taking codeine may have an increased risk of morphine overdose if the mother is an ultrarapid metabolizer of codeine. Codeine is excreted into human breast milk. Codeine is partially metabolized by cytochrome P4502D6 (CYP2D6) into morphine, which is excreted into breast milk. If nursing mothers are CYP2D6 ultra-rapid metabolisers, higher levels of morphine may be present in their breast milk. This may result in symptoms of opioid toxicity in both mother and the breastfed infant. Life-threatening adverse events or neonatal death may occur even at therapeutic doses.
Therefore, Nurofen Plus is contraindicated for use during breastfeeding. However, in circumstances where a breastfeeding mother requires codeine therapy, breastfeeding should be suspended and alternative arrangement should be made for feeding the infant for any period during codeine treatment. Breastfeeding mothers should be told how to recognize signs of high morphine levels in themselves and their babies. For example, in a mother, symptoms include extreme sleepiness and trouble caring for the baby. In the baby, symptoms include signs of increased sleepiness (more than usual), difficulty breastfeeding, breathing difficulties, or limpness. Medical advice should be sought immediately.
4.7 Effects on Ability to Drive and Use Machines
Codeine may cause drowsiness.
Opioid analgesics can impair mental function and cause blurred vision and dizziness. Rare side effects may include convulsions, hallucinations, blurred or double vision and orthostatic hypotension. Patients should be advised not to drive or operate machinery.
4.8 Adverse Effects (Undesirable Effects)
The most commonly observed adverse events are gastrointestinal in nature. Adverse events are mostly dose-dependent, in particular the risk of occurrence of gastrointestinal bleeding which is dependent on the dosage range and duration of treatment.
Side effects from codeine are theoretical warnings based on drug class. No clinical data is available to determine frequency.
Regular prolonged use of codeine is known to lead to addiction and symptoms of restlessness and irritability may result when treatment is then stopped.
Prolonged use of a painkiller for headache can make them worse.
The list of the following adverse events relates to those experienced with ibuprofen and codeine (maximum of 1200 mg ibuprofen per day), for short-term use. In the treatment of chronic conditions, under long-term treatment, additional adverse events may occur.
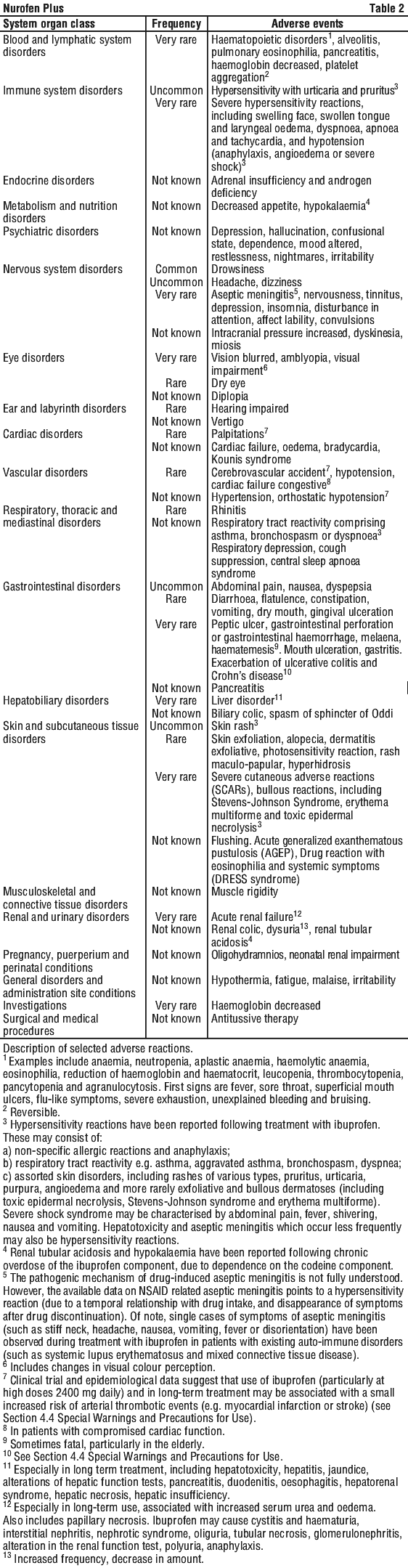
Adverse events which have been associated with ibuprofen and codeine are given in Table 2, tabulated by System Organ Class (SOC) and frequency.
The frequencies of adverse effects are defined as follows.
Very common: ≥ 1/10. Common: ≥ 1/100, < 1/10. Uncommon: ≥ 1/1,000, < 1/100. Rare: ≥ 1/10,000, < 1/1,000. Very rare: < 1/10,000, including isolated reports. Not known: cannot be estimated from the available data.
Within each frequency grouping, adverse events are presented in order of decreased seriousness.
4.9 Overdose
Overuse of this product, defined as consumption of quantities in excess of the recommended dose, or consumption for a prolonged period, may lead to physical or psychological dependency. Symptoms of restlessness and irritability may result when treatment is stopped.
Symptoms. The symptoms of overdose with ibuprofen include nausea, vomiting, abdominal pain, diarrhoea (rarely), headache, dizziness, drowsiness, nystagmus, vertigo, blurred vision, tinnitus and rarely, hypertension, metabolic acidosis, convulsions, excitation, disorientation, coma, renal failure, liver damage, hypotension, respiratory depression, cyanosis and loss of consciousness. Exacerbation of asthma is possible in asthmatics.
Nausea and vomiting are prominent features of codeine overdose. Respiratory depression, excitability, convulsions, hypotension and loss of consciousness may occur with large codeine overdose. Central nervous system depression, including respiratory depression, may develop but is unlikely to be severe unless other sedative agents have been co-ingested, including alcohol, or the overdose is very large. The pupils may be pinpoint in size. Hypotension and tachycardia are possible. Toxic leukoencephalopathy has been observed with opioid overdose.
Prolonged use at higher than recommended doses may result in severe hypokalaemia and renal tubular acidosis. Symptoms may include reduced level of consciousness and generalised weakness (see Section 4.4 Special Warnings and Precautions for Use; Section 4.8 Adverse Effects (Undesirable Effects)).
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. It is thought that ibuprofen produces an anti-inflammatory effect at least in part by inhibiting prostaglandin synthetase. Ibuprofen has shown anti-inflammatory, analgesic and antipyretic activity in both animal and human studies.
Codeine phosphate hemihydrate is a narcotic analgesic acting on central opiate receptors, although its pharmacological effects are thought to be largely due to its biotransformation to morphine.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Ibuprofen. Absorption. Ibuprofen is well absorbed after oral administration with peak serum levels occurring after 1 to 2 hours.
Distribution. Apparent volume of distribution is 0.14 L/kg. Ibuprofen and its metabolites readily cross the placental barrier in pregnant rabbits and rats. It is not known if the drug enters the cerebrospinal fluid.
99% of ibuprofen is protein bound. The high protein binding of the drug should be borne in mind when prescribing ibuprofen together with other protein bound drugs that bind to the same site on human serum albumin.
Metabolism. 90% of ibuprofen is metabolised in the liver to produce two major metabolites, a hydroxylated and carboxylated compound.
Excretion. Both the inactive metabolites and a small amount of unchanged ibuprofen are excreted rapidly and completely by the kidney with 95% of the administered dose eliminated in the urine within four hours of ingestion.
The elimination half-life of ibuprofen is in the range 1.9 to 2.2 hours.
Codeine. Absorption. Codeine is well absorbed from the gastrointestinal tract and peak plasma concentrations are reached one hour after oral administration. Onset of action occurs in 15 to 30 minutes and analgesia is maintained for 4 to 6 hours.
Distribution. Codeine is rapidly distributed to skeletal muscles, kidneys, liver, gastrointestinal tract, lungs, spleen and brain. It crosses the placenta and is distributed in low levels in breast milk.
Metabolism. Codeine is metabolised mainly in the liver. The major metabolic pathway involves glucuronidation of codeine to codeine-6-glucuronide. Codeine can also undergo O- and N-demethylation catalysed by CYP2D6 and CYP3A4 respectively.
About 10% of an administered dose of codeine is converted by O-demethylation to morphine, which subsequently undergoes glucuronidation to morphine-3 or morphine-6 glucuronide, or N-demethylation to normorphine. Approximately 8% of the general Australian population cannot convert codeine to the active metabolite morphine as they are deficient in the CYP2D6 enzyme. These patients are likely to obtain reduced pain relief from codeine. Codeine is also converted by N-demethylation to norcodeine, which subsequently undergoes glucuronidation to norcodeine glucuronide or O-demethylation to normorphine.
Excretion. Codeine is excreted mainly by the kidneys. Of the excreted material in the urine 40-70% is free or conjugated codeine, 5-15% is free or conjugated morphine, and 10-20% is free or conjugated norcodeine. The plasma half-life of codeine is 2 to 4 hours. Only traces of codeine and its metabolites are found in the faeces.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Also containing microcrystalline cellulose, sodium starch glycollate, hypromellose, pregelatinised maize starch, talc and Opaspray white colouring.
Nurofen Plus tablets are gluten-free and lactose-free.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 25 degrees Celsius.
6.5 Nature and Contents of Container
Aluminium blister packed in a carton.
Packs of *4, *6, *12, *24 and 30 tablets.
*Not available in Australia.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Ibuprofen. Chemical name: 2-(4-Isobutylphenyl) propionic acid. It is a white or almost white powder or crystals with a characteristic odour. Practically insoluble in water, soluble 1 in 1.5 of alcohol, 1 in 1 of chloroform, 1 in 2 of ether and 1 in 1.5 of acetone; soluble in aqueous solutions of alkali hydroxides and carbonates.
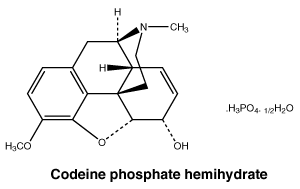
Codeine phosphate hemihydrate. Chemical name: (5R,6S)-7,8-didehydro-4,5-epoxy-3-methoxy-N-methylmorphinan-6-ol dihydrogen orthophosphate hemihydrate. It is a small, colourless, odourless crystal or a white, odourless crystalline powder. Codeine phosphate is soluble in four parts water, slightly soluble in ethanol (96%), practically insoluble in chloroform and ether.
Chemical structure.
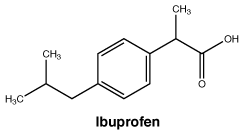
Chemical formula. Ibuprofen: C13H18O2.
Codeine phosphate hemihydrate: C18H21NO3.H3PO4.1/2 H2O.
CAS number. Ibuprofen CAS: 15687-27-1.
Codeine phosphate hemihydrate CAS: 41444-62-6.
7 Medicine Schedule (Poisons Standard)
S4 (Prescription Only Medicine).
Date of First Approval
17 February 2005
Date of Revision
19 November 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.