Ocuflox
Brand Information
| Brand name | Ocuflox |
| Active ingredient | Ofloxacin |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Ocuflox.
Summary CMI
OCUFLOX®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using OCUFLOX®?
OCUFLOX® eye drops contain the active ingredient ofloxacin. OCUFLOX® is used in adults to treat ulcers on the front layer of the eye (the cornea) and severe eye infection (conjunctivitis).
For more information, see Section 1. Why am I using OCUFLOX®? in the full CMI.
2. What should I know before I use OCUFLOX®?
Check the list of ingredients at the end of the CMI. Do not use OCUFLOX® if you have ever had an allergic reaction to any of them.
Talk to your doctor before you use this medicine if he/she is not aware that you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use OCUFLOX®? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with OCUFLOX® and affect how it works, or OCUFLOX® may interfere with other medicines and affect how they work. For more information see Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use OCUFLOX®?
- Your doctor will advise how many drops to use depending on your condition
- Your doctor will advise when to start using the drops and when to finish.
- Do not stop using OCUFLOX® earlier than instructed without checking with your doctor, even if you feel better.
More instructions can be found in Section 4. How do I use OCUFLOX®? in the full CMI.
5. What should I know while using OCUFLOX®?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using OCUFLOX®? in the full CMI.
6. Are there any side effects?
Burning, stinging, watering eye, itchy eye, eyelid, feeling something in the eye, unable to tolerate sunlight, eye redness, inflamed eye, swelling of eyelid, face, dry eye, eye pain, skin rash, blurred vision, feeling dizzy, severe allergic reaction.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
OCUFLOX® (ock-you-flox)
Active ingredient: ofloxacin (oh-flox-ah-sin)
Consumer Medicine Information (CMI)
This leaflet provides important information about using OCUFLOX® eye drops. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using OCUFLOX®.
Where to find information in this leaflet:
1. Why am I using OCUFLOX®?
2. What should I know before I use OCUFLOX®?
3. What if I am taking other medicines?
4. How do I use OCUFLOX®?
5. What should I know while using OCUFLOX®?
6. Are there any side effects?
7. Product details
1. Why am I using OCUFLOX®?
OCUFLOX® contains the active ingredient ofloxacin.
Ofloxacin is an antibiotic and acts by killing a wide range of bacteria that cause certain eye infections.
OCUFLOX® is used in adults to treat ulcers on the front layer of the eye (corneal ulcers) and severe eye infections (conjunctivitis).
2. What should I know before I use OCUFLOX®?
Warnings
Do not use OCUFLOX® if:
- you are allergic to ofloxacin, or any of the ingredients listed at the end of this leaflet.
- you are allergic to any similar antibiotics, such as ciprofloxacin, moxifloxacin, nalidixic acid.
- the seal around the cap is broken.
- the bottle/packaging shows signs of tampering.
- the product does not look quite right.
- the expiry date on the bottle has passed.
Check with your doctor if you:
- have or have had unusual sensitivity to sunlight or artificial ultraviolet light
- take any medicines for any other condition.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Make sure your doctor is aware that you are pregnant or plan to become pregnant or are breastfeeding or intend to breastfeed.
OCUFLOX® is not recommended in pregnant or breastfeeding women.
Children
OCUFLOX® should not be used in children younger than 12 years of age.
3. What if I am taking other medicines?
It is not expected that OCUFLOX will interact with other medicines, however tell your optical practitioner, doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins, or supplements that you buy without a prescription from your pharmacy, supermarket, or health food store.
Check with your optical practitioner, doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect OCUFLOX®.
4. How do I use OCUFLOX®?
How much to use, and when to use OCUFLOX®
Your doctor will tell you how many drops you need to use each day. Use OCUFLOX® as prescribed by your doctor.
Do not stop using OCUFLOX® earlier than instructed without checking with your doctor, even if you feel better.
Corneal ulcer:
- 1 to 2 drops in the affected eye(s) every 30 minutes (during waking hours) and also 1 to 2 drops into the affected eye(s) during the night 4 hours after retiring and 2 hours after this, for 2 days, then
- 1 to 2 drops into the affected eye(s) every hour (during waking hours) for 5 days, then
- 1 to 2 drops into the affected eye(s) 4 times each day until your doctor tells you to stop.
Conjunctivitis:
- 1 drop in the affected eye(s) every 4 hours for the first 2 days, then
- 1 drop every 6 hours for up to 8 days
- You should not use these drops for longer than 10 days without checking with your doctor.
How to use OCUFLOX®
- These eye drops are for topical use only.
- Wash your hands well with soap and water.
- If you wear soft contact lenses, remove them before using OCUFLOX®.
- Tear off the protective seal on any new bottle and throw the seal away. Remove the lid/cap.
- Hold the bottle upside down in one hand between your thumb and index finger.
- Using your other hand, gently pull down your lower eyelid to form a pouch or pocket.
- Tilt your head back and look up. This might be easier if you are sitting or lying down.
- Put the tip of the bottle close to your lower eyelid. Do not let the tip touch your eye, eyelid or any other surface.
- Release one drop into the pouch or pocket formed between your eye and eyelid by gently squeezing the bottle.
- If a drop misses your eye, try again, and wipe off any excess that runs down your cheek.
- Close your eye. Do not blink or rub your eye.
- While your eye is closed, place your index finger against the inside corner of your eye and press against your nose for about two minutes. This will help to stop the medicine from draining through the tear duct to the nose and throat, from where it can be absorbed into other parts of your body.
- Replace the lid/cap, sealing it tightly.
- Wash your hands again with soap and water.
- If you wear soft contact lenses, wait 15 minutes before replacing your contact lenses.
If you forget to use OCUFLOX®
It is important that you use OCUFLOX® strictly as instructed.
If you forget to use OCUFLOX® and it is almost time for your next dose, skip the missed dose and use the drops when you are next meant to.
Otherwise, use the drops as soon as you remember, and then go back to using them as you would normally. If you are not sure whether to skip the dose, talk to your doctor or pharmacist.
Do not use a double dose to make up for the dose you missed.
If you use too much OCUFLOX®
Immediately rinse your eyes with warm water.
If OCUFLOX® is accidentally swallowed, immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there appear to be no signs of discomfort or poisoning.
5. What should I know while using OCUFLOX®?
Things you should do
- If you wear soft contact lenses, remove them before using OCUFLOX®, and wait at least 15 minutes before inserting them again. The preservative in OCUFLOX® eye drops (benzalkonium chloride) may be absorbed and cause discolouration of soft contact lenses.
- Avoid direct exposure to sunlight or artificial ultraviolet light when using OCUFLOX®.
- Follow all your doctor's instructions carefully.
- Tell your doctor if your eye(s) infection seem(s) to be getting worse or is/are not improving.
Call your doctor straight away if you:
- experience an eye injury, or have eye surgery
- become pregnant while using OCUFLOX®.
Remind any doctor, dentist, pharmacist, or optometrist you visit that you are using OCUFLOX®.
Things you should not do
- Do not to touch the dropper tip against your eye, eyelid, or anything else, to avoid contaminating the eye drops and to avoid injury to the eye caused by the tip.
- Do not stop using OCUFLOX® earlier than instructed, or use it for longer than instructed or change the dose without checking with your doctor, even if you think you are better.
Driving or using machines
Wait for your vision to clear after using OCUFLOX® before you drive or use machines.
Looking after your medicine
Follow the instructions on the carton and bottle label on how to take care of your medicine properly.
Store it in a cool place away from heat or sunlight; for example:
- do not store it in the car or on window sills.
Keep it where young children cannot reach it.
When to discard your medicine
Discard the bottle 4 weeks after first opening it.
Getting rid of any unwanted medicine
If your doctor advises that you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
The most common side effects when using OCUFLOX® are burning or stinging eye. These side effects are usually temporary and mild. While less common, the most serious side effects of OCUFLOX® are severe allergic reactions. See the information below and, if you need to, ask your optical practitioner, doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Effects on the eye:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Effects on the eye:
| Call your doctor straight away, if you notice any of these serious side effects. |
Effects on the body as a whole:
| Go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What OCUFLOX® contains
| Active ingredient (main ingredient) | ofloxacin |
| Preservative |
|
| Other ingredients (inactive ingredients) |
|
Do not take this medicine if you are allergic to any of these ingredients.
What OCUFLOX® looks like
OCUFLOX® is a clear, pale to light yellow-green solution. It is supplied in a white plastic dropper bottle containing 5 mL of liquid, which is enclosed in a carton.
(Aust R 47485).
Who distributes OCUFLOX®?
OCUFLOX® is distributed in Australia by:
AbbVie Pty Ltd
Mascot NSW 2020
AUSTRALIA
Ph: 1800 252 224
This leaflet was prepared in July 2025.
© 2025 AbbVie. All rights reserved.
OCUFLOX® and its design are trademarks of Allergan, Inc., an AbbVie company.
Brand Information
| Brand name | Ocuflox |
| Active ingredient | Ofloxacin |
| Schedule | S4 |
MIMS Revision Date: 01 October 2023
1 Name of Medicine
Ofloxacin.
2 Qualitative and Quantitative Composition
Ocuflox eye drops contain 3 mg/mL ofloxacin.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Eye drops, solution.
4 Clinical Particulars
4.1 Therapeutic Indications
Ocuflox eye drops are indicated for the treatment of corneal ulcers (bacterial keratitis) and severe bacterial conjunctivitis caused by ofloxacin sensitive organisms in adults.
4.2 Dose and Method of Administration
Corneal ulcers (bacterial keratitis). Days 1 and 2. Instill one to two drops into the affected eye(s) every 30 minutes while awake. Instill a further one to two drops into the affected eye(s) during the night, four hours after retiring, and again two hours after this.
Days 3 to 7. Instill one to two drops into the affected eye(s) every hour while awake.
Days 7 to completion of treatment (usually within 21 days). Instill one to two drops into the affected eye(s) four times daily until the ulcer is completely healed (complete epithelialisation and no progression of infiltrate).
Bacterial conjunctivitis. The dosage recommendation is one drop every four hours for the first two days, and then one drop every six hours into the affected eye(s) for up to eight days. Dosage should not normally be continued for more than 10 days without an ophthalmic review (see Section 4.4 Special Warnings and Precautions for Use).
In order to minimise systemic absorption of Ocuflox eye drops, apply pressure to the tear duct immediately following administration of the drug.
Ocuflox eye drops have been assessed in clinical studies for up to 23 days treatment; safety has not been adequately demonstrated for longer periods of use.
4.3 Contraindications
Ocuflox eye drops are contraindicated in patients sensitive to ofloxacin or any other component of the solution. A history of hypersensitivity to other quinolone anti-infectives, including nalidixic acid, may also contraindicate the use of Ocuflox eye drops.
4.4 Special Warnings and Precautions for Use
Identified precautions. The preservative in Ocuflox eye drops, benzalkonium chloride, may be absorbed by and cause discoloration of soft contact lenses. Ocuflox eye drops should not be administered while wearing soft contact lenses. Patients should be instructed to remove contact lenses prior to the administration of Ocuflox and wait at least 15 minutes following administration before reinserting soft contact lenses.
Patients should be instructed to avoid allowing the tip of the dispensing container to contact the eye or surrounding structures to avoid eye injury and contamination of eye drops.
Ocuflox eye drops are not for injection.
Serious and occasionally fatal hypersensitivity (anaphylactic/ anaphylactoid) reactions, some following the first dose, have been reported in patients receiving systemic quinolones, including ofloxacin. Some reactions were accompanied by cardiovascular collapse, loss of consciousness, angioedema (including laryngeal, pharyngeal or facial edema), airway obstruction, dyspnea, urticaria, and itching. Serious, acute hypersensitivity reactions may require immediate emergency treatment. Oxygen and airway management should be administered as clinically indicated.
There have been rare reports of Stevens-Johnson syndrome and toxic epidermal necrolysis reported in association with topical ophthalmic ofloxacin.
Hypersensitivity reactions including angioedema, dyspnea, anaphylactic reaction/ shock, oropharyngeal swelling, and tongue swollen have been reported with Ocuflox eye drops (see Section 4.8 Adverse Effects (Undesirable Effects), Postmarketing experience).
If an allergic reaction to ofloxacin occurs, discontinue the drug. Use Ocuflox eye drops with caution in patients who have exhibited sensitivities to other quinolone antibacterial agents.
As with other anti-infectives, prolonged use may result in overgrowth of non-susceptible organisms. If superinfection occurs, or if clinical improvement is not noted within a reasonable period, discontinue use and institute appropriate therapy.
Corneal precipitates, and corneal perforation in patients with pre-existing corneal epithelial defect/ corneal ulcers, have been reported during treatment with topical ophthalmic ofloxacin. However, a causal relationship has not been established.
Quinolones induce phototoxicity in a number of in vitro and in vivo animal models. Quinolones have the potential to produce phototoxic reactions in sensitive individuals following systemic administration. Patients taking ofloxacin should avoid direct exposure to sunlight or artificial ultraviolet light. Therapy should be discontinued if photosensitivity occurs.
While systemic concentrations of ofloxacin are low following topical dosing, neurological adverse reactions (including convulsions, increased intra-cranial pressure and toxic psychosis) have been associated with oral administration.
Long-term, high dose use of other fluoroquinolones in experimental animals has caused lenticular opacities. However, this effect has not been reported in human patients, nor has it been noted following topical ophthalmic treatment with ofloxacin for up to six months in animal studies including studies in monkeys.
Use in the elderly. No comparative data are available with topical dosing in elderly versus other age groups.
Paediatric use. Adequate clinical studies of the safety of topical ophthalmic treatment with ofloxacin in children have not been conducted. Ocuflox eye drops should be avoided in children who have not attained joint maturity. The oral administration of quinolones (including norfloxacin, ciprofloxacin, ofloxacin, nalidixic acid and cinoxacin) has led to lesions or erosions of the cartilage in weight-bearing joints and other signs of arthropathy in immature animals of various species.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
It has been shown that the systemic administration of some quinolones inhibits the metabolic clearance of caffeine and theophylline. Drug interaction studies conducted with systemic ofloxacin have demonstrated that metabolic clearance of caffeine and theophylline are not significantly affected by ofloxacin.
Although there have been reports of an increased prevalence of CNS toxicity with systemic dosing of fluoroquinolones when used concomitantly with systemic nonsteroidal anti-inflammatory drugs (NSAIDs), this has not been reported with the concomitant systemic use of NSAIDs and ofloxacin.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category B3)
There were no adequate and well-controlled studies performed in pregnant women. Since systemic quinolones have been shown to cause arthropathy in immature animals, it is recommended that Ocuflox eye drops not be used in pregnant women.
Ofloxacin has not been shown to have any teratogenic effects at oral dose up to 810 mg/kg/day and 160 mg/kg/day when administered to pregnant rats and rabbits, respectively. Additional studies in rats with oral doses up to 360 mg/kg/day demonstrated no adverse effects on late foetal development, labour, delivery, lactation, neonatal viability or growth of the newborn. Doses of 810 mg/kg/day and 160 mg/kg/day resulted in decreased fetal bodyweight and increased fetal mortality in rats and rabbits, respectively. Minor fetal skeletal variations were reported in rats receiving doses of 810 mg/kg/day.
Use in lactation. Because ofloxacin and other quinolones taken systemically are excreted in breast milk and there is potential for harm to nursing infants, a decision should be made whether to temporarily discontinue nursing or not to administer the drug, taking into account the importance of the drug to the mother.
4.7 Effects on Ability to Drive and Use Machines
As with any ocular medication, if transient blurred vision occurs at instillation, the patient should wait until the vision clears before driving or using machinery.
4.8 Adverse Effects (Undesirable Effects)
The following adverse reactions have been reported in association with use of ofloxacin.
Transient side effects reported with Ocuflox eye drops include burning/ stinging (10-14% of treated eyes), tearing (6-11% of treated eyes), itching, foreign body sensation, photophobia, blurred vision, hyperemia, conjunctivitis, chemical conjunctivitis/ keratitis, periocular/ facial oedema, eye oedema, eye pruritus, eyelid pruritus, dry eyes, eye pain (1-5% of treated eyes) and dizziness. These symptoms led to cessation of treatment in 1.6% of patients.
Adverse events such as burning and stinging, tearing, photophobia and foreign body sensation occur more frequently in patients treated for corneal ulcer. The incidence of discomfort is likely to be a result of the underlying condition.
Gastrointestinal disorders reported include nausea.
Since a small amount of ofloxacin is systemically absorbed after topical administration, side effects reported with systemic use could possibly occur.
Postmarketing experience. The following adverse reactions have been identified during post-marketing use of Ocuflox eye drops in clinical practice. Because they are reported voluntarily from a population of unknown size, estimates of frequency cannot be made.
Eye disorders. Hypersensitivity, lacrimation increased, eyelid oedema and ocular hyperemia.
Immune system disorders. Hypersensitivity (including angioedema, dyspnea, anaphylactic reaction/ shock, oropharyngeal swelling and tongue swollen) and allergic dermatitis.
Nervous system disorders. Dizziness.
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
The acute oral LD50 values in male/ female mice and male/ female rats exceed 5 g/kg and 3 g/kg respectively. In monkeys, the acute oral LD50 value is greater than 0.5 g/kg. Acute overdosage information for humans is not available.
Signs of toxicity after oral or subcutaneous administration included hypoactivity, ptosis, hypopnoea, convulsion and tremor in rats, mice, dogs and monkeys. In addition, emesis was observed in dogs and monkeys.
In the event of accidental ingestion of 5 mL of Ocuflox eye drops, 15 mg of ofloxacin would be ingested. This amount does not appear to be clinically significant in terms of overdosage. However, there would be an increased potential for systemic reactions (see Section 4.8 Adverse Effects (Undesirable Effects)).
In the event of a topical overdosage, flush eye with a sterile topical ocular irrigant.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Ofloxacin is a third generation fluorinated 4-quinolone having broad spectrum in vitro bactericidal activity against certain aerobic Gram-positive, Gram-negative and some anaerobic bacteria.
Ofloxacin appears to have more than one mechanism contributing to its bactericidal action. The primary mechanism of action is believed to be the inhibition of bacterial DNA gyrase, the enzyme responsible for inserting negative supercoils into bacterial DNA. Apparently, this enzyme inhibition leads to bacterial death through a complex process in which DNA synthesis is arrested and regulation of normal gene expression is disrupted. Ofloxacin, unlike most of the other quinolones, possesses an additional bactericidal mechanism which is not dependent on protein or RNA synthesis. It is bactericidal in both the replicating and nonreplicating stages of bacterial growth.
Ofloxacin, in vitro maintains an inhibitory effect on cell growth of susceptible bacteria for 6-8 hours after drug removal.
Ofloxacin is not subject to degradation by beta-lactamase enzymes nor is it modified by enzymes such as aminoglycoside adenylases or phosphorylases, or chloramphenicol acetyltransferase. Spontaneous resistance is rare and occurs in only 1 in 1010 to 1011 sensitive bacteria under routine laboratory conditions. Development of resistance to greater than 8 microgram/mL of ofloxacin typically requires two independent genetic mutations under aerobic conditions. The viability and pathogenicity of most resistant mutants are reduced. Resistant mutants are typically unstable and most readily revert to full sensitivity to ofloxacin when cultured without quinolones.
Clinical trials. Corneal ulcer. In a randomised, double-blind, parallel group trial of 134 patients with positive bacterial cultures, Ocuflox treated patients had an overall clinical success rate of 86%. The median time to clinical success was 11 days of treatment (range 8-14 days).
5.2 Pharmacokinetic Properties
Absorption. Tear film ofloxacin concentrations ranged from 5.67 to 31.0 microgram/g during the 40-minute period following the last dose in the 11-day study. In 5 subjects mean tear film levels measured four hours after topical dosing (9.16 ± 8.24 microgram/g) were higher than the 2 microgram/mL minimum concentration of ofloxacin necessary to inhibit 90% of most bacterial strains (MIC90) in vitro.
Distribution. In rabbits, an eye-drop instillation produced therapeutically effective concentrations of ofloxacin in tears (i.e. above MIC90 for most ocular pathogens) for four to six hours.
Metabolism. Serum, urine and tear film concentrations of ofloxacin were measured in 30 healthy women at various time points during a ten-day course of treatment with 3 mg/mL ofloxacin eye drops. The mean serum ofloxacin concentration ranged from 0.4 nanogram/mL to 1.9 nanogram/mL. Maximum ofloxacin concentration increased from 1.1 nanogram/mL on day one to 1.9 nanogram/mL on day 11 after QID dosing for 10 to 12 days. Maximum serum ofloxacin concentrations (1.89 ± 1.13 nanogram/mL) after ten days of topical dosing were more than 1000 times lower than those reported after standard oral doses of ofloxacin.
Excretion. Topical ofloxacin was excreted in the urine primarily in unmodified form.
5.3 Preclinical Safety Data
Genotoxicity. Ofloxacin shows high selectivity for the bacterial DNA gyrase enzyme while showing little activity against mammalian topoisomerase (counterpart mammalian target) enzyme.
Cross-resistance has been observed between ofloxacin and other fluoroquinolones. Ofloxacin has shown in vitro efficacy against certain organisms resistant to other types of antimicrobials, including aminoglycosides, chloramphenicol, macrolides (erythromycin), sulfacetamide, penicillins and tetracycline.
Carcinogenicity. Long term studies to determine the carcinogenic potential of ofloxacin have not been conducted.
6 Pharmaceutical Particulars
6.1 List of Excipients
Ocuflox eye drops contain 3 mg/mL ofloxacin and are formulated as an isotonic solution using 9 mg/mL sodium chloride preserved with 0.05 mg/mL benzalkonium chloride. The pH of Ocuflox eye drops range from 6.0 - 7.0.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
2 years.
6.4 Special Precautions for Storage
Store below 25°C. Protect from light and excessive heat.
To avoid contamination of the solution, keep container tightly closed.
Do not touch dropper tip to any surface.
Discard contents 4 weeks after opening the bottle. Contents are sterile if seal is intact.
6.5 Nature and Contents of Container
Eye drops: 5 mL (dropper bottle). AUST R 47485.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Chemical structure.
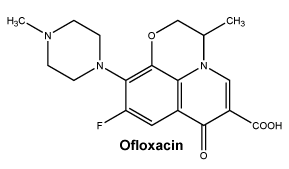
Chemical name: (±)-9-Fluoro-2,3-dihydro-3-methyl-10-(4-methyl-1-piperazinyl)-7-oxo-7H-pyrido [1, 2, 3-de]-1, 4 benzoxazine-6-carboxylic acid.
CAS number. 82419-36-1.
7 Medicine Schedule (Poisons Standard)
S4 - Prescription Only Medicine.
Date of First Approval
02 December 1997
Date of Revision
23 August 2023
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.