Omegapharm Acetylcysteine Solution
Brand Information
| Brand name | Omegapharm Acetylcysteine Solution |
| Active ingredient | Acetylcysteine |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Omegapharm Acetylcysteine Solution.
What is in this leaflet
This leaflet answers some common questions about Omegapharm Acetylcysteine Solution for Inhalation.
It does not contain all the available information.
It does not take the place of talking with your doctor or pharmacist.
All medicines have risks and benefits. Your doctor has weighed the risks of your treatment using Omegapharm Acetylcysteine Solution for Inhalation against the benefits they expect it will have for you.
If you have any concerns about taking this medicine, ask your doctor or pharmacist.
Keep this leaflet with the medicine. You may need to read it again.
What Omegapharm Acetylcysteine Solution for Inhalation is used for
The name of your medicine is Omegapharm Acetylcysteine Solution for Inhalation. It contains the active ingredient called acetylcysteine.
Omegapharm Acetylcysteine Solution for Inhalation reduces the stickiness of fluid in your airways and lungs, making it more liquid.
Omegapharm Acetylcysteine Solution for Inhalation is used in patients with abnormal, sticky or thick mucous secretions in conditions such as:
- ongoing lung disease such as pneumonia, bronchitis, emphysema, tuberculosis and other conditions that affect your breathing
- short term lung disease such as pneumonia or bronchitis
- lung problems of cystic fibrosis
- lung problems occurring after an accident or after surgery
- other lung problems associated with having mucus in your airway
Omegapharm Acetylcysteine Solution for Inhalation may also be used:
- when you are having tests carried out on your lungs
- as part of tracheostomy care
- when you are receiving an anaesthetic
Your doctor may have prescribed Omegapharm Acetylcysteine Solution for Inhalation for another reason. Ask your doctor if you have any questions about why Omegapharm Acetylcysteine Solution for Inhalation has been prescribed for you.
There is no evidence that acetylcysteine is addictive.
This medicine is available only with a doctor's prescription.
Before you use Omegapharm Acetylcysteine Solution for Inhalation
When you must not use it
Do not use Omegapharm Acetylcysteine Solution for Inhalation if you have an allergy to acetylcysteine or any of the ingredients listed at the end of this leaflet. Symptoms of an allergic reaction may include shortness of breath, wheezing or difficulty breathing.
Do not use Omegapharm Acetylcysteine Solution for Inhalation after the expiry date printed on the pack. If you take this medicine after the expiry date has passed, it may not work as well.
Do not use Omegapharm Acetylcysteine Solution for Inhalation if the packaging is torn or shows signs of tampering.
If you are not sure whether you should start using Omegapharm Acetylcysteine Solution for Inhalation, talk to your doctor.
Before you start to use it
Tell your doctor if you have allergies to:
- acetylcysteine or any of the ingredients listed at the end of this leaflet
- any other medicines
- any other substances, such as foods, preservatives or dyes
Tell your doctor if you are pregnant or intend to become pregnant. Your doctor will discuss the risks and benefits of using Omegapharm Acetylcysteine Solution for Inhalation during pregnancy.
Tell your doctor if you are breast-feeding or plan to breast-feed. It is recommended that if Omegapharm Acetylcysteine Solution for Inhalation must be given to breast-feeding mothers, the infant should not be breast-fed during therapy.
Tell your doctor if you have or have had any other medical conditions, especially the following:
- asthma
- difficulty in coughing
- kidney disease
- liver disease
If you have not told your doctor about any of the above, tell them before you start using Omegapharm Acetylcysteine Solution for Inhalation.
Taking other medicines
Tell your doctor if you are taking any other medicines, including any that you buy without a prescription from your pharmacy, supermarket or health food shop. Some medicines may be affected by acetylcysteine, or may affect how well it works. You may need to take different amounts of your medicine, or you may need to take different medicines. Your doctor will advise you.
Your doctor and pharmacist may have more information on medicines to be careful with or avoid while taking Omegapharm Acetylcysteine Solution for Inhalation.
How to use Omegapharm Acetylcysteine Solution for Inhalation
How much to use
Omegapharm Acetylcysteine Solution for Inhalation may be used undiluted or diluted with an equal volume of either sterile normal saline or sterile water for injections.
The recommended dose for most patients is 3 to 5 mL of the undiluted solution or 5 to 10 mL of the diluted solution, three to four times a day.
Follow your doctor's instructions about how much Omegapharm Acetylcysteine Solution for Inhalation you should use, how often you should take it and for how long. They may differ from the information contained in this leaflet.
If you do not understand the instructions on the box, ask your doctor or pharmacist for help.
How to use it
Omegapharm Acetylcysteine Solution for Inhalation may be administered in the hospital, in your doctor's office, or in your home.
Outside the hospital, Omegapharm Acetylcysteine Solution for Inhalation is taken by inhalation through a device called a nebuliser. Your doctor will tell you where to obtain the necessary nebulisation equipment. The following procedure should be followed:
- Place the solution in the small chamber called the nebuliser.
- Attach the nebuliser to an oxygen mask.
- Attach the tubing from the oxygen mask to the container of compressed air.
- Place the mask over your nose and mouth, making sure that it fits firmly.
- Feed the compressed air into the tubing at the pressure level your doctor told you.
- Once all of the solution is gone from the nebuliser, turn off the compressed air and remove the mask.
After using Omegapharm Acetylcysteine Solution for Inhalation with a face mask there may be some stickiness remaining on the face. This is easily removed by washing with water.
You may initially notice a slight odour when the Omegapharm Acetylcysteine Solution for Inhalation is nebulised. This is normal and you will very quickly get used to it.
If you have any questions about how to use Omegapharm Acetylcysteine Solution for Inhalation, ask your doctor or pharmacist.
When to use it
Omegapharm Acetylcysteine Solution for Inhalation is usually used three to four times a day. Your doctor's instructions may be different to this.
Always follow your doctor's instructions on when to use Omegapharm Acetylcysteine Solution for Inhalation.
Use Omegapharm Acetylcysteine Solution for Inhalation at the same time every day.
If you forget to use it
If it is almost time for your next dose, skip the dose you missed and use your next dose when you are meant to.
Otherwise, use it as soon as you remember and then go back to taking your medicine as you would normally.
Do not use a double dose to make up for the dose that you missed. This may increase the chance of you getting an unwanted side effect.
If you are not sure what to do, ask your pharmacist.
How long to use it
Omegapharm Acetylcysteine Solution for Inhalation treats the symptoms of your condition, but it does not cure it. Continue taking Omegapharm Acetylcysteine Solution for Inhalation for as long as your doctor tells you.
Do not stop using it unless your doctor tells you to - even if you feel better.
If you use it too much (overdose)
Immediately telephone your doctor or the Poisons Information Centre (telephone 13 11 26), or go to Accident and Emergency at your nearest hospital, if you think that you or anyone else may have taken too much Omegapharm Acetylcysteine Solution for Inhalation. Do this even if there are no signs of discomfort or poisoning. You may need urgent medical attention.
While you are using Omegapharm Acetylcysteine Solution for Inhalation
Things you must do
Tell any other doctors, dentists and pharmacists who are treating you that you are using Omegapharm Acetylcysteine Solution for Inhalation.
If you are about to be started on any new medicine, tell your doctor, dentist or pharmacist that you are using Omegapharm Acetylcysteine Solution for Inhalation.
If you plan to have surgery that needs a general anaesthetic, tell your doctor or dentist that you are using Omegapharm Acetylcysteine Solution for Inhalation.
If you become pregnant while using Omegapharm Acetylcysteine Solution for Inhalation, tell your doctor immediately.
Things you must not do
Do not give Omegapharm Acetylcysteine Solution for Inhalation to anyone else, even if they have the same condition as you.
Do not use Omegapharm Acetylcysteine Solution for Inhalation to treat any other complaints unless your doctor tells you to.
Do not stop using Omegapharm Acetylcysteine Solution for Inhalation, or lower the dosage, without first checking with your doctor.
Things to be careful of
Tell your doctor if you are not coughing properly. Omegapharm Acetylcysteine Solution for Inhalation may increase the amount of runny mucous in your lungs. If you are not coughing properly, you should let your doctor know as he/she may wish to remove the mucus mechanically.
Be careful driving or operating machinery until you know how Omegapharm Acetylcysteine Solution for Inhalation affects you. As with some medicines, Omegapharm Acetylcysteine Solution for Inhalation may cause drowsiness in some people. Make sure you know how you react to Omegapharm Acetylcysteine Solution for Inhalation before you drive a car, operate machinery, or do anything else that could be dangerous if you are affected. If this occurs do not drive. If you drink alcohol, symptoms may become worse.
Side Effects
Tell your doctor as soon as possible if you do not feel well while you are using Omegapharm Acetylcysteine Solution for Inhalation. Omegapharm Acetylcysteine Solution for Inhalation helps most people with mucous problems, but it may have unwanted side effects in a few people. All medicines can have side effects.
Sometimes they are serious, most of the time they are not. You may need medical treatment if you get some of the side effects. If you are over 65 years of age you may have an increased chance of getting side effects.
Ask your doctor or pharmacist to answer any questions you may have.
Tell your doctor if you notice any of the following and they worry you:
- sore mouth
- nausea or vomiting
- runny nose
- drowsiness
- clamminess
These side effects are usually mild.
Tell your doctor immediately if you notice any of the following:
- chest tightness
- wheezing, coughing or difficulty breathing
These may be serious side effects. You may need urgent medical attention. Serious side effects are rare.
Other side effects not listed above may occur in some patients. Tell your doctor if you notice anything else that is making you feel unwell.
Do not be alarmed by this list of side effects. You may not experience any of them.
After using Omegapharm Acetylcysteine Solution for Inhalation
Storage
Store unopened Omegapharm Acetylcysteine Solution for Inhalation vials in a cool dry place, where the temperature stays below 25°C. Do not refrigerate or freeze. Protect from light.
This product contains no antimicrobial preservative and is recommended for single use only. Discard the container after opening.
Do not store Omegapharm Acetylcysteine Solution for Inhalation or any other medicine in the bathroom or near a sink.
Do not leave it on a window sill or in the car on hot days. Heat and dampness can destroy some medicines.
Keep it where children cannot reach it. A locked cupboard at least one-and-a-half metres above the ground is a good place to store medicines.
Disposal
If your doctor tells you to stop using Omegapharm Acetylcysteine Solution for Inhalation or if it has passed its expiry date, ask your pharmacist what to do with any that is left over.
Product Description
What it looks like
Omegapharm Acetylcysteine Solution for Inhalation is a clear, colourless liquid.
Under certain conditions, a light purple colour may appear in the acetylcysteine solution. This does not affect its safety or effectiveness.
Omegapharm Acetylcysteine Solution for Inhalation comes in vials of 4 mL (800 mg/4 mL) or 10 mL (2000 mg/10 mL) and are supplied in a boxes containing 1 or 10 vials.
Ingredients
Active ingredient:
- acetylcysteine
Other ingredients:
- disodium edetate
- sodium hydroxide
- hydrochloric acid
- water for injections
Supplier
Omegapharm Pty Ltd
P O Box 18
Ormond, Victoria 3204
AUSTRALIA
Australian Registration Numbers:
800 mg/4 mL (AUST R 172101)
2000 mg/10 mL (AUST R 180343)
This leaflet was prepared March 2011.
Brand Information
| Brand name | Omegapharm Acetylcysteine Solution |
| Active ingredient | Acetylcysteine |
| Schedule | S4 |
MIMS Revision Date: 01 December 2012
Name of the medicine
Acetylcysteine.
Excipients. Disodium edetate, sodium hydroxide and water for injections. The product may also contain hydrochloric acid for pH adjustment. The pH of the product is maintained in the range of 6.0-7.5.
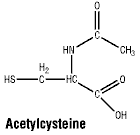
Description
Chemical name: (2R)-2-acetamino- 3-sulfanylpropanoic acid. Molecular formula: C5H9NO3S. MW: 163.2. CAS: 616-91-1. Acetylcysteine is a white, crystalline powder with a slight acetic odour. It is soluble in water and alcohol and practically insoluble in chloroform, dichloromethane and ether. Acetylcysteine is the N-acetyl derivative of the naturally occurring amino acid, L-cysteine. The chemical name is N-acetyl-mercapto-alanine. This product is not for injection.
Pharmacology
Acetylcysteine, the N-acetyl derivative of the naturally occurring amino acid L-cysteine, is neither an enzyme nor a detergent. The viscosity of pulmonary mucous secretions depends on the concentrations of mucoprotein and to a lesser extent deoxyribonucleic acid (DNA). The latter increases with increasing purulence owing to cellular debris. The mucolytic action of acetylcysteine is related to the sulfhydryl group in the molecule. This group probably opens disulfide linkages in mucus, thereby lowering the viscosity. The mucolytic activity of acetylcysteine is unaltered by the presence of DNA, and increases with increasing pH. Significant mucolysis occurs between pH 7 and 9.
Pharmacokinetics. Acetylcysteine is the acetyl derivative of the normally occurring amino acid, L-cysteine. Any absorbed acetylcysteine probably undergoes deacetylation and the remaining cysteine follows the normal pathway of this amino acid.
Indications
Adjuvant therapy for patients with abnormal, viscid or inspissated mucous secretions in such conditions as: chronic bronchopulmonary disease (chronic emphysema, emphysema with bronchitis, chronic asthmatic bronchitis, tuberculosis, bronchiectasis and primary amyloidosis of the lung); acute bronchopulmonary disease (pneumonia, bronchitis, tracheobronchitis); pulmonary complications of cystic fibrosis; pulmonary complications associated with surgery; post-traumatic chest conditions; atelectasis due to mucus obstruction; tracheostomy care; anaesthesia.
Acetylcysteine may also be used as an adjunct to diagnostic bronchial studies (bronchograms, bronchospirometry and bronchial wedge catheterisation).
Contraindications
History of hypersensitivity to acetylcysteine or any of the ingredients in the formulation.
Precautions
After proper administration of acetylcysteine, an increased volume of liquified bronchial secretions may occur. When cough is inadequate, the open airway must be maintained by mechanical suction if necessary. When there is a large mechanical block due to foreign body or local accumulation, the airway should be cleared by endotracheal aspiration, with or without bronchoscopy.
Asthmatic patients under treatment with acetylcysteine should be watched carefully. If bronchospasm progresses, this medication should be immediately discontinued.
If encephalopathy due to hepatic failure becomes evident in patients treated chronically with acetylcysteine, treatment should be discontinued to avoid further administration of nitrogenous substances.
In patients with hepatic failure or renal failure, acetylcysteine should be avoided in order to avoid further administration of nitrogenous substances.
With the administration of acetylcysteine, the patient may observe initially a slight odour that is soon not noticeable. With a face mask there may be stickiness on the face after nebulisation, which is easily removed by washing with water.
Under certain conditions, a light purple colour may develop in the solution of acetylcysteine. This does not significantly affect safety or mucolytic effectiveness.
To prevent the accumulation of acetylcysteine in the solution after prolonged nebulisation, dilute with an equal volume of sterile water for injection, when three quarters of the initial volume has been nebulised.
Use in pregnancy. (Category B2)
There are no well controlled studies in pregnant women, but experience does not include any positive evidence of adverse effects on the fetus. Acetylcysteine should only be used when the expected benefit clearly outweighs the potential risk.
Use in lactation. As it is not known whether this drug is excreted in breast milk, it is recommended that if acetylcysteine must be given to breastfeeding mothers, the infant should not be breastfed during therapy.
Instructions to patients. In cases where domestic inhalation therapy is undertaken, the use of a Maxi-Myst or similar compressor and nebuliser is recommended. This equipment is simple to use, and contains adequate instructions.
Domicilliary patients should be advised of precautions listed above.
Adverse effects
Adverse effects have included stomatitis, nausea, rhinorrhoea, vomiting, other gastrointestinal symptoms, fever, drowsiness, clamminess, chest tightness and bronchoconstriction. Sensitivity and sensitisation to acetylcysteine have been reported very rarely. A few susceptible patients, particularly asthmatics (see Precautions) may experience varying degrees of bronchospasm associated with the administration of nebulised acetylcysteine. Most patients with bronchospasm are quickly relieved by the use of a bronchodilator given by nebulisation. Anaphylaxis, anaphylactoid reaction and angioedema have been reported.
Clinically overt acetylcysteine induced bronchospasm occurs infrequently and unpredictably even in patients with asthmatic bronchitis or bronchitis complicating bronchial asthma. Acquired sensitisation to acetylcysteine has been reported rarely. Sensitisation has been confirmed in several inhalation therapists who reported a history of dermal eruptions after frequent and extended exposure to acetylcysteine.
Dosage and Administration
Acetylcysteine may be administered by direct application, intratracheal instillation or nebulisation. Although acetylcysteine may be administered undiluted, the dilution with an equal volume of either sterile normal saline or sterile water for injections may lessen the occasional occurrence of bronchospasm in susceptible individuals.
Direct application. Acetylcysteine may be introduced directly into a particular segment of the bronchopulmonary tree by inserting (under local anaesthesia and direct vision) a small plastic catheter into the trachea. 2 to 5 mL of 20% solution may then be instilled by means of a syringe connected to the catheter.
Direct instillation: catheter. Acetylcysteine may also be given through a percutaneous intratracheal catheter. 1 to 2 mL of the 20% or 2 to 4 mL of the diluted solution every one to four hours may then be given by a syringe attached to the catheter.
Direct instillation: tracheostomy. When used for the routine nursing care of patients with tracheostomy, 1 to 2 mL of 20% solution may be given every one to four hours by instillation into the tracheostomy.
Nebulisation. (See Precautions.) Nebulisation via face mask, mouth piece or tracheostomy. When nebulised into a face mask, mouth piece or tracheostomy, 1 to 10 mL of 20% solution or 2 to 20 mL of the diluted solution may be given every two to six hours; the recommended dose for most patients is 3 to 5 mL of 20% solution, or 5 to 10 mL of the diluted solution three to four times a day.
Administration of aerosol. Materials. Acetylcysteine may be administered using conventional nebulisers made of plastic or glass. Certain materials used in nebulisation equipment react with acetylcysteine. The most reactive ofthese are certain metals (notably iron and copper) and rubber. Where materials may come into contact with acetylcysteine solution, parts made of the following acceptable materials should be used: glass, plastic, aluminium, anodised aluminium, chromed metal, tantalum, sterling silver or stainless steel. Silver may become tarnished after exposure, but this is not harmful to the drug action nor the patient.
Nebulising gases. Air is recommended. Compressed air from a Maxi-Myst unit is recommended, or a cylinder of compressed air may be used but should be used for nebulising the solution. Oxygen may also be used, but should be used with caution in patients with severe respiratory disease and carbon dioxide retention.
Apparatus. Acetylcysteine is usually administered as fine nebulae for its local effect and the nebuliser used should be capable of providing optimal quantities within a suitable range of particle sizes. Commercially available nebulisers will produce nebulae of acetylcysteine satisfactory for retention in the respiratory tract. Most of the nebulisers tested will supply a high proportion of the drug solution as particles of less than 10 micron in diameter which should be satisfactorily retained in the respiratory tract. Hand bulbs are not recommended for routine use for nebulising acetylcysteine because their output is generally too small. Some hand operated nebulisers deliver particles that are larger than optimum for inhalation therapy. Acetylcysteine should not be used in heated nebulisers. The nebulising equipment should be cleaned immediately after use; the residues may occlude the fine orifices or corrode metal parts.
Compatibility. The physical and chemical compatibility of acetylcysteine with other drugs commonly administered by nebulisation, direct instillation or topical application has been studied. A compatible rating means that there was no visible physical change in the admixture, and that there was no predicted chemical incompatibility. All of the mixtures have been tested for short-term chemical compatibility by assaying for the concentration of acetylcysteine after mixing. The supplying of these data should not be interpreted as a recommendation for combining acetylcysteine with other drugs. The following listing is not presented as positive assurance that no incompatibility will be present, since these data are based only on short-term compatibility studies carried out in the Mead Johnson Research Centre. Manufacturers of pharmaceutical products may change formulations. This could alter compatibilities. These data are intended to serve only as a guide. If it is deemed advisable to prepare an admixture, it should be administered as soon as possible after preparation. Do not store unused mixtures.
Anaesthetic, gas. Halothane, nitrous oxide.
Anaesthetic, local. Lignocaine hydrochloride.
Antibacterials. Neomycin sulfate; streptomycin sulfate; penicillin G potassium (benzylpenicillin potassium) (mix and use at once); polymyxin N sulfate; methicillin sodium; kanamycin sulfate.
Bronchodilators. Isoprenaline hydrochloride; adrenaline.
Contrast media. Propyliodone.
Decongestants. Phenylephrine hydrochloride.
Solvents. Propylene glycol; alcohol.
Steroids. Prednisolone sodium phosphate; dexamethasone 21-phosphate.
Note. Acetylcysteine may not always be pyrogen free and is therefore not suitable for injection.
Overdosage
No treatment known, other than removal of unabsorbed material from the gastrointestinal tract. Oral doses as high as 18 mg of acetylcysteine daily have been reported in adult human beings without ill effects.
For general advice on overdose management contact the Poisons Information Centre on 131 126.
Presentation
Solution for inhalation, 200 mg/mL, 4 mL (AUST R 172101): 1's*,10's; 10 mL* (AUST R 180343): 1's, 10's (clear glass vial).
* Not currently marketed in Australia.
Storage
Store below 25°C. Do not refrigerate or freeze. Protect from light. Shelf life: 18 months.
This product contains no antimicrobial agent and is recommended for single use only. Discard container after opening.
Poison schedule
S4.
Date of First Approval
24 February 2011
Date of Revision
24 February 2011
Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.