Omnaris Nasal Spray
Brand Information
| Brand name | Omnaris Nasal Spray |
| Active ingredient | Ciclesonide |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Omnaris Nasal Spray.
Summary CMI
OMNARIS® Nasal Spray
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using OMNARIS?
OMNARIS contains the active ingredient ciclesonide, a corticosteroid. When sprayed into your nose, OMNARIS helps to stop hayfever (allergic rhinitis) symptoms that are due to pollen (seasonal allergic rhinitis) in people 6 years of age and older, or things in the environment such as house dust mites, pets such as cats or dogs, or moulds (perennial rhinitis) in people 12 years of age and older.
For more information, see Section 1. Why am I using OMNARIS? in the full CMI.
2. What should I know before I use OMNARIS?
Do not use if you have ever had an allergic reaction to ciclesonide, other steroids or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions (e.g., immune compromised, not vaccinated for chickenpox or measles or have any type of infection), damage to or recent surgery on the nose, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
Tell your doctor if you are switching from steroids that you take internally (e.g., by injection or by mouth), to OMNARIS.
For more information, see Section 2. What should I know before I use OMNARIS? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with OMNARIS and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use OMNARIS?
Use OMNARIS in each nostril, once every day, for as long as the doctor tells you. Do not suddenly stop using it.
More instructions can be found in Section 4. How do I use OMNARIS? in the full CMI.
5. What should I know while using OMNARIS?
| Things you should do |
|
| Things you must not do |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using OMNARIS? in the full CMI.
6. Are there any side effects?
Most side effects, if any, will be mild, and include headache, occasional nosebleed, stuffy, dry or painful nose or throat. Serious side effects that you need urgent attention for may be: frequent nosebleed, wheezing or problems breathing and serious allergic reactions (swollen nose, lips, face, eye or throat).
For more details about side effects, and what to do, see Section 6. Are there any side effects? in the full CMI.
Full CMI
OMNARIS® Nasal Spray
Active ingredient: ciclesonide
Consumer Medicine Information (CMI)
This leaflet provides important information about using OMNARIS. You should speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using OMNARIS.
Where to find information in this leaflet:
1. Why am I using OMNARIS?
2. What should I know before I use OMNARIS?
3. What if I am taking other medicines?
4. How do I use OMNARIS?
5. What should I know while using OMNARIS?
6. Are there any side effects?
7. Product details
1. WHY AM I USING OMNARIS?
OMNARIS contains the active ingredient ciclesonide.
Ciclesonide is a corticosteroid which are used to reduce inflammation.
OMNARIS When sprayed into your nose helps to stop seasonal allergic rhinitis in people 6 years of age and older and perennial rhinitis in people 12 years of age and older.
Allergic Rhinitis (hayfever) may cause the inside of your nose to become inflamed or swollen, blocked, runny and itchy and you may sneeze.
You may have symptoms only during spring and summer (seasonal rhinitis) when it is caused by pollens from grasses or trees. Some people may experience symptoms all year round (perennial rhinitis). This is usually caused by house dust mites, pets or moulds.
2. What should I know before I use OMNARIS?
Warnings
Do not use OMNARIS if:
- you are allergic to ciclesonide or any of the ingredients listed at the end of this leaflet
- you are under 6 years of age
Always check the ingredients to make sure you can use this medicine.
Check with your doctor if you:
- have frequent nose bleeds
- have a nasal or sinus infection
- had a recent injury or surgery to your nose
- have ulcers or sores in your nose
- have any type of infection caused by bacteria, virus, fungus, or parasites
- have a history of tuberculosis (TB) infections
- have herpes simplex (virus) infection of the eye
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
They will look at whether the benefits of having OMNARIS outweigh any possible effects on unborn or newborn babies.
Having an operation
If you are going to have surgery, tell the surgeon or anaesthetist that you are using this medicine.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with OMNARIS and affect how it works. These include:
- other corticosteroid medicines such as tablets, asthma inhalers, nasal sprays, or eye/nose drops
- medicines that suppress the immune system (e.g., ciclosporin)
- some antifungal medicines (e.g., ketoconazole, itraconazole)
- some antiviral medicines (e.g., ritonavir, nelfinavir, cobicistat)
- some medicines for heart problems (such as diltiazem, verapamil)
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect with OMNARIS.
4. How do I use OMNARIS?
How much to use
- OMNARIS is for use in the nose only.
- Use two sprays in each nostril once a day.
- Do not use any more than this.
- Elderly people have the same dose as other adults.
Your doctor may change this dosage, depending on how you respond to this medicine, so that you use the lowest dose that controls your symptoms.
Follow the instructions provided and use OMNARIS until your doctor tells you to stop.
How to use your OMNARIS inhaler
- Remove OMNARIS from its foil pouch. Count 4 months from this date and write the date (that is 4 months from removing the bottle from the foil pouch) on the space provided on the nasal spray bottle. It is important to throw away the nasal spray bottle after this date.
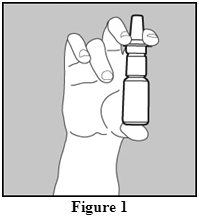
- Before you use OMNARIS for the first time, you will need to prime the bottle. Hold the bottle upright and shake the bottle gently. To prime OMNARIS Nasal Spray, fully press down on the finger rests of the applicator eight times (See Figure 1). If you do not use the nasal spray for 4 days, you will need to shake the bottle gently, and prime the pump again by spraying one time, or until you see a fine mist.
Using the spray
- Gently blow your nose to clear your nostrils if needed
- Shake the bottle gently and remove the cap
- Hold the bottle firmly with your index and middle finger on either side of the nozzle while supporting the base of the bottle with your thumb (Figure 1)
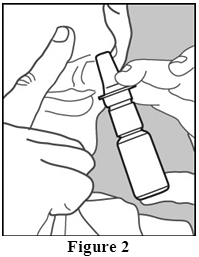
- Insert the nozzle tip into one nostril, and close the other nostril with your finger (Figure 2)
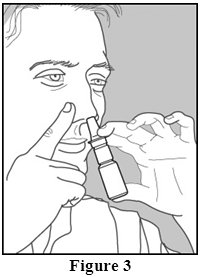
- Tilt your head forward slightly and keeping the bottle upright, press the pump quickly and firmly and inhale through your nose as you spray (Figure 3).
Do not spray in eyes or directly onto the nasal septum (the wall between the two nostrils).
- Repeat steps 3-5 for the second spray in the same nostril and for each spray in the other nostril.
How to clean it
After daily use of your nasal spray, wipe the nozzle tip with a clean tissue and replace the cap.
If the nozzle is clogged or requires more thorough cleaning, use the following cleaning instructions:
- Remove the cap, grasp the white plastic pump firmly with one hand and then carefully pull upwards on the shoulders of the nozzle with the other hand to free the nozzle
- Wash the cap and nozzle with warm water
- Dry and replace the nozzle. The nozzle will snap into place when properly positioned
- Prime the pump by spraying one time or until a fine mist appears
- Replace the cap.
Do not try to unblock the tiny spray hole on the nozzle tip with a pin or other sharp object.
Do not twist or attempt to remove the white plastic pump attached to the medication bottle.
How to know when your nasal spray bottle is empty
The amount of nasal spray left can be seen through a window on the bottle.
If you forget to use OMNARIS
OMNARIS should be used regularly at the same time each day.
If you miss your dose at the usual time, have it as soon as you remember.
If it is almost time for your next dose, skip the dose you missed and have your next dose when you are meant to.
Do not have a double dose to make up for the dose you missed.
If you use too much OMNARIS
If you think that you have used too much OMNARIS on one occasion, there is nothing to worry about.
If you use too much of it over a long time (months or years), you may start to get unwanted side effects inside your nose and body.
You should immediately:
- phone the Poisons Information Centre (by calling 13 11 26), or
- contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using OMNARIS?
Things you should do
Call your doctor straight away if:
- your nose starts to bleed or becomes infected, red, itchy or swollen
- you have any problems with your eyesight (i.e blurred vision). You may need to have your eyes checked by an eye specialist
- you have wheezing or other problems breathing
- you become pregnant while using OMNARIS
Other things you should do:
- keep all of your doctors appointments so that your progress can be checked. Your doctor may also change the dose if needed and check any effects on the nose and your general health.
- children and younger adults especially should see their doctor regularly so that they can be watched carefully for any effects on their growth.
- remind any doctor, dentist or pharmacist you visit that you are using OMNARIS
Things you should not do
- Do not stop using this medicine suddenly.
- Do not use OMNARIS to treat any other complaints unless your doctor tells you to.
- Do not give your medicine to anyone else, even if they have the same condition as you.
Do not change the amount you use without speaking to your doctor first.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how OMNARIS affects you.
Looking after your medicine
- Store the pack at room temperature, below 30°C. Do not freeze.
- Keep the spray in its foil pouch until you are ready to use it.
Follow the instructions above on how to look after your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on windowsills
Keep it where young children cannot reach it.
When to discard your medicine
- If your doctor or pharmacist tells you to stop taking this medicine.
- If the expiry date has passed.
- Four months after you first open the pouch, or when you have used all of the doses, whichever is first.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Digestive system
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Lungs
May include rash, itching or hives on the skin, skin redness, swollen face, lips, eyes, tongue or throat or other parts of the body. | Call your doctor straight away or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed may occur in some people.
Always speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
Reporting side effects
After you have spoken to your doctor or pharmacist about any side effects you have, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
7. Product details
This medicine is only available with a doctor's prescription.
What OMNARIS contains
| Active ingredient (main ingredient) |
|
| Other ingredients (inactive ingredients) |
|
| Potential allergens |
|
Do not use this medicine if you are allergic to any of these ingredients.
OMNARIS does not contain gluten, sucrose, lactose, tartrazine or any other azo dyes.
What OMNARIS looks like
OMNARIS is a white to off-white milky liquid in a brown glass bottle with a pump. The bottle is sealed in a foil pouch.
There is also an oxygen-absorber sachet in the pouch that you should throw away after you open it.
Each spray gives you 50 micrograms ciclesonide.
There are two sizes:
- 10 mL bottle containing 60 sprays
- 15 mL bottle containing 120 sprays.
Not all pack sizes may be available.
ARTG Number: Aust R 167910.
Who distributes OMNARIS
Chiesi Australia Pty Ltd
Level 7, Suite 1, 500 Bourke Street,
Melbourne, VIC 3000
Australia
E: medinfo.au@chiesi.com
W: www.chiesi.com.au
This leaflet was prepared in September 2025
Brand Information
| Brand name | Omnaris Nasal Spray |
| Active ingredient | Ciclesonide |
| Schedule | S4 |
MIMS Revision Date: 01 November 2025
1 Name of Medicine
Ciclesonide.
2 Qualitative and Quantitative Composition
Omnaris Nasal Spray is a metered-dose, manual-pump spray. Once primed (see Section 4.2 Dose and Method of Administration), each actuation of the pump delivers 50 microgram ciclesonide in a volume of 70 microL from the nasal actuator.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Nasal spray presented as a white to off-white suspension in brown glass bottle with a pump.
4 Clinical Particulars
4.1 Therapeutic Indications
Omnaris Nasal Spray is indicated for:
the treatment of seasonal allergic rhinitis in adults and children 6 years of age and older;
the treatment of perennial allergic rhinitis in adults and adolescents 12 years of age and older.
4.2 Dose and Method of Administration
Omnaris should only be administered by the intranasal route.
Dosage consideration. The recommended dose of Omnaris is 200 microgram per day administered as 2 actuations (50 microgram/actuation) in each nostril once daily.
The maximum total daily dosage should not exceed 2 actuations in each nostril (200 microgram/day).
Administration. Prior to initial use, Omnaris must be shaken gently and then the pump must be primed by actuating 8 times. If not used for 4 or more consecutive days, it should be shaken gently and reprimed with 1 actuation or until a fine mist appears.
During dosing, users are advised to tilt the head forward slightly and, while keeping the bottle upright, users are advised to press the pump quickly and firmly and inhale through the nose as they spray.
4.3 Contraindications
Patients with a hypersensitivity to ciclesonide or any of its ingredients.
4.4 Special Warnings and Precautions for Use
Immunosuppression and risk of infection. Rarely, immediate hypersensitivity reactions or contact dermatitis may occur after the administration of intranasal corticosteroids. Patients with a known hypersensitivity reaction to other corticosteroid preparations should use caution when using ciclesonide nasal spray since cross reactivity to other corticosteroids including ciclesonide may also occur. Patients who are using drugs that suppress the immune system are more susceptible to infections than healthy individuals. Chickenpox and measles, for example, can have a more serious or even fatal course in susceptible children or adults using corticosteroids. In children or adults who have not had these diseases or have not been properly immunised, particular care should be taken to avoid exposure. How the dose, route, and duration of corticosteroid administration affect the risk of developing a disseminated infection is not known. The contribution of the underlying disease and/or prior corticosteroid treatment to the risk is also not known. If exposed to chickenpox, prophylaxis with varicella zoster immune globulin (VZIG) may be indicated. If exposed to measles, prophylaxis with pooled intramuscular immunoglobulin (IG) may be indicated. If chickenpox develops, treatment with antiviral agents may be considered.
Local effects. Epistaxis and rare instances of nasal septum perforation have been reported following the intranasal application of corticosteroids.
Because of the inhibitory effect of corticosteroids on wound healing, patients who have experienced recent nasal septal ulcers, nasal surgery, or nasal trauma should not use a nasal corticosteroid until healing has occurred.
Respiratory. Rare instances of wheezing have been reported following the intranasal application of corticosteroids.
Infection. In clinical studies with corticosteroids administered intranasally, the development of localised infections of the nose and pharynx with Candida albicans have been reported only rarely. When such an infection develops, it may require treatment with appropriate local therapy and discontinuation of treatment with the intranasal corticosteroid. Therefore, patients using intranasal corticosteroids over several months or longer should be examined periodically for evidence of Candida infection or other signs of adverse effects on the nasal mucosa.
Corticosteroid should be used with caution, if at all, in patients with active or quiescent tuberculosis infections of the respiratory tract; or in patients with untreated local or systemic fungal or bacterial infections; systemic viral or parasitic infections; or ocular herpes simplex because of the potential for worsening of these infections.
Systemic effects. Although systemic effects have been minimal with recommended doses of Omnaris, any such effect is likely to be dose dependent. Therefore, larger than recommended doses of Omnaris should be avoided. If recommended doses of intranasal corticosteroids are exceeded or if individuals are particularly sensitive or predisposed by virtue of recent systemic steroid therapy, symptoms of hypercorticism may occur, including very rare cases of menstrual irregularities, acneiform lesions, and cushingoid features. If such changes occur, topical corticosteroids should be discontinued slowly, consistent with accepted procedures for discontinuing oral steroid therapy.
Visual disturbance. Visual disturbance may be reported with systemic and topical corticosteroid use. Rare instances of cataracts, glaucoma, and increased intraocular pressure have been reported following the intranasal application of corticosteroids. If a patient presents with symptoms such as blurred vision or other visual disturbances, the patient should be considered for referral to an ophthalmologist for evaluation of possible causes which may include cataract, glaucoma or rare diseases such as central serous chorioretinopathy (CSCR) which have been reported after use of systemic and topical corticosteroids.
The risk of glaucoma was evaluated by assessments of intraocular pressure in 3 studies including 943 patients. Of these, 390 adolescents or adults were treated for up to 52 weeks and 186 children ages 2 to 11 received treatment with ciclesonide 200 microgram daily for up to 12 weeks. In these trials, no significant differences in intraocular pressure changes were observed between ciclesonide- and placebo-treated patients. Additionally, no significant differences between the two patient groups were noted during the 52-week study of adults and adolescent patients in whom thorough ophthalmologic assessments were performed including evaluation of cataract formation using slit lamp examinations.
Growth. Intranasal corticosteroids may cause a reduction in growth velocity when administered to paediatric patients. Physicians should closely follow the growth of children and adolescents taking corticosteroids by any route (see Paediatric use).
Systemic steroid replacement by a topical steroid. The replacement of a systemic corticosteroid with a topical corticosteroid can be accompanied by signs of adrenal insufficiency. In addition, some patients may experience symptoms of corticosteroid withdrawal, e.g. joint and/or muscular pain, lassitude, and depression. Patients previously treated for prolonged periods with systemic corticosteroids and transferred to topical corticosteroids should be carefully monitored for acute adrenal insufficiency in response to stress. In those patients who have asthma or other clinical conditions requiring long-term systemic corticosteroid treatment, rapid decreases in systemic corticosteroid dosages may cause a severe exacerbation of their symptoms.
Use in the elderly. A total of 31 patients above 65 years of age (age range 65 to 75 years) have been treated with Omnaris 200 microgram/day for up to one year. The adverse reactions reported in this population were similar in type and incidence to those reported by younger patients, but greater sensitivity of some older individuals cannot be ruled out.
Paediatric use. The efficacy of Omnaris in children 6 years of age and older for the treatment of the symptoms of allergic rhinitis is supported by evidence from four adequate and well-controlled studies in adults and adolescents 12 years of age and older with seasonal or perennial allergic rhinitis and one study in patients 6 to 11 years of age with seasonal allergic rhinitis (see Section 5.1 Pharmacodynamic Properties, Clinical trials). The efficacy of Omnaris in children under 5 years of age has not been established. The safety of Omnaris in children 2 to 11 years of age was evaluated in four controlled clinical studies of 2 to 12 weeks duration (see Section 5.1 Pharmacodynamic Properties, Clinical trials). The growth of paediatric patients receiving intranasal corticosteroids, including Omnaris, should be monitored routinely (e.g. via stadiometry). The potential growth effects of prolonged treatment should be weighed against clinical benefits obtained and the availability of safe and effective noncorticosteroid treatment alternatives. To minimise the systemic effects of intranasal corticosteroids, each patient should be titrated to the lowest dose that effectively controls his/her symptoms (see Section 4.4 Special Warnings and Precautions for Use, Systemic effects; Section 4.8 Adverse Effects (Undesirable Effects)).
Effects on laboratory tests. Interactions with laboratory tests have not been established. Drug-laboratory interactions are unlikely for intranasal corticosteroids.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Based on in vitro studies in human liver microsomes and hepatocytes, des-ciclesonide is not an inhibitor of cytochrome (CYP) isoenzymes 1A2, 2A6, 2C9, 2C19, 2D6, 2E1 or 3A4 at therapeutic concentrations, and ciclesonide is not an inducer of CYP1A2, 2C9, 2C19 or 3A4. The inhibitory potential of ciclesonide on CYP450 isoenzymes has not been studied. In vitro studies demonstrated that the plasma protein binding of des-ciclesonide was not affected by warfarin or salicylic acid, indicating that protein binding-based drug interactions are unlikely.
In vitro data indicate that CYP 3A4 is the major enzyme involved in the metabolism of the active metabolite, des-ciclesonide, in man. A drug interaction study with orally inhaled ciclesonide and oral erythromycin, a substrate and weak inhibitor of CYP 3A4, had no relevant effect on the pharmacokinetics of either des-ciclesonide or erythromycin. In another drug interaction study at steady state with orally inhaled ciclesonide and oral ketoconazole, a potent CYP 3A4 inhibitor, the exposure (AUC) of des-ciclesonide increased by approximately 3.5-fold, while levels of ciclesonide remained unchanged.
The serum levels of ciclesonide and des-ciclesonide are negligible following administration of ciclesonide nasal spray. Therefore, the potential for clinically relevant drug-drug interactions is very low. However, co-administration with potent inhibitors of the CYP 3A4 (e.g. protease inhibitors for the treatment of HIV infections) should be considered with caution because there might be an increase in systemic drug levels of des-ciclesonide.
Concomitant treatment with ketoconazole or other potent CYP 3A4 inhibitors should be avoided unless the benefit outweighs this increased risk of systemic side effects of corticosteroids.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No evidence of impairment of fertility was observed in a reproductive study conducted in male and female rats both dosed orally with ciclesonide at up to 900 microgram/kg/day (approximately 41 times the maximum human daily intranasal dose in adults based on microgram/m2 body surface area in a 50 kg adult).
Use in pregnancy. (Category B3)
No adverse effects on embryofoetal development were observed in rats treated with ciclesonide at oral doses up to 900 microgram/kg/day (41 times the maximum human daily intranasal dose in adults on a body surface area basis) during the period of organogenesis. In a study in rats in which this dose was administered throughout gestation and lactation, pup birth weight and postnatal bodyweight gain were reduced; this occurred in the context of maternotoxicity. In rabbits, adverse effects on embryofoetal development occurred at subcutaneous doses ≥ 5 microgram/kg/day (0.6 times the maximum adult human dose based on body surface area), and comprised cleft palate, fore and/or hind leg flexure, enlarged fontanelle, incomplete ossification of skull bones, parchment-like skin and decreased foetal weight. Ciclesonide increased foetal loss in the rabbit with subcutaneous administration at doses ≥ 100 microgram/kg/day (11 times the maximum adult human dose based on body surface area).
There are no adequate and well controlled studies with Omnaris in pregnant women. As with other corticosteroids, ciclesonide should only be used during pregnancy when the potential benefit to the mother justifies the potential risk to the mother, foetus or infant. Infants born to mothers who received corticosteroids during pregnancy should be observed carefully for hypoadrenalism.
Use in lactation. It is unknown if ciclesonide is excreted in human milk. There was limited excretion of ciclesonide and/or its metabolites in to milk in lactating rats after intravenous or oral administration. Oral administration of ciclesonide to rats from early pregnancy until weaning was associated with reduced bodyweight in pups (see Use in pregnancy - Category B3). As with other corticosteroids, Omnaris should only be used in nursing women when the potential benefit to the mother justifies the potential risk to the mother and/or infant.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Clinical trials experience. In controlled clinical studies with patients receiving ciclesonide aqueous nasal spray, the overall incidence of adverse events for patients treated with ciclesonide aqueous nasal spray was comparable to that in patients treated with placebo. Adverse reactions reported at an incidence of 1% or greater and more commonly reported in ciclesonide aqueous nasal spray versus placebo were as follows:
Nervous system disorders. Headache.
Respiratory, thoracic and mediastinal disorders. Nasopharyngitis, epistaxis, sinusitis.
Adult and adolescent patients aged 12 years and older. In controlled clinical studies, a total of 1524 patients ages 12 years and older received treatment with ciclesonide administered intranasally. In studies of 2 to 6 weeks duration in patients 12 years and older, 546 patients were treated with Omnaris 200 microgram daily, and in a study of up to one year in duration, 441 patients were treated with Omnaris 200 microgram daily. The overall incidence of adverse events for patients treated with Omnaris was comparable to that in patients treated with placebo. Adverse events did not differ appreciably based on age, gender, or race. Approximately 2% of patients treated with Omnaris in clinical trials discontinued because of adverse events; this rate was similar for patients treated with placebo. Table 1 displays adverse events, irrespective of drug relationship, that occurred with an incidence of 2% or greater and more frequently with Omnaris than with placebo in clinical trials of 2 to 6 weeks in duration.

Less common adverse reactions reported in controlled clinical trials 2 to 52 weeks in duration in patients 12 years of age and older with seasonal or perennial allergic rhinitis were:
Gastrointestinal disorders. Dry mouth (0.2%), dyspepsia (0.2%).
Infections and infestations. Candidiasis (0.2%), rhinitis (0.2%).
Investigations. Laboratory test abnormal NOS (0.2%), white blood cell count increased (0.3%).
Nervous system disorders. Dysgeusia (0.2%).
Respiratory, thoracic and mediastinal disorders. Nasal dryness (0.4%), pharyngolaryngeal pain (0.4%), rhinorrhoea* (0.3%), nasal septum disorder (0.2%), throat irritation* (0.2%).
* Occurred at rates ≤ placebo.
Paediatric patients aged 6 to 11 years. Two controlled clinical studies 2 and 12 weeks in duration were conducted in a total of 1282 patients with allergic rhinitis ages 6 to 11 years, of which 913 were treated with Omnaris 200 microgram, 100 microgram or 25 microgram daily. The overall incidence of adverse events for patients treated with Omnaris was comparable to that in patients treated with placebo. Adverse events did not differ appreciably based on age, gender, or race. In clinical trials, 1.6% and 2.7% of patients treated with Omnaris 200 microgram or 100 microgram, respectively, discontinued because of adverse events; these rates were lower than the rate in patients treated with placebo (2.8%). Table 2 displays adverse events, irrespective of drug relationship, that occurred with an incidence of 3% or greater and more frequently with Omnaris 200 microgram than with placebo.
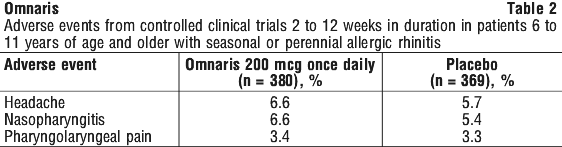
These effects described above were observed with ciclesonide administered as a metered dose inhaler utilising a different formulation and at different dosages to Omnaris.
Post-marketing experience. Hypersensitivity reactions, including angioedema, loss of consciousness, nasal oedema, dyspnoea, and nasal septum perforation have been reported in association with post-market use of Omnaris. Because these reactions are reported voluntarily from a population of uncertain size and are not always confirmed with a health care professional, it is not possible to reliably estimate their frequency or to establish a causal relationship to drug exposure.
Eye disorders, such as blurred vision with frequency unknown, have been reported with systemic and topical corticosteroid use (see Section 4.4 Special Warnings and Precautions for Use, Systemic effects).
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
There are no data available on the effects of acute or chronic overdosage with Omnaris. Because of low systemic bioavailability, acute overdosage is unlikely to require any therapy other than observation. A single oral dose of up to 10 mg of ciclesonide in healthy volunteers was well tolerated and serum cortisol levels were virtually unchanged in comparison with placebo treatment. Chronic overdosage with any corticosteroid may result in signs or symptoms of hypercorticism.
For information on the management of overdose, contact the Poisons Information Centre on 131126 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Ciclesonide is a pro-drug that is enzymatically hydrolysed to a pharmacologically active metabolite, C21-desisobutyryl-ciclesonide (des-ciclesonide) following intranasal application. Des-ciclesonide has anti-inflammatory activity with affinity for the glucocorticoid receptor that is 120 times higher than the parent compound.
The precise mechanism through which ciclesonide affects allergic rhinitis symptoms is not known. Corticosteroids have been shown to have a wide range of effects on multiple cell types (e.g. mast cells, eosinophils, neutrophils, macrophages, and lymphocytes) and mediators (e.g. histamine, eicosanoids, leukotrienes, and cytokines) involved in allergic inflammation.
The anti-inflammatory properties of ciclesonide and des-ciclesonide were shown in several in vitro and in vivo investigations, including experiments using a guinea pig model of allergic rhinitis and several investigations in primary human nasal epithelial cells, bronchial epithelial and smooth muscle cells.
Pharmacodynamics. In a study of 40 healthy adult volunteers and 8 asymptomatic seasonal allergic rhinitis patients, no significant differences between the active and placebo groups were observed in 24-hour plasma or urine cortisol after administration of 50-800 microgram daily of ciclesonide for 14 days. In a 1-year safety study including 174 patients treated with ciclesonide 200 microgram once daily and 92 patients treated with placebo who had cortisol assessments, no significant differences in morning plasma and 24 hour urine cortisol levels were observed with ciclesonide versus placebo treatment.
In two studies conducted in children with perennial allergic rhinitis, daily doses of 200 microgram, 100 microgram, and 25 microgram of ciclesonide were compared to placebo nasal spray. The ciclesonide-treated groups had a numerically greater decline in 24 hour urinary free cortisol compared to the placebo group. In the 12 week study in children 6-11 years of age, the difference (and 95% confidence intervals) from placebo in the mean change from baseline to 12 weeks was -0.81 (-4.0, 2.4) microgram/day for the 200 microgram dose group. The mean morning plasma cortisol value did not show any consistent treatment effect. In the 6 week study in children 2-5 years of age, the difference (and 95% confidence intervals) from placebo in the mean change from baseline to 6 weeks was -2.04 (-4.4, 0.3) microgram/day for the 200 microgram dose groups. The plasma cortisol decreased numerically after treatment with ciclesonide with the difference (and 95% confidence intervals) from placebo in the mean change in plasma cortisol from baseline to 6 weeks being -1.04 (-2.7, 0.7) microgram/dL for the 200 microgram dose group. In the studies, serum was assayed for ciclesonide and des-ciclesonide (see Section 5.2 Pharmacokinetic Properties, Absorption).
Clinical trials. Seasonal allergic rhinitis and perennial allergic rhinitis. Adult and adolescent patients aged 12 years and older. The efficacy and safety of Omnaris were evaluated in 4 randomised, double-blind, parallel group, multi-centre, placebo controlled clinical trials of 2 weeks to 1 year in duration conducted in adults and adolescents with allergic rhinitis. Three of these trials were 2 to 6 weeks in duration and primarily designed to assess efficacy. One of these trials was 1 year in duration and primarily designed to assess safety. The three trials of 2 to 6 weeks duration included a total of 1524 patients (495 males and 1029 females) of whom 79 were adolescents, ages 12 to 17 years. Of the 1524 patients, 546 patients received Omnaris 200 microgram once daily. Patients enrolled in the studies were 12 to 86 years of age with a history of seasonal or perennial allergic rhinitis, a positive skin test to at least one relevant allergen, and active symptoms of allergic rhinitis at study entry. Assessment of efficacy in these trials was based on patient recording of four nasal symptoms (runny nose, nasal itching, sneezing, and nasal congestion) on a 0-3 categorical severity scale (0 = absent, 1 = mild, 2 = moderate, and 3 = severe) as reflective or instantaneous scores. Reflective scoring required the patients to record symptom severity over the previous 12 hours; the instantaneous scoring required patients to record symptom severity at the time of recording. The results of these trials showed that patients treated with Omnaris 200 microgram once daily exhibited statistically significantly greater decreases in total nasal symptom scores than placebo treated patients. Secondary measures of efficacy were generally supportive.
In the 2-week dose-ranging trial that evaluated efficacy of Omnaris in patients with seasonal allergic rhinitis, the primary efficacy endpoint was the difference from placebo in the change from baseline of the sum of morning and evening reflective total nasal symptom score averaged over the 2-week treatment period. In this trial Omnaris 200 microgram once daily was statistically significantly different from placebo.
In the 4-week single dose level trial conducted in patients with seasonal allergic rhinitis and the 6 week single dose level trial conducted in patients with perennial allergic rhinitis, the primary efficacy endpoints were the difference from placebo in the change from baseline of the average of morning and evening reflective total nasal symptom score averaged over the first 2 weeks of treatment and over the 6 weeks of treatment, respectively. In these trials, Omnaris 200 microgram once daily was statistically significantly different from placebo. Statistically significant differences in the morning pre-dose instantaneous total nasal symptom score indicate that the effect was maintained over the full 24 hour dosing interval.
Results of the primary efficacy endpoint in these trials are shown in Table 3.
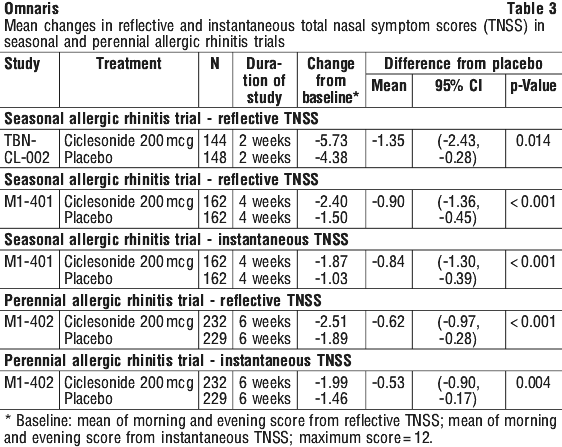
Onset of action was evaluated in two environmental exposure unit studies with a single dose of Omnaris 200 microgram. Results from these two studies did not demonstrate a replicate onset of action within the assessment period. Onset of action was also evaluated in the 4-week seasonal allergic rhinitis and in the 6-week perennial allergic rhinitis trial by frequent recording of instantaneous symptom score after the first dose. In these trials, onset of effect was seen within 24 to 48 hours with further symptomatic improvement observed over 1 to 2 weeks in seasonal allergic rhinitis and 5 weeks in perennial allergic rhinitis.
Paediatric patients aged 6 to 11 years. The efficacy of Omnaris was evaluated in 618 children aged 6 to 11 years old with seasonal allergic rhinitis in a randomised, double-blind, parallel group, multi-centre, placebo-controlled clinical trial. The 2-week trial conducted in patients compared the efficacy of ciclesonide 200 microgram and 100 microgram once daily nasal spray. The primary efficacy endpoint was the difference from placebo in the change from baseline of the average of morning and evening reflective total nasal symptom score averaged over 2 weeks of treatment. In the study, the ciclesonide 200 microgram once daily dose was statistically significantly different from placebo, but the 100 microgram once daily dose was not statistically significantly different from placebo. The efficacy results for the seasonal allergic rhinitis trial are shown in Table 4.

5.2 Pharmacokinetic Properties
Absorption. Ciclesonide and des-ciclesonide have negligible oral bioavailability (both less than 1%) due to low gastrointestinal absorption and high first pass metabolism. The intranasal administration of ciclesonide at recommended doses results in negligible serum concentrations of ciclesonide. However, the known active metabolite (des-ciclesonide) is detected in the serum of some patients after nasal inhalation of ciclesonide. The bioanalytical assay used has a lower limit of quantification of 25 picogram/mL and 10 picogram/mL, for ciclesonide and des-ciclesonide, respectively.
In healthy adults treated for two weeks with 50 to 800 microgram of ciclesonide nasal spray daily, the peak serum concentrations of des-ciclesonide in all subjects were found to be below 30 picogram/mL. Of those treated with 800 microgram and 400 microgram daily, 100% and 67% had detectable levels of des-ciclesonide, respectively. With daily doses of 200 microgram or less, detectable serum levels of des-ciclesonide were not observed. The low systemic exposure following ciclesonide nasal spray administration was confirmed in a crossover study in twenty-nine healthy adults. The median Cmax was less than 10 picogram/mL and 602 picogram/mL following a single dose of ciclesonide nasal spray (300 microgram) and orally inhaled ciclesonide (320 microgram, Alvesco), respectively.
In paediatric subjects treated with 25 to 200 microgram of ciclesonide nasal spray daily, serum concentrations of des-ciclesonide were below 45 picogram/mL, with the exception of one value of 64.5 picogram/mL. In a 12 week study in children 6 to 11 years of age with perennial allergic rhinitis, des-ciclesonide was detected in 50% of the subjects treated with 200 microgram and in 5% of those treated with 100 microgram ciclesonide nasal spray daily.
Distribution. Following intravenous administration of 800 microgram of ciclesonide, the volumes of distribution of ciclesonide and des-ciclesonide were approximately 2.9 L/kg and 12.1 L/kg, respectively. The percentage of ciclesonide and des-ciclesonide bound to human plasma proteins averaged ≥ 99% each, with approximately 1% of unbound drug detected in the systemic circulation. Des-ciclesonide is not significantly bound to human transcortin.
Metabolism. Intranasal ciclesonide is hydrolysed to a biologically active metabolite des-ciclesonide by esterases in the nasal mucosa. Des-ciclesonide undergoes further metabolism in the liver to additional metabolites mainly by the CYP 450 3A4 isozyme and to a lesser extent by CYP 2D6. The full range of potentially active metabolites of ciclesonide has not been characterised. After intravenous administration of 14C-ciclesonide, 19.3% of the resulting radioactivity in the plasma is accounted for by ciclesonide or des-ciclesonide; the remainder may be a result of other, as yet, unidentified multiple metabolites.
Excretion. Following intravenous administration of 800 microgram of ciclesonide, the clearance values of ciclesonide and des-ciclesonide were high (approximately 152 L/h and 228 L/h, respectively). 14C-labelled ciclesonide was predominantly excreted via the faeces after intravenous administration (66%) indicating that excretion through bile is the major route of elimination. Approximately 20% or less of drug related radioactivity was excreted in the urine.
Special populations. The pharmacokinetics of intranasally administered ciclesonide have not been assessed in patient subpopulations because the resulting blood levels of ciclesonide and des-ciclesonide are insufficient for pharmacokinetic calculations. However, population pharmacokinetic analysis showed that characteristics of des-ciclesonide after oral inhalation of ciclesonide were not appreciably influenced by a variety of subject characteristics such as bodyweight, age, race, and gender.
Studies in renal impaired patients were not conducted since renal excretion of des-ciclesonide is a minor route of elimination (≤ 20%).
5.3 Preclinical Safety Data
Genotoxicity. Ciclesonide did not induce gene mutations in bacterial (Ames) or mammalian (HPRT) tests in vitro, nor induce chromosomal aberrations in human lymphocytes or micronuclei in Chinese hamster V79 cells in vitro. Racemic ciclesonide was also negative in the Ames test. In contrast, ciclesonide induced micronuclei in mouse bone marrow in vivo (at ≥ 75 mg/kg in females and > 1000 mg/kg in males). Positive in vivo clastogenicity results have also been observed with high doses of other corticosteroids and may reflect effects on erythrocyte differentiation. The clinical relevance of these clastogenicity findings is unknown but likely limited.
Carcinogenicity. The carcinogenic potential of ciclesonide was investigated in a 2 year oral study in mice and in a 2 year inhalation study in rats. Gastric adenomas (benign tumour) were significantly increased in female mice at 900 microgram/kg/day (approximately 20 and 11 times the maximum human daily intranasal dose in adults and children, respectively, based on microgram/m2 body surface area).
This effect may arise from a local action in the stomach, with local exposure (based on microgram/kg doses being ≥ 90 times higher in the animals compared with humans receiving the maximum recommended dose of Omnaris. No tumourigenicity was observed in rats administered ciclesonide by inhalation at up to 89 microgram/kg/day (males) or 104 microgram/kg/day (females) (approximately 4 and 2 times the maximum human dose in adults and children, respectively, based on body surface area).
6 Pharmaceutical Particulars
6.1 List of Excipients
Omnaris Nasal Spray contains cellulose microcrystalline, carmellose sodium, hypromellose, potassium sorbate, disodium edetate, hydrochloric acid to adjust the pH to 4.5, purified water and nitrogen.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C. Do not freeze.
Store in the foil pouch and only open pouch immediately before first use. Discard 4 months after first opening of pouch.
6.5 Nature and Contents of Container
Omnaris Nasal Spray is a metered-dose, manual-pump spray formulation containing a hypotonic aqueous suspension of ciclesonide. Each pack of Omnaris contains one spray pump bottle containing 60 actuations (10 mL bottle) or 120 actuations (15 mL bottle) actuations of 50 microgram/actuation of ciclesonide after initial priming (see Section 4.2 Dose and Method of Administration).
* Not all pack sizes may be available in Australia.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Ciclesonide is a white to yellow-white powder, practically insoluble in water and freely soluble in ethanol and acetone.
Chemical name (CAS): [11β,16α(R)] -16,17-[(Cyclohexylmethylene)bis(oxy)]-11-hydroxy-21-(2-methyl-1-oxopropoxy) pregna-1,4-diene-3,20-dione.
Chemical structure.
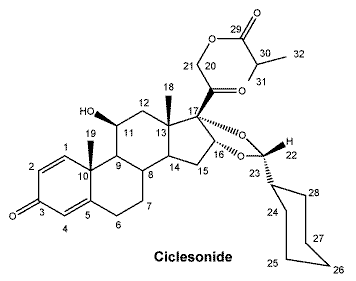
Molecular formula: C32H44O7.
Molecular weight: 540.7.
7 Medicine Schedule (Poisons Standard)
Prescription only medicine (Schedule 4).
Date of First Approval
16 December 2011
Date of Revision
01 September 2025
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.