Opsumit
Brand Information
| Brand name | Opsumit |
| Active ingredient | Macitentan |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Opsumit.
Summary CMI
OPSUMIT®
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
▼ This medicine is new or being used differently. Please report side effects. See the full CMI for further details.
WARNING: Important safety information is provided in a boxed warning in the full CMI. Read before using this medicine.
1. Why am I using OPSUMIT?
OPSUMIT contains the active ingredient macitentan. OPSUMIT is used to treat pulmonary arterial hypertension (PAH). It can be used on its own or with other drugs to treat PAH.
For more information, see Section 1. Why am I using OPSUMIT? in the full CMI.
2. What should I know before I use OPSUMIT?
Do not use if you have ever had an allergic reaction to macitentan, soya or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding. Women of childbearing potential must use reliable birth control (contraception).
For more information, see Section 2. What should I know before I use OPSUMIT? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with OPSUMIT and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use OPSUMIT?
For adults, the recommended dose of OPSUMIT is one 10 mg film-coated tablet, once a day with or without food.
For children over 2 years of age, the dose of OPSUMIT dispersible tablets is based on the child's body weight. Children weighing 40 kg or more may use a 10 mg film-coated tablet as a replacement.
More instructions can be found in Section 4. How do I use OPSUMIT? in the full CMI.
5. What should I know while using OPSUMIT?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using OPSUMIT? in the full CMI.
6. Are there any side effects?
The common but less serious side effects you may experience include low number of red blood cells (anaemia), swelling caused by fluid buildup in the body, infection of nose or throat. The serious side effects that require medical attention include severe allergic reaction.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
▼ This medicine is subject to additional monitoring due to approval of an extension of indications. This will allow quick identification of new safety information. You can help by reporting any side effects you may get. You can report side effects to your doctor, or directly at www.tga.gov.au/reporting-problems.
WARNING: OPSUMIT may cause birth defects (harm to unborn babies). Do not take OPSUMIT if you are pregnant or plan to become pregnant.
OPSUMIT®
Active ingredient(s): macitentan (ma-SEE-ten-tan)
Consumer Medicine Information (CMI)
This leaflet provides important information about using OPSUMIT. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using OPSUMIT.
Where to find information in this leaflet:
1. Why am I using OPSUMIT?
2. What should I know before I use OPSUMIT?
3. What if I am taking other medicines?
4. How do I use OPSUMIT?
5. What should I know while using OPSUMIT?
6. Are there any side effects?
7. Product details
1. Why am I using OPSUMIT?
OPSUMIT contains the active ingredient macitentan. OPSUMIT belongs to a group of medicines called endothelin receptor antagonists (ERA).
OPSUMIT is used to treat pulmonary arterial hypertension (PAH). It can be used on its own or with other drugs to treat PAH.
PAH is high blood pressure in the blood vessels (the pulmonary arteries) that carry blood from the heart to the lungs. In people with PAH, these arteries get narrower, so the heart has to work harder to pump blood through them. This causes people to feel tired, dizzy, and short of breath.
OPSUMIT widens the pulmonary arteries, making it easier for the heart to pump blood through them. This lowers the blood pressure, relieves the symptoms and improves the course of the disease.
2. What should I know before I use OPSUMIT?
Warnings
Do not use OPSUMIT if:
- you are allergic to macitentan, soya or any of the other ingredients listed at the end of this leaflet.
Always check the ingredients to make sure you can use this medicine.
Symptoms of an allergic reaction may be mild or severe. They usually include some or all of the following:
wheezing, swelling of the lips/mouth, difficulty in breathing, hay fever, lumpy rash (hives) or fainting. - you are pregnant, if you are planning to become pregnant, or if you could become pregnant because you are not using reliable birth control (contraception).
- you have serious liver disease or very high levels of liver enzymes in your blood.
Check with your doctor if you:
- have an intolerance to lactose or any other sugars. OPSUMIT film-coated tablets contain small amounts of a sugar called lactose.
- have anaemia (low number of red blood cells)
- have liver or kidney problems
- have pulmonary veno-occlusive disease (obstruction of the lung veins)
- have low blood pressure
- are 75 years or older
Your doctor will order blood test before you start treatment and also during treatment with OPSUMIT to check if you have anaemia (low number of red blood cells) or if your liver is working properly.
Signs that your liver may not be working properly include:
- nausea (urge to vomit)
- vomiting
- fever (high temperature)
- pain in your stomach
- jaundice (yellowing of your skin or the whites of your eyes)
- dark-coloured urine
- itching of your skin
- lethargy or fatigue (unusual tiredness or exhaustion)
- flu-like syndrome (joint and muscle pain with fever)
If you notice any of these signs, tell your doctor immediately.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Do not take OPSUMIT if you or your partner are pregnant or planning to become pregnant as it may harm unborn babies conceived before, during or soon after treatment.
Do not become pregnant for at least 3 months after stopping OPSUMIT.
If you are a woman who could become pregnant, your doctor will ask you to take a pregnancy test before you start taking OPSUMIT and regularly (once a month) while you are taking OPSUMIT.
If it is possible you could become pregnant, use at least two reliable forms of birth control (contraception) while you are taking OPSUMIT. You must continue to use contraception for at least 3 months after stopping OPSUMIT. Talk to your doctor about this.
If you become pregnant or think that you may be pregnant while you are taking OPSUMIT, or shortly after stopping OPSUMIT (up to three month), see your doctor immediately.
It is not known if OPSUMIT is transferred to breast milk. Do not breastfeed while you are taking OPSUMIT. Talk to your doctor the best way to feed your baby if you take OPSUMIT.
Male fertility
If you are a male you should avoid exposing your partner to your semen by use of appropriate contraception.
OPSUMIT may lower your sperm count. Talk to your doctor if you have any questions or concerns about this.
Children and adolescents
OPSUMIT should not be used in children under 2 years of age. The safety of OPSUMIT and how effective it is have not been established in this age group.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with OPSUMIT and affect how it works. You must tell you doctor if you are taking:
- rifampicin, clarithromycin, ciprofloxacin, erythromycin (antibiotics used to treat infections)
- ritonavir (a medicine for HIV infections)
- ketoconazole (except shampoo), itraconazole, voriconazole, fluconazole, miconazole (medicines used against fungal infections)
- phenytoin (a medicine used to treat seizures)
- carbamazepine (a medicine used to treat depression and epilepsy)
- amiodarone (medicines to treat irregular heartbeat)
- ciclosporin (medicine used to prevent organ rejection after transplant)
- diltiazem, verapamil (medicine used to treat high blood pressure or specific heart problems)
- St. John's Wort (a herbal medicine used to support healthy mood balance)
- piperine (a herbal dietary supplement)
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect OPSUMIT.
4. How do I use OPSUMIT?
How much to take
Adults
- The recommended dose of OPSUMIT in adults is one 10 mg film-coated tablet, once a day. Follow the instructions provided and take OPSUMIT until your doctor tells you to stop.
- Swallow the whole film-coated tablet, with a glass of water. Do not chew or break the tablet.
Children over 2 years to less than 18 years of age
Your doctor will tell you the number of OPSUMIT dispersible tablets to take or give for children over 2 years of age. This is based on the body weight of the child.
For children less than 18 years weighing 40 kg or more, 10 mg film-coated tablet may be used as a direct replacement for dispersible tablets.
OPSUMIT is given once a day. Follow the instructions provided and take OPSUMIT until your doctor tells you to stop.
Take or give OPSUMIT dispersible tablets as an oral suspension only
OPSUMIT dispersible tablets must be dispersed in liquids to form an oral suspension before they can be given. The oral suspension can be prepared in either a stainless-steel spoon or in a small glass. Take care that the entire dose is swallowed. Hands must be thoroughly washed and dried before and after preparation of the medicine.
How to prepare and take or give the oral suspension using a stainless-steel spoon
- Prepare the oral suspension by adding the prescribed number of dispersible tablets to room temperature drinking water in a stainless-steel spoon.
- Gently stir the liquid for 1 to 3 minutes using the tip of a knife. Either give the resulting white cloudy liquid to the child right away or mix it further with a small portion of apple sauce or yoghurt to aid with administration.
- Add a little more water or apple sauce or yogurt to the spoon and have the child swallow it to make sure all the medicine has been taken.
- If not taken right away, discard the medicine and prepare a new dose.
- Alternatively, instead of drinking water, the oral suspension can be prepared in orange juice or apple juice or skimmed milk.
How to prepare and take or give the oral suspension using a small glass
- Prepare the oral suspension by adding the prescribed number of dispersible tablets to a small amount (from 10 mL to maximum 100 mL) of room temperature drinking water in a small glass.
- Gently stir with a spoon for 1 to 2 minutes. Have the child drink the resulting white cloudy liquid right away.
- Add a little more water (minimum 5 mL) to the small glass and stir with the same spoon and have the child drink the entire contents of the glass to make sure all the medicine has been taken.
- If not taken right away, discard the medicine and prepare a new dose.
Special information for caregivers
Caregivers are advised to avoid contact with suspensions of OPSUMIT dispersible tablets. Wash hands thoroughly before and after preparation of the suspension.
When to take OPSUMIT
- OPSUMIT can be taken with or without food.
- It is best to take the tablet at the same time each day.
If you forget to take or give OPSUMIT
OPSUMIT should be taken or given regularly at the same time each day.
If you forget to take or give a dose of OPSUMIT at the usual time and it is almost time for the next dose, skip the missed dose and take or give the next dose when you are meant to.
Do not take or give a double dose to make up for the missed dose.
If you take or give too much OPSUMIT
If you think that you have taken or given too much OPSUMIT, urgent medical attention may be needed.
You should immediately:
- phone the Poisons Information Centre (in Australia telephone 13 11 26 and in New Zealand telephone 0800 POISON or 0800 764 766), or
- contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using OPSUMIT?
Things you should do
If it is possible you could become pregnant, use at least two reliable forms of birth control (contraception) while you are taking OPSUMIT. You must continue to use contraception for at least 3 months after stopping OPSUMIT. Talk to your doctor about this.
Call your doctor straight away if you:
- if you become pregnant or are trying to become pregnant.
- have signs of pulmonary oedema when using OPSUMIT, such as a sudden, important increase in breathlessness and low oxygen. In patients with pulmonary veno-occlusive disease (obstruction of the lung veins), the use of medicines for treatment of PAH, including OPSUMIT, may cause a build-up of fluid in your lungs (pulmonary oedema).
Remind any doctor, dentist or pharmacist you visit that you are using OPSUMIT.
Things you should not do
- Do not stop taking this medicine without talking to your doctor. OPSUMIT is a treatment that you will need to keep on taking to control your PAH.
- Do not breastfeed while you are taking OPSUMIT.
- Do not give this medicine to anyone else, even if their symptoms seem similar to yours.
- Do not use OPSUMIT to treat any other complaints unless your doctor says to.
Driving or using machines
OPSUMIT may cause headaches in some people.
Be careful before you drive or use any machines or tools until you know how OPSUMIT affects you.
Looking after your medicine
- Store below 30°C. Keep tablets in the original pack.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Keep it where young children cannot reach it.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Allergic reaction:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Allergic reaction:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems or in New Zealand at pophealth.my.site.com/carmreportnz/s/. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What OPSUMIT contains
Film-coated tablets
| Active ingredient (main ingredient) | macitentan |
| Other ingredients (inactive ingredients) | lactose monohydrate microcrystalline cellulose sodium starch glycollate type A povidone magnesium stearate polysorbate 80 The film-coating contains: OPADRY AMB complete film coating system OY-B-28920 White, ARTG PI No. 4271 |
| Potential allergens | lactose soya bean products |
Dispersible tablets
| Active ingredient (main ingredient) | macitentan |
| Other ingredients (inactive ingredients) | croscarmellose sodium isomalt magnesium stearate mannitol |
Do not take this medicine if you are allergic to any of these ingredients.
What OPSUMIT looks like
Film-coated tablets
OPSUMIT 10 mg tablets are white to off white, round, biconvex, film-coated tablets with "10" on both sides, supplied in blister packs of 9 or 30 tablets. (AUST R 205624)
Dispersible tablets
2.5 mg - white to almost white, round, immediate-release dispersible tablet, with a "2.5" on one side and with "Mn" on the other side. It is supplied in blister pack of 30 tablets (AUST R 457898)
Who distributes OPSUMIT?
JANSSEN-CILAG Pty Ltd
17 Khartoum Road
Macquarie Park NSW 2113 Australia
Telephone: 1800 226 334
NZ Office: Auckland New Zealand
Telephone: 0800 800 806
This CMI was prepared on 30 January 2026.
Brand Information
| Brand name | Opsumit |
| Active ingredient | Macitentan |
| Schedule | S4 |
▼ This medicinal product is subject to additional monitoring in Australia. This will allow quick identification of new safety information. Healthcare professionals are asked to report any suspected adverse events at www.tga.gov.au/reporting-problems.
MIMS Revision Date: 01 April 2026
1 Name of Medicine
Macitentan.
2 Qualitative and Quantitative Composition
Opsumit is available as a film-coated tablet containing 10 mg of macitentan or as a dispersible tablet containing 2.5 mg of macitentan.
Excipients with known effects. 10 mg film-coated tablet. Lactose and soya bean products.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Film-coated tablet. 10 mg - white to off-white, round, biconvex film-coated tablet, debossed with "10" on both sides.
Dispersible tablet. 2.5 mg - white to almost white, round, immediate-release dispersible tablet, debossed with a "2.5" on one side and with "Mn" on the other side.
4 Clinical Particulars
4.1 Therapeutic Indications
Film-coated tablets. Opsumit, as monotherapy or in combination with approved PAH treatments (phosphodiesterase-5 inhibitors or inhaled prostanoids), is indicated for the treatment of:
idiopathic pulmonary arterial hypertension;
heritable pulmonary arterial hypertension;
pulmonary arterial hypertension associated with connective tissue disease;
pulmonary arterial hypertension associated with congenital heart disease with repaired shunts in patients with WHO Functional class II, III or IV symptoms.
Dispersible tablets. Paediatric (2 years to less than 18 years of age). Opsumit, as monotherapy or in combination, is indicated for the treatment of:
idiopathic pulmonary arterial hypertension;
heritable pulmonary arterial hypertension;
pulmonary arterial hypertension associated with connective tissue disease;
pulmonary arterial hypertension associated with congenital heart disease with repaired shunts;
pulmonary arterial hypertension with co-incidental congenital heart disease in paediatric patients with WHO Functional class I, II, III or IV symptoms.
4.2 Dose and Method of Administration
Treatment with Opsumit should only be initiated and monitored by a physician experienced in the treatment of PAH.
Dosage. Adults. The recommended dose of Opsumit in adults is one 10 mg film-coated tablet taken orally once daily. Doses higher than 10 mg daily have not been studied and are not recommended.
Opsumit is also available as 2.5 mg dispersible tablets. Opsumit administered in the form of 4 x 2.5 mg dispersible tablets is bioequivalent to 1 x 10 mg film-coated tablet. Therefore, 4 x 2.5 mg dispersible tablets may be used as direct replacements for patients who are unable to swallow a 10 mg film-coated tablet (see Section 5.2 Pharmacokinetic Properties, Paediatrics (aged ≥ 1 month to less than 18 years)).
Paediatrics (2 years to less than 18 years of age). The recommended dose of Opsumit in paediatric patients aged 2 years to less than 18 years is based on body weight (see Table 1). Opsumit should be taken once daily.
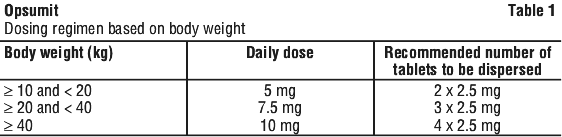
Special populations. Dosage adjustment in elderly patients. No dose adjustment is required in patients over the age of 65 years.
Dosage adjustment in patients with hepatic impairment. Based on pharmacokinetic data, no dose adjustment is required in patients with mild or moderate hepatic impairment. There is no clinical experience with the use of Opsumit in PAH patients with moderate or severe hepatic impairment.
Opsumit is contraindicated in patients with severe hepatic impairment, or clinically significant elevated hepatic aminotransferases (greater than 3 times the Upper Limit of Normal (> 3 x ULN)); see Section 4.3 Contraindications.
Dosage adjustment in patients with renal impairment. Based on pharmacokinetic data, no dose adjustment is required in patients with renal impairment. There is no clinical experience with the use of Opsumit in PAH patients with severe renal impairment. The use of Opsumit is not recommended in patients undergoing dialysis.
Paediatric population (below 2 years). The safety and efficacy of Opsumit in children below 2 years of age have not been established. Currently available data are described in Section 4.8 Adverse Effects (Undesirable Effects); Section 5.1 Pharmacodynamic Properties; Section 5.2 Pharmacokinetic Properties but no recommendation on a posology can be made.
Administration. Film-coated tablets. Opsumit should be taken orally once a day with or without food. The film-coated tablets are not breakable and are to be swallowed whole, with water.
Dispersible tablets. Opsumit should be taken orally once a day at about the same time, with or without food. Opsumit dispersible tablet(s) must be dispersed in room temperature liquids and are to be taken as an oral suspension only. The oral suspension must be prepared and administered using either a stainless-steel spoon or a small glass. Care should be taken to ensure the entire dose of medicine has been taken. If not administered right away the medicine should be discarded and a new dose of medicine should be prepared. Hands must be thoroughly washed and dried before and after preparation of the medicine.
Administration by a stainless-steel spoon. The prescribed daily dose of dispersible tablet(s) should be added to room temperature drinking water in a stainless-steel spoon to form a white cloudy liquid. The liquid can be gently stirred for 1 to 3 minutes using a knife tip to speed up dissolution. Either administer the medicine to the patient right away or mix it further with a small portion of apple sauce or yoghurt to aid with administration. A little more water, apple sauce or yoghurt should be added to the spoon and administered to the patient to make sure the entire dose of medicine has been taken.
Alternatively, instead of drinking water, the oral suspension can be prepared in orange juice, apple juice or skimmed milk.
Administration by a glass. The prescribed daily dose of dispersible tablet(s) should be placed in a small glass containing a small volume (from 10 mL up to maximum 100 mL) of room temperature drinking water to form a white cloudy liquid. The liquid can be gently stirred with a spoon for 1 to 2 minutes. Administer the medicine to the patient right away. A little more water (minimum 5 mL) should be added to the glass and stirred with the same spoon to re-suspend any remaining medicine. The entire contents of the glass should be administered to the patient to make sure all the medicine has been taken.
4.3 Contraindications
Opsumit is contraindicated in:
Women who are or may become pregnant (see Boxed Warnings; see Section 4.6 Fertility, Pregnancy and Lactation).
Women of child-bearing potential who are not using reliable contraception (see Section 4.6 Fertility, Pregnancy and Lactation). Women must not become pregnant for at least 3 months after stopping treatment with Opsumit.
Hypersensitivity to the active substance or to any of the excipients listed, see Section 6.1 List of Excipients.
Patients with severe hepatic impairment (with or without cirrhosis) (see Section 4.4 Special Warnings and Precautions for Use, Liver function).
Patients with baseline values of hepatic aminotransferases (aspartate aminotransferase [AST] and/or alanine aminotransferase [ALT]) greater than 3 times the Upper Limit of Normal (ULN) (see Section 4.4 Special Warnings and Precautions for Use, Liver function).
4.4 Special Warnings and Precautions for Use
Macitentan has only been studied in a limited number of patients with WHO functional class IV symptoms.
Macitentan has only been studied in a limited number of patients with PAH due to HIV, drugs or toxins.
The efficacy and safety of macitentan when co-administered with epoprostenol has not been specifically studied in controlled clinical trials.
Liver function. Hepatic enzyme elevations, and in some cases serious hepatic events, potentially related to therapy have been observed with endothelin receptor antagonists (ERAs).
The incidence of aminotransferase elevations (ALT/AST) > 3 x ULN was 3.4% on macitentan 10 mg and 4.5% on placebo in a double-blind study in patients with PAH. The incidence of elevations in ALT > 3 x ULN were 3.4% on macitentan 10 mg and 1.6% on placebo (see Section 4.8 Adverse Effects (Undesirable Effects)). The incidence of ALT > 8 x ULN were 2.1% on macitentan 10 mg and 0.4% on placebo.
Opsumit is not to be initiated in patients with severe hepatic impairment or elevated aminotransferases (> 3 x ULN) (see Section 4.3 Contraindications) and is not recommended in patients with moderate hepatic impairment. Liver enzyme tests should be obtained prior to initiation of macitentan and monthly monitoring of aminotransferases during treatment with macitentan is recommended. Patients should be monitored for signs of hepatic injury.
If clinically relevant aminotransferase elevations occur, or if elevations are accompanied by an increase in bilirubin > 2 x ULN, or by clinical symptoms of hepatic injury (e.g. jaundice), macitentan treatment should be discontinued. Re-initiation of macitentan may be considered following the return of hepatic enzyme levels to within the normal range in patients who have not experienced clinical symptoms of hepatic injury and following the advice of a liver specialist.
Caution should be exercised when Opsumit is used concomitantly with medicinal products known to be associated with hepatic injury as the additive effects of Opsumit with these agents are not known.
Haematological changes. Decreases in haemoglobin concentration and haematocrit have occurred following administration of other ERAs and were observed in clinical studies with Opsumit (see Section 4.8 Adverse Effects (Undesirable Effects)).
In placebo-controlled studies, macitentan-related decreases in haemoglobin concentration were not progressive, stabilised after the first 4-12 weeks of treatment and remained stable during chronic treatment. Cases of anaemia requiring blood cell transfusion have been reported with Opsumit and other ERAs. Initiation of Opsumit is not recommended in patients with clinically significant anaemia. It is recommended that haemoglobin concentrations be measured prior to initiation of treatment and tests repeated during treatment as clinically indicated.
Fluid retention. Oedema or fluid retention has been observed with ERAs and may also be a clinical consequence of PAH. Opsumit 10 mg was not associated with increased incidences of treatment-emergent oedema or fluid retention in a long-term placebo-controlled trial (see Section 4.8 Adverse Effects (Undesirable Effects)).
If clinically significant fluid retention develops during therapy with Opsumit, with or without associated weight gain, further evaluation should be undertaken to determine the cause, such as underlying heart failure, and the possible need for specific treatment should be considered.
Pulmonary veno-occlusive disease. Cases of pulmonary oedema have been reported with vasodilators (mainly prostacyclins) when used in patients with pulmonary veno-occlusive disease. Consequently, if signs of pulmonary oedema occur when Opsumit is administered in patients with PAH, the possibility of pulmonary veno-occlusive disease should be considered. Discontinuation of Opsumit should be considered in patients with treatment related pulmonary veno-occlusive disease.
Use in renal impairment. Patients with renal impairment may run a higher risk of experiencing hypotension and anaemia during treatment with macitentan. Therefore, monitoring of blood pressure and haemoglobin should be considered. There is no clinical experience with the use of Opsumit in patients with severe renal impairment. Caution is recommended in this population. There is no experience with the use of Opsumit in patients undergoing dialysis, therefore Opsumit is not recommended in this population.
Use in patients with pre-existing hypotension. Hypotension has been associated with the use of ERAs. Caution should be exercised when initiating macitentan in patients with pre-existing hypotension and blood pressure in such patients should be monitored closely.
Use in the elderly. Of the total number of subjects in the clinical study of Opsumit for PAH, 14% were 65 and over. No overall differences in safety or effectiveness were observed between these subjects and younger subjects. Limited data are available in those > 75 years of age, therefore caution is recommended.
Paediatric use. The safety and efficacy of Opsumit in children below 2 years of age have not been established. Currently available data are described in Section 4.8 Adverse Effects (Undesirable Effects); Section 5.1 Pharmacodynamic Properties; Section 5.2 Pharmacokinetic Properties but no recommendation on a posology can be made.
Effects on laboratory tests. See Section 4.8 Adverse Effects (Undesirable Effects), Laboratory abnormalities.
4.5 Interactions with Other Medicines and Other Forms of Interactions
In vitro studies. The metabolism of macitentan to its active metabolite aprocitentan is catalysed by CYP3A with minor contributions from CYP2C8, CYP2C9 and CYP2C19.
Macitentan and its active metabolite do not have clinically relevant inhibitory or inducing effects on CYP enzymes.
Macitentan and its active metabolite are not substrates of the multi-drug resistance protein (P-gp, MDR-1) or organic anion transporting polypeptides (OATP1B1 and OATP1B3).
Macitentan and its active metabolite are unlikely to inhibit hepatic or renal drug transporters at clinically relevant concentrations, including the multi-drug resistance protein (P-gp, MDR-1), the multidrug and toxin extrusion transporters (MATE1 and MATE2-K), the organic anion transporters (OAT1 and OAT2), the organic cation transporters (OCT1 and OCT2), the bile salt export pump (BSEP), the sodium-dependent co-transporting polypeptide (NTCP), and the organic anion transporting polypeptides (OATP1B1 and OATP1B3).
In vivo studies. Warfarin. Macitentan given as multiple doses of 10 mg once daily had no effect on exposure to S-warfarin (CYP2C9 substrate) or R-warfarin (CYP3A4 substrate) after a single dose of 25 mg warfarin. The pharmacodynamic effect of warfarin on International Normalised Ratio (INR) was not affected by macitentan. The pharmacokinetics of macitentan and its active metabolite were not affected by warfarin.
Sildenafil. At steady-state, the exposure to sildenafil 20 mg t.i.d. was increased by 15% during concomitant administration of macitentan 10 mg once daily. Sildenafil, a CYP3A4 substrate, did not affect the pharmacokinetics of macitentan, while there was a 15% reduction in the exposure to the active metabolite of macitentan. These changes are not considered clinically relevant. In a placebo controlled trial in patients with PAH, the efficacy and safety of macitentan in combination with sildenafil were demonstrated.
Strong CYP3A4 inhibitors. In the presence of ketoconazole 400 mg once daily, a strong CYP3A4 inhibitor, exposure to macitentan increased approximately 2-fold. The predicted increase was approximately 3-fold in the presence of ketoconazole 200 mg twice daily using physiologically based pharmacokinetic (PBPK) modelling. Exposure to the active metabolite of macitentan was reduced by 26%. Caution should be exercised when macitentan is administered concomitantly with strong CYP3A4 inhibitors (e.g. itraconazole, ketoconazole, voriconazole, clarithromycin, ritonavir, and saquinavir).
Fluconazole. In the presence of fluconazole 400 mg daily, a moderate dual inhibitor of CYP3A4 and CYP2C9, exposure to macitentan may increase approximately 3.8-fold based on physiologically based pharmacokinetic (PBPK) modelling. Caution should be exercised when macitentan is administered concomitantly with moderate dual inhibitors of CYP3A4 and CYP2C9 (e.g. fluconazole and amiodarone).
Caution should also be exercised when macitentan is administered concomitantly with both a moderate CYP3A4 inhibitor (e.g. ciprofloxacin, ciclosporin, diltiazem, erythromycin, verapamil) and moderate CYP2C9 inhibitor (e.g. miconazole, piperine).
HIV drugs. Effects of other strong CYP3A4 inhibitors such as ritonavir on macitentan were not studied, but are likely to result in an increase in macitentan exposure at steady-state similar to that seen with ketoconazole.
Ciclosporin A. Concomitant treatment with ciclosporin A 100 mg b.i.d., combined CYP3A4 and OATP inhibitor, did not alter the steady-state exposure to macitentan and its active metabolite to a clinically relevant extent.
Strong CYP3A4 inducers. Concomitant treatment with rifampicin 600 mg daily, a potent inducer of CYP3A4, reduced the steady-state exposure to macitentan by 79% but did not affect the exposure to the active metabolite. Reduced efficacy of macitentan in the presence of a potent inducer of CYP3A4 such as rifampicin should be considered. The combination of macitentan with strong CYP3A4 inducers (e.g. rifampicin, St. John's wort, carbamazepine, and phenytoin) should be avoided.
Hormonal contraceptives. Macitentan does not affect the exposure to CYP3A4 substrates. In healthy subjects, macitentan 10 mg once daily did not affect the pharmacokinetics of a single dose of an oral contraceptive (norethisterone 1 mg and ethinylestradiol 35 microgram).
Breast cancer resistance protein (BCRP) substrate drugs. Macitentan 10 mg once daily did not affect the pharmacokinetics of oral riociguat or rosuvastatin (riociguat 1 mg, rosuvastatin 10 mg).
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. Male fertility. Reversible testicular tubular dilatation was observed in chronic toxicity studies at exposures greater than 7-fold and 23-fold the human exposure in rats and dogs, respectively. After 2 years of treatment, tubular atrophy was seen in rats at 4-fold the human exposure. Macitentan did not affect male or female fertility in rats at exposures ranging from approximately 18 to 44-fold the human exposure, respectively. In a 26 week study in male rats treated with macitentan, there was no effect on sperm count or motility but there was a dose-dependent increase in the incidence of morphologically abnormal sperm at or above 7-fold the human exposure. No testicular findings were noted in mice after treatment up to 2 years.
Decreases in sperm count have been observed in patients taking ERAs. Opsumit, like other ERAs, may have an adverse effect on spermatogenesis.
Use in pregnancy. (Category X)
Due to a high mortality risk to both mother and foetus, pregnancy is considered contraindicated in PAH.
Teratogenicity is a class effect of endothelin receptor antagonists.
There are no data on the use of macitentan in pregnant women. Opsumit is contraindicated during pregnancy and in women of childbearing potential who are not using reliable contraception. If Opsumit is used during pregnancy, or if the patient becomes pregnant while taking Opsumit, advise the patient of the potential harm to the foetus.
Macitentan was teratogenic in rabbits and rats at all doses tested. In both rabbits and rats, there were cardiovascular and mandibular arch fusion abnormalities. A no-effect level for teratogenicity has not been established. Administration of macitentan to female rats from late pregnancy through lactation caused reduced pup survival and impairment of the male fertility of the offspring at all dose levels tested.
Use in women of child-bearing potential. In females of child bearing potential, pregnancy should be excluded before the start of treatment with macitentan and prevented thereafter by the use of two reliable methods of contraception. If necessary, patients should discuss with their doctor or gynaecologist which methods would be most suitable for them. Given the teratogenic nature of the drug, women should not become pregnant for 3 months after discontinuation of Opsumit. Monthly pregnancy tests during treatment with Opsumit are recommended to allow the early detection of pregnancy.
It is not known whether macitentan is present in semen. It is therefore not known whether there is the potential for foetal harm (teratogenicity) resulting from transfer of macitentan via semen.
Use in lactation. It is not known whether Opsumit is excreted into human breast milk. In rats, Opsumit and its metabolites were excreted into milk during lactation. Breast-feeding is not recommended during treatment with Opsumit. A risk to newborns/infants cannot be excluded.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration. See Section 4.8 Adverse Effects (Undesirable Effects).
4.8 Adverse Effects (Undesirable Effects)
Experience from clinical studies. The safety of macitentan has been evaluated in a long-term placebo-controlled trial of 742 adult and adolescent patients with symptomatic PAH (SERAPHIN study). The mean treatment duration was 103.9 weeks in the macitentan 10 mg group, and 85.3 weeks in the placebo group. The majority of adverse events (AEs) were mild to moderate in intensity. The most commonly reported AEs were nasopharyngitis (14.0% vs 10.4%), headache (13.6% vs 8.8%) and anaemia (13.2% vs 3.2%) (Table 2).
Table 3 presents adverse reactions occurring in macitentan-treated subjects at an incidence < 3% and with a placebo-corrected difference ≥ 1% (during treatment and up to 28 days after treatment discontinuation). Adverse reactions are listed by system organ class and frequency category, using the convention: common (≥ 1/100 and < 1/10). Within each frequency group, adverse reactions are presented in order of decreasing seriousness.
Frequency determination does not account for other factors including varying study duration, pre-existing conditions, and baseline patient characteristics.
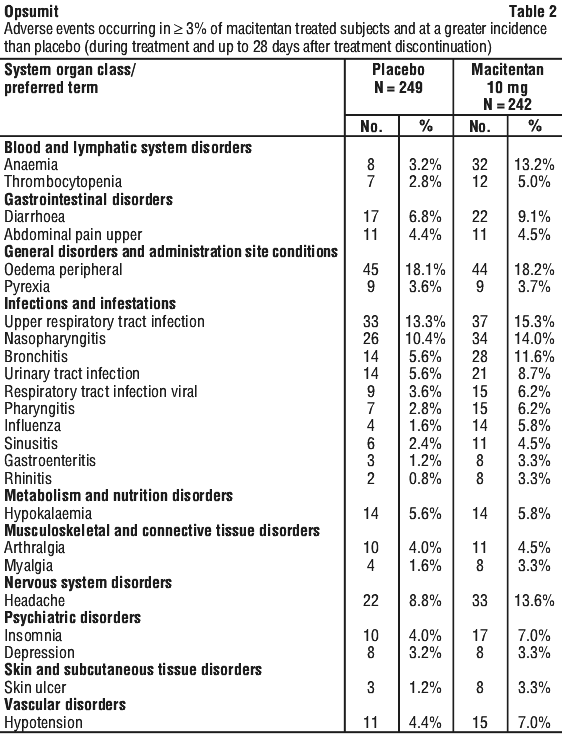
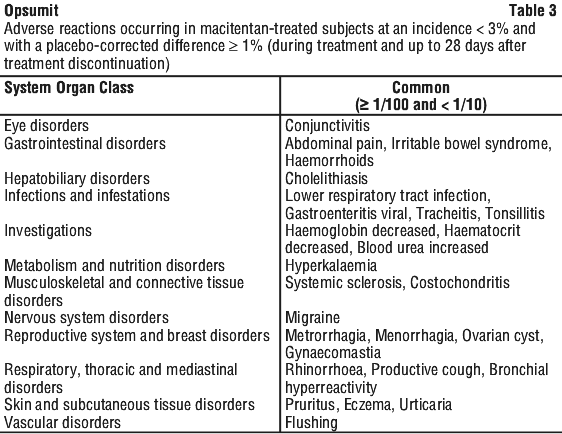
Overall, the safety profile in this paediatric population was consistent with that observed in the adult population. The following adverse drug reactions have been reported with higher frequency in paediatric patients: upper respiratory tract infection (31.9%), rhinitis (8.3%), and gastroenteritis (11.1%). The majority of adverse reactions were mild to moderate in intensity.
Paediatric population (aged ≥ 1 month to < 2 years). An additional 11 patients, aged ≥ 1 month to less than 2 years old were enrolled to receive Opsumit without randomisation, 9 patients from the open-label arm of the TOMORROW study and 2 Japanese patients from the PAH3001 study. At enrolment, the age range of the patients from the TOMORROW study was 1.2 years to 1.9 years and the median duration of treatment was 37.1 weeks (range 7.0 - 72.9 weeks). At enrolment, the ages of the 2 patients from PAH3001 were 21 months and 22 months. Overall, the safety profile in this paediatric population was consistent with that observed in the adult population and paediatric population aged ≥ 2 years to less than 18 years.
Description of selected adverse reactions. Oedema/fluid retention has been associated with the use of ERAs and is also a clinical manifestation of right heart failure and underlying PAH disease. In SERAPHIN, a long-term double-blind study in patients with PAH, the incidence of oedema AEs in the macitentan 10 mg and placebo treatment groups was 21.9% and 20.5%, respectively. This corresponded to 11.0 events/100 patient-years on macitentan 10 mg compared to 12.5 events/100 patient-years on placebo.
Laboratory abnormalities. Liver aminotransferases. The incidence of elevated aminotransferases in the study of Opsumit in PAH is shown in Tables 4 and 5.


Haemoglobin. In SERAPHIN, a double-blind study in patients with PAH, macitentan 10 mg was associated with a mean decrease in haemoglobin versus placebo of 10 g/L. A decrease from baseline in haemoglobin concentration to below 100 g/L was reported in 8.7% of patients treated with macitentan 10 mg and 3.4% of placebo-treated patients.
White blood cells. In SERAPHIN, a double-blind study in patients with PAH, macitentan 10 mg was associated with a decrease in mean leucocyte count from baseline of 0.7 x 109/L versus no change in placebo-treated patients.
Platelets. In SERAPHIN, a double-blind study in patients with PAH, macitentan 10 mg was associated with a decrease in mean platelet count of 17 x 109/L, versus a mean decrease of 11 x 109/L in placebo-treated patients.
Long-term safety. 550 patients entered a long-term open-label extension study (182 patients who continued on Opsumit 10 mg and 368 patients crossed over to Opsumit 10 mg from either placebo or macitentan 3 mg) and were followed for a median exposure of 3.3 years and a maximum exposure of 10.9 years. 242 patients received Opsumit 10 mg in the double-blind study with 182 continuing in the open label study, giving a median exposure of 4.6 years and a maximum exposure of 11.8 years. The safety profile was consistent with that described above.
Post-marketing experience. See Table 6.

4.9 Overdose
Macitentan has been administered as a single dose of up to and including 600 mg to healthy adult subjects. AEs of headache, nausea, and vomiting were observed. In the event of an overdose, standard supportive measures must be taken, as required. Due to the high degree of protein binding of macitentan, dialysis is unlikely to be effective.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Endothelin (ET)-1 and its receptors (ETA and ETB) mediate a variety of effects such as vasoconstriction, fibrosis, proliferation, hypertrophy, and inflammation. In disease conditions such as pulmonary arterial hypertension (PAH), the local ET system is upregulated and is involved in vascular hypertrophy and in organ damage.
Macitentan is an orally active, dual ETA and ETB receptor antagonist that prevents the binding of ET-1 to its receptors. Macitentan displays high affinity for and sustained occupancy of the ET receptors in human pulmonary arterial smooth muscle cells and has physicochemical properties favouring penetration into lung tissue, particularly in disease conditions.
In animal models of pulmonary hypertension, macitentan selectively decreased mean pulmonary arterial pressure without affecting systemic blood pressure, prevented pulmonary arterial hypertrophy and right ventricular remodeling, and significantly increased survival.
Cardiac electrophysiology. In a randomised, placebo-controlled four-way crossover study with a positive control in healthy adult subjects, repeated doses of macitentan 10 and 30 mg (3 times the recommended dosage) had no significant effect on the QTc interval.
Clinical trials. Efficacy in patients with pulmonary arterial hypertension. A multi-centre, double-blind, placebo-controlled, parallel-group, event-driven, Phase 3 outcome study (AC-055-302/SERAPHIN) was conducted in 742 patients with symptomatic PAH, who were randomised to three treatment groups (placebo [N = 250], 3 mg [N = 250] or 10 mg [N = 242] of macitentan once daily), to assess the long-term effect on morbidity or mortality. At baseline, the majority of enrolled patients (64%) were treated with a stable dose of specific therapy for PAH, either oral phosphodiesterase inhibitors (61%) and/or inhaled/oral prostanoids (6%). The primary study endpoint was the time to first occurrence of a morbidity or mortality event, up to end of treatment (EOT), defined as death, or atrial septostomy, or lung transplantation, or initiation of intravenous (i.v.) or subcutaneous (s.c.) prostanoids, or other worsening of PAH. Other worsening of PAH was defined as the presence of all of the three following components: a sustained decrease in 6-minute walk distance (6MWD) of at least 15% from baseline; worsening of PAH symptoms (worsening of WHO Functional Class [FC] or right heart failure); and need for new treatment for PAH. All events were confirmed by an independent adjudication committee, blinded to treatment allocation.
The median treatment duration was 101, 116, and 118 weeks in the placebo, macitentan 3 mg, and 10 mg group, respectively, up to a maximum of 188 weeks on macitentan.
Efficacy was evaluated up to the end of double-blind treatment (EOT). The EOT either coincided with end of study (EOS) for patients who completed the study as scheduled or occurred earlier in case of premature discontinuation of study drug. For those patients who stopped treatment prior to EOS, PAH therapy, including macitentan, may have been initiated. All patients were followed up to EOS for vital status. The ascertainment rate for vital status at the EOS was greater than 95%.
The mean age of all patients was 46 years (range 12-85 years of age) with the majority of subjects being Caucasian (55%) and female (77%). Approximately 52%, 46%, and 2% of patients were in WHO FC II, III, and IV, respectively. Patients with WHO Functional Class I were excluded from the study.
Idiopathic or heritable PAH was the most common aetiology in the study population (57%), followed by PAH due to connective tissue disorders (31%), PAH associated with congenital heart disease with shunts (8%), and PAH associated with other aetiologies (drugs and toxins [3%] and HIV [1%]).
Outcome endpoints. Treatment with Opsumit 10 mg resulted in a 45% reduction (HR 0.55, 97.5% CI: 0.39-0.76; logrank p < 0.0001) in the occurrence of the primary endpoint up to end of double-blind treatment compared to placebo (Figure 1 and Table 7). The beneficial effect of Opsumit 10 mg was primarily attributable to a reduction in clinical worsening events (deterioration in 6MWD and worsening of PAH symptoms and need for additional PAH treatment). The treatment effect was established early and sustained for a median duration of 2 years.
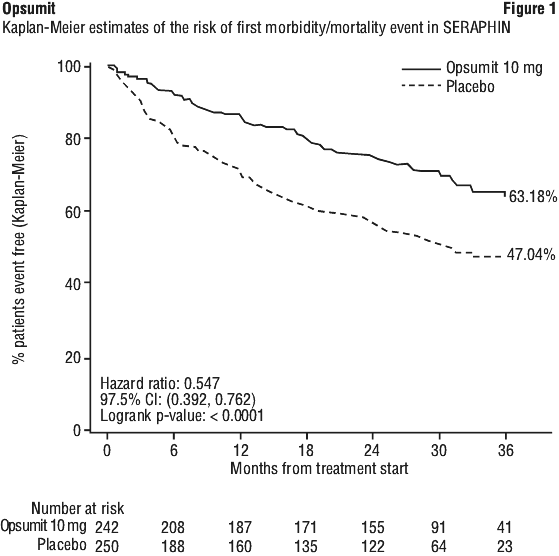
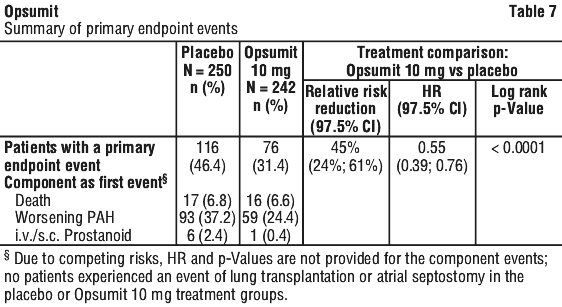
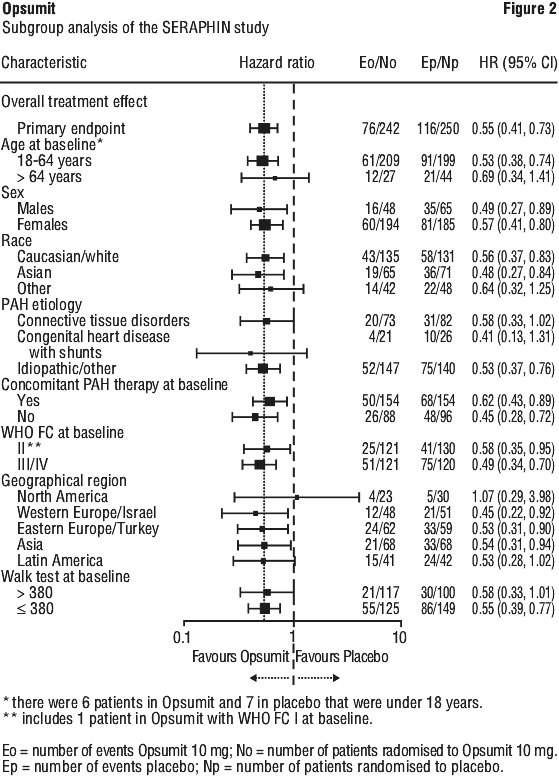
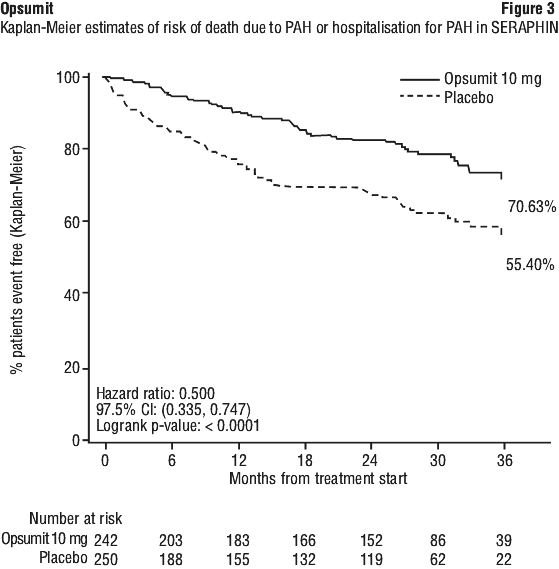
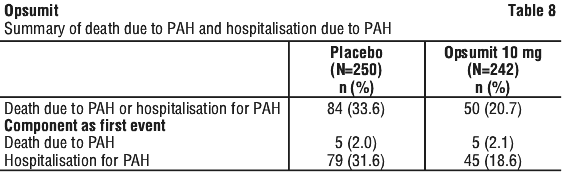
The risk of PAH-related death or hospitalisation for PAH up to the end of double-blind treatment was reduced by 50% in patients receiving macitentan 10 mg (50 events) (HR 0.50; 97.5% CI: 0.34-0.75; logrank p < 0.0001) compared to placebo (84 events) (Figure 3 and Table 8).
Treatment with macitentan 10 mg led to a 74% higher chance of WHO FC improvement relative to placebo (risk ratio 1.74; 97.5% CI: 1.10-2.74). Treatment with macitentan 10 mg led to an improvement of at least one WHO FC at Month 6 in 22% of patients compared to 13% of patients treated with placebo.
Macitentan 10 mg improved quality of life assessed by the SF-36 questionnaire.
Haemodynamic endpoints. Haemodynamic parameters were assessed in a subset of patients (placebo, N = 67, macitentan 10 mg, N = 57) after 6 months of treatment. Patients treated with macitentan 10 mg achieved a median reduction of 36.5% (CI: 21.7-49.2%) in pulmonary vascular resistance and an increase of 0.58 L/min/m2 (CI: 0.28-0.93 L/min/m2) in cardiac index compared to placebo.
Long-term treatment of PAH. In long-term follow-up of patients who were treated with Opsumit 10 mg in the double-blind/ open-label extension studies (N = 242), Kaplan-Meier estimates of survival at 1, 2, 3, 5 and 7 years were 95%, 89%, 84%, 73% and 63%. The median follow-up time was 5.9 years. Without a control group, these data must be interpreted cautiously.
Efficacy in paediatric patients with pulmonary arterial hypertension. A multi-centre, open-label, randomised, Phase 3 study with an open-label single-arm extension period (AC-055-312/TOMORROW) was conducted to assess pharmacokinetics and efficacy in paediatric patients with symptomatic PAH.
The primary endpoint was the characterisation of pharmacokinetics (see Section 5.2 Pharmacokinetic Properties).
The key secondary endpoint was the time to first Clinical Events Committee (CEC) confirmed disease progression occurring between randomisation and the end of the core period (EOCP) visit defined as, deaths (all causes), or atrial septostomy or Potts' anastomosis, or registration on lung transplant list, or hospitalisation due to worsening PAH or clinical worsening of PAH. Clinical worsening of PAH was defined as: need for, or initiation of new PAH-specific therapy or IV diuretics or continuous oxygen use AND at least 1 of the following: worsening in WHO FC, or new occurrence or worsening of syncope, or new occurrence or worsening of at least 2 PAH symptoms or new occurrence or worsening of signs of right heart failure not responding to oral diuretics.
Other secondary endpoints included time to first CEC-confirmed hospitalisation for PAH, time to CEC confirmed death due to PAH both between randomisation and EOCP, time to all cause death between randomisation and EOCP, change in WHO FC, and N-terminal prohormone of brain natriuretic peptide (NT-proBNP) data.
Paediatric population (aged ≥ 2 years to less than 18 years). A total of 148 patients aged ≥ 2 years to less than 18 years were randomised 1:1 to receive either Opsumit or Standard of Care (SoC). In the TOMORROW study, Opsumit doses were given based on weight (3.5 mg for ≥ 10 - < 15 kg, 5 mg for ≥ 15 - < 25 kg, 7.5 mg for ≥ 25 - < 50 kg and 10.0 mg for ≥ 50 kg)*. The majority of patients received at least one PAH-specific concomitant therapy (82.2% Opsumit, 100% SoC). The most common therapies were sildenafil (72.6% Opsumit, 92.0% SoC), tadalafil (11.0% Opsumit, 5.3% SoC), and bosentan (4.1% Opsumit, 48.0% SoC). The mean age was 9.8 years (range 2.1 years 17.9 years), with 35 (23.6%) aged ≥ 2 to < 6 years, 61 (41.2%) aged ≥ 6 to < 12 years, and 52 (35.1%) aged ≥ 12 to < 18 years. The majority of patients were white (51.4%) and female (59.5%). Patients were either WHO FC I (25.0%), FC II (56.1%), or FC III (18.9%).
*The doses recommended for clinical use (see Section 4.2 Dose and Method of Administration) slightly differ from those used in the TOMORROW study because PK modelling indicated that administering slightly higher doses will result in better exposure matching to the exposure achieved in adults.
Idiopathic PAH was the most common aetiology in the study population (48.0%), followed by PAH associated with post-operative congenital heart disease (28.4%), PAH with co-incidental congenital heart disease (17.6%), heritable PAH (4. 1%) and PAH associated with connective tissue disease (2.0%). The mean treatment duration in the randomised study was 183.4 weeks in the Opsumit arm and 130.6 weeks in the SoC arm.
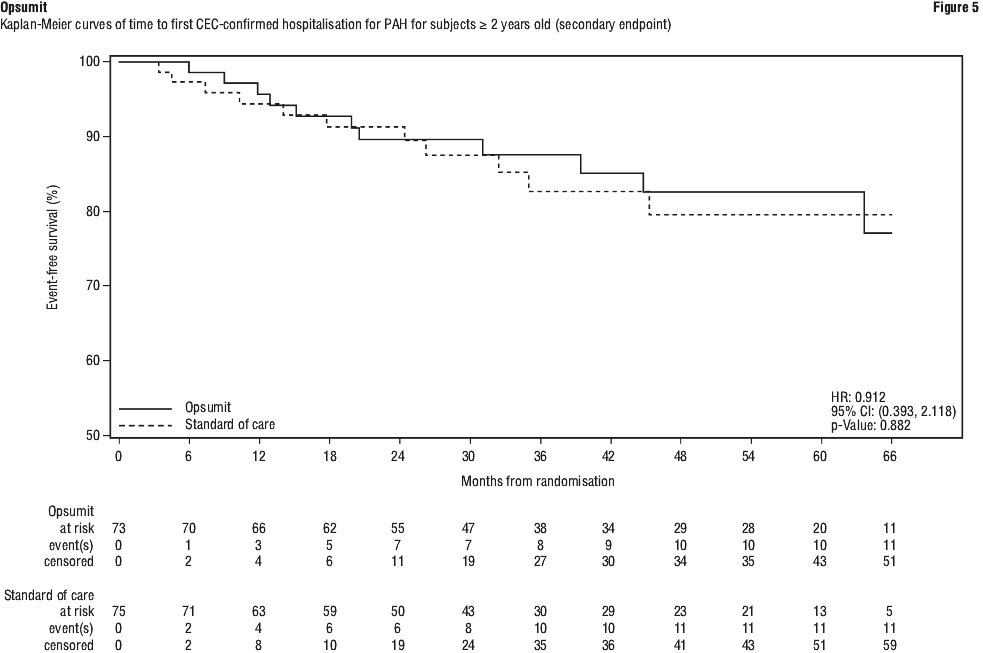
Treatment with Opsumit exhibited a trend toward clinical benefit for time to first CEC-confirmed disease progression (HR 0.828; 95% CI 0.460; 1.492; 2-sided stratified p-Value = 0.567) (Figure 4), as compared to the SoC arm. The same number of events for first-confirmed hospitalisation for PAH were observed in both groups (Opsumit 11 vs SoC 11; adjusted HR 0.912; 95% CI 0.393; 2.118; 2-sided stratified p-Value = 0.882) (Figure 5).
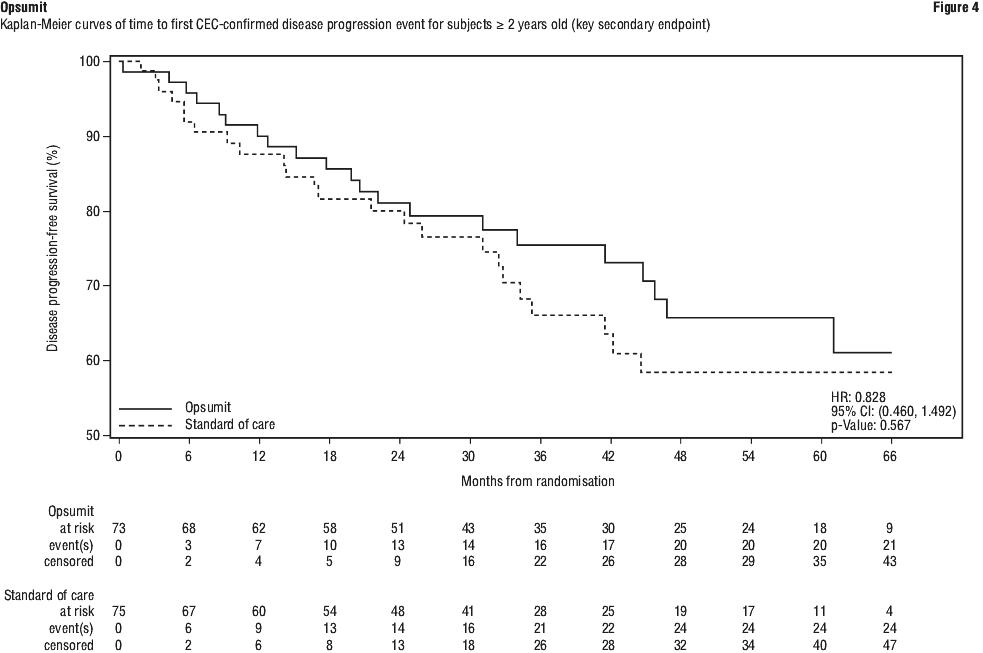
Opsumit exhibited a beneficial trend for WHO FC score I or II compared to SoC at week 12 (88.7% in macitentan arm versus 81.7% in SoC arm) and at week 24 (90.0% in macitentan arm versus 82.5% in SoC arm).
Opsumit treatment tended to reduce the percent of baseline NT-proBNP (pmol/L) at week 12 compared with the SoC arm (geometric mean ratio equal to 0.72; 95% CI 0.49 to 1.05) but the results were not statistically significant (2-sided p-Value of 0.086). The non-significant trend was less pronounced at week 24 (geometric mean ratio equal to 0.97; 95% CI 0.66 to 1.43; 2-sided p-Value of 0.884).
Paediatric population (aged ≥ 1 month less than 2 years). An additional 11 patients, aged ≥ 1 month to less than 2 years old were enrolled to receive Opsumit without randomisation, 9 patients from the open-label arm of the TOMORROW study and 2 Japanese patients from the PAH3001 study. PAH3001 was a multi-centre, open-label, single arm, Phase 3 study in Japanese paediatric participants (between ≥ 3 months and < 15 years of age) with PAH, conducted to assess the pharmacokinetics and efficacy of macitentan.
At baseline, 6 patients from the TOMORROW study were on phosphodiesterase-5 inhibitors (PDE5i) therapy. At enrolment, the age of the patients ranged from 1.2 years - 1.9 years. Patients were either WHO FC II (4 patients) or FC I (5 patients). PAH associated with congenital heart disease was the most common aetiology (5 patients), followed by idiopathic PAH (4 patients). The initially administered daily dose was 2.5 mg macitentan until the patients reached the 2 years of age. After a median follow-up of 37.3 weeks, none of the patients had experienced a CEC-confirmed disease progression event, a CEC-confirmed hospitalisation for PAH, a CEC-confirmed death due to PAH, or an event of death from all causes. NT-proBNP was reduced by 42.9% (n = 6) at week 12, 53.2% (n = 5) at week 24 and 26.1% (n = 6) at week 36.
At baseline, 1 Japanese patient from the PAH3001 study was on PDE5i therapy. Both Japanese patients were male and their ages at enrolment were 21 months and 22 months. Both patients were in Panama FC I and II and the leading aetiology was post-operative PAH. At week 24, a reduction in baseline NT-proBNP levels of -3.894 pmol/L and -16.402 pmol/L was observed.
Exposure-matching to adult patients was not established in this age group (see Section 4.2 Dose and Method of Administration; Section 5.2 Pharmacokinetic Properties).
5.2 Pharmacokinetic Properties
The pharmacokinetics of macitentan and its active metabolite aprocitentan have mainly been documented in healthy adult subjects. Exposure to macitentan in patients with PAH was approximately 1.2-fold greater than in healthy subjects. The exposure to the active metabolite in patients, which is approximately 5-fold less potent than macitentan, was approximately 1.3-fold higher than in healthy subjects. The pharmacokinetics of macitentan in PAH patients were not influenced by the severity of the disease.
After repeated administration, the pharmacokinetics of macitentan are dose-proportional up to and including 30 mg.
Absorption. Maximum plasma concentrations of macitentan are achieved about 8 to 9 hours after administration for film-coated tablets and dispersible tablets. The absolute bioavailability after oral administration is not known. Thereafter, plasma concentrations of macitentan and its active metabolite decrease slowly, with an apparent elimination half-life of approximately 16 hours and 48 hours, respectively.
In healthy subjects, the exposure to macitentan and its active metabolite is unchanged in the presence of food and, therefore, macitentan may be taken with or without food.
Distribution. Macitentan and its active metabolite aprocitentan are highly bound to plasma proteins (> 99%), primarily to albumin and to a lesser extent to alpha-1-acid glycoprotein. The apparent volumes of distribution (Vss/F) of macitentan and its active metabolite were about 50 L and 40 L, respectively, in healthy subjects.
Metabolism. Macitentan has four primary metabolic pathways. The most important is oxidative depropylation of the sulfamide to yield a pharmacologically active metabolite aprocitentan. This reaction is dependent on the cytochrome P450 system, mainly CYP3A4 (approximately 99%) with minor contributions from CYP2C8, CYP2C9 and CYP2C19. The active metabolite circulates in human plasma and may contribute to the pharmacological effect.
Other metabolic pathways yield products without pharmacological activity. For these pathways, CYP2C9 plays a predominant role with minor contributions from CYP2C8, CYP2C19 and CYP3A.
Excretion. Macitentan is only excreted after extensive metabolism. The major excretion route is via urine, accounting for about 50% of the dose.
Comparison between film-coated tablet and dispersible tablet formulations. Bioequivalence of macitentan 10 mg was established between the film-coated tablet and 4 x 2.5 mg dispersible tablets in a study with 28 healthy subjects.
Special populations. For film-coated tablets, there is no clinically relevant effect of age, sex or ethnic origin on the pharmacokinetics of macitentan and its active metabolite.
Similarly, for dispersible tablets, sex or ethnic origin do not have a clinically relevant impact on the pharmacokinetics of macitentan and its active metabolite.
Paediatrics (aged ≥ 1 month to less than 18 years). Pharmacokinetics of macitentan and its active metabolite aprocitentan were characterised in 47 paediatric patients ≥ 2 years or older and in 11 patients who are ≥ 1 month to less than 2 years old. Weight-based dose regimens of macitentan resulted in observed / simulated exposures in paediatric patients aged 2 years to less than 18 years that were comparable to exposures observed in adult PAH patients and healthy subjects who received 10 mg once daily.
Exposures of macitentan comparable to that of adult PAH patients receiving 10 mg once daily were not achieved for the age group of ≥ 1 month to less than 2 years old (see Section 4.2 Dose and Method of Administration).
Renal impairment. Exposure to macitentan and its active metabolite was increased by 1.3 and 1.6-fold, respectively, in adult patients with severe renal impairment.
Hepatic impairment. Exposure to macitentan was decreased by 21%, 34%, and 6% and for the active metabolite by 20%, 25%, and 25% in adult subjects with mild, moderate or severe hepatic impairment, respectively.
5.3 Preclinical Safety Data
Genotoxicity. Macitentan was not genotoxic in a standard battery of in vitro and in vivo assays.
Carcinogenicity. Studies of 2 years duration did not reveal a carcinogenic potential at exposures 18-fold and 116-fold the human exposure in rats and mice, respectively.
6 Pharmaceutical Particulars
6.1 List of Excipients
Film-coated tablets. Tablet core. Lactose monohydrate, microcrystalline cellulose, sodium starch glycollate Type A, povidone, magnesium stearate, polysorbate 80.
Film coating. Opadry AMB complete film coating system OY-B-28920 white, ARTG PI No. 4271.
Dispersible tablets. Croscarmellose sodium, isomalt, magnesium stearate, mannitol.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store Opsumit below 30°C, protect from moisture.
6.5 Nature and Contents of Container
Film-coated tablets. Blisters: PVC/PE/PVdC/Aluminium foil blisters in cartons containing 3^, 6^, 9 or 30 film-coated tablets.
Dispersible tablets. Aluminium cold form blisters with integrated desiccant and an aluminium push-through lidding foil in cartons containing 30 tablets.
^ Not marketed.
6.6 Special Precautions for Disposal
No special precautions for disposal.
6.7 Physicochemical Properties
Active: macitentan.
Opsumit is the brand name for macitentan and is a dual ETA and ETB endothelin receptor antagonist. The chemical name of macitentan is N-[5-(4-bromophenyl)-6-[2-[(5-bromo-2-pyrimidinyl)oxy]ethoxy]-4-pyrimidinyl]-N'-propylsulfamide.
The molecular formula: C19H20Br2N6O4S.
Relative molecular mass: 588.3 g/mol, the molecule is achiral.
Chemical structure.
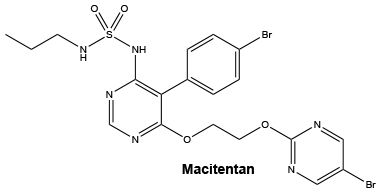
Macitentan is a white to off white crystalline powder that is insoluble in water and slightly soluble in ethanol (approx 2 mg/mL). In the solid state macitentan is very stable, is not hygroscopic, and is not light sensitive. The melting temperature of macitentan is 135°C, Partition coefficient (1-octanol/ aqueous phosphate buffer, pH 7.4): log D = 2.9 and ionization constant pKa is 6.2.
7 Medicine Schedule (Poisons Standard)
Schedule 4 - Prescription Only Medicine.
Date of First Approval
05 February 2014
Date of Revision
30 January 2026
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.