Oraqix Periodontal Gel
Brand Information
| Brand name | Oraqix Periodontal Gel |
| Active ingredient | Lidocaine (lignocaine) + Prilocaine |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Oraqix Periodontal Gel.
Summary CMI
ORAQIX® Periodontal Gel
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using ORAQIX?
ORAQIX periodontal gel is used to provide local anaesthesia in periodontal pockets during dental procedures. It contains lidocaine (lignocaine) and prilocaine, which belong to a group of medicines called local anaesthetics.
For more information, see Section 1. Why am I using ORAQIX? in the full CMI.
2. What should I know before I use ORAQIX?
Do not use if you have ever had an allergic reaction to ORAQIX or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use ORAQIX? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with ORAQIX and affect how it works.
Tell your doctor or dentist about all the medicines you take, including those bought without a prescription.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How ORAQIX is given?
ORAQIX is administered by a dental professional. It is applied directly into the periodontal pockets using a special applicator.
More instructions can be found in Section 4. How ORAQIX is given? in the full CMI.
5. What should I know while using ORAQIX?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using ORAQIX? in the full CMI.
6. Are there any side effects?
Tell your doctor or nurse as soon as possible if you do not feel well while you are being given ORAQIX.
Common side effects may include irritation or numbness at the application site. Serious allergic reactions are rare but require immediate medical attention.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
ORAQIX® Periodontal Gel
Active ingredients: lidocaine (lignocaine) and prilocaine
Consumer Medicine Information (CMI)
This leaflet provides important information about using ORAQIX. You should also speak to your doctor or pharmacist if you would like further information or if you have any concerns or questions about using ORAQIX.
Where to find information in this leaflet:
1. Why am I using ORAQIX?
2. What should I know before I use ORAQIX?
3. What if I am taking other medicines?
4. How ORAQIX is given?
5. What should I know while using ORAQIX?
6. Are there any side effects?
7. Product details
1. Why am I using ORAQIX?
ORAQIX periodontal gel contains the active ingredients lidocaine (lignocaine) and prilocaine. ORAQIX belongs to a group of medicines called local anaesthetics.
This medicine is used in adults to stop or relieve pain during certain types of dental procedures, such as scaling, probing and root planing.
It is injected into the mouth, where it makes the nerves unable to pass messages to the brain. Depending on the amount used, ORAQIX will either totally stop pain or will cause a partial loss of feeling.
Your dentist or healthcare professionals will have explained why you are being treated with ORAQIX. Follow all directions given to you by your dentist carefully. They may differ from the information contained in this leaflet. Ask your dentist or healthcare professionals if you want more information.
ORAQIX is not addictive.
Note, there is not enough information to recommend the use of this medicine for children and adolescents under the age of 18.
2. What should I know before I use ORAQIX?
Warnings
Do not use ORAQIX if:
- you are allergic to lidocaine (lignocaine), prilocaine, or any of the ingredients listed at the end of this leaflet.
- the expiry date printed on the pack has passed.
- the solution is not clear, or the packaging is torn or shows signs of tampering.
If you have an allergic reaction, you may experience:
- a shortness of breath
- wheezing or difficulty breathing
- swelling of the face, lips, tongue or other parts of the body
- rash, itching or hives on the skin.
Congenital or idiopathic methaemoglobinaemia
You must not be given ORAQIX if you have congenital or idiopathic methaemoglobinaemia.
A small amount of haemoglobin is normally present in the blood in a modified form called methaemoglobin. Methaemoglobinaemia is a condition where an excess of haemoglobin has changed into methaemoglobin. If too much methaemoglobin is formed, it becomes more difficult for the blood to provide the tissue with oxygen.
Porphyria
You must not be given ORAQIX if you have porphyria that keeps going away and coming back.
Porphyrias are a group of conditions that affect the way your blood is made. Porphyrias may mean your blood is not as red as it should be. You may also have liver, skin or nerve problems.
Check with your doctor if you:
- have any other medical conditions
- glucose-6-phospatase dehydrogenase deficiencies
- kidney or liver disease
- irregular heart activity
- mouth ulcers or an infection in your mouth
It may not be safe for you to be given ORAQIX if you have any of these conditions.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Talk to your doctor if you are pregnant or breastfeeding, or planning to. Your dental practitioner can discuss with you the risks and benefits involved.
ORAQIX should not be used in pregnancy unless clearly necessary.
You can continue to breastfeed after treatment with ORAQIX.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with ORAQIX and affect how it works. These include:
- medicines used to treat irregular heart activity (antiarrhythmics)
- other medicines that can cause methaemoglobinaemia (e.g. certain kinds of antibiotics known as sulfonamides).
You may need different amounts of your medicines, or you may need to take different medicines.
Your dental practitioner and pharmacist have more information on medicines to be careful with or avoid while being given this medicine.
4. How ORAQIX is given?
Follow all directions given to you by your dental practitioner carefully. They may differ from the information contained in this leaflet.
How much is given
Your dental practitioner will give you ORAQIX. The dosage is decided by the dental practitioner and depends on how many and which teeth are to be treated.
The maximum dose in a single treatment is 5 cartridges.
How it is given/administered
The gel is applied inside the tooth pocket by the ORAQIX dispenser and a blunt-tipped applicator.
Note, ORAQIX is not injected.
The full effect is achieved after about half a minute and the dental practitioner can start further treatment.
You may receive other local anaesthetics at the same treatment session.
Frequent use of large amounts of ORAQIX is not recommended.
If you have the impression that the effect of ORAQIX is too strong or too weak, talk to your dental practitioner.
If you are given too much ORAQIX
The dentist giving you ORAQIX will be experienced in the use of local anaesthetics, so it is unlikely that you will be given an overdose.
However, if too much local anaesthetic (i.e. ORAQIX in combination with dental injection) has been given, the following side effects may occur: numbness of the lips and around the mouth, nervousness, light-headedness, dizziness, shakiness, or sometimes blurred vision, drowsiness and loss of consciousness. Other rare effects are fits, breathlessness and lowered blood pressure.
Tell your dental practitioner immediately if you have any of the above symptoms.
Too much prilocaine (ORAQIX in combination with dental injection) may also increase the methaemoglobin level and cause methaemoglobinaemia. Methaemoglobinaemia is characterised by slate-grey cyanosis, a bluish-grey discoloration of the lips and the skin. See additional information under Section 2. What should I know before I use ORAQIX?
If you have any of the above symptoms, contact your dental practitioner or doctor, or go to Accident and Emergency at your nearest hospital for assessment of the risk and advice. You may need to be watched for several hours.
5. What should I know while using ORAQIX?
Things you should do
- Remind any doctor, dentist or pharmacist you visit that you have been recently given ORAQIX.
- If the gel comes into accidental contact with the eye, the eye must be washed with water immediately or saline solution and protected until you recover feeling in it.
Things you should not do
- ORAQIX may occasionally block all feeling in the treated area, so be careful to avoid accidental injury. Do not eat or drink anything until the feeling has returned to your mouth. You may burn or bite yourself.
- ORAQIX may interfere with tests for substances that sportswomen and sportsmen are banned from taking. ORAQIX may give a false positive test result for these substances
Driving or using machines
This medicine is not expected to affect your ability to drive a car or operate machinery.
Looking after your medicine
ORAQIX will be stored by your dental practitioner under the recommended conditions.
It should be kept in a cool dry place where the temperature stays below 24°C.
When to discard your medicine
Any ORAQIX from a single dose which is not used, will be disposed of in a safe manner by your dentist.
Do not use this medicine after the expiry date.
6. Are there any side effects?
Tell your doctor or nurse as soon as possible if you do not feel well while you are being given ORAQIX. This medicine helps most people have pain free visits to the dental practitioner, but it may have unwanted side-effects in a few people. All medicines can have side effects.
Sometimes they are serious, most of the time they are not. You may need medical attention if you get some of the side effects.
Do not be alarmed by the following lists of side effects. You may not experience any of them.
Ask your dental practitioner to answer any questions you may have.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor as soon as possible, if you notice any of these serious side effects. You may need urgent medical attention. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What ORAQIX contains
| Active ingredients (main ingredients) |
|
| Other ingredients (inactive ingredients) |
|
What ORAQIX looks like
ORAQIX is a liquid at room temperature and a gel at the temperature in the tooth pockets.
It is packed in dental cartridges intended for single use. Each cartridge contains 1.7 g gel containing the active ingredients: lidocaine (lignocaine) 42.5 mg and prilocaine 42.5 mg.
Australian Registration Number: AUST R 143855
New Zealand Registration Number: TT50-7897
Who sponsors/distributes ORAQIX
Australia
Dentsply Sirona Pty Ltd
11 – 21 Gilby Road
Mount Waverley, VIC 3149
Australia
Tel: 1300 552 929
New Zealand
Dentsply Sirona (NZ) Limited
c/o- Lowndes Jordan
Level 15, PWC Tower
188 Quay Street
Auckland 1010
Tel: 0800 336 877
® Trade Mark herein is the property of the AstraZeneca group
This leaflet was revised in December 2025 (Version 3).
Brand Information
| Brand name | Oraqix Periodontal Gel |
| Active ingredient | Lidocaine (lignocaine) + Prilocaine |
| Schedule | S4 |
MIMS Revision Date: 01 April 2026
1 Name of Medicine
Lidocaine.
Prilocaine.
2 Qualitative and Quantitative Composition
Oraqix periodontal gel contains lidocaine (25 mg/g) and prilocaine (25 mg/g) as the active substances.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Oraqix periodontal gel is a clear, colourless, oil-in-water microemulsion. Oraqix is a low viscosity fluid at room temperature and an elastic gel at the temperature in the periodontal pockets.
Oraqix Periodontal Gel is supplied in dental cartridges that provide 1.7 g Oraqix gel (corresponding to 42.5 mg lidocaine and 42.5 mg prilocaine). Individually blister-packed cartridges of Oraqix are distributed in a carton of 20. Each individual blister pack also contains a sterile blunt-tipped applicator.
4 Clinical Particulars
4.1 Therapeutic Indications
Oraqix is indicated in adults for localised anaesthesia in periodontal pockets for probing, scaling and/or root planing.
4.2 Dose and Method of Administration
Do not inject.
Administer Oraqix as a liquid. Oraqix will gel upon contact with warm oral mucosa.
Oraqix is not intended for use with standard dental anaesthetic syringes. This product should only be used with the Oraqix Dispenser, which is available from Dentsply Sirona Pty Ltd.
Adults. On average, one cartridge (1.7 g) or less of Oraqix will be sufficient for one quadrant of the dentition. The maximum recommended dose of Oraqix at one treatment session is five cartridges, i.e. 8.5 g gel containing 212.5 mg lidocaine base and 212.5 mg prilocaine base.
Clinicians should administer Oraqix in the following sequence.
1. Apply Oraqix gel around the gingival margin of 2 or 3 teeth. Gingivae will be anaesthetised after 30 seconds.
2. Insert blunt tipped cannula subgingivally and deposit Oraqix directly into periodontal pockets of the same 2 or 3 teeth. Completely fill the pockets for optimal anaesthesia. Subgingival anaesthesia will be complete after 30 seconds (a longer waiting time does not enhance the anaesthesia).
3. You may now remove Oraqix gel by having the patient rinse, though this is not necessary.
4. Commence scaling.
5. The Oraqix gel will be removed from the periodontal pocket by the directional strokes of handscaling, and/or by the lavage and tip motion of an ultrasonic scaler. This is normal and will not affect the anaesthesia duration.
6. Gel should be suctioned from the mouth as it is removed from pockets.
7. Move onto the next 2 or 3 teeth once complete.
The duration of anaesthesia, as assessed by probing of pocket depths, is about 20 minutes. If the anaesthesia starts to wear off, reapply Oraqix as needed.
When administered, Oraqix should be a liquid. If it has formed a gel, it should be placed in a refrigerator until it becomes a liquid again. The air bubble visible in the cartridge will then move if the cartridge is tilted.
The cartridge and applicator are intended for single use.
Discard any unused Oraqix.
Oraqix should not be used after the expiry date printed on the container.
4.3 Contraindications
Oraqix is contraindicated in patients with a known history of hypersensitivity to local anaesthetics of the amide type or to any other component of the product.
4.4 Special Warnings and Precautions for Use
Oraqix must not be injected.
Patients with glucose-6-phosphate dehydrogenase deficiency or congenital or idiopathic methaemoglobinaemia are more susceptible to drug induced methaemoglobinaemia (see Section 5.2 Pharmacokinetic Properties).
Care should be taken not to allow Oraqix to come in contact with the eyes as it may cause eye irritation. Also the loss of protective reflexes may allow corneal irritation and potential abrasion. If eye contact occurs, immediately rinse the eye in water or sodium chloride solution and protect it until sensation returns.
When Oraqix is used, the patient should be aware that its use may be accompanied by a block of all sensations in the treated area and, if inadvertently spread may induce numbness of the oral mucosa. Care should be taken to avoid excess Oraqix gel from spreading to the oropharyngeal mucosa. The patient should avoid inadvertent trauma to the treated area, exposure to extreme hot or cold temperatures and refrain from eating and drinking until complete sensation has returned.
Avoid contact with Oraqix to prevent the development of possible allergy.
Do not use cartridge warmers with Oraqix. The heat will cause the product to gel.
Oraqix does not contain a preservative. The product and the blunt-tipped applicator are for single use in one patient only. Discard any unused portion.
Use in hepatic impairment. Due to the extensive liver metabolism the pharmacokinetics of lidocaine and prilocaine is dependant on liver function. The lidocaine half-life may be doubled or more in patients with impaired liver function.
Use in renal impairment. Lidocaine and prilocaine and their metabolites are known to be excreted by the kidney, and the metabolites may accumulate in patients with impaired renal function.
Use in the elderly. There is no data on plasma levels of lidocaine and prilocaine following application of Oraqix in elderly patients. However, data on EMLA cream (eutectic mixture of lidocaine and prilocaine) used on intact skin do not indicate higher plasma levels in geriatric compared to non-geriatric patients.
Paediatric use. The use of Oraqix in children and adolescents has not been assessed. Isolated cases of methaemoglobinaemia in children using the combination of lidocaine and prilocaine in other drugs have been reported.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Oraqix, i.e. lidocaine and prilocaine, should be used with caution in combination with dental injection anaesthesia, other local anaesthetics or agents structurally related to amide-type local anaesthetics, e.g. antiarrhythmics such as mexiletine, since the toxic effects of these drugs are additive (see Section 4.9 Overdose).
In view of the low systemic exposure and short duration of Oraqix application, metabolic drug-drug interactions of clinical significance with lidocaine or prilocaine seem unlikely.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. The potential effects of the lidocaine/ prilocaine combination on fertility have not been evaluated.
Use in pregnancy. (Category A)
Lidocaine and prilocaine have been used by a large number of pregnant women and women of childbearing age without an increased incidence of malformations or other direct or indirect harmful effects on the foetus having been observed.
No embryofoetal effects were observed in rats dosed subcutaneously with 40 mg/kg/day lidocaine and 40 mg/kg/day prilocaine during organogenesis.
Use in lactation. Lidocaine, and in all probability, prilocaine are excreted in breast milk, though in such small quantities that there is generally no risk of the child being affected after therapeutic use of Oraqix.
4.7 Effects on Ability to Drive and Use Machines
Oraqix has no known influence on the ability to drive and use machines.
4.8 Adverse Effects (Undesirable Effects)
Clinical trial data. Although no major differences in adverse events between Oraqix and placebo treated subjects were observed, all patients in the placebo controlled studies received either Oraqix or a placebo gel (consisting of the vehicle in Oraqix without lidocaine or prilocaine). Therefore, it is not possible to determine if adverse events in each treatment group were attributable to the inactive ingredients comprising the Oraqix vehicle or if adverse event rates were higher than expected background rates. Therefore, a causal relationship between the reported adverse reactions and Oraqix could neither be established nor ruled out.
Following SRP treatment with Oraqix in 391 patients, the most frequent adverse events were local reactions in the oral cavity (see Table 1). These events, which occurred in approximately 15% of patients, included pain, soreness, irritation, numbness, vesicles, ulcerations, oedema and/or redness in the treated area. Of the 391 patients treated with Oraqix, five developed ulcerative lesions and two developed vesicles of mild to moderate severity near the site of SRP. In addition, ulcerative lesions in or near the treated area were also reported for three out of 168 patients who received placebo. Other symptoms reported in more than one patient were headache, taste perversion, nausea, fatigue, flu, respiratory infection, musculoskeletal pain and accident/ injury.
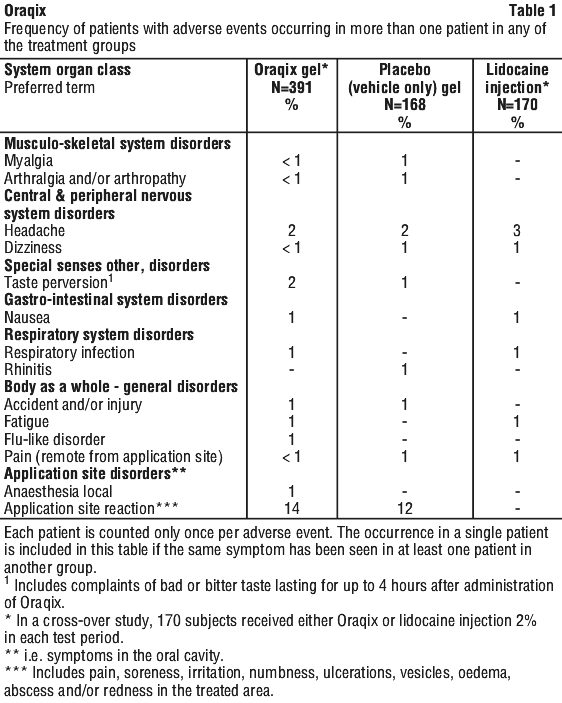
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
For information on the management of overdose, contact the Poison Information Centre on 13 11 26 (Australia) and 0800 764 766 (New Zealand).
Oraqix alone and used as recommended is not likely to cause toxic plasma levels (> 5 mg/L). However if other local anaesthetics are administered at the same time, e.g. topically or by dental injection, the toxic effects are additive and may cause an overdose with systemic toxic reactions.
Should symptoms of systemic toxicity occur, the signs are anticipated to be similar in nature to those following the administration of local anaesthetics by other routes. Local anaesthetic toxicity is manifested by symptoms of nervous system excitation and, in severe cases, central nervous and cardiovascular depression.
Severe CNS symptoms (convulsions, CNS depression) or cardiovascular symptoms must be treated symptomatically by the administration of e.g. anticonvulsive drugs, respiratory support and/or cardiovascular resuscitation as necessary.
Prilocaine in high doses may cause an increase in the methaemoglobin level, particularly in conjunction with other methaemoglobin inducing agents. Clinically significant methaemoglobinaemia should be treated with a slow intravenous injection of methylene blue.
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Lidocaine and prilocaine belong to the amide class of local anaesthetic agents which produce a local blockade of nerve impulses. Local anaesthetics affect the micro-vascular bed, which may cause a transient paleness or redness.
Oraqix is applied directly into the periodontal pockets to provide localised anaesthesia. The onset of local anaesthesia after application of Oraqix in tooth pockets is rapid, about 30 seconds, and a longer waiting time does not seem to enhance the anaesthesia. The median duration of anaesthesia, as assessed by probing of pocket depths, is 20 minutes.
Clinical trials. A total of 337 patients (146 men and 191 women; 169 Oraqix and 168 placebo) were studied in three randomised, double blind, placebo controlled trials. Subjects received a median dose of approximately 1 cartridge (1.7 g gel), ranging from ¼-2½ cartridges per quadrant treated. The primary objective of these clinical studies was to estimate the analgesic effect of Oraqix by asking subjects to rate their pain on a continuous visual analog scale (VAS) from 0 (no pain) to 100 mm (worst pain imaginable). Patients were asked to report overall procedural pain 5 minutes following manual scaling and/or root planing (SRP) in a single quadrant that had been pretreated with Oraqix or placebo (vehicle only, without lidocaine or prilocaine). In all three studies, subjects who were given Oraqix reported less pain during the procedure than those given placebo. Study B3 recruited patients with a known sensitivity to mechanical probing of dental pockets, whereas in studies B1 and B2, this was not a requirement. Results of B1, B2 and B3 are summarised in Table 2.


5.2 Pharmacokinetic Properties
Prilocaine base and lidocaine base are both relatively hydrophilic amino-amides.
Absorption. Lidocaine and prilocaine are absorbed from the oral mucous membranes to a similar extent. The systemic bioavailability after the highest recommended dose, 8.5 g, is estimated to be 20 to 40% (95% confidence interval) for both drugs. A low bioavailability is expected from the gel if swallowed, as both lidocaine and prilocaine show a substantial first-pass hepatic elimination. The median Tmax of both drugs is approximately 30 minutes.
Distribution. Lidocaine and prilocaine have an intermediate degree of plasma binding, mainly to α1-acid glycoprotein, with protein binding of 70% and 40% respectively. The plasma concentration of lidocaine is higher than that of prilocaine, with mean Cmax values of 0.17 and 0.08 mg/L respectively after single application of 0.9-3.5 g, and of 0.28 and 0.11 mg/L after a cumulative dose of 8.5 g Oraqix administered as repeated applications during 3 hours.
Metabolism. Lidocaine is mainly metabolised in the liver and has a high hepatic extraction ratio (0.65). Prilocaine has a high clearance in excess of normal hepatic blood flow, which suggests extensive extrahepatic metabolism.
The main metabolism of lidocaine is through N-dealkylation to monoethylglycinexylidide (MEGX) and glycinexylidide (GX), which is mainly mediated by CYP3A4. These are hydrolysed to 2,6-xylidine, which is converted to 4-hydroxy-2,6-xylidine, the major urinary metabolite in man. MEGX has an antiarrhythmic and convulsant activity similar to that of lidocaine and GX has a weak antiarrhythmic effect but lacks convulsant activity.
Prilocaine is split at the amide linkage to o-toluidine, which is converted further to 4 and 6-hydroxytoluidine. The formation of methaemoglobin during treatment with prilocaine is related to the plasma concentration of o-toluidine and its metabolites. However, even after the maximum recommended dose of 8.5 g Oraqix, individual maximum plasma concentrations of methaemoglobin were within the normal range (< 2% of haemoglobin).
Excretion. Lidocaine and prilocaine have mean total plasma clearances of 0.95 L/min and 2.37 L/min respectively. The terminal half-life of both drugs after IV administration is 1.6 h. After application of Oraqix, the mean terminal half-life of lidocaine is 3.6 h and of prilocaine 2.8 h, which indicates absorption dependent elimination. However, the duration of anaesthesia at the application site is only 20 minutes.
Linearity. The increase in Cmax of both lidocaine and prilocaine is proportional to the dose; at the maximum recommended dose the increase is less than proportional.
5.3 Preclinical Safety Data
Genotoxicity. There is equivocal evidence for genotoxic potential of the metabolites. See Section 5.3 Preclinical Safety Data, Carcinogenicity.
Carcinogenicity. Carcinogenicity potential of the lidocaine/prilocaine combination or each agent alone has not been studied. There is evidence that the major metabolites of these compounds, 2,6-xylidine and o-toluidine, have carcinogenic potential. In a rat Carcinogenicity study, carcinomas and adenomas of the nasal cavity and subcutaneous fibromas and/or fibrosarcomas were increased at high doses. In Carcinogenicity studies in mice and rats, the following tumours associated with o-toluidine administration were observed: hepatocarcinomas/adenomas in female mice, multiple occurrences of hemangiosarcomas/hemangiomas in both sexes of mice, sarcomas of multiple organs, transitional cell carcinomas/papillomas of urinary bladder in both sexes of rats, subcutaneous fibromas/fibrosarcomas and mesotheliomas in male rats, and mammary gland fibroadenomas/adenomas in female rates.
The carcinogenic potential of Oraqix from infrequent use at low doses is likely to be low.
6 Pharmaceutical Particulars
6.1 List of Excipients
Excipients: poloxamer (containing butylated hydroxytoluene), hydrochloric acid for pH adjustment to pH 7.5-8.0 and purified water.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 24°C to avoid gel forming in the cartridge. Refrigerate any gelated product to return it to the liquid state. Do not freeze. At temperatures below 5°C opaqueness may occur. This opaqueness will disappear when the cartridge is warmed to room temperature.
Keep out of reach of children.
6.5 Nature and Contents of Container
Each carton contains 20 x 1.7 g blister-packed cartridges.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure. The chemical structure of lidocaine is:
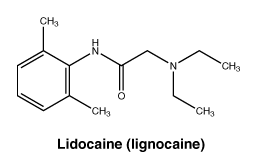
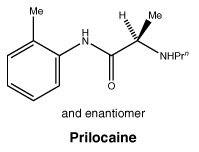
Prilocaine has a molecular weight of 220.3. The CAS number for prilocaine is 721-50-6.
7 Medicine Schedule (Poisons Standard)
S4 - Prescription Only Medicine.
Date of First Approval
06 June 2008
Date of Revision
27 January 2026
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.