Orgaran Injection
Brand Information
| Brand name | Orgaran Injection |
| Active ingredient | Danaparoid sodium |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Orgaran Injection.
Summary CMI
ORGARAN injection
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or nurse.
1. Why am I being given ORGARAN injection?
ORGARAN contains the active ingredient danaparoid sodium. It is used to help prevent blood clots forming in your veins. If you are bedridden, you have an increased risk of clot formation in the veins of the legs, especially if you have undergone an operation.
For more information, see Section 1. Why am I being given ORGARAN? in the full CMI.
2. What should I know before I receive ORGARAN injection?
Do not use if you have ever had an allergic reaction to danaparoid or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I receive ORGARAN injection? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with ORGARAN and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How is ORGARAN injection given?
- ORGARAN is administered by a subcutaneous (under the skin) injection by a doctor or nurse.
- The normal dose administered is 750 anti-Xa units (one 0.6 mL ampoule), twice a day for up to 10 postoperative days.
More instructions can be found in Section 4. How is ORGARAN injection given? in the full CMI.
5. What should I know while using ORGARAN injection?
| Things you should do |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using ORGARAN injection? in the full CMI.
6. Are there any side effects?
Common side effects of ORGARAN injection include injection site reactions (such as pain and bruising) and increased bleeding. Serious side effects of ORGARAN injection include an allergic reaction, or severe bleeding.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
ORGARAN injection
Active ingredient: danaparoid sodium
Consumer Medicine Information (CMI)
This leaflet provides important information about using ORGARAN.
You should also speak to your doctor or nurse if you would like further information or if you have any concerns or questions about using ORGARN.
Where to find information in this leaflet:
1. Why am I being given ORGARAN injection?
2. What should I know before I receive ORGARAN injection?
3. What if I am taking other medicines?
4. How is ORGARAN injection given?
5. What should I know while being given ORGARAN injection?
6. Are there any side effects?
7. Product details
1. Why am I being given ORGARAN?
ORGARAN contains the active ingredient danaparoid sodium. It is used to help prevent blood clots forming in your veins.
Blood clots which form in veins may restrict the blood flow resulting in tissue death through lack of blood. Furthermore, parts of the clot are liable to break off and may block the blood circulation in the lungs. This may have fatal consequences.
If you are bedridden, you have an increased risk of clot formation in the veins of the legs, especially if you have undergone an operation.
Ask your doctor if you have any questions about why ORGARAN injection has been prescribed for you.
Your doctor may have decided to give this medicine to you for another purpose.
2. What should I know before I receive ORGARAN?
Warnings
You must not be given ORGARAN injection if:
- you are allergic to danaparoid sodium or any of the ingredients listed at the end of this leaflet.
Symptoms of an allergic reaction may include shortness of breath, wheezing or difficulty in breathing; swelling of the face, lips, tongue, or any other parts of the body; rash, itching or hives on the skin. - you have any of the following health conditions:
- a birth condition which causes you to bleed severely e.g. haemophilia.
- a severe kidney and/or liver disorder
- severe high blood pressure
- a severe gastric or duodenal ulcer (unless it is the reason for your operation)
- positive test to an in vitro aggregation test in the presence of ORGARAN
- retina problems, caused by diabetes.
- acute infection of the inner lining and valves of the heart (acute bacterial endocarditis)
- acute stage of stroke caused by bleeding.
- bleeding uncontrollably.
Check with your doctor if you:
- have or have had any other medical conditions, including the following:
- kidney or liver damage with impaired blood circulation.
- ulcers of the stomach or intestine
- other conditions or diseases which may lead to an increased risk of bleeding into a vital organ or site.
- a sensitivity to sulfite, which, especially in asthma patients can result in severe allergic reactions.
- any other undesirable effect that you think may be due to this medication. - take any medicines for any other condition.
- have had any allergy to any other medicines, or any other substances, such as foods, preservatives, or dyes.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding.
3. What if I am taking other medicines?
Tell your doctor or nurse if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with ORGARAN and affect how it works. These include:
- medicines which interfere with platelet function such as aspirin and non -steroidal anti-inflammatory drugs (e.g. these include common painkillers such as ibuprofen and naproxen).
The combination of ORGARAN with aspirin may cause an extension to bleeding time. - medicines which may cause peptic ulcers such as corticosteroids.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect ORGARAN.
4. How is ORGARAN given?
How much is given
The normal dose is 750 anti-Xa units (one 0.6 mL ampoule), twice a day, for up to 10 postoperative days.
How it is given
ORGARAN is administered by subcutaneous (under the skin) injection by a doctor or nurse.
Extra medical supervision by your doctor or nurse may be necessary in certain circumstances.
If you are given too much
As ORGARAN is given to you under the supervision of your doctor or nurse, it is unlikely that you will receive too much.
However, if it does occur, an overdose of ORGARAN may result in an unusual loss of blood which in the first instance is corrected by stopping further administration of ORGARAN. In some cases, a blood transfusion will be necessary.
5. What should I know while being given ORGARAN injection?
Looking after this medicine
ORGARAN injection will usually be stored in the hospital ward.
It should be kept in a cool dry place, protected from light, where the temperature stays below 30°C.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or nurse if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Speak to your doctor or nurse if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Signs of an allergic reaction
| Call your doctor immediately or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
If you are on ORGARAN and are given an epidural anaesthetic (a medicine injected in the spine during surgery to reduce pain), there is an increased rare risk of developing a spinal bruise. This could result in:
| Your doctor can advise you further on this. |
Tell your doctor or nurse if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
7. Product details
This medicine is only available with a doctor's prescription.
What ORGARAN contains
| Active ingredient (main ingredient) |
|
| Other ingredients (inactive ingredients) |
|
| Potential allergens |
|
Do not use this medicine if you are allergic to any of these ingredients.
What ORGARAN injection looks like
ORGARAN injection is a clear, colourless to slightly yellow sterile liquid in a 0.6 mL glass ampoule. Available in packs of 10 ampoules.
Australian Registration number: AUST R 46096
Storage
It is unlikely that you will have to store ORGARAN as it is not self-administered.
ORGARAN is a prescription only product with a shelf-life of 3 years when stored below 30°C.
ORGARAN must not be administered beyond the expiry date shown on the ampoule and carton.
Who distributes ORGARAN injection
Aspen Pharmacare Australia Pty Ltd
34-36 Chandos St
St Leonards NSW 2065
Australia
This leaflet was revised in September 2025.
Brand Information
| Brand name | Orgaran Injection |
| Active ingredient | Danaparoid sodium |
| Schedule | S4 |
MIMS Revision Date: 01 October 2024
1 Name of Medicine
Danaparoid sodium.
2 Qualitative and Quantitative Composition
Orgaran is supplied in 0.6 mL ampoules each containing 750 anti-Xa units (approx. 55 mg) danaparoid sodium, as a sterile, isotonic solution of pH 7.
List of excipients with known effect. Sodium sulfite.
For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
Clear, colourless to slightly yellow aqueous solution.
4 Clinical Particulars
4.1 Therapeutic Indications
Orgaran is indicated for the prevention of postoperative venous thromboembolism in patients undergoing general or orthopaedic surgery.
4.2 Dose and Method of Administration
Orgaran is administered by subcutaneous injection at a dose of 750 anti-Factor Xa units twice daily for 7 to 10 days. In human studies, its safety has only been demonstrated for the use of up to 10 postoperative days duration.
In surgical patients. It is recommended to give the last pre-operative dose 1-4 hours before surgery.
Dosage in the elderly. Clearance of anti-Factor Xa activity has not been shown to be markedly reduced in the elderly and the usual dosage is recommended.
Plasma anti-Xa activity is linearly related to the dose of Orgaran given. The steady state plasma anti-Xa response is 0.15-0.35 units per mL following the recommended subcutaneous dose of Orgaran. If it is necessary to monitor anticoagulant activity, and for individual dose setting, a functional anti-Factor Xa test using a chromogenic peptide substrate should be used. In this test, Orgaran should be used as the standard.
4.3 Contraindications
Severe haemorrhagic diathesis e.g. haemophilia and idiopathic thrombo-cytopenic purpura.
Haemorrhagic stroke in the acute phase.
Uncontrollable active bleeding state.
Hypersensitivity to Orgaran.
Hypersensitivity to sulfite.
A positive in vitro aggregation test in the presence of Orgaran in patients with a history of thrombocytopenia induced by heparin-like anticoagulants.
Severe renal and/or hepatic insufficiency.
Severe hypertension.
Severe gastric or duodenal ulcer, unless it is the reason for operating.
Acute bacterial endocarditis.
Diabetic retinopathy.
4.4 Special Warnings and Precautions for Use
Identified precautions. Orgaran and low molecular weight heparins are not interchangeable clinically.
Orgaran should not be given by the intramuscular route.
Orgaran contains sodium sulfite. In asthmatic patients hypersensitive to sulfite, the latter can result in bronchospasm and/or anaphylactic shock.
Orgaran has very little cross-reactivity in producing thrombocytopenia in patients with a previous history of heparin-induced thrombocytopenia syndrome (HITS). Cross-reactivity in vitro between danaparoid sodium and heparin in producing immune platelet aggregation occurred in only 9% of patients tested. Orgaran can be administered to patients sensitised by heparin after cross-reactivity with Orgaran has been ruled out by an in vitro test.
The anticoagulant activity of Orgaran is characterised by a very flat dose-response curve in clotting assays such as prothrombin time, activated partial thromboplastin time, kaolin cephalin clotting time and thrombin clotting time, therefore these routine clotting assays are unsuitable for monitoring its anticoagulant activity.
No incidences of osteoporosis have been reported in patients treated with the recommended dose of Orgaran. However, as for heparin, treatment with a glycosaminoglucoronan may result in osteoporosis if the dosage is inappropriate.
It should be noted that the anti-Xa units of Orgaran have a different relationship to clinical efficacy than those of heparin and low molecular weight heparins.
Spinal/epidural haematomas. When neuraxial anaesthesia (epidural/spinal anaesthesia) or spinal puncture is employed, patients anticoagulated or scheduled with low molecular weight heparins or heparinoids for prevention of thromboembolic complications are at risk of developing an epidural or spinal haematoma which can result in long-term or permanent paralysis.
The risk of these events is increased by the use of indwelling epidural catheters for administration of analgesia or by the concomitant use of drugs affecting haemostasis such as non-steroidal anti-inflammatory drugs (NSAIDs), platelet inhibitors, or other anti-coagulants. The risk also appears to be increased by traumatic or repeated epidural or spinal puncture.
Patients should be frequently monitored for signs and symptoms of neurological impairment. If neurologic compromise is noted, urgent treatment is necessary.
The physician should consider the potential benefit versus risk before neuraxial intervention in patients anticoagulated or to be anticoagulated for thromboprophylaxis.
Use in hepatic and renal impairment. Orgaran should be used with caution in patients with moderately impaired renal and/or liver function with impaired haemostasis, ulcerative lesions of the gastro-intestinal tract or other diseases/conditions which may lead to an increased danger of haemorrhage into a vital organ or site.
Use in the elderly. No data available.
Paediatric use. No data available.
Effects on laboratory tests. No data available.
4.5 Interactions with Other Medicines and Other Forms of Interactions
In clinical studies no clinically significant interactions with other medications have been found.
Orgaran may be used together with oral anticoagulants, or drugs which interfere with platelet function, such as aspirin and non-steroidal anti-inflammatory drugs, or potentially ulcerogenic drugs (such as corticosteroids), but caution remains necessary. Combination of Orgaran with aspirin may cause a prolongation of the bleeding time.
Monitoring of anticoagulant activity of oral anticoagulants by prothrombin time and Thrombotest is unreliable within 5 hours following Orgaran administration.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category C)
In rats, doses of danaparoid greater than 100 anti-Xa units/kg/day administered IV during gestation reduced postnatal weight gain of the offspring. Higher doses, up to 1600 anti-Xa units/kg/day, did not cause any other maternotoxic or teratogenic effects. In rabbits, IV doses up to 800 anti-Xa units/kg/day did not cause any foetal malformations, but the highest dose used was insufficient to rule out the possibility of teratogenic activity in this species. The use of Orgaran in human pregnancy has only been studied incidentally. Observations in pregnant women have so far given no indication that the use of Orgaran during pregnancy leads to foetal abnormalities or to exacerbation of bleeding in mother or infant during delivery.
Use in lactation. There is no data available on Orgaran secretion into breast milk.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
Bruising and/or pain may occur at injection sites. Skin rashes and other local or generalised hypersensitivity reactions have occasionally been observed (see Table 1).
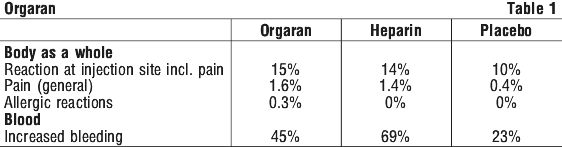
Liver abnormalities such as changes in transaminase and alkaline phosphates, have been observed, but no clinical significance has been demonstrated.
4.9 Overdose
In the event of serious bleeding other than caused by a surgical error, Orgaran should be stopped and blood transfusion considered. Although protamine partially neutralises the anticoagulant activity of Orgaran, it is likely to be of little use in reversing the potential bleeding complications associated with overdosage and therefore cannot be recommended.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Pharmacology. Danaparoid sodium has been shown both in animal models and in human studies to be an effective antithrombotic substance. Danaparoid sodium has a negligible effect on haemostatic plug formation, platelet function and platelet aggregability. In animal models it produces less bleeding enhancing activity than heparin whilst in human studies the effects of Orgaran and heparin were found to be similar.
The ultimate step in blood coagulation, the fibrinogen-fibrin conversion, is critically dependent on thrombin generation, to which Factor Xa and thrombin contribute substantially. The anticoagulant profile of danaparoid sodium is characterised by a high ratio of anti-Factor Xa/antithrombin activities (approx. 20/1), resulting in an effective inhibition of thrombin generation and thrombus formation with minimal bleeding enhancing activity. The anti-Xa activity is mediated by antithrombin-III and is not inactivated by endogenous heparin-neutralising factors. The small antithrombin activity is mediated by heparin co-factor II and antithrombin-III. The heparan sulfate fraction with low affinity for antithrombin-III, lacking significant effects on coagulation Factors Xa and IIa in vitro, has been shown in animal studies to contribute substantially (approx. 50%) to the antithrombotic activity by an as yet unexplained mechanism. The risk of haemorrhage with this drug is not related to plasma anti-Xa activity, within the therapeutic dose range.
Clinical trials. Clinical studies have demonstrated that the clinical activity of danaparoid sodium may not relate to the dose expressed as either milligrams or anti-Xa activity.
5.2 Pharmacokinetic Properties
Pharmacokinetic studies were primarily based on the kinetics of relevant anticoagulant activities of danaparoid sodium, as no specific chemical assay methods are available.
The absolute bioavailability of danaparoid sodium after subcutaneous administration approaches 100%. In humans, the time to reach peak plasma anti-Xa activity levels is approximately 4-5 hours.
The half-lives of elimination of anti-Xa and thrombin generation inhibiting activities of approximately 25 hours and 7 hours, respectively, after both subcutaneous and intravenous administration, are independent of the dose and route of administration. Although plasma clearance of anti-Xa activity was found to be 20% lower in female volunteers than in male volunteers, this difference is not significant. Steady-state levels of plasma anti-Xa activity are usually reached within 4-5 days of dosing. Measured by thrombin generation inhibiting activity, steady-state levels are reached earlier, i.e. within 1-2 days. Danaparoid sodium is mainly eliminated by renal excretion. Animal experiments indicate that the liver is not involved in its metabolism. In patients with severely impaired renal function, the half-life of elimination of plasma anti-Factor Xa activity may be prolonged (see Section 4.2 Dose and Method of Administration).
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. Long-term animal carcinogenicity studies of Orgaran have not been done. A standard battery of mutagenicity tests has not shown any evidence of genotoxic potential.
6 Pharmaceutical Particulars
6.1 List of Excipients
Hydrochloric acid, sodium chloride, sodium sulfite and water for injections.
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C. Protect from light.
6.5 Nature and Contents of Container
Pack of ten 0.6 mL ampoules.
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
CAS number. 83513-48-8.
Orgaran is supplied in 0.6 mL ampoules each containing 750 anti-Xa units (approx. 55 mg) danaparoid sodium, as a sterile, isotonic solution of pH 7.
Orgaran contains danaparoid sodium, which is a mixture of low molecular weight sulfated glycosaminoglycuronans derived from animal mucosa, comprising heparan sulfate, dermatan sulfate and a minor amount of chondroitin sulfate. One mL contains 1250 amidolytic anti-Factor Xa units (approx. 90 mg) danaparoid sodium and 0.15% (w/v) sodium sulfite. The anti-Xa unit is derived from the international heparin standard in an antithrombin-III containing buffer system.
7 Medicine Schedule (Poisons Standard)
S4 - Prescription Medicine.
Date of First Approval
06 April 1994
Date of Revision
18 July 2024
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.