Osmitrol Intravenous Infusion
Brand Information
| Brand name | Osmitrol Intravenous Infusion |
| Active ingredient | Mannitol |
| Schedule | Unscheduled |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Osmitrol Intravenous Infusion.
Summary CMI
Osmitrol Intravenous Infusion
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I receiving Osmitrol?
Osmitrol Intravenous Infusion contains mannitol. It is used to help the body make more urine to prevent or treat early kidney failure before it becomes permanent. It is also used to lower high pressure inside the eye (when other treatments don't work), reduce pressure inside the head and swelling of the brain or help the body get rid of harmful substances through the urine.
For more information, see Section 1. Why am I receiving Osmitrol? in the full CMI.
2. What should I know before I receive Osmitrol?
Do not use if you have ever had an allergic reaction to any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I receive Osmitrol? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with Osmitrol and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How is Osmitrol given?
- Your doctor or trained nurse will give Osmitrol by an infusion (drip) into a vein (intravenously).
- Your doctor will decide how much Osmitrol to give. The amount depends on your need and condition.
More instructions can be found in Section 4. How is Osmitrol given? in the full CMI.
5. What should I know while receiving Osmitrol?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while receiving Osmitrol? in the full CMI.
6. Are there any side effects?
Serious side effects, such as allergic reactions, cardiac failure and kidney failure, are possible.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
Osmitrol Intravenous Infusion
Active ingredient: mannitol
Consumer Medicine Information (CMI)
This leaflet provides important information about using Osmitrol Intravenous Infusion (Osmitrol). You should also speak to your doctor, nurse or pharmacist if you would like further information or if you have any concerns or questions about using Osmitrol.
Where to find information in this leaflet:
1. Why am I receiving Osmitrol?
2. What should I know before I receive Osmitrol?
3. What if I am taking other medicines?
4. How is Osmitrol given?
5. What should I know while receiving Osmitrol?
6. Are there any side effects?
7. Product details
1. Why am I receiving Osmitrol?
Osmitrol contains the active ingredient mannitol. Mannitol is a type of sugar alcohol that is used as a medicine to help remove extra water from the body. It works by making your kidneys produce more urine.
Osmitrol is used to:
- Help the body make more urine to prevent or treat early kidney failure before it becomes permanent.
- Lower high pressure inside the eye when other treatments don't work.
- Reduce pressure inside the head and swelling of the brain.
- Help the body get rid of harmful substances through the urine.
2. What should I know before I receive Osmitrol?
Warnings
Do not use Osmitrol if:
- you are allergic to mannitol or any of the ingredients listed at the end of this leaflet.
- Always check the ingredients to make sure you can use this medicine.
- you have higher-than-normal amounts of salts, sugars or other substances in your blood.
- you have severe heart failure, or heart or lung issues following the start of Osmitrol therapy.
- you have severe kidney problems where you are passing little to no urine, or kidney damage following the start of Osmitrol therapy.
- you did not respond to the Osmitrol test dose.
- you have fluid in your lungs, causing severe breathing difficulties.
- you have active bleeding on the brain, or a condition that allows harmful substances to cross from your bloodstream into your brain.
- you are very dehydrated.
Check with your doctor if you:
- have kidney problems
- have heart problems
- are in shock (a medical condition where your body doesn't have enough blood to keep your organs working properly)
- have recently had an operation
- drink water compulsively
- have a condition that affects your central nervous system
- have any other medical conditions
- take any medicines for any other condition
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Check with your doctor if you are pregnant or intend to become pregnant.
Talk to your doctor if you are breastfeeding or intend to breastfeed.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines and Osmitrol may interfere with each other and put your health at risk. These include:
- diuretics (a group of medicines used to remove fluid from the body)
- lithium (a medicine used to treat bipolar disorder)
- methotrexate (a medicine used to treat some autoimmune conditions and cancers)
- Cyclosporin (a medicine used to prevent rejection of organ transplants)
- certain types of antibiotic medicines (aminoglycosides)
- some medicines used to relax muscles
- some medicines used to prevent blood clots
- digoxin (a medicine used to treat heart problems)
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect Osmitrol.
4. How is Osmitrol given?
How it is given
- Your doctor or trained nurse will give Osmitrol by an infusion (drip) into a vein (intravenously).
- It is usually given in the hospital, clinic or nursing home.
How much is given
- Your doctor will decide how much Osmitrol to give.
The amount depends on your need and condition.
If you are given too much Osmitrol
The doctor or nurse giving you Osmitrol is trained and experienced in the use of this sort of medicine, so it is unlikely that you will be given an overdose.
If you think that you have been given too much, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while receiving Osmitrol?
Things you should do
During and after treatment, discuss your progress with your doctor. Tell them if you've had any side effects or concerns, especially during the first few days of therapy.
Your doctor and nurse will closely monitor you during treatment, and may use laboratory tests to make sure you are safely receiving the right amount of medicine.
Remind any doctor, nurse, dentist or pharmacist you visit that you have used Osmitrol.
Things you should not do
- Osmitrol has been prescribed for you and should not be used by anyone else.
- Do not use Osmitrol to treat any other condition unless your doctor tells you to.
Driving or using machines
Be careful before you drive or use any machines or tools until you know how Osmitrol affects you.
The effects of Osmitrol on driving and using machines or tools have not been studied.
Drinking alcohol
Tell your doctor if you drink alcohol.
Osmitrol may cause dizziness or tiredness in some people.
Looking after your medicine
Osmitrol will be stored in the pharmacy or the hospital ward, below 30°C.
Getting rid of any unwanted medicine
Osmitrol is for single use, and for one person only. Your doctor or nurse will discard any unused medicine. They will not save unused portions for later, either for you or anyone else.
6. Are there any side effects?
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
Infusion site-related:
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
Infusion site-related:
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people. Some side effects, such as imbalances in body salt levels, can only be detected using laboratory tests.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What Osmitrol contains
| Active ingredients (main ingredients) | Mannitol |
| Other ingredients (inactive ingredients) | Hydrochloric acid/Sodium hydroxide (for pH adjustment) |
Do not take this medicine if you are allergic to any of these ingredients.
What Osmitrol looks like
Osmitrol is a clear, colourless solution in VIAFLEX plastic bags. It is available as:
- 10% Osmitrol (Mannitol) IV Infusion, 1000 mL – Aust R 19479
- 20% Osmitrol (Mannitol) IV Infusion, 500 mL – Aust R 19496
Who distributes Osmitrol
Baxter Healthcare Pty Ltd
1 Baxter Drive
Old Toongabbie
NSW 2146
Australia
This leaflet was prepared in November 2025.
Brand Information
| Brand name | Osmitrol Intravenous Infusion |
| Active ingredient | Mannitol |
| Schedule | Unscheduled |
MIMS Revision Date: 01 March 2021
1 Name of Medicine
Mannitol.
2 Qualitative and Quantitative Composition
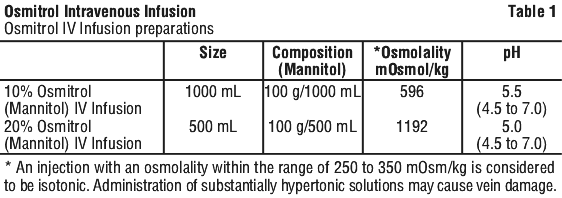
Osmitrol Intravenous (IV) Infusion (Mannitol IV Infusion,) is a sterile, nonpyrogenic solution of Mannitol, in a single dose container for intravenous administration. It contains no antimicrobial agents. Mannitol is a six carbon sugar alcohol prepared commercially by the reduction of glucose. Although virtually inert metabolically in humans, it occurs naturally in fruits and vegetables. Mannitol is an obligatory osmotic diuretic. The pH is adjusted with sodium hydroxide and hydrochloric acid. Composition, osmolality and pH are shown in Table 1.
3 Pharmaceutical Form
Solution for Intravenous Infusion.
4 Clinical Particulars
4.1 Therapeutic Indications
Osmitrol IV Infusion can be used in:
the promotion of diuresis, in the prevention and/or treatment of the oliguric phase of acute renal failure before irreversible renal failure becomes established;
the reduction of elevated intraocular pressure when the pressure cannot be lowered by other means;
the reduction of intracranial pressure and treatment of cerebral oedema by reducing brain mass;
promoting the urinary excretion of toxic substances.
4.2 Dose and Method of Administration
Dosage. Osmitrol (IV) Infusion should be administered only by intravenous Infusion. The total dosage, concentration, and rate of administration should be governed by the nature and severity of the condition being treated, fluid requirement, and urinary output. There should be a dosage limit of 50 g of Osmitrol IV Infusion on any one occasion. The usual adult dosage ranges from 20 to 100 g in a 24 hour period, but in most instances an adequate response will be achieved at a dosage of approximately 50 to 100 g in a 24 hour period. The rate of administration is usually adjusted to maintain a urine flow of at least 30 to 50 mL/hour. This outline of administration and dosage is only a general guide to therapy.
Parenteral medicine products should be inspected visually for particulate matter and discolouration prior to administration whenever solution and container permit. Use of a final filter is recommended during administration of all parenteral solutions, where possible.
Test dose. A test dose of mannitol should be given prior to instituting Osmitrol IV Infusion therapy for patients with marked oliguria, or those believed to have inadequate renal function. Such a test dose may be approximately 0.2 g/kg bodyweight (about 75 mL of a 20% solution) infused in a period of three to five minutes to produce a urine flow of at least 30 to 50 mL/hour. If urine flow does not increase, a second test dose may be given; if there is an inadequate response, the patient should be re-evaluated.
Prevention of acute renal failure (oliguria). When used during cardiovascular and other types of surgery, 50 to 100 g of mannitol as a 10% or 20% solution may be given. The concentration will depend upon the fluid requirements of the patient.
Treatment of oliguria. The usual dose for treatment of oliguria is 100 g administered as a 20% solution.
Reduction of intraocular pressure. A dose of 1.5 to 2.0 g/kg as a 20% solution (7.5 to 10 mL/kg) may be given over a period as short as 30 minutes in order to obtain a prompt and maximum effect. When used preoperatively the dose should be given one to one and a half hours before surgery to achieve maximum reduction of intraocular pressure before operation.
Reduction of intracranial pressure. Usually a maximum reduction in intracranial pressure in adults can be achieved with a dose of 0.25 g per kg given not more frequently than every six to eight hours. An osmotic gradient between the blood and cerebrospinal fluid of approximately 10 mOsmL per litre will yield a satisfactory reduction in intracranial pressure.
Adjunctive therapy for intoxication. As an agent to promote diuresis in intoxications, 10% or 20% mannitol is indicated. The concentration will depend upon the fluid requirement and urinary output of the patient. Measurement of glomerular filtration rate by creatine clearance may be useful for determination of dosage.
All intravenous infusions in Viaflex containers are intended for intravenous administration using sterile equipment.
Directions for use. Warning. Mannitol solutions may crystallize when exposed to low temperature. At higher concentrations, the solutions have a greater tendency to crystallize. Inspect for crystals prior to administration. If crystals are visible, re-dissolve by warming the solution up to 70°C, with agitation. Solutions should not be heated in water or in a microwave oven due to the potential for product contamination or damage. Allow the solution to cool to room or body temperature before reinspection for crystals and use.
Do not use plastic container in series connections. Such use could result in air embolism due to residual air being drawn from the primary container before administration of the fluid from the secondary container is completed.
Pressurising intravenous solutions contained in flexible plastic containers to increase flow rate can result in air embolism if the residual air in the container is not fully evacuated prior to administration.
Use of a vented intravenous administration set with the vent in the open position could results in air embolism. Vented intravenous administration sets with the vent in the open position should not be used in flexible plastic container.
To open. Tear overwrap down side at slit and remove solution container. Some opacity of the plastic due to moisture absorption during the sterilisation process may be observed. This is normal and does not affect the solution quality or safety. The opacity will diminish gradually. Check for minute leaks by squeezing the inner bag firmly. If leaks are found, discard solution as sterility may be impaired.
Preparation for administration. Osmitrol IV Infusion is a sterile preparation. Thus, aseptic technique must be applied throughout the administration.
1. Suspend container from eyelet support, at bottom of container.
2. Remove plastic protector from outlet port at bottom of container.
3. Attach administration set. Refer to complete directions accompanying set.
4.3 Contraindications
Osmitrol IV Infusion is contraindicated in patients with:
hypersensitivity to mannitol;
pre-existing plasma hyperosmolarity;
severe heart failure;
disturbance of the blood brain barrier;
well established anuria due to severe renal disease;
no response to test dose;
severe pulmonary congestion or frank pulmonary oedema;
active intracranial bleeding except during craniotomy;
severe dehydration;
progressive renal damage or dysfunction after institution of mannitol therapy, including increasing oliguria and azotemia;
progressive heart failure or pulmonary congestion after institution of mannitol therapy.
4.4 Special Warnings and Precautions for Use
General. Osmitrol IV Infusion is hypertonic. Hypertonic solutions should be administered via a large peripheral and preferably central vein. Rapid infusion in peripheral veins may be harmful.
Hypersensitivity reactions. Anaphylactic/ anaphylactoid reactions, including anaphylaxis, as well as other hypersensitivity/ infusion reactions have been reported with mannitol. Fatal outcome has been reported. (See Section 4.8 Adverse Effects (Undesirable Effects)).
The infusion must be stopped immediately if any signs or symptoms of a suspected hypersensitivity reaction develop. Appropriate therapeutic countermeasures must be instituted as clinically indicated.
Risk of renal complications. Reversible, acute oligoanuric renal failure has occurred in patients with normal pretreatment renal function who received large intravenous doses of mannitol.
In patients with severe impairment of renal function, a test dose should be utilised (see Section 4.2 Dose and Method of Administration). A second test dose may be tried if there is inadequate response, but no more than two test doses should be attempted.
Patients with pre-existing renal disease, or those receiving potentially nephrotoxic drugs, are at increased risk of renal failure following administration of Osmitrol IV Infusion. Serum osmolarity, urine flow and renal function should be monitored particularly closely.
The acid/ base, renal function and serum osmolarity must be monitored carefully when Osmitrol IV Infusion is used. Should patient serum osmolarity increase during treatment, the effects of Osmitrol IV Infusion on diuresis and reduction of intracranial and intraocular pressures may be impaired.
Osmotic nephrosis, a reversible vacuolisation of the tubules of unknown clinical significance, may proceed to severe irreversible nephrosis, so that the renal function must be closely monitored during mannitol infusion. If urine output continues to decline during mannitol infusion, the patient's clinical status should be closely reviewed and mannitol infusion suspended if necessary. Accumulation of mannitol may result in overexpansion of the extracellular fluid, which may intensify existing or latent congestive heart failure.
CNS toxicity. CNS toxicity manifested by, e.g. confusion, lethargy and coma has been reported in patients treated with mannitol, in particular in the presence of impaired renal function. Fatal outcomes have been reported. CNS toxicity may result from:
high serum mannitol concentrations;
serum hyperosmolarity resulting in intracellular dehydration within the CNS;
hyponatraemia or other disturbances of electrolyte and acid/ base balance;
secondary to mannitol administration.
At high concentrations, mannitol may cross the blood brain barrier and interfere with the ability of the brain to maintain the pH of the cerebrospinal fluid especially in the presence of acidosis.
A rebound increase in intracranial pressure may occur approximately 12 hours after the use of mannitol for the reduction of intracranial pressure.
The use of mannitol in acute traumatic brain injury and acute stroke is not recommended. This is based on two systematic reviews that indicate the potential for harm and lack of sufficient data for a definitive assessment of the risk or benefit for using mannitol in these two clinical conditions.
Risk of water and electrolyte imbalances, hyperosmolarity. The obligatory diuretic response following rapid infusion of a 20% mannitol IV Infusion may further aggravate pre-existing haemoconcentration. Excessive loss of water and electrolytes, may lead to serious imbalances such as hypernatraemia. Electrolyte measurements, including serum sodium and potassium are of vital importance and should be carefully monitored during mannitol administration.
Mannitol induced osmotic diuresis may cause or worsen dehydration/ hypovolaemia and haemoconcentration.
Osmitrol IV Infusion should not be administered in patients with shock and renal dysfunction until volume (fluid; blood) and electrolytes have been replaced.
In addition, depending on dosage and duration of administration, electrolyte and acid/ base imbalances may result from transcellular shifts of water and electrolytes, osmotic diuresis and/or other mechanisms. Such imbalances may be severe and potentially fatal.
Imbalances that may result from mannitol treatment include:
hypernatraemia, dehydration and haemoconcentration;
hyponatraemia, can lead to headache, nausea, seizures, lethargy, coma, cerebral edema, and death. Acute symptomatic hyponatraemic encephalopathy is considered a medical emergency. The risk for developing hyponatraemia is increased, for example in children, elderly patients, women, postoperatively and in persons with psychogenic polydipsia. The risk for developing encephalopathy as a complication of hyponatraemia is increased, for example in pediatric patients (≤ 16 years of age), women (in particular, premenopausal women), patients with hypoxemia and patients with underlying central nervous system disease;
hypokalemia;
hyperkalemia;
other electrolytes imbalances;
metabolic acidosis;
metabolic alkalosis.
By sustaining diuresis, mannitol administration may obscure and intensify inadequate hydration or hypovolemia.
The use of supplemental additive medication is not recommended.
Risk of hypervolaemia. The cardiovascular status of the patient should be carefully evaluated before rapidly administrating mannitol since sudden expansion of the extracellular fluid may lead to fulminating congestive heart failure.
Shift of sodium free intracellular fluid into the extracellular compartment following mannitol infusion may lower serum sodium concentration and aggravate pre-existing hyponatremia.
Patients receiving Osmitrol IV Infusion should be monitored for any deterioration in renal, cardiac or pulmonary function and treatment discontinued in the case of adverse events.
Incompatibility with blood. Electrolyte and mannitol IV infusions should be not given conjointly with blood. If it is essential that blood be given simultaneously, at least 20 mEq of sodium chloride should be added to each litre of mannitol solution to avoid pseudoagglutination.
Incompatibility with additives. There may be potential incompatibility with additives which include the risk of precipitation if potassium or sodium chloride is added to mannitol. Also some antibiotics including cefepime, imipenem or cilastatin may be incompatible with mannitol.
Crystallisation. When exposed to low temperatures, solutions of mannitol may crystalise. Concentrations of 20% have a greater tendency to crystallisation. Inspect for crystals prior to administration. If crystals are visible, redissolve by warming the solution up to 70°C, with agitation. Solutions should not be heated in water or in a microwave oven due to the potential for product contamination or damage. Allow the solution to cool to room temperature before reinspection for crystals. Administer intravenously using a sterile, filter type set.
Infusion site reactions. Infusion site reactions have occurred with the use of mannitol including signs and symptoms of infusion site irritation and inflammation as well as severe reactions (compartment syndrome and bullous eruptions) when associated with extravasation.
Use in the elderly. As for adults, the dosage depends on the weight, clinical and biological condition of the patient and concomitant therapy. The general dose range is the same as for adults (50 to 200 g in 24 hour period), with a dosage limit of 50 g on any one occasion. Since incipient renal insufficiency may be present, caution should be used when reviewing patient's status prior to dose selection.
Paediatric use. Dosage requirements for patients 12 years of age and under have not been established. Safety and effectiveness in this population have not been established.
Effects on laboratory tests. Although blood levels of mannitol can be measured there is little if any clinical virtue in doing so. The appropriate monitoring of blood levels of sodium and potassium; degree of haemoconcentration and haemodilution, if any, indices of renal, cardiac and pulmonary function are paramount in avoiding excess fluid and electrolyte shifts. The routine features of physical examination and clinical chemistries suffice in achieving an adequate degree of appropriate patient monitoring.
Mannitol can cause false low results in some tests systems for inorganic phosphorus blood concentrations.
Mannitol produces false positive results in tests for blood ethylene glycol concentrations in which mannitol is initially oxidised to an aldehyde.
4.5 Interactions with Other Medicines and Other Forms of Interactions
Potentiation effects. Concurrent use of other diuretics may potentiate the effects of mannitol and dose adjustments may be required.
Inhibition effects. Concomitant use of mannitol impairs the response to lithium and methotrexate due to the increases in urinary excretion.
Cumulative nephrotoxicity. Patients receiving concomitant cyclosporin should be closely monitored for signs of nephrotoxicity.
Other potential interactions. Caution regarding concomitant use with aminoglycosides (potentiation of ototoxic effects), depolarising neuromuscular blocking agents (enhancement of their effects), oral anticoagulants (reduce their effects by increasing concentration of clotting factors secondary to dehydration), and digoxin (digoxin toxicity if hypokalaemia follows mannitol treatment).
See Section 6.2 Incompatibilities.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category B2)
Teratogenic effects. Animal reproduction studies have not been conducted with mannitol. It is also not known whether mannitol can cause foetal harm when administered to a pregnant woman or can affect reproduction capacity. Mannitol should be given to a pregnant woman only if clearly needed.
Use in lactation. It is not known whether this medicine is excreted in human milk. Because many medicines are excreted in human milk, caution should be exercised when mannitol is administered to a nursing woman.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Extensive use of mannitol over the last several decades has produced recorded adverse events, in a variety of clinical settings that are isolated or idiosyncratic in nature. None of these adverse reactions have occurred with any great frequency or with any security in attributing them to mannitol.
The inability to clearly exclude the medicine related nature of such events in these isolated reports prompts the necessity to list the reactions that have been observed in patients during the following mannitol infusions.
Immediate reactions: can be noted, very rarely and in the same manner than with all Osmitrol IV Infusion solutions. (In these cases the infusion must be discontinued.)
Gastrointestinal disorder. Nausea; vomiting.
Hypersensitivity reactions. Local pain; skin necrosis; thrombophlebitis at the site if intravenous infusion; rhinitis; angioedema; allergic reaction; anaphylactic shock.
Neurological reactions. Chills; dizziness; urticaria; fever; headache; blurred vision; intracranial pressure increase.
Circulatory effects. Hypotension; hypertension; tachycardia; cardiac arrhythmia; angina-like chest pain; pulmonary congestion; oedema; convulsions; congestive cardiac failure.
Renal effects. Nephrosis osmotic; alveolar nephrosis. Large doses of mannitol have been known to cause acute renal failure even in patients with satisfactory pretreatment renal function. Excessive diuresis; urinary retention.
Blood disturbances. Acidosis; fluid and electrolyte imbalance.
Metabolic/ nutritional disorder. Dehydration; oedema; cramps; thirst; dryness of mouth.
Of far greater clinical significance are a variety of events that are related to inappropriate recognition and monitoring of fluid shifts. These are not intrinsic adverse reactions to the medicine but the consequence of manipulating osmolarity by an agency in a therapeutically inappropriate manner. Failure to recognise severe impairment of renal function with the high likelihood of nondiuretic response can lead to aggravated dehydration of tissues and increased vascular fluid load. Induced diuresis in the presence of pre-existing haemoconcentration and pre-existing deficiency of water and electrolytes can lead to serious imbalances. Expansion of the extracellular space can aggravate cardiac decompensation or induce it in the presence of latent heart failure. Pulmonary congestion or oedema can be seriously aggravated with the expansion of the extracellular fluid space by osmotic shift of water can induce or aggravate pre-existing hyponatremia.
These are not truly adverse reactions to the medicine and can be appropriately prevented by evaluation of degree of renal failure with a test dose response to mannitol when indicated; evaluation of hypervolemia and hypovolemia; sodium and potassium levels; haemodilution or haemoconcentration and evaluation of renal, cardiac and pulmonary function at the onset of therapy.
The following adverse reactions have been reported in the postmarketing experience listed by MedDRA System Organ Class (SOC).
Immune system disorders. Anaphylactic/ anaphylactoid reactions, including anaphylaxis, with skin, gastrointestinal, and severe circulatory (hypotension), and respiratory manifestations (e.g. dyspnea). Other hypersensitivity/ infusion reactions include hypertension, pyrexia, chills, sweating, cough, musculoskeletal stiffness and myalgia, urticaria/ rash, pruritus, generalized pain, discomfort, nausea, vomiting, and headache.
Metabolism and nutrition disorders. Fluid and electrolyte imbalances, including hypervolaemia, peripheral edema, dehydration, hyponatraemia, hypernatraemia, hyperkalaemia, hypokalaemia; metabolic acidosis, metabolic alkalosis.
Nervous system disorders. CNS toxicity manifested by, e.g. coma, convulsion, confusion, lethargy; rebound increase in intracranial pressure; dizziness.
Cardiac disorders. Congestive cardiac failure; palpitations.
Respiratory, thoracic and mediastinal disorders. Pulmonary edema.
Gastrointestinal disorders. Thirst, dry mouth.
Renal and urinary disorders. Renal failure acute, osmotic nephrosis, renal impairment, azotemia, anuria, hematuria, oliguria, polyuria.
General disorders and administration site conditions. Asthenia, malaise; infusion site reactions, including infusion site phlebitis, infusion site inflammation, infusion site pain, infusion site rash, infusion site erythema, infusion site pruritus; compartment syndrome, bullous eruptions, and swelling at the injection site associated with extravasation (see Section 4.4 Special Warnings and Precautions for Use, Infusion site reactions).
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
4.9 Overdose
In case of suspected overdose, treatment with Osmitrol IV Infusion should be stopped immediately.
Prolonged administration or rapid infusion of large volumes of hyperosmotic solutions may result in circulatory overload and acidosis. Headache, nausea and shivering without temperature change may represent initial signs/ symptoms. Confusion, lethargy, convulsions, stupor and coma may follow.
Signs and symptoms of overdose with mannitol may include acute renal failure, electrolytes imbalance, hypervolaemia and CNS toxicity.
Management is symptomatic and supportive, with monitoring of fluid electrolyte balance. Mannitol is dialyzable; haemodialysis may be useful in eliminating mannitol.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Osmitrol IV Infusion is one of the nonelectrolyte, obligatory, osmotic diuretics. It is freely filterable at the renal glomerulus, is poorly reabsorbed by the renal tubule, is not secreted by the tubule, and is pharmacologically inert.
The increase in extracellular osmolarity affected by the intravenous administration of mannitol will induce the movement of intracellular water to the extracellular and vascular spaces. The action underlies the role of mannitol in reducing intracranial pressure, intracranial oedema, and reducing elevated intraocular pressure.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Mannitol, when administered intravenously, exerts its osmotic effect as a solute of relatively small molecular size being largely confined to the extracellular space. Only relatively small amounts of the dose administered are metabolised. Mannitol is readily diffused through the glomerulus of the kidney over a wide range of normal and impaired kidney function. In this fashion, approximately 80% of a 100 g dose of mannitol will appear in the urine in three hours with lesser amounts thereafter. Even at peak concentrations, mannitol will exhibit less than 10% of tubular reabsorption and is not secreted by the tubular cells. Mannitol will hinder tubular reabsorption of water and enhance excretion of sodium and chloride by elevating the osmolarity of the glomerular filtrate.
5.3 Preclinical Safety Data
Genotoxicity. No data available.
Carcinogenicity. No data available.
6 Pharmaceutical Particulars
6.1 List of Excipients
Hydrochloric acid (for pH adjustment), sodium hydroxide (for pH adjustment).
6.2 Incompatibilities
There may be potential incompatibility with additives which include the risk of precipitation if potassium or sodium chloride is added to mannitol. Also, some antibiotics including cefepime, imipenem or cilastatin may be incompatible with mannitol.
Electrolyte and mannitol IV Infusions should be not given co-jointly with blood. If it is essential that blood be given simultaneously, at least 20 mEq of sodium chloride should be added to each litre of mannitol solution to avoid pseudoagglutination.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C.
Exposure of pharmaceutical products to heat should be minimised. Avoid excessive heat, brief exposure up to 40°C does not adversely affect the product.
6.5 Nature and Contents of Container
The Viaflex plastic container is fabricated from a specially formulated polyvinyl chloride (PL 146 Plastic). The amount of water that can permeate from inside the container into the overwrap is insufficient to affect the solution significantly. Solutions in contact with the plastic container can leach out certain of its chemical components in very small amounts within the expiration period, e.g. di-2-ethylhexyl phthalate (DEHP), up to 5 parts per million. However, the safety of the plastic has been confirmed in tests in animals according to USP biological tests for plastic containers as well as tissue culture toxicity studies.
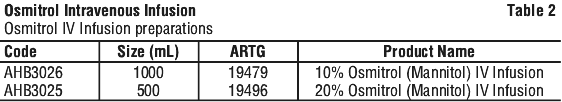
Osmitrol (Mannitol) IV Infusion in Viaflex plastic containers is available as shown in Table 2:
6.6 Special Precautions for Disposal
Any unused product or waste material should be disposed of in accordance with local requirements.
6.7 Physicochemical Properties
Chemical structure.
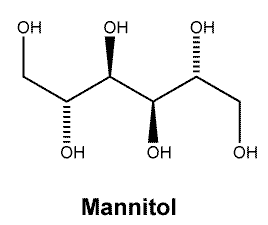
Molecular Weight: 182.2.
Appearance: white or almost white crystals or powder.
Solubility: freely soluble in water, practically insoluble in ethanol (96%).
CAS number. CAS No.: 69-65-8.
7 Medicine Schedule (Poisons Standard)
Not scheduled.
Date of First Approval
21 March 2007
Date of Revision
11 January 2021
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.