Panafcort
Brand Information
| Brand name | Panafcort |
| Active ingredient | Prednisone |
| Schedule | S4 |
Consumer Medicine Information (CMI) leaflet
Please read this leaflet carefully before you start using the Panafcort.
Summary CMI
Panafcort
Consumer Medicine Information (CMI) summary
The full CMI on the next page has more details. If you are worried about using this medicine, speak to your doctor or pharmacist.
1. Why am I using PANAFCORT?
PANAFCORT contains the active ingredient prednisone. PANAFCORT is used to treat many different conditions that involve inflammation, as well as to prevent or reduce symptoms associated with these conditions.
For more information, see Section 1. Why am I using PANAFCORT? in the full CMI.
2. What should I know before I use PANAFCORT?
Do not use if you have ever had an allergic reaction to prednisone, prednisolone, or any of the ingredients listed at the end of the CMI.
Talk to your doctor if you have any other medical conditions, take any other medicines, or are pregnant or plan to become pregnant or are breastfeeding.
For more information, see Section 2. What should I know before I use PANAFCORT? in the full CMI.
3. What if I am taking other medicines?
Some medicines may interfere with PANAFCORT and affect how it works.
A list of these medicines is in Section 3. What if I am taking other medicines? in the full CMI.
4. How do I use PANAFCORT?
- The dose of PANAFCORT varies from patient to patient.
- Your doctor will tell you how much to take each day, and when to take it.
- PANAFCORT is best taken with or immediately after food. Swallow the tablet whole with a glass of water.
More instructions can be found in Section 4. How do I use PANAFCORT? in the full CMI.
5. What should I know while using PANAFCORT?
| Things you should do |
|
| Things you should not do |
|
| Driving or using machines |
|
| Drinking alcohol |
|
| Looking after your medicine |
|
For more information, see Section 5. What should I know while using PANAFCORT? in the full CMI.
6. Are there any side effects?
Short term use of PANAFCORT is unlikely to cause any problems. But if problems do occur, they may likely involve mood changes and disturbances in your digestive system such as nausea, vomiting, anorexia, increased appetite, stomach bloating/irritation, or diarrhoea or constipation. Long term use and higher doses of PANAFCORT may have a greater risk of side effects. Serious side effects include severe stomach/intestinal pain, epileptic fits, psychiatric disturbances, sudden changes in your vision, or severe dizziness, fainting, weakness, chest pain, or irregular heartbeat.
For more information, including what to do if you have any side effects, see Section 6. Are there any side effects? in the full CMI.
Full CMI
1. Why am I using PANAFCORT?
PANAFCORT contains the active ingredient prednisone. PANAFCORT belongs to a group of medicines called corticosteroids. It is a synthetic version of a naturally occurring body hormone called cortisol.
PANAFCORT is used in the treatment of many different conditions. Some of these conditions include: severe allergies, severe or chronic asthma, skin problems, arthritis, inflammatory diseases of the bowel, cancer and "auto-immune" diseases.
PANAFCORT is also used to prevent or reduce the symptoms of inflammation (such as swelling, redness, pain, tenderness or itching) in any part of the body. These symptoms can occur in response to injury or can be caused by many different conditions.
It works by entering inflammatory cells and blocking the inflammatory reaction. PANAFCORT is only able to prevent or reduce symptoms of your condition, it does not cure it.
Ask your doctor if you have any questions about why PANAFCORT has been prescribed for you.
Your doctor may have prescribed it for another reason.
This medicine is only available with a doctor's prescription.
There is no evidence that it is addictive.
2. What should I know before I use PANAFCORT?
Warnings
Do not use PANAFCORT if:
- You are allergic to prednisolone, prednisone or any of the ingredients listed at the end of this leaflet.
Some symptoms of an allergic reaction may include shortness of breath, wheezing or difficulty in breathing; swelling of the face, lips, tongue or any other parts of the body; rash, itching or hives on the skin.
Always check the ingredients to make sure you can use this medicine. - You have a current serious or uncontrolled infection, including fungal infections.
- The expiry date on the pack has passed.
It may have no effect at all or an entirely unexpected effect if you take it after the expiry date. - The bottle shows signs of having been tampered with.
- You are using it to treat any other complaints unless your doctor has instructed you to do so.
Do not give this medicine to anyone else.
Check with your doctor if you:
Have any other medical conditions, especially the following:
- a current serious or uncontrolled infection, including fungal infections
- recent surgery or serious injury
- diabetes mellitus (sugar diabetes)
- osteoporosis (softening of the bone)
- a stomach ulcer or other stomach or intestinal problems
- liver, kidney or heart disease
- tuberculosis
- epilepsy
- muscle weakness
- glaucoma (high pressure in the eye) or cataracts
- thyroid disease
- high blood pressure
It may not be safe for you to take PANAFCORT if you have any of these medical conditions.
Take any medicines for any other condition
Have had any allergy to any other medicines, or any other substances, such as foods, preservatives, or dyes.
During treatment, you may be at risk of developing certain side effects. It is important you understand these risks and how to monitor for them. See additional information under Section 6. Are there any side effects?
Pregnancy and breastfeeding
Do not take PANAFCORT if you are pregnant or plan to become pregnant unless you and your doctor have discussed the risks and benefits involved.
Do not take it if you are breastfeeding or intend to breastfeed.
It is not recommended for use while breastfeeding as it is found in breast milk.
Use in children
Take special care when giving PANAFCORT to children. It should only be given under your doctor's supervision.
If possible, children should not be exposed to common childhood illnesses such as chickenpox or measles while they are taking this medicine. They may suffer from more serious attacks of these illnesses if such exposure occurs.
Children should not be vaccinated with "live" vaccines against common childhood illnesses while they are taking it, as this may result in severe attacks of these illnesses.
Potentially serious side effects can occur in children and growing teenagers who are taking corticosteroids. Some of these include obesity, slowed growth, osteoporosis (softening of the bone) and changes to the adrenal glands.
Use in the elderly
Elderly patients may be more sensitive to the effects or side effects of this medicine.
3. What if I am taking other medicines?
Tell your doctor or pharmacist if you are taking any other medicines, including any medicines, vitamins or supplements that you buy without a prescription from your pharmacy, supermarket or health food shop.
Some medicines may interfere with PANAFCORT and affect how it works. These include:
- antacids (in large amounts)
- medicines for diabetes
- some medicines for heart disease
- medicines for removal of fluid (diuretics)
- some medicines for epilepsy
- some types of antibiotics
- potassium or salt supplements
- immunisations or vaccines.
You may need to take different amounts of your medicine, or you may need to take different medicines. Your doctor or pharmacist has a more complete list of medicines to avoid while taking this medicine.
Check with your doctor or pharmacist if you are not sure about what medicines, vitamins or supplements you are taking and if these affect PANAFCORT.
4. How do I use PANAFCORT?
How much to take
- Your doctor will tell you how much PANAFCORT to take.
- The dose will depend on the condition being treated and your response to the treatment. Your initial dose will be maintained or adjusted until a satisfactory response is noted.
How to take it
- Swallow the tablet whole with a glass of water.
When to take it
- PANAFCORT is best taken with or immediately after food.
- How often PANAFCORT can be taken may vary depending on what condition is being treated. It can be taken once daily, several times a day or on alternate days (every other day).
- If you take it once a day or every second day, then it is best taken at breakfast time.
- If it needs to be taken more than once a day, then space it out during the day.
How long to take it for
- Continue taking PANAFCORT for as long as your doctor or pharmacist tells you.
This will depend on your condition and on your response to treatment. Some people will need this medicine for only short periods of time whilst others may require long term therapy. - Do not miss any doses and do not stop taking the medicine even if you feel better.
Missing doses may make your symptoms worse.
What to expect
Individuals will vary greatly in their response to PANAFCORT. Your doctor will check your progress at regular intervals.
If you forget to take PANAFCORT
If you miss a dose of this medicine the decision of whether you should take it or not will depend on how many times a day you take PANAFCORT.
If you are taking PANAFCORT:
- Once a day -
If you miss a dose and remember in less than 12 hours, take it straight away, then continue as normal the next day. Otherwise, skip that day's dose but be sure to take the next day's dose when it is due. - Several times a day -
If you miss a dose and it is more than 2 hours before your next dose is due, take it straight away, then continue as you normally would. If it is less than 2 hours to your next dose, skip the dose you have missed but be sure to take the next dose when you are meant to. - On alternate days -
If you miss a dose and remember it the same morning, take it straight away then continue as you normally would. If you do not remember the missed dose until later, wait and take it the following morning. Then skip a day before continuing your regular dosage schedule.
Do not try to make up for missed doses by taking more than one dose at a time.
If you have trouble remembering when to take your medicine, ask your pharmacist for some hints.
If you use too much PANAFCORT
If you think that you have used too much PANAFCORT, you may need urgent medical attention.
You should immediately:
- phone the Poisons Information Centre
(by calling 13 11 26), or - contact your doctor, or
- go to the Emergency Department at your nearest hospital.
You should do this even if there are no signs of discomfort or poisoning.
5. What should I know while using PANAFCORT?
Things you should do
- Take PANAFCORT exactly as your doctor has prescribed.
If you do not follow your doctor's instructions, you may not get improvement in the symptoms of your condition. Try not to miss any doses and take the medicine even if you feel well. - Tell your doctor if your condition returns or becomes worse after your dose of PANAFCORT has been reduced or treatment has been stopped.
- Tell any other doctors, dentists and pharmacists who are treating you that you are taking PANAFCORT, especially if you are being started on any new medicines.
- Tell your doctor, surgeon, or dentist that you are taking PANAFCORT if you are about to undergo surgery or an operation.
Your dose of this medicine may need to be increased to cover you during the stress of the operation. - Ask your doctor when and how you should stop taking PANAFCORT.
If you have been taking it for a long time your doctor may gradually reduce the amount you are taking over a period of several days, weeks or months before stopping completely. If you have been taking PANAFCORT for a short period of time, this may not apply. - Tell your doctor straight away if you are a diabetic, and you notice a change in the results of your blood or urine sugar tests.
This medicine may affect your blood sugar levels as it can affect the body's ability to handle glucose. For diabetics, this means that your diabetes may become more severe. For others, diabetes may develop for the first time while taking corticosteroids such as PANAFCORT. - Tell your doctor immediately if you become pregnant while taking PANAFCORT.
Things you should not do
- Do not stop taking PANAFCORT suddenly unless advised by your doctor.
If you stop taking it suddenly, the symptoms of your condition may return or you may develop symptoms of cortisol deficiency such as fainting, weakness, restlessness, nausea, vomiting, headache, dizziness, muscle weakness or joint pain. - Do not have any immunisations (particularly with "live" vaccines such as measles, oral polio or yellow fever) without your doctor's approval while you are being treated with PANAFCORT.
- Do not take PANAFCORT to treat any other complaints unless your doctor has instructed you to do so.
- Do not give this medicine to anyone else, even if their symptoms seem similar to yours.
Things to be careful of
- Avoid close contact with anyone who has a contagious disease such as measles or chickenpox.
Exposure to such diseases while you are taking PANAFCORT, particularly if large doses are being taken, can put you at greater risk of developing these diseases if you have not had them before. - Tell your doctor straight away if you think you have been exposed to chickenpox or measles.
The signs and symptoms of infections such as fever or inflammation may be hidden by the anti-inflammatory action of PANAFCORT. You should see your doctor for medical advice for any but the most minor infections.
Driving or using machines
As with any new medicine, you should take care when driving or operating machinery until you know how PANAFCORT affects you.
Drinking alcohol
Check with your doctor or pharmacist before drinking alcohol while you are taking PANAFCORT.
If you drink alcohol while taking PANAFCORT you may find that stomach problems occur.
Looking after your medicine
Keep PANAFCORT in a cool dry place, protected from light, where the temperature stays below 30°C.
Follow the instructions in the carton on how to take care of your medicine properly.
Store it in a cool dry place away from moisture, heat or sunlight; for example, do not store it:
- in the bathroom or near a sink, or
- in the car or on window sills.
Heat and dampness can destroy some medicines.
Keep it where young children cannot reach it.
A locked cupboard at least one-and-a-half metres above ground is a good place to store medicines.
Getting rid of any unwanted medicine
If you no longer need to use this medicine or it is out of date, take it to any pharmacy for safe disposal.
Do not use this medicine after the expiry date.
6. Are there any side effects?
Tell your doctor or pharmacist as soon as possible if you do not feel well while you are taking PANAFCORT.
All medicines can have side effects. If you do experience any side effects, most of them are minor and temporary. However, some side effects may need medical attention.
See the information below and, if you need to, ask your doctor or pharmacist if you have any further questions about side effects.
Less serious side effects
| Less serious side effects | What to do |
| Short term use When PANAFCORT is taken for short periods of time it is unlikely to cause any problems.
| Speak to your doctor if you have any of these less serious side effects and they worry you. |
| Long term use When PANAFCORT is taken for long periods of time and in high doses the risk of side effects is greater. general changes to the body:
| Tell your doctor if you have any of these less serious side effects and they worry you. |
Serious side effects
| Serious side effects | What to do |
| Call your doctor straight away, or go straight to the Emergency Department at your nearest hospital if you notice any of these serious side effects. |
Some side effects can only be detected by your doctor. So it is important to visit your doctor for regular check-ups when PANAFCORT is taken for long periods of time.
Such side effects can include:
- osteoporosis or other changes in bone which can result in an increased chance of fractures due to brittleness or softening of the bone.
- changes in other hormone levels in your body
- changes in the body's ability to handle glucose (steroid diabetes)
- effects on the parathyroid and thyroid glands which control calcium and body metabolism
- increased amounts of cholesterol in the blood
- changes to your white blood cells
- changes to your nervous system which may affect the way your nerves work
- changed sperm count
- increased blood pressure
- slow heart rate
- increased pressure in the skull
- increased pressure in the eye (glaucoma).
Tell your doctor or pharmacist if you notice anything else that may be making you feel unwell.
Other side effects not listed here may occur in some people.
Some people may get other side effects while using PANAFCORT.
Do not be alarmed by this list of possible side effects.
You may not experience any of them.
Reporting side effects
After you have received medical advice for any side effects you experience, you can report side effects to the Therapeutic Goods Administration online at www.tga.gov.au/reporting-problems. By reporting side effects, you can help provide more information on the safety of this medicine.
Always make sure you speak to your doctor or pharmacist before you decide to stop taking any of your medicines.
7. Product details
This medicine is only available with a doctor's prescription.
What PANAFCORT contains
| Active ingredient (main ingredient) |
|
| Other ingredients (inactive ingredients) |
|
Do not take this medicine if you are allergic to any of these ingredients.
PANAFCORT tablets do not contain gluten, sucrose, tartrazine or any other azo dyes.
What PANAFCORT looks like
PANAFCORT tablets are available in three strengths: 1 mg, 5 mg and 25 mg.
The 1 mg tablets are white, scored, round and biconvex. They are marked with "PN/1" on one side.
Available in HDPE bottles of 100.
(AUST R 39879)
The 5 mg tablets are white, scored, round and biconvex. They are marked with "PN/5" on one side.
Available in HDPE bottles of 60.
(AUST R 27974)
The 25 mg tablets are white, scored, round and biconvex. They are marked with "PN/25" on one side.
Available in HDPE bottles of 30.
(AUST R 27957)
Who distributes PANAFCORT
Aspen Pharmacare Australia Pty Ltd
34-36 Chandos St
St Leonards NSW 2065
Australia
This leaflet was prepared in August 2025.
Brand Information
| Brand name | Panafcort |
| Active ingredient | Prednisone |
| Schedule | S4 |
MIMS Revision Date: 01 June 2022
1 Name of Medicine
Prednisone.
2 Qualitative and Quantitative Composition
Panafcort tablets contain either 1 mg, 5 mg or 25 mg of the active prednisone.
List of excipients with known effects: lactose monohydrate. For the full list of excipients, see Section 6.1 List of Excipients.
3 Pharmaceutical Form
1 mg: Round, biconvex, white scored tablet with 'PN/1' on one side and plain on the other.
5 mg: Round, biconvex, white scored tablet debossed with 'PN/5' on one side and plain on the other.
25 mg: Round, biconvex, white scored tablet debossed with 'PN/25' on one side and plain on the other.
4 Clinical Particulars
4.1 Therapeutic Indications
Wherever corticosteroid therapy is indicated such as: pemphigus vulgaris, allergic dermatitis, eczema, exfoliative dermatitis, dermatitis herpetiformis, dermatitis medicamentosa, erythema multiforme; disseminated lupus erythematosus, dermatomyositis, polyarteritis nodosa; severe bronchial asthma and status asthmaticus, emphysema, pulmonary fibrosis; adrenal hyperplasia (adrenogenital syndrome); idiopathic thrombocytopenic purpura, acquired haemolytic anaemia, acute leukaemia; nephrotic syndrome; iridochoroiditis; ulcerative colitis; rheumatoid arthritis; ankylosing spondylitis, rheumatic fever, gout, periarthritis of the shoulder.
4.2 Dose and Method of Administration
The severity, prognosis, expected duration of the disease, and the patient's reaction to medication are primary factors in determining dosage.
Despite the 1 mg tablet being scored, it should not be broken.
Adults. The initial adult dosage may range from 20 to 40 mg daily, but can be 60 to 80 mg daily if necessary, depending on the disease being treated.
Maintenance dosage. Usually 5 to 20 mg daily. In long term therapy the ideal dosage should not be greater than 40 mg per day so as to minimise side-effects. It is usually administered in 2-4 divided doses or as a single daily dose after breakfast or on alternate days.
Alternate-day therapy. Alternate-day therapy is the dosage regimen of choice for long-term oral glucocorticoid treatment of most conditions. In alternate-day therapy, a single dose is administered every other morning. This regimen provides relief of symptoms while minimizing adrenal suppression, protein catabolism, and other adverse effects. However, some patients may require daily glucocorticoid therapy because symptoms of the underlying disease cannot be controlled by alternate-day therapy.
Elderly. As for adults - though the dose should be the minimum necessary to achieve the desired therapeutic effect.
Children. Initial dosage. 0.5 mg/kg daily in three or four divided doses after food as in adults. This dosage can be doubled or trebled if necessary.
Maintenance dosage. 0.125 to 0.25 mg/kg daily.
For infants and children, the recommended dosage should be governed by the same considerations as adults rather than by strict adherence to the ratio indicated by age or body weight.
The following therapeutic guidelines should be kept in mind for all therapy with corticosteroids. Dosage should be decreased or discontinued gradually when the drug has been administered for more than a few days to avoid the risk of relative adrenal insufficiency (see Section 4.4 Special Warnings and Precautions for Use).
Continued supervision of the patient after cessation of corticosteroids is essential, since there may be a reappearance of severe manifestations of the disease for which the patient was treated.
In general, initial dosage should be maintained or adjusted until the anticipated response is observed. The dose should then be gradually reduced until the lowest dose which will maintain an adequate clinical response is reached.
Stress and intercurrent illness. In patients on long term corticosteroid therapy subjected to stress from trauma or infection, steroid dosage should generally be increased to cover the stressful period. For mild infections without fever, no increase is necessary. For more serious infections, the dose of prednisone/prednisolone should be doubled (to a maximum of 20 mg daily, if the usual dosage was below this).
Adrenocortical insufficiency. Drug induced secondary adrenocortical insufficiency may result from too rapid withdrawal of corticosteroids and may be minimised by gradual reduction of dosage (see Section 4.4 Special Warnings and Precautions for Use). This type of relative insufficiency may persist for months after discontinuation of therapy; therefore, in any situation of stress occurring during that period, hormone therapy may need to be reinstituted. If the patient is receiving steroids already, dosage may have to be increased.
4.3 Contraindications
Uncontrolled infections; known hypersensitivity to prednisone or prednisolone, or any of the excipients in the tablet.
4.4 Special Warnings and Precautions for Use
Scleroderma renal crisis. Caution is required in patients with systemic sclerosis because of an increased incidence of (possibly fatal) scleroderma renal crisis with hypertension and decreased urinary output observed with a daily dose of 15 mg or more prednisone or prednisolone. Blood pressure and renal function (s-creatinine) should therefore be routinely checked. When renal crisis is suspected, blood pressure should be carefully controlled.
During prolonged corticosteroid therapy, adrenal suppression and atrophy may occur and secretion of corticotrophin may be suppressed.
Duration of treatment and dosage appear to be important factors in determining suppression of the pituitary adrenal axis and response to stress on cessation of steroid treatment. The patient's liability to suppression is also variable. Some patients may recover normal function rapidly. In others, the production of hydrocortisone in response to the stress of infections, surgical operations or accident may be insufficient, and death results. Therefore, withdrawal of corticosteroids should always be gradual.
Abrupt withdrawal of corticosteroid therapy may precipitate acute adrenal insufficiency (see Section 4.8 Adverse Effects (Undesirable Effects)). In some cases, withdrawal symptoms may simulate a clinical relapse of the disease for which the patient has been under treatment.
Use with caution in the following circumstances. Use with caution in nonspecific ulcerative colitis, if there is a probability of impending perforation, abscess or other pyogenic infection. Caution must also be used in diverticulitis, fresh intestinal anastomoses, active or latent peptic ulcer, renal insufficiency, hypertension and myasthenia gravis, when steroids are used as direct or adjunctive therapy.
Use with caution in patients with epilepsy, diabetes mellitus, uraemia and in the presence of diminished cardiac reserve or congestive heart failure (see Section 4.8 Adverse Effects (Undesirable Effects)).
The possibility of development of osteoporosis should be an important consideration in initiating and managing corticosteroid therapy, especially in post menopausal women (see Section 4.8 Adverse Effects (Undesirable Effects)).
The risk of gastrointestinal ulceration or hemorrhage is increased when alcohol is used concurrently with glucocorticoids.
Infection. Corticosteroids may mask some signs of infection (such as fever and inflammation), and new infections may appear during their use. There may be decreased resistance and inability to localise infection when corticosteroids are used. Susceptibility to infection is not specific for any particular bacterial or fungal pathogen.
Patients should not be vaccinated with live vaccines while on corticosteroid therapy. Other immunisation procedures should not be undertaken in patients on corticosteroid therapy, especially on high doses, because of possible hazards of neurological complications and lack of antibody response. Immunization procedures may be undertaken in patients receiving corticosteroids as replacement therapy.
Children who are on immunosuppressant drugs are more susceptible to infections than healthy children. Chickenpox and measles, for example, can have a more serious or even fatal course in children on immunosuppressant corticosteroids. In such children, or in adults who have not had these diseases, particular care should be taken to avoid exposure. If exposed, therapy with varicella zoster immune globulin (VZIG) or pooled intravenous immunoglobulin (IVIG), as appropriate, may be indicated. If chickenpox develops, treatment with antiviral agents may be considered.
Tuberculosis. Patients with active or doubtfully quiescent tuberculosis should not be given Panafcort except as adjuncts to treatment with tuberculostatic drugs as reactivation of the disease may occur. Chemoprophylaxis is indicated during prolonged corticosteroid therapy.
Visual disturbance. Visual disturbance may be reported with systemic and topical corticosteroid use. If a patient presents with symptoms such as blurred vision or other visual disturbances, the patient should be considered for referral to an ophthalmologist for evaluation of possible causes which may include cataract, glaucoma or rare diseases such as central serous chorioretinopathy (CSCR) which have been reported after use of systemic and topical corticosteroids.
Check the following before use. During long courses of treatment, laboratory and metabolic studies should be made. Fluid retention should be watched for via a fluid balance chart and daily weighing. Sodium intake may need to be reduced to less than 1 g daily and potassium supplements may be necessary.
Use in hepatic impairment. Use with caution in patients with impaired hepatic function, a reduction of dosage may be necessary. In treating chronic active liver disease with the drug, major adverse reactions such as vertebral collapse, diabetes, hypertension, cataracts and Cushing's syndrome occur in about 30% of patients.
Use in renal impairment. See Section 4.4, Scleroderma renal crisis.
Use in the elderly. Caution is recommended for elderly patients as they are more susceptible to adverse reactions.
Paediatric use. Children on long term steroids must be carefully observed for potential serious adverse reactions such as obesity, growth retardation, osteoporosis and adrenal suppression.
Effects on laboratory tests. Glucocorticoids may decrease I131 uptake and protein-bound iodine concentrations, making it difficult to monitor the therapeutic response of patients receiving the drugs for thyroiditis. Glucocorticoids may produce false-negative results in the nitroblue tetrazolium test for systemic bacterial infection. Glucocorticoids may suppress reactions to skin tests.
4.5 Interactions with Other Medicines and Other Forms of Interactions
The following drug interactions with corticosteroids have been selected on the basis of their potential clinical significance: antacids, antidiabetic agents (oral or insulin), digitalis glycosides, diuretics, drugs which induce hepatic microsomal enzymes, such as barbiturates, phenytoin and rifampicin; potassium supplements, ritodrine, sodium-containing medications or foods, somatrem or somatropin, vaccines, live viruses or other immunisations.
4.6 Fertility, Pregnancy and Lactation
Effects on fertility. No data available.
Use in pregnancy. (Category A)
In animal experiments, corticosteroids have been found to cause malformations of various kinds (cleft palate, skeletal malformations) and abortion. These findings do not seem to be relevant to humans. Reduced placental and birth weight have been recorded in animals and humans after long term treatment. Since the possibility of suppression of the adrenal cortex in the new born baby after long term treatment must be considered, the needs of the mother must be carefully weighed against the risk to fetus when prescribing corticosteroids. The short term use of corticosteroids antepartum for the prevention of respiratory distress syndrome, does not seem to pose a risk to the fetus or the newborn infant. Maternal pulmonary oedema has been reported with tocolysis and fluid overload.
Use in lactation. The drug is excreted in breast milk; therefore, administration to nursing mothers is not recommended.
4.7 Effects on Ability to Drive and Use Machines
The effects of this medicine on a person's ability to drive and use machines were not assessed as part of its registration.
4.8 Adverse Effects (Undesirable Effects)
Reporting suspected adverse effects. Reporting suspected adverse reactions after registration of the medicinal product is important. It allows continued monitoring of the benefit-risk balance of the medicinal product. Healthcare professionals are asked to report any suspected adverse reactions at www.tga.gov.au/reporting-problems.
Short-term administration of Panafcort, even in massive dosages, is unlikely to produce harmful effects. The majority of adverse reactions from corticosteroids are those resulting from withdrawal or from prolonged use of high doses.
More common reactions. Gastrointestinal. Adverse gastrointestinal effects of corticosteroids include nausea, vomiting, anorexia (which may result in weight loss), increased appetite (which may result in weight gain), diarrhoea or constipation, abdominal distension and gastric irritation.
Cardiovascular. The mineralocorticoid activity of a steroid may lead to salt and water retention which can also result in hypertension. Hypokalaemia can lead to arrhythmias and cardiac arrest.
Neurological. Adverse neurological effects have included headache, vertigo, insomnia, restlessness and increased motor activity, ischemic neuropathy, EEG abnormalities and seizures. Large doses can cause behavioural and personality changes ranging from nervousness, euphoria or mood swings to psychotic episodes which can include both manic and depressive states, paranoid states and acute toxic psychoses.
It is no longer believed that previous psychiatric problems predispose to behavioural disturbances during therapy with glucocorticoids. Conversely, the absence of a history of psychiatric illness is no guarantee against the occurrence of psychosis during hormonal therapy.
Dermatological. Dermatological adverse effects of corticosteroids include impaired wound healing, facial plethora, increased sweating, easy bruising, hirsutism, an acneiform eruption on the face, chest and back, red striae on the thighs, buttocks and shoulders. Several months of high dose therapy can often result in thinning of skin. Dermatologic manifestations of hypersensitivity to the corticosteroids include hives and/or allergic dermatitis, urticaria, and angioedema.
Corticosteroid induced purpura resembles senile purpura. This purpura usually occurs on extensor surfaces, dorsum of the hand, and radial aspect of the forearm.
Endocrine. The endocrine effects of the glucocorticoids involve variously the hypothalamic pituitary adrenal axis; the parathyroid and thyroid. There are also metabolic effects, primarily involving the carbohydrates. Suppression of growth may occur in children.
Cushing's syndrome may result from prolonged elevation of plasma glucocorticoid levels.
Corticosteroids have also been reported to increase or decrease motility and number of sperm in some men. Disorders of menstruation are common.
Antagonism occurs between the parathyroids and hypercorticism. Latent hypoparathyroidism may be unmasked by administration of corticosteroids. The phosphate retention occurring in renal failure caused by adrenal insufficiency may also make hypoparathyroidism manifest.
Biochemical. All glucocorticoids increase gluconeogenesis. Glucose tolerance and sensitivity to insulin are decreased but provided pancreatic islet function is normal carbohydrate metabolism will not be noticeably deranged. Steroid diabetes, has been reported to develop in one fifth of patients treated with high glucocorticoid dosage.
High dose corticosteroid therapy may induce marked hypertriglyceridaemia with milky plasma.
General. Retardation of growth by long term corticosteroid treatment in children.
Haematological. Corticosteroids will increase the total WBC count, with an increase in neutrophils and a decrease in monocytes, lymphocytes and eosinophils.
Immunological. The frequency and severity of clinical infections increase during glucocorticoid therapy.
Musculoskeletal. Osteoporosis and vertebral compression fractures can occur in patients of all ages. Osteoporosis is an indication for withdrawal of therapy.
Myopathy, characterised by weakness of the proximal musculature of arms and legs and their associated shoulder and pelvic muscles, is occasionally reported in patients taking large doses of corticosteroids. It may occur soon after treatment is begun and be sufficiently severe to prevent ambulation. It is an indication for withdrawal of therapy.
Avascular aseptic necrosis of bone has often been described and preferentially involves the femoral and humeral head.
Serious or life threatening reactions. Suppression of the hypothalamic pituitary adrenal axis is one of the consequences of repeated administration of glucocorticoids (see Section 4.4 Special Warnings and Precautions for Use). In some cases acute adrenal insufficiency after a period of glucocorticoid treatment has proved fatal.
Less common reactions. Gastrointestinal. Pancreatitis and ulcerative oesophagitis can occur. Peptic ulceration is an occasional complication. The high incidence of haemorrhage and perforation in these ulcers and the insidious nature of their development make them severe therapeutic problems. Some investigators believe the available evidence does not support the conclusion that steroids cause ulcers. Others feel that only patients with rheumatoid arthritis have an increased incidence of ulcers. It has been proposed that the glucocorticoids alter the mucosal defense mechanism.
Neurological. Latent epilepsy can be rendered manifest by corticosteroid treatment. Long term treatment may result in benign intracranial hypertension.
Ophthalmological. Prolonged use of glucocorticoids may result in posterior subcapsular cataracts (particularly in children), exophthalmos, or increased intraocular pressure which may result in glaucoma or may occasionally damage the optic nerve and in rare cases, lead to blindness. Establishment of secondary fungal and viral infections of the eye may also be enhanced.
Post marketing. Eye disorders. Blurred vision.
Scleroderma renal crisis. Frequency 'unknown'. Amongst the different subpopulations the occurrence of scleroderma renal crisis varies. The highest risk has been reported in patients with diffuse systemic sclerosis. The lowest risk has been reported in patients with limited systemic sclerosis (2%) and juvenile onset systemic sclerosis (1%).
Cardiac disorders. Frequency 'unknown'. Bradycardia has been reported following high doses.
Withdrawal adverse effects. Muscle weakness, hypotension, hypoglycaemia, headache, nausea, vomiting, restlessness and muscle and joint pain. Muscle weakness and stiff joints may persist for three to six months after discontinuation of treatment.
4.9 Overdose
Treatment is symptomatic with the dosage being reduced or the drug withdrawn.
For information on the management of overdose, contact the Poisons Information Centre on 13 11 26 (Australia).
5 Pharmacological Properties
5.1 Pharmacodynamic Properties
Mechanism of action. Prednisone is a synthetic corticosteroid with glucocorticoid and anti-inflammatory effects. Prednisone has the same chemical relationship to prednisolone as cortisone has to hydrocortisone.
The mechanism of action of corticosteroids is thought to be by control of protein synthesis. Corticosteroids react with receptor proteins in the cytoplasm of sensitive cells in many tissues to form a steroid-receptor complex.
Corticosteroids are palliative symptomatic treatment by virtue of their anti-inflammatory effects; they are never curative.
Clinical trials. No data available.
5.2 Pharmacokinetic Properties
Prednisone is readily absorbed from the gastrointestinal tract, but whereas prednisolone already exists in a metabolically active form, prednisone must be converted in the liver to its active metabolite, prednisolone.
Absorption. The plasma half-life after oral administration ranges from 3 to 4 hours. Oral bioavailability varies widely between subjects.
Metabolism. The conversion from prednisone into prednisolone is rapid so that prednisone has a preconversion biological half-life of only about 60 minutes.
Excretion. Little Panafcort is excreted unchanged in the urine.
5.3 Preclinical Safety Data
Genotoxicity. In male rats, administration of prednisolone in the drinking water at a daily dose level of 0.4 mg/kg for two years caused an increased incidence of hepatocellular tumours. Similar results were obtained with triamcinolone acetonide and budesonide, indicating a class effect of glucocorticosteroids. The hepatocarcinogenic response to these drugs does not appear to be related to genotoxic activity.
Carcinogenicity. The carcinogenic potential of prednisone has been evaluated in mice at oral doses up to 5 mg/kg/day for 18 months. No carcinogenic effect was noted in the mouse.
6 Pharmaceutical Particulars
6.1 List of Excipients
Panafcort tablets contain lactose monohydrate, povidone, maize starch, magnesium stearate, crospovidone and macrogol 6000 (Panafcort 25 mg tablets only).
6.2 Incompatibilities
Incompatibilities were either not assessed or not identified as part of the registration of this medicine.
6.3 Shelf Life
In Australia, information on the shelf life can be found on the public summary of the Australian Register of Therapeutic Goods (ARTG). The expiry date can be found on the packaging.
6.4 Special Precautions for Storage
Store below 30°C.
6.5 Nature and Contents of Container
1 mg [AUST R 39879]: HDPE bottles of 100 and bulk* tablets.
5 mg [AUST R 27974]: HDPE bottles of 30*, 60 and 90* and bulk* tablets.
25 mg [AUST R 27957]: HDPE bottles of 30 and bulk* tablets.
(*Not currently distributed in Australia).
6.6 Special Precautions for Disposal
In Australia, any unused medicine or waste material should be disposed of by taking to your local pharmacy.
6.7 Physicochemical Properties
Prednisone occurs as a white to practically white, odourless, crystalline powder. It is very slightly soluble in water; slightly soluble in alcohol, in chloroform, in dioxane, and in methanol.
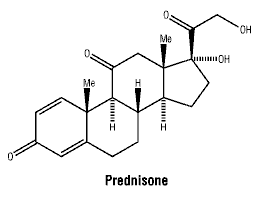
Chemical name: 17, 21-Dihydroxypregna-1, 4-diene-3,11,20-trione. Molecular weight: 358.4.
Chemical structure.
7 Medicine Schedule (Poisons Standard)
S4.
Date of Revision
09 May 2022
Summary Table of Changes

Reasonable care is taken to provide accurate information at the time of creation. This information is not intended as a substitute for medical advice and should not be exclusively relied on to manage or diagnose a medical condition. The Australian Commission on Safety and Quality in Health Care disclaims all liability (including for negligence) for any loss, damage, injury or any other negative effects resulting from reliance on or use of this information. Read our full disclaimer. This website uses cookies. Read our privacy policy.